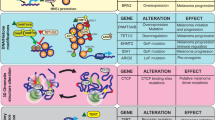
Abstract
Melanoma is a common cancer in the Western world with an increasing incidence. Sun exposure is still considered to be the major risk factor for melanoma. The prognosis of patients with malignant (advanced-stage) melanoma differs widely between countries, but public campaigns advocating early detection have led to significant reductions in mortality rates. As well as sun exposure, distinct genetic alterations have been identified as associated with melanoma. For example, families with melanoma who have germline mutations in CDKN2A are well known, whereas the vast majority of sporadic melanomas have mutations in the mitogen-activated protein kinase cascade, which is the pathway with the highest oncogenic and therapeutic relevance for this disease. BRAF and NRAS mutations are typically found in cutaneous melanomas, whereas KIT mutations are predominantly observed in mucosal and acral melanomas. GNAQ and GNA11 mutations prevail in uveal melanomas. Additionally, the PI3K–AKT–PTEN pathway and the immune checkpoint pathways are important. The finding that programmed cell death protein 1 ligand 1 (PDL1) and PDL2 are expressed by melanoma cells, T cells, B cells and natural killer cells led to the recent development of programmed cell death protein 1 (PD1)-specific antibodies (for example, nivolumab and pembrolizumab). Alongside other new drugs — namely, BRAF inhibitors (vemurafenib and dabrafenib) and MEK inhibitors (trametinib and cobimetinib) — these agents are very promising and have been shown to significantly improve prognosis for patients with advanced-stage metastatic disease. Early signs are apparent that these new treatment modalities are also improving long-term clinical benefit and the quality of life of patients. This Primer summarizes the current understanding of melanoma, from mechanistic insights to clinical progress. For an illustrated summary of this Primer, visit: http://go.nature.com/vX2N9s
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Schadendorf, D. & Hauschild, A. Melanoma in 2013: Melanoma—the run of success continues. Nature Rev. Clin. Oncol. 11, 75–76 (2014).
Lawrence, M. S. et al. Mutational heterogeneity in cancer and the search for new cancer-associated genes. Nature 499, 214–218 (2013).
Si, L., Wang, X. & Guo, J. Genotyping of mucosal melanoma. Chin. Clin. Oncol. 3, 27 (2014).
De Angelis, R. et al. Cancer survival in Europe 1999–2007 by country and age: results of EUROCARE—5-a population-based study. Lancet Oncol. 15, 23–34 (2014).
Pflugfelder, A. et al. Malignant melanoma S3-guideline “diagnosis, therapy and follow-up of melanoma”. J. Dtsch. Dermatol. Ges. 11 (Suppl. 6), 1–116, 1–126 (2013).
Garbe, C. & Bauer, J. in Dermatology 3rd edn (eds Bolognia, J. L., Jorizzo, J. L. & Schaffer, J. V. ) 1885–1914 (Elsevier, 2012).
Erdmann, F. et al. International trends in the incidence of malignant melanoma 1953-2008—are recent generations at higher or lower risk? Int. J. Cancer 132, 385–400 (2013). A paper describing the incidence rates of melanoma across the world.
Geller, A. C. et al. Melanoma epidemic: an analysis of six decades of data from the Connecticut Tumor Registry. J. Clin. Oncol. 31, 4172–4178 (2013).
Garbe, C. & Leiter, U. Melanoma epidemiology and trends. Clin. Dermatol. 27, 3–9 (2009).
MacKie, R. M., Hauschild, A. & Eggermont, A. M. Epidemiology of invasive cutaneous melanoma. Ann. Oncol. 20 (Suppl. 6), vi1–vi7 (2009).
Hausauer, A. K., Swetter, S. M., Cockburn, M. G. & Clarke, C. A. Increases in melanoma among adolescent girls and young women in California: trends by socioeconomic status and UV radiation exposure. Arch. Dermatol. 147, 783–789 (2011).
Breitbart, E. W. et al. Systematic skin cancer screening in Northern Germany. J. Am. Acad. Dermatol. 66, 201–211 (2012).
Iannacone, M. R., Youlden, D. R., Baade, P. D., Aitken, J. F. & Green, A. C. Melanoma incidence trends and survival in adolescents and young adults in Queensland, Australia. Int. J. Cancer 136, 603–609 (2014).
Czarnecki, D. The incidence of melanoma is increasing in the susceptible young Australian population. Acta Derm. Venereol. 94, 539–541 (2014).
Downing, A., Yu, X. Q., Newton-Bishop, J. & Forman, D. Trends in prognostic factors and survival from cutaneous melanoma in Yorkshire, UK and New South Wales, Australia between 1993 and 2003. Int. J. Cancer 123, 861–866 (2008).
Livingstone, E. et al. A first prospective population-based analysis investigating the actual practice of melanoma diagnosis, treatment and follow-up. Eur. J. Cancer 47, 1977–1989 (2011).
Gandini, S. et al. Meta-analysis of risk factors for cutaneous melanoma: II. Sun exposure. Eur. J. Cancer 41, 45–60 (2005).
Gandini, S. et al. Meta-analysis of risk factors for cutaneous melanoma: I. Common and atypical naevi. Eur. J. Cancer 41, 28–44 (2005). A paper that describes the association of UV exposure, melanocytic naevus count and melanoma development.
Boniol, M., Autier, P., Boyle, P. & Gandini, S. Cutaneous melanoma attributable to sunbed use: systematic review and meta-analysis. BMJ 345, e4757 (2012). A systematic review and meta-analysis that links usage of sunbeds to the development of cutaneous melanoma.
Gandini, S. et al. Melanoma attributable to sunbed use and tan seeking behaviours: an Italian survey. Eur. J. Dermatol. 24, 35–40 (2014).
Burnet, N. G., Jefferies, S. J., Benson, R. J., Hunt, D. P. & Treasure, F. P. Years of life lost (YLL) from cancer is an important measure of population burden—and should be considered when allocating research funds. Br. J. Cancer 92, 241–245 (2005).
Joosse, A. et al. Superior outcome of women with stage I/II cutaneous melanoma: pooled analysis of four European Organisation for Research and Treatment of Cancer phase III trials. J. Clin. Oncol. 30, 2240–2247 (2012).
Joosse, A. et al. Sex is an independent prognostic indicator for survival and relapse/progression-free survival in metastasized stage III to IV melanoma: a pooled analysis of five European organisation for research and treatment of cancer randomized controlled trials. J. Clin. Oncol. 31, 2337–2346 (2013).
Bastian, B. C. The molecular pathology of melanoma: an integrated taxonomy of melanocytic neoplasia. Annu. Rev. Pathol. 9, 239–271 (2014).
FitzGerald, M. G. et al. Prevalence of germ-line mutations in p16, 19ARF, and CDK4 in familial melanoma: analysis of a clinic-based population. Proc. Natl Acad. Sci. USA 93, 8541–8545 (1996).
Goldstein, A. M. et al. High-risk melanoma susceptibility genes and pancreatic cancer, neural system tumors, and uveal melanoma across GenoMEL. Cancer Res. 66, 9818–9828 (2006).
Chin, L., Garraway, L. A. & Fisher, D. E. Malignant melanoma: genetics and therapeutics in the genomic era. Genes Dev. 20, 2149–2182 (2006).
Sheppard, K. E. & McArthur, G. A. The cell-cycle regulator CDK4: an emerging therapeutic target in melanoma. Clin. Cancer Res. 19, 5320–5328 (2013).
Zuo, L. et al. Germline mutations in the p16INK4a binding domain of CDK4 in familial melanoma. Nat. Genet. 12, 97–99 (1996).
Wiesner, T. et al. Germline mutations in BAP1 predispose to melanocytic tumors. Nat. Genet. 43, 1018–1021 (2011).
Robles-Espinoza, C. D. et al. POT1 loss-of-function variants predispose to familial melanoma. Nat. Genet. 46, 478–481 (2014).
Shi, J. et al. Rare missense variants in POT1 predispose to familial cutaneous malignant melanoma. Nat. Genet. 46, 482–486 (2014).
Eggermont, A. M. M., Spatz, A. & Robert, C. Cutaneous melanoma. Lancet 383, 816–827 (2014).
Hawryluk, E. B. & Tsao, H. Melanoma: clinical features and genomic insights. Cold Spring Harb. Perspect. Med. 4, a015388 (2014).
Ward, K. A., Lazovich, D. & Hordinsky, M. K. Germline melanoma susceptibility and prognostic genes: a review of the literature. J. Am. Acad. Dermatol. 67, 1055–1067 (2012).
Raimondi, S. et al. MC1R variants, melanoma and red hair color phenotype: a meta-analysis. Int. J. Cancer 122, 2753–2760 (2008).
García-Borrón, J. C., Sánchez-Laorden, B. L. & Jiménez-Cervantes, C. Melanocortin-1 receptor structure and functional regulation. Pigment Cell Res. 18, 393–410 (2005).
Garraway, L. A. et al. Integrative genomic analyses identify MITF as a lineage survival oncogene amplified in malignant melanoma. Nature 436, 117–122 (2005).
Hodis, E. et al. A landscape of driver mutations in melanoma. Cell 150, 251–263 (2012).
Alexandrov, L. B. et al. Signatures of mutational processes in human cancer. Nature 500, 415–421 (2013).
Berger, M. F. et al. Melanoma genome sequencing reveals frequent PREX2 mutations. Nature 485, 502–506 (2012).
Krauthammer, M. et al. Exome sequencing identifies recurrent somatic RAC1 mutations in melanoma. Nat. Genet. 44, 1006–1014 (2012). References 39, 41 and 42 are landmark papers describing the mutational landscape of melanoma.
Flaherty, K. T., Hodi, F. S. & Fisher, D. E. From genes to drugs: targeted strategies for melanoma. Nat. Rev. Cancer 12, 349–361 (2012).
Kamb, A. Role of a cell cycle regulator in hereditary and sporadic cancer. Cold Spring Harb. Symp. Quant. Biol. 59, 39–47 (1994).
Hussussian, C. J. et al. Germline p16 mutations in familial melanoma. Nat. Genet. 8, 15–21 (1994).
Kwong, L. N. et al. Oncogenic NRAS signaling differentially regulates survival and proliferation in melanoma. Nat. Med. 18, 1503–1510 (2012).
Jakob, J. A. et al. NRAS mutation status is an independent prognostic factor in metastatic melanoma. Cancer 118, 4014–4023 (2012).
Griewank, K. G. et al. Genetic alterations and personalized medicine in melanoma: progress and future prospects. J. Natl. Cancer Inst. 106, djt435 (2014).
Van Raamsdonk, C. D. et al. Mutations in GNA11 in uveal melanoma. N. Engl. J. Med. 363, 2191–2199 (2010).
Noonan, F. P. et al. Melanoma induction by ultraviolet A but not ultraviolet B radiation requires melanin pigment. Nat. Commun. 3, 884 (2012).
Lito, P., Rosen, N. & Solit, D. B. Tumor adaptation and resistance to RAF inhibitors. Nat. Med. 19, 1401–1409 (2013).
McArthur, G. A. et al. Safety and efficacy of vemurafenib in BRAFV600E and BRAFV600K mutation-positive melanoma (BRIM-3): extended follow-up of a phase 3, randomised, open-label study. Lancet. Oncol. 15, 323–332 (2014).
Hauschild, A. et al. Dabrafenib in BRAF-mutated metastatic melanoma: a multicentre, open-label, phase 3 randomised controlled trial. Lancet 380, 358–365 (2012).
Flaherty, K. T. et al. Combined BRAF and MEK inhibition in melanoma with BRAF V600 mutations. N. Engl. J. Med. 367, 1694–1703 (2012).
Ribas, A. et al. Combination of vemurafenib and cobimetinib in patients with advanced BRAFV600-mutated melanoma: a phase 1b study. Lancet. Oncol. 15, 954–965 (2014).
Hartsough, E., Shao, Y. & Aplin, A. E. Resistance to RAF inhibitors revisited. J. Invest. Dermatol. 134, 319–325 (2014).
Roesch, A. Tumor heterogeneity and plasticity as elusive drivers for resistance to MAPK pathway inhibition in melanoma. Oncogenehttp://dx.doi.org/10.1038/onc.2014.249 (2014).
Shi, H. et al. Melanoma whole-exome sequencing identifies V600EB-RAF amplification-mediated acquired B-RAF inhibitor resistance. Nat. Commun. 3, 724 (2012).
Poulikakos, P. I. et al. RAF inhibitor resistance is mediated by dimerization of aberrantly spliced BRAF(V600E). Nature 480, 387–390 (2011).
Johannessen, C. M. et al. COT drives resistance to RAF inhibition through MAP kinase pathway reactivation. Nature 468, 968–972 (2010).
Montagut, C. et al. Elevated CRAF as a potential mechanism of acquired resistance to BRAF inhibition in melanoma. Cancer Res. 68, 4853–4861 (2008).
Nazarian, R. et al. Melanomas acquire resistance to B-RAF(V600E) inhibition by RTK or N-RAS upregulation. Nature 468, 973–977 (2010).
Sun, C. et al. Reversible and adaptive resistance to BRAF(V600E) inhibition in melanoma. Nature 508, 118–122 (2014).
Villanueva, J. et al. Acquired resistance to BRAF inhibitors mediated by a RAF kinase switch in melanoma can be overcome by cotargeting MEK and IGF-1R/PI3K. Cancer Cell 18, 683–695 (2010).
Michaloglou, C. et al. BRAFE600-associated senescence-like cell cycle arrest of human naevi. Nature 436, 720–724 (2005).
Gaffal, E. et al. Neonatal UVB exposure accelerates melanoma growth and enhances distant metastases in Hgf-Cdk4R24C C57BL/6 mice. Int. J. Cancer 129, 285–294 (2011).
Bald, T. et al. Ultraviolet-radiation-induced inflammation promotes angiotropism and metastasis in melanoma. Nature 507, 109–113 (2014).
Viros, A. et al. Ultraviolet radiation accelerates BRAF-driven melanomagenesis by targeting TP53. Nature 511, 478–482 (2014).
Mitra, D. et al. An ultraviolet-radiation-independent pathway to melanoma carcinogenesis in the red hair fair skin background. Nature 491, 449–453 (2012).
Dankort, D. et al. BrafV600E cooperates with Pten loss to induce metastatic melanoma. Nat. Genet. 41, 544–552 (2009).
Tsao, H., Goel, V., Wu, H., Yang, G. & Haluska, F. G. Genetic interaction between NRAS and BRAF mutations and PTEN/MMAC1 inactivation in melanoma. J. Invest. Dermatol. 122, 337–341 (2004).
Nathanson, K. L. et al. Tumor genetic analyses of patients with metastatic melanoma treated with the BRAF inhibitor dabrafenib (GSK2118436). Clin. Cancer Res. 19, 4868–4878 (2013).
Pardoll, D. M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 12, 252–264 (2012).
Topalian, S. L. et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J. Clin. Oncol. 32, 1020–1030 (2014).
Wolchok, J. D. et al. Nivolumab plus ipilimumab in advanced melanoma. N. Engl. J. Med. 369, 122–133 (2013).
Umansky, V. & Sevko, A. Melanoma-induced immunosuppression and its neutralization. Semin. Cancer Biol. 22, 319–326 (2012).
Quail, D. F. & Joyce, J. A. Microenvironmental regulation of tumor progression and metastasis. Nat. Med. 19, 1423–1437 (2013).
Damsky, W. E., Theodosakis, N. & Bosenberg, M. Melanoma metastasis: new concepts and evolving paradigms. Oncogene 33, 2413–2422 (2014).
Hüsemann, Y. et al. Systemic spread is an early step in breast cancer. Cancer Cell 13, 58–68 (2008).
Klein, C. A. Parallel progression of primary tumours and metastases. Nat. Rev. Cancer 9, 302–312 (2009).
Bernards, R. & Weinberg, R. A. A progression puzzle. Nature 418, 823 (2002).
Gartner, J. J. et al. Comparative exome sequencing of metastatic lesions provides insights into the mutational progression of melanoma. BMC Genomics 13, 505 (2012).
Pleasance, E. D. et al. A comprehensive catalogue of somatic mutations from a human cancer genome. Nature 463, 191–196 (2010).
Turajlic, S. et al. Whole genome sequencing of matched primary and metastatic acral melanomas. Genome Res. 22, 196–207 (2012).
Meacham, C. E. & Morrison, S. J. Tumour heterogeneity and cancer cell plasticity. Nature 501, 328–337 (2013).
Roesch, A. et al. A temporarily distinct subpopulation of slow-cycling melanoma cells is required for continuous tumor growth. Cell 141, 583–594 (2010).
Roesch, A. et al. Overcoming intrinsic multidrug resistance in melanoma by blocking the mitochondrial respiratory chain of slow-cycling JARID1Bhigh cells. Cancer Cell 23, 811–825 (2013).
Ramirez, R. D. et al. Progressive increase in telomerase activity from benign melanocytic conditions to malignant melanoma. Neoplasia 1, 42–49 (1999).
Rudolph, P. et al. Telomerase activity in melanocytic lesions: A potential marker of tumor biology. Am. J. Pathol. 156, 1425–1432 (2000).
Horn, S. et al. TERT promoter mutations in familial and sporadic melanoma. Science 339, 959–961 (2013).
Huang, F. W. et al. Highly recurrent TERT promoter mutations in human melanoma. Science 339, 957–959 (2013).
Griewank, K. G. et al. TERT promoter mutation status as an independent prognostic factor in cutaneous melanoma. J. Natl. Cancer Inst. 106, dju246 (2014).
Hartman, M. L. & Czyz, M. Anti-apoptotic proteins on guard of melanoma cell survival. Cancer Lett. 331, 24–34 (2013).
Liu, J., Fukunaga-Kalabis, M., Li, L. & Herlyn, M. Developmental pathways activated in melanocytes and melanoma. Arch. Biochem. Biophys. 563C, 13–21 (2014).
Curtin, J. A. et al. Distinct sets of genetic alterations in melanoma. N. Engl. J. Med. 353, 2135–2147 (2005).
McPherson, M. et al. Presentation and detection of invasive melanoma in a high-risk population. J. Am. Acad. Dermatol. 54, 783–792 (2006).
Brady, M. S. et al. Patterns of detection in patients with cutaneous melanoma. Cancer 89, 342–347 (2000).
Criscione, V. D. & Weinstock, M. A. Melanoma thickness trends in the United States, 1988–2006. J. Invest. Dermatol. 130, 793–797 (2010).
Argenziano, G. et al. Accuracy in melanoma detection: a 10-year multicenter survey. J. Am. Acad. Dermatol. 67, 54–59 (2012).
Marghoob, A. A. & Scope, A. The complexity of diagnosing melanoma. J. Invest. Dermatol. 129, 11–13 (2009).
Vestergaard, M. E., Macaskill, P., Holt, P. E. & Menzies, S. W. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br. J. Dermatol. 159, 669–676 (2008). After excluding two outlier studies, this meta-analysis of seven prospective studies with consecutively recruited patients showed that dermoscopy has a relative diagnostic odds ratio of 9.0 (95%CI 1.5–54.6; P = 0.03) for primary melanoma detection compared with naked-eye examination alone.
Carli, P. et al. Addition of dermoscopy to conventional naked-eye examination in melanoma screening: a randomized study. J. Am. Acad. Dermatol. 50, 683–689 (2004).
Carli, P. et al. Improvement of malignant/benign ratio in excised melanocytic lesions in the “dermoscopy era”: a retrospective study 1997–2001. Br. J. Dermatol. 150, 687–692 (2004).
Tromme, I. et al. Availability of digital dermoscopy in daily practice dramatically reduces the number of excised melanocytic lesions: results from an observational study. Br. J. Dermatol. 167, 778–786 (2012).
Kittler, H. et al. Identification of clinically featureless incipient melanoma using sequential dermoscopy imaging. Arch. Dermatol. 142, 1113–1119 (2006).
Haenssle, H. A. et al. Results from an observational trial: digital epiluminescence microscopy follow-up of atypical nevi increases the sensitivity and the chance of success of conventional dermoscopy in detecting melanoma. J. Invest. Dermatol. 126, 980–985 (2006).
Kelly, J. W., Yeatman, J. M., Regalia, C., Mason, G. & Henham, A. P. A high incidence of melanoma found in patients with multiple dysplastic naevi by photographic surveillance. Med. J. Aust. 167, 191–194 (1997).
Feit, N. E., Dusza, S. W. & Marghoob, A. A. Melanomas detected with the aid of total cutaneous photography. Br. J. Dermatol. 150, 706–714 (2004).
Goodson, A. G., Florell, S. R., Hyde, M., Bowen, G. M. & Grossman, D. Comparative analysis of total body and dermatoscopic photographic monitoring of nevi in similar patient populations at risk for cutaneous melanoma. Dermatol. Surg. 36, 1087–1098 (2010).
Rhodes, A. R. Intervention strategy to prevent lethal cutaneous melanoma: use of dermatologic photography to aid surveillance of high-risk persons. J. Am. Acad. Dermatol. 39, 262–267 (1998).
Moloney, F. J. et al. Detection of primary melanoma in individuals at extreme high risk: a prospective 5-year follow-up study. JAMA Dermatol. 150, 819–827 (2014).
Salerni, G. et al. Benefits of total body photography and digital dermatoscopy (“two-step method of digital follow-up”) in the early diagnosis of melanoma in patients at high risk for melanoma. J. Am. Acad. Dermatol. 67, e17–e27 (2012).
Guitera, P. et al. In vivo reflectance confocal microscopy enhances secondary evaluation of melanocytic lesions. J. Invest. Dermatol. 129, 131–138 (2009).
Pellacani, G., Pepe, P., Casari, A. & Longo, C. Reflectance confocal microscopy as a second-level examination in skin oncology improves diagnostic accuracy and saves unnecessary excisions: a longitudinal prospective study. Br. J. Dermatol.http://dx.doi.org/10.1111/bjd.13148 (2014).
Monheit, G. et al. The performance of MelaFind: a prospective multicenter study. Arch. Dermatol. 147, 188–194 (2011).
Malvehy, J. et al. Clinical performance of the Nevisense system in cutaneous melanoma detection: an international, multicentre, prospective and blinded clinical trial on efficacy and safety. Br. J. Dermatol. 171, 1099–1107 (2014).
Thomas, N. E. et al. Comparison of clinicopathologic features and survival of histopathologically amelanotic and pigmented melanomas: a population-based study. JAMA Dermatol. 150, 12 (2014).
Lodha, S., Saggar, S., Celebi, J. T. & Silvers, D. N. Discordance in the histopathologic diagnosis of difficult melanocytic neoplasms in the clinical setting. J. Cutan. Pathol. 35, 349–352 (2008).
Shoo, B. A., Sagebiel, R. W. & Kashani-Sabet, M. Discordance in the histopathologic diagnosis of melanoma at a melanoma referral center. J. Am. Acad. Dermatol. 62, 751–756 (2010).
Cerroni, L. et al. Melanocytic tumors of uncertain malignant potential: results of a tutorial held at the XXIX Symposium of the International Society of Dermatopathology in Graz, October 2008. Am. J. Surg. Pathol. 34, 314–326 (2010).
Levell, N. J., Beattie, C. C., Shuster, S. & Greenberg, D. C. Melanoma epidemic: a midsummer night's dream? Br. J. Dermatol. 161, 630–634 (2009).
Ohsie, S. J., Sarantopoulos, G. P., Cochran, A. J. & Binder, S. W. Immunohistochemical characteristics of melanoma. J. Cutan. Pathol. 35, 433–444 (2008).
Bauer, J. & Bastian, B. C. Distinguishing melanocytic nevi from melanoma by DNA copy number changes: comparative genomic hybridization as a research and diagnostic tool. Dermatol. Ther. 19, 40–49.
Luo, S., Sepehr, A. & Tsao, H. Spitz nevi and other Spitzoid lesions part I. Background and diagnoses. J. Am. Acad. Dermatol. 65, 1073–1084 (2011).
Gerami, P. et al. Fluorescence in situ hybridization (FISH) as an ancillary diagnostic tool in the diagnosis of melanoma. Am. J. Surg. Pathol. 33, 1146–1156 (2009).
Massi, D. et al. Atypical Spitzoid melanocytic tumors: a morphological, mutational, and FISH analysis. J. Am. Acad. Dermatol. 64, 919–935 (2011).
Vergier, B. et al. Fluorescence in situ hybridization, a diagnostic aid in ambiguous melanocytic tumors: European study of 113 cases. Mod. Pathol. 24, 613–623 (2011).
Raskin, L. et al. Copy number variations and clinical outcome in atypical spitz tumors. Am. J. Surg. Pathol. 35, 243–252 (2011).
Gaiser, T. et al. Classifying ambiguous melanocytic lesions with FISH and correlation with clinical long-term follow up. Mod. Pathol. 23, 413–419 (2010).
Gerami, P. et al. Sensitivity of fluorescence in situ hybridization for melanoma diagnosis using RREB1, MYB, Cep6, and 11q13 probes in melanoma subtypes. Arch. Dermatol. 146, 273–278 (2010).
Pouryazdanparast, P. et al. Distinctive clinical and histologic features in cutaneous melanoma with copy number gains in 8q24. Am. J. Surg. Pathol. 36, 253–264 (2012).
Gammon, B., Beilfuss, B., Guitart, J. & Gerami, P. Enhanced detection of spitzoid melanomas using fluorescence in situ hybridization with 9p21 as an adjunctive probe. Am. J. Surg. Pathol. 36, 81–88 (2012).
Gerami, P. et al. Fluorescence in situ hybridization for distinguishing nevoid melanomas from mitotically active nevi. Am. J. Surg. Pathol. 33, 1783–1788 (2009).
Busam, K. J. Molecular pathology of melanocytic tumors. Semin. Diagn. Pathol. 30, 362–374 (2013).
Rock, C. et al. Development and validation of a gene expression signature to distinguish malignant melanoma from benign nevi. ASCO Meet. Abstr. 32, 9021 (2014).
Van Kempen, L. C. & Spatz, A. From biomarker development towards implementation of multidimensional biomarker panels in a clinical setting. Mol. Oncol. 8, 781–782 (2014).
U.S. Preventive Services Task Force. Screening for skin cancer. U.S. Preventive Services Task Force recommendation statement. Ann. Intern. Med. 150, 188–193 (2009).
Ferrini, R. L., Perlman, M. & Hill, L. American College of Preventive Medicine policy statement: screening for skin cancer. Am. J. Prev. Med. 14, 80–82 (1998).
Kefford, R. et al. Genetic testing for melanoma. Lancet. Oncol. 3, 653–654 (2002).
Hansen, C. B., Wadge, L. M., Lowstuter, K., Boucher, K. & Leachman, S. A. Clinical germline genetic testing for melanoma. Lancet. Oncol. 5, 314–319 (2004).
Welch, H. G. & Black, W. C. Overdiagnosis in cancer. J. Natl Cancer Inst. 102, 605–613 (2010).
Beddingfield, F. C. The melanoma epidemic: res ipsa loquitur. Oncologist 8, 459–465 (2003).
Welch, H. G., Woloshin, S. & Schwartz, L. M. Skin biopsy rates and incidence of melanoma: population based ecological study. BMJ 331, 481 (2005).
Swerlick, R. A. & Chen, S. The melanoma epidemic: more apparent than real? Mayo Clin. Proc. 72, 559–564 (1997).
Epstein, D. S., Lange, J. R., Gruber, S. B., Mofid, M. & Koch, S. E. Is physician detection associated with thinner melanomas? JAMA 281, 640–643 (1999).
Katalinic, A. et al. Does skin cancer screening save lives?: an observational study comparing trends in melanoma mortality in regions with and without screening. Cancer 118, 5395–5402 (2012). This paper reports that melanoma-specific mortality decreased by 47% in the years after a population-based skin cancer screening programme was conducted in the German state of Schleswig-Holstein from July 2003 to June 2004.
U.S. Preventive Services Task Force. Skin cancer: screening. Summary of recommendations and evidence. [online], (2009).
U.S. Department of Health and Human Services. The Surgeon General's call to action to prevent skin cancer. [online], (2014).
Lu, C. et al. The genomic landscape of childhood and adolescent melanoma. J. Invest. Dermatol. 135, 816–823 (2014).
El Ghissassi, F. et al. A review of human carcinogens—part D: radiation. Lancet Oncol. 10, 751–752 (2009).
Colantonio, S., Bracken, M. B. & Beecker, J. The association of indoor tanning and melanoma in adults: systematic review and meta-analysis. J. Am. Acad. Dermatol. 70, 847–857.e1-18 (2014).
Green, A. C., Williams, G. M., Logan, V. & Strutton, G. M. Reduced melanoma after regular sunscreen use: randomized trial follow-up. J. Clin. Oncol. 29, 257–263 (2011). In this prospective randomized controlled trial conducted in Queensland, Australia, daily sunscreen application to the head and arms reduced the risk of all melanomas by 50% (P = 0.051) and invasive melanomas by 73% (P = 0.045) compared with discretionary sunscreen application.
Goldenhersh, M. A. & Koslowsky, M. Increased melanoma after regular sunscreen use? J. Clin. Oncol. 29, e557–e558 (2011).
Bigby, M. & Kim, C. C. A prospective randomized controlled trial indicates that sunscreen use reduced the risk of developing melanoma. Arch. Dermatol. 147, 853–854 (2011).
Matsuoka, L. Y., Wortsman, J., Hanifan, N. & Holick, M. F. Chronic sunscreen use decreases circulating concentrations of 25-hydroxyvitamin D. A preliminary study. Arch. Dermatol. 124, 1802–1804 (1988).
Holick, M. F. & Chen, T. C. Vitamin D deficiency: a worldwide problem with health consequences. Am. J. Clin. Nutr. 87, 1080S–1086S (2008).
Marks, R. et al. The effect of regular sunscreen use on vitamin D levels in an Australian population. Results of a randomized controlled trial. Arch. Dermatol. 131, 415–421 (1995).
Holick, M. F., Matsuoka, L. Y. & Wortsman, J. Regular use of sunscreen on vitamin D levels. Arch. Dermatol. 131, 1337–1339 (1995).
Vieth, R. Vitamin D supplementation, 25-hydroxyvitamin D concentrations, and safety. Am. J. Clin. Nutr. 69, 842–856 (1999).
Mosher, C. E. & Danoff-Burg, S. Addiction to indoor tanning: relation to anxiety, depression, and substance use. Arch. Dermatol. 146, 412–417 (2010).
Harrington, C. R. et al. Addictive-like behaviours to ultraviolet light among frequent indoor tanners. Clin. Exp. Dermatol. 36, 33–38 (2011).
Fell, G. L., Robinson, K. C., Mao, J., Woolf, C. J. & Fisher, D. E. Skin β-endorphin mediates addiction to UV light. Cell 157, 1527–1534 (2014).
The Alpha-Tocopherol, Beta Carotene Cancer Prevention Study Group. The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. N. Engl. J. Med. 330, 1029–1035 (1994).
Solomon, S. D. et al. Cardiovascular risk associated with celecoxib in a clinical trial for colorectal adenoma prevention. N. Engl. J. Med. 352, 1071–1080 (2005).
Bonovas, S. et al. Can statin therapy reduce the risk of melanoma? A meta-analysis of randomized controlled trials. Eur. J. Epidemiol. 25, 29–35 (2010).
Freeman, S. R. et al. Statins, fibrates, and melanoma risk: a systematic review and meta-analysis. J. Natl Cancer Inst. 98, 1538–1546 (2006).
Cook, N. R. et al. Low-dose aspirin in the primary prevention of cancer: the Women's Health Study: a randomized controlled trial. JAMA 294, 47–55 (2005).
Jacobs, E. J. et al. A large cohort study of long-term daily use of adult-strength aspirin and cancer incidence. J. Natl Cancer Inst. 99, 608–615 (2007).
Asgari, M. M., Maruti, S. S. & White, E. A large cohort study of nonsteroidal anti-inflammatory drug use and melanoma incidence. J. Natl Cancer Inst. 100, 967–971 (2008).
Francis, S. O., Mahlberg, M. J., Johnson, K. R., Ming, M. E. & Dellavalle, R. P. Melanoma chemoprevention. J. Am. Acad. Dermatol. 55, 849–861 (2006).
Uzarska, M. et al. Chemoprevention of skin melanoma: facts and myths. Melanoma Res. 23, 426–433 (2013).
Gershenwald, J. E. & Ross, M. I. Sentinel-lymph-node biopsy for cutaneous melanoma. N. Engl. J. Med. 364, 1738–1745 (2011). A review of current practice of SNB.
Morton, D. L. et al. Technical details of intraoperative lymphatic mapping for early stage melanoma. Arch. Surg. 127, 392–399 (1992).
Wong, S. L. et al. Sentinel lymph node biopsy for melanoma: American Society of Clinical Oncology and Society of Surgical Oncology joint clinical practice guideline. Ann. Surg. Oncol. 19, 3313–3324 (2012).
Gershenwald, J. E., Coit, D. G., Sondak, V. K. & Thompson, J. F. The challenge of defining guidelines for sentinel lymph node biopsy in patients with thin primary cutaneous melanomas. Ann. Surg. Oncol. 19, 3301–3303 (2012).
NCCN Clinical Practice Guidelines in Oncology: Melanoma. National Comprehensive Cancer Network[online], (2012).
Gershenwald, J. E. et al. Multi-institutional melanoma lymphatic mapping experience: the prognostic value of sentinel lymph node status in 612 stage I or II melanoma patients. J. Clin. Oncol. 17, 976–983 (1999).
Morton, D. L. et al. Final trial report of sentinel-node biopsy versus nodal observation in melanoma. N. Engl. J. Med. 370, 599–609 (2014).
Mocellin, S., Lens, M. B., Pasquali, S., Pilati, P. & Chiarion Sileni, V. Interferon alpha for the adjuvant treatment of cutaneous melanoma. Cochrane Database Syst. Rev. 6, CD008955 (2013). A meta-analysis of IFNα use in adjuvant trials.
Grob, J. J. et al. Adjuvant therapy with pegylated interferon alfa-2b (36 months) versus low-dose interferon alfa-2b (18 months) in melanoma patients without macrometastatic nodes: an open-label, randomised, phase 3 European Association for Dermato-Oncology (EADO) study. Eur. J. Cancer 49, 166–174 (2013).
Ascierto, P. A. et al. Adjuvant interferon alfa in malignant melanoma: an interdisciplinary and multinational expert review. Crit. Rev. Oncol. Hematol. 85, 149–161 (2013).
Eggermont, A. M. M. et al. Ulceration and stage are predictive of interferon efficacy in melanoma: results of the phase III adjuvant trials EORTC 18952 and EORTC 18991. Eur. J. Cancer 48, 218–225 (2012). This study describes ulceration as an important biomarker for IFN treatment and clinical benefit.
Jemal, A. et al. Recent trends in cutaneous melanoma incidence and death rates in the United States, 1992–2006. J. Am. Acad. Dermatol. 65, S17–S25.e1–e3 (2011).
Corrie, P. G. et al. Adjuvant bevacizumab in patients with melanoma at high risk of recurrence (AVAST-M): preplanned interim results from a multicentre, open-label, randomised controlled phase 3 study. Lancet Oncol. 15, 620–630 (2014).
Eggermont, A. M. et al. Ipilimumab versus placebo after complete resection of stage III melanoma: Initial efficacy and safety results from the EORTC 18071 phase III trial. ASCO Meet. Abstr. 32, LBA9008 (2014).
Garraway, L. A. & Baselga, J. Whole-genome sequencing and cancer therapy: is too much ever enough? Cancer Discov. 2, 766–768 (2012).
Hsueh, E. C., Famatiga, E., Gupta, R. K., Qi, K. & Morton, D. L. Enhancement of complement-dependent cytotoxicity by polyvalent melanoma cell vaccine (CancerVax): correlation with survival. Ann. Surg. Oncol. 5, 595–602.
Hodi, F. S. et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 363, 711–723 (2010).
Robert, C. et al. Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N. Engl. J. Med. 364, 2517–2526 (2011).
Bristol-Myers Squibb. YERVOY® (ipilimumab). [online], (2013).
Ribas, A. & Flaherty, K. T. BRAF targeted therapy changes the treatment paradigm in melanoma. Nat. Rev. Clin. Oncol. 8, 426–433 (2011).
McArthur, G. A. & Ribas, A. Targeting oncogenic drivers and the immune system in melanoma. J. Clin. Oncol. 31, 499–506 (2013).
Chapman, P. B. et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N. Engl. J. Med. 364, 2507–2516 (2011). The first paper to describe an overall survival benefit using targeted therapy (the selective BRAF inhibitor vemurafenib) compared with chemotherapy in untreated metastatic melanoma.
Hauschild, A. 1092PD: an update on overall survival (OS) and follow-on therapies in BREAK-3, a phase III, randomized trial: dabrafenib (D) versus dacarbazine (DTIC) in patients (pts) with BRAF V600E mutation-positive metastatic melanoma (MM). Ann. Oncol. 25 (Suppl. 4), iv374–iv393 (2014).
Hauschild, A. et al. An update on BREAK-3, a phase III, randomized trial: Dabrafenib (DAB) versus dacarbazine (DTIC) in patients with BRAF V600E-positive mutation metastatic melanoma (MM). ASCO Meet. Abstr. 31, 9013 (2013).
Sosman, J. A. et al. Survival in BRAF V600-mutant advanced melanoma treated with vemurafenib. N. Engl. J. Med. 366, 707–714 (2012).
Zimmer, L. et al. Atypical melanocytic proliferations and new primary melanomas in patients with advanced melanoma undergoing selective BRAF inhibition. J. Clin. Oncol. 30, 2375–2383 (2012).
Oberholzer, P. A. et al. RAS mutations are associated with the development of cutaneous squamous cell tumors in patients treated with RAF inhibitors. J. Clin. Oncol. 30, 316–321 (2012).
Su, F. et al. RAS mutations in cutaneous squamous-cell carcinomas in patients treated with BRAF inhibitors. N. Engl. J. Med. 366, 207–215 (2012).
Solit, D. B. et al. BRAF mutation predicts sensitivity to MEK inhibition. Nature 439, 358–362 (2006).
Von Euw, E. et al. Antitumor effects of the investigational selective MEK inhibitor TAK733 against cutaneous and uveal melanoma cell lines. Mol. Cancer 11, 22 (2012).
Flaherty, K. T. et al. Improved survival with MEK inhibition in BRAF-mutated melanoma. N. Engl. J. Med. 367, 107–114 (2012).
Van Allen, E. M. et al. The genetic landscape of clinical resistance to RAF inhibition in metastatic melanoma. Cancer Discov. 4, 94–109 (2014).
Shi, H. et al. Acquired resistance and clonal evolution in melanoma during BRAF inhibitor therapy. Cancer Discov. 4, 80–93 (2014).
Long, G. V. et al. Combined BRAF and MEK Inhibition versus BRAF Inhibition Alone in Melanoma. N. Engl. J. Med. 371, 1877–1888 (2014).
Larkin, J. et al. Combined vemurafenib and cobimetinib in BRAF-mutated melanoma. N. Engl. J. Med. 371, 1867–1876 (2014).
Robert, C. et al. Improved overall survival in melanoma with combined dabrafenib and trametinib. N. Eng. J. Med. 372, 30–39 (2015). The first study to demonstrate an overall survival benefit in patients with BRAFV600-mutant melanoma using a combined BRAF inhibitor plus MEK inhibitor compared with BRAF-inhibitor monotherapy.
Carvajal, R. D. et al. KIT as a therapeutic target in metastatic melanoma. JAMA 305, 2327–2334 (2011).
Brahmer, J. R. et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N. Engl. J. Med. 366, 2455–2465 (2012).
Topalian, S. L. et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N. Engl. J. Med. 366, 2443–2454 (2012).
Hamid, O. et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N. Engl. J. Med. 369, 134–144 (2013).
Robert, C. et al. Anti-programmed-death-receptor-1 treatment with pembrolizumab in ipilimumab-refractory advanced melanoma: a randomised dose-comparison cohort of a phase 1 trial. Lancet 384, 1109–1117 (2014).
Robert, C. et al. Nivolumab in previously untreated melanoma without BRAF mutation. N. Eng. J. Med. 372, 320–330 (2015). The first clinical study to demonstrate a clinically meaningful benefit using a PD1-specific antibody in untreated melanoma patients with no BRAF mutation compared with dacarbazine chemotherapy, leading to an increase in 1-year survival rate from 43% to 73%.
Tumeh, P. C. et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 515, 568–571 (2014).
Rosenberg, S. A. Cell transfer immunotherapy for metastatic solid cancer—what clinicians need to know. Nat. Rev. Clin. Oncol. 8, 577–585 (2011).
Rosenberg, S. A. et al. Durable complete responses in heavily pretreated patients with metastatic melanoma using T-cell transfer immunotherapy. Clin. Cancer Res. 17, 4550–4557 (2011).
Hersh, E. M. et al. A phase 2 clinical trial of nab-paclitaxel in previously treated and chemotherapy-naive patients with metastatic melanoma. Cancer 116, 155–163 (2010).
Hauschild, A. et al. Results of a phase III, randomized, placebo-controlled study of sorafenib in combination with carboplatin and paclitaxel as second-line treatment in patients with unresectable stage III or stage IV melanoma. J. Clin. Oncol. 27, 2823–2830 (2009).
Atkins, M. B. et al. High-dose recombinant interleukin 2 therapy for patients with metastatic melanoma: analysis of 270 patients treated between 1985 and 1993. J. Clin. Oncol. 17, 2105–2116 (1999).
Andtbacka, R. H. I. et al. OPTiM: A randomized phase III trial of talimogene laherparepvec (T-VEC) versus subcutaneous (SC) granulocyte-macrophage colony-stimulating factor (GM-CSF) for the treatment (tx) of unresected stage IIIB/C & IV melanoma. ASCO Meet. Abstr. 31, LBA9008 (2013).
Burton, A. W., Chai, T. & Smith, L. S. Cancer pain assessment. Curr. Opin. Support. Palliat. Care 8, 112–116 (2014).
Cleeland, C. S. & Ryan, K. M. Pain assessment: global use of the Brief Pain Inventory. Ann. Acad. Med. Singapore 23, 129–138 (1994).
Kvaal, K., Ulstein, I., Nordhus, I. H. & Engedal, K. The Spielberger State-Trait Anxiety Inventory (STAI): the state scale in detecting mental disorders in geriatric patients. Int. J. Geriatr. Psychiatry 20, 629–634 (2005).
Spielberger, C. D., Gorsuch, R. L. & Lushene, R. E. Manual for the State–Trait Anxiety Inventory. (Palo Alto, CA: Consulting Psychologists Press, 1970).
Manocchia, M. et al. SF-36 Health Survey Annotated Bibliography: Second Edition (1988–1996). (The Health Assessment Lab, New England Medical Center, 1998).
Aaronson, N. K. et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J. Natl Cancer Inst. 85, 365–376 (1993).
Holterhues, C. et al. Impact of melanoma on patients' lives among 562 survivors: a Dutch population-based study. Arch. Dermatol. 147, 177–185 (2011).
De Vries, M., Hoekstra, H. J. & Hoekstra-Weebers, J. E. H. M. Quality of life after axillary or groin sentinel lymph node biopsy, with or without completion lymph node dissection, in patients with cutaneous melanoma. Ann. Surg. Oncol. 16, 2840–2847 (2009).
Hamidou, Z., Dabakuyo, T. S. & Bonnetain, F. Impact of response shift on longitudinal quality-of-life assessment in cancer clinical trials. Expert Rev. Pharmacoecon. Outcomes Res. 11, 549–559 (2011).
Brandberg, Y. et al. Health-related quality of life in patients with high-risk melanoma randomised in the Nordic phase 3 trial with adjuvant intermediate-dose interferon alfa-2b. Eur. J. Cancer 48, 2012–2019 (2012).
Mohr, P., Hauschild, A., Trefzer, U. & Weichenthal, M. Quality of life in patients receiving high-dose interferon alfa-2b after resected high-risk melanoma. J. Clin. Oncol. 27, e70; author reply e71 (2009).
Bottomley, A. et al. Adjuvant therapy with pegylated interferon alfa-2b versus observation in resected stage III melanoma: a phase III randomized controlled trial of health-related quality of life and symptoms by the European Organisation for Research and Treatment of Cancer. J. Clin. Oncol. 27, 2916–2923 (2009).
De Kock, I. et al. Conversion of Karnofsky Performance Status (KPS) and Eastern Cooperative Oncology Group Performance Status (ECOG) to Palliative Performance Scale (PPS), and the interchangeability of PPS and KPS in prognostic tools. J. Palliat. Care 29, 163–169 (2013).
Mor, V., Laliberte, L., Morris, J. N. & Wiemann, M. The Karnofsky Performance Status Scale. An examination of its reliability and validity in a research setting. Cancer 53, 2002–2007 (1984).
Hatswell, A. J. et al. Patient-reported utilities in advanced or metastatic melanoma, including analysis of utilities by time to death. Health Qual. Life Outcomes 12, 140 (2014).
Schadendorf, D. et al. 1091PD COMBI-D: quality of life (QOL) impact of the combination of dabrafenib and trametinib (D + T) versus dabrafenib monotherapy (D) in patients with BRAF V600E/K unresectable or metastatic melanoma in a Phase III trial. Ann. Onc. 25 (Suppl. 4), iv377–iv393 (2014).
Grob, J.-J. et al. Patient perception of the benefit of a BRAF inhibitor in metastatic melanoma: quality-of-life analyses of the BREAK-3 study comparing dabrafenib with dacarbazine. Ann. Oncol. 25, 1428–1436 (2014).
Schadendorf, D. et al. Functional and symptom impact of trametinib versus chemotherapy in BRAF V600E advanced or metastatic melanoma: quality-of-life analyses of the METRIC study. Ann. Oncol. 25, 700–706 (2014).
Flaherty, K. T. et al. Surrogate endpoints for overall survival in metastatic melanoma: a meta-analysis of randomised controlled trials. Lancet. Oncol. 15, 297–304 (2014).
Ives, N. J., Stowe, R. L., Lorigan, P. & Wheatley, K. Chemotherapy compared with biochemotherapy for the treatment of metastatic melanoma: a meta-analysis of 18 trials involving 2,621 patients. J. Clin. Oncol. 25, 5426–5434 (2007).
Zimmer, L. et al. Phase II DeCOG-study of ipilimumab in pretreated and treatment-naïve patients with metastatic uveal melanoma. PLoS ONE (in the press).
Chen, X. et al. Combined PKC and MEK inhibition in uveal melanoma with GNAQ and GNA11 mutations. Oncogene 33, 4724–4734 (2014).
Feng, X. et al. Hippo-independent activation of YAP by the GNAQ uveal melanoma oncogene through a trio-regulated rho GTPase signaling circuitry. Cancer Cell 25, 831–845 (2014).
Yu, F.-X. et al. Mutant Gq/11 promote uveal melanoma tumorigenesis by activating YAP. Cancer Cell 25, 822–830 (2014).
Martin, M. et al. Exome sequencing identifies recurrent somatic mutations in EIF1AX and SF3B1 in uveal melanoma with disomy 3. Nat. Genet. 45, 933–936 (2013).
Harbour, J. W. et al. Frequent mutation of BAP1 in metastasizing uveal melanomas. Science 330, 1410–1413 (2010).
Prescher, G. et al. Prognostic implications of monosomy 3 in uveal melanoma. Lancet 347, 1222–1225 (1996).
Matatall, K. A. et al. BAP1 deficiency causes loss of melanocytic cell identity in uveal melanoma. BMC Cancer 13, 371 (2013).
Landreville, S. et al. Histone deacetylase inhibitors induce growth arrest and differentiation in uveal melanoma. Clin. Cancer Res. 18, 408–416 (2012).
Schadendorf, D. et al. Pooled analysis of long-term survival data from phase II and phase III trials of ipilimumab in unresectable or metastatic melanoma. J. Clin. Oncol.http://dx.doi.org/10.1200/JCO.2014.59.5041 (2015). An analysis of almost 5,000 patients with advanced-stage melanoma treated with ipilimumab, showing for the first time a long-term clinical benefit of a treatment and a 5-year survival rate of 20%.
Schadendorf, D. et al. Overall survival (OS) update on METRIC (NCT01245062), a randomized phase 3 study to assess efficacy of trametinib (T) compared with chemotherapy (C) in patients (pts) with BRAFV600E/K mutation-positive (+) advanced or metastatic melanoma (MM). Pigment Cell Melanoma Res, 26, 997 (2013).
Maio, M. et al. Five-year survival rates for treatment-naive patients with advanced melanoma who received ipilimumab plus dacarbazine in a Phase III trial. J. Clin. Oncol.http://dx.doi.org/10.0.4.176/JCO.2014.56.6018 (2015).
Daud, A. et al. Overall survival update for BRF113220 Part C, a Phase II three-arm randomized study of dabrafenib alone (D) versus a combination of dabrafenib and trametinib (D+T) in pts with BRAF V600 mutation-positive metastatic melanoma. Society for Melanoma Research 2013 International Congress (17–20 Nov 2013).
Acknowledgements
This work was not supported by a third party.
Author information
Authors and Affiliations
Contributions
Introduction (D.S. and A. Hauschild); Epidemiology (C.G.); Mechanisms/pathophysiology (M.H. and A. Roesch); Diagnosis, screening and prevention (D.F., A. Halpern and M.A.M.); Management (J.E.G., J.-J.G., G.M. and A. Ribas); Quality of life (J.-J.G. and D.S.); Outlook (D.S. and A. Hauschild); and overview of Primer (D.S.).
Corresponding author
Ethics declarations
Competing interests
D.S. and A. Hauschild declare an association with the following companies: Amgen, Bristol–Myers Squibb, Genentech, GlaxoSmithKline, Merck/MSD, Novartis, Pfizer, Boehringer Ingelheim and Roche. C.G. declares personal fees from Amgen, Merck/MSD and Novartis, and declares grants and personal fees from Bristol–Myers Squibb, GlaxoSmithKline and Roche outside of the submitted work. A. Roesch has received travel grants and honoraria from Roche and TEVA, and research grants from Novartis. M.A.M. has received honoraria from Next Meeting Generation for speaking on the topic of dermoscopy at the American Dermoscopy Meeting. A. Halpern serves as a consultant to Caliber Imaging and Diagnostics, Canfield Scientific, DermTech and SciBase AB, and serves on the data safety and monitoring board of Quintiles and Janssen Research and Development LLC. J.-J.G. has received fees for advisory boards and lectures from Amgen, GlaxoSmithKline, MSD, Novartis and Roche, and has received research grants from Bristol–Myers Squibb and Roche. J.E.G. serves on the global advisory board for Merck. A. Ribas has served as consultant for Amgen, Astellas, Genentech-Roche, GlaxoSmithKline, Merck, Novartis and Pierre Fabre, and serves on the scientific advisory board and has stock options for Compugen, Flexus Biosciences and Kite Pharma. G.M. has received consulting income from Provectus, and has received research support from Celgene and Pfizer. M.H. and D.E.F. declare no competing interests.
Rights and permissions
About this article
Cite this article
Schadendorf, D., Fisher, D., Garbe, C. et al. Melanoma. Nat Rev Dis Primers 1, 15003 (2015). https://doi.org/10.1038/nrdp.2015.3
Published:
DOI: https://doi.org/10.1038/nrdp.2015.3
This article is cited by
-
Augmenting MEK inhibitor efficacy in BRAF wild-type melanoma: synergistic effects of disulfiram combination therapy
Journal of Experimental & Clinical Cancer Research (2024)
-
Alternative Wnt-signaling axis leads to a break of oncogene-induced senescence
Cell Death & Disease (2024)
-
Mitofusin 1 silencing decreases the senescent associated secretory phenotype, promotes immune cell recruitment and delays melanoma tumor growth after chemotherapy
Scientific Reports (2024)
-
Ruthenium(II) complex with 2-mercaptothiazoline ligand induces selective cytotoxicity involving DNA damage and apoptosis in melanoma cells
JBIC Journal of Biological Inorganic Chemistry (2024)
-
Targeting CRAF kinase in anti-cancer therapy: progress and opportunities
Molecular Cancer (2023)