Abstract
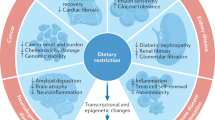
Caloric restriction, be it constant or intermittent, is reputed to have health-promoting and lifespan-extending effects. Caloric restriction mimetics (CRMs) are compounds that mimic the biochemical and functional effects of caloric restriction. In this Opinion article, we propose a unifying definition of CRMs as compounds that stimulate autophagy by favouring the deacetylation of cellular proteins. This deacetylation process can be achieved by three classes of compounds that deplete acetyl coenzyme A (AcCoA; the sole donor of acetyl groups), that inhibit acetyl transferases (a group of enzymes that acetylate lysine residues in an array of proteins) or that stimulate the activity of deacetylases and hence reverse the action of acetyl transferases. A unifying definition of CRMs will be important for the continued development of this class of therapeutic agents.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Fontana, L., Partridge, L. & Longo, V. D. Extending healthy life span — from yeast to humans. Science 328, 321–326 (2010).
Rubinsztein, D. C., Mariño, G. & Kroemer, G. Autophagy and aging. Cell 146, 682–695 (2011).
Libert, S. & Guarente, L. Metabolic and neuropsychiatric effects of calorie restriction and sirtuins. Annu. Rev. Physiol. 75, 669–684 (2013).
Colman, R. J. et al. Caloric restriction delays disease onset and mortality in rhesus monkeys. Science 325, 201–204 (2009).
Mattison, J. A. et al. Impact of caloric restriction on health and survival in rhesus monkeys from the NIA study. Nature 489, 318–321 (2012).
Anson, R. M. et al. Intermittent fasting dissociates beneficial effects of dietary restriction on glucose metabolism and neuronal resistance to injury from calorie intake. Proc. Natl Acad. Sci. USA 100, 6216–6220 (2003).
Li, L., Wang, Z. & Zuo, Z. Chronic intermittent fasting improves cognitive functions and brain structures in mice. PLoS ONE 8, e66069 (2013).
Heilbronn, L. K., Smith, S. R., Martin, C. K., Anton, S. D. & Ravussin, E. Alternate-day fasting in nonobese subjects: effects on body weight, body composition, and energy metabolism. Am. J. Clin. Nutr. 81, 69–73 (2005).
Klempel, M. C., Kroeger, C. M., Bhutani, S., Trepanowski, J. F. & Varady, K. A. Intermittent fasting combined with calorie restriction is effective for weight loss and cardio-protection in obese women. Nutr. J. 11, 4 (2012).
Magkos, F., Yannakoulia, M., Chan, J. L. & Mantzoros, C. S. Management of the metabolic syndrome and type 2 diabetes through lifestyle modification. Annu. Rev. Nutr. 29, 223–256 (2009).
Roth, L. W. & Polotsky, A. J. Can we live longer by eating less? A review of caloric restriction and longevity. Maturitas 71, 315–319 (2012).
Lee, S.-H. & Min, K.-J. Caloric restriction and its mimetics. BMB Rep. 46, 181–187 (2013).
Mariño, G. et al. Regulation of autophagy by cytosolic acetyl-coenzyme A. Mol. Cell 53, 710–725 (2014).
Feng, Y., He, D., Yao, Z. & Klionsky, D. J. The machinery of macroautophagy. Cell Res. 24, 24–41 (2014).
Choi, A. M. K., Ryter, S. W. & Levine, B. Autophagy in human health and disease. N. Engl. J. Med. 368, 1845–1846 (2013).
Eisenberg, T. et al. Nucleocytosolic depletion of the energy metabolite acetyl-coenzyme a stimulates autophagy and prolongs lifespan. Cell. Metab. 19, 431–444 (2014).
Lum, J. J. et al. Growth factor regulation of autophagy and cell survival in the absence of apoptosis. Cell 120, 237–248 (2005).
Boya, P. et al. Inhibition of macroautophagy triggers apoptosis. Mol. Cell. Biol. 25, 1025–1040 (2005).
Eisenberg, T. et al. Induction of autophagy by spermidine promotes longevity. Nature Cell Biol. 11, 1305–1314 (2009).
Morselli, E. et al. Caloric restriction and resveratrol promote longevity through the sirtuin-1-dependent induction of autophagy. Cell Death Dis. 1, e10 (2010).
Jia, K. & Levine, B. Autophagy is required for dietary restriction-mediated life span extension in C. elegans. Autophagy 3, 597–599 (2007).
Meléndez, A. et al. Autophagy genes are essential for dauer development and life-span extension in C. elegans. Science 301, 1387–1391 (2003).
Pyo, J.-O. et al. Overexpression of Atg5 in mice activates autophagy and extends lifespan. Nature Commun. 4, 2300 (2013).
Hebert, A. S. et al. Calorie restriction and SIRT3 trigger global reprogramming of the mitochondrial protein acetylome. Mol. Cell 49, 186–199 (2013).
Pietrocola, F. et al. Pro-autophagic polyphenols reduce the acetylation of cytoplasmic proteins. Cell Cycle 11, 3851–3860 (2012).
Wellen, K. E. & Thompson, C. B. A two-way street: reciprocal regulation of metabolism and signalling. Nature Rev. Mol. Cell. Biol. 13, 270–276 (2012).
Yeh, L. A. & Kim, K. H. Regulation of acetyl-coA carboxylase: properties of coA activation of acetyl-coA carboxylase. Proc. Natl Acad. Sci. USA 77, 3351–3355 (1980).
Imamura, H. et al. Visualization of ATP levels inside single living cells with fluorescence resonance energy transfer-based genetically encoded indicators. Proc. Natl Acad. Sci. USA 106, 15651–15656 (2009).
Weinert, B. T. et al. Acetylation dynamics and stoichiometry in Saccharomyces cerevisiae. Mol. Syst. Biol. 10, 716 (2014).
Wellen, K. E. et al. ATP-citrate lyase links cellular metabolism to histone acetylation. Science 324, 1076–1080 (2009).
Takahashi, H., McCaffery, J. M., Irizarry, R. A. & Boeke, J. D. Nucleocytosolic acetyl-coenzyme a synthetase is required for histone acetylation and global transcription. Mol. Cell 23, 207–217 (2006).
Roberts, D. J., Tan-Sah, V. P., Ding, E. Y., Smith, J. M. & Miyamoto, S. Hexokinase-II positively regulates glucose starvation-induced autophagy through TORC1 inhibition. Mol. Cell 53, 521–533 (2014).
Mohanti, B. K. et al. Improving cancer radiotherapy with 2-deoxy-D-glucose: Phase I/II clinical trials on human cerebral gliomas. Int. J. Radiat. Oncol. Biol. Phys. 35, 103–111 (1996).
Xie, Z. et al. Improvement of cardiac functions by chronic metformin treatment is associated with enhanced cardiac autophagy in diabetic OVE26 mice. Diabetes 60, 1770–1778 (2011).
Mariño, G. et al. Autophagy is essential for mouse sense of balance. J. Clin. Invest. 120, 2331–2344 (2010).
Onakpoya, I., Hung, S. K., Perry, R., Wider, B. & Ernst, E. The use of garcinia extract (hydroxycitric acid) as a weight loss supplement: a systematic review and meta-analysis of randomised clinical trials. J. Obes. 2011, 509038 (2011).
Saito, M. et al. High dose of Garcinia cambogia is effective in suppressing fat accumulation in developing male Zucker obese rats, but highly toxic to the testis. Food Chem. Toxicol. 43, 411–419 (2005).
Haberland, M., Montgomery, R. L. & Olson, E. N. The many roles of histone deacetylases in development and physiology: implications for disease and therapy. Nature Rev. Genet. 10, 32–42 (2009).
Scott, I., Webster, B. R., Li, J. H. & Sack, M. N. Identification of a molecular component of the mitochondrial acetyltransferase programme: a novel role for GCN5L1. Biochem. J. 443, 655–661 (2012).
Ko, M. H. & Puglielli, L. Two endoplasmic reticulum (ER)/ER Golgi intermediate compartment-based lysine acetyltransferases post-translationally regulate BACE1 levels. J. Biol. Chem. 284, 2482–2492 (2009).
Wang, Q. et al. Acetylation of metabolic enzymes coordinates carbon source utilization and metabolic flux. Science 327, 1004–1007 (2010).
Zhao, S. et al. Regulation of cellular metabolism by protein lysine acetylation. Science 327, 1000–1004 (2010).
Alavez, S., Vantipalli, M. C., Zucker, D. J. S., Klang, I. M. & Lithgow, G. J. Amyloid-binding compounds maintain protein homeostasis during ageing and extend lifespan. Nature 472, 226–229 (2011).
Soh, J.-W. et al. Curcumin is an early-acting stage-specific inducer of extended functional longevity in Drosophila. Exp. Gerontol. 48, 229–239 (2013).
Bjedov, I. et al. Mechanisms of life span extension by rapamycin in the fruit fly Drosophila melanogaster. Cell. Metab. 11, 35–46 (2010).
Abbas, S. & Wink, M. Epigallocatechin gallate from green tea (Camellia sinensis) increases lifespan and stress resistance in Caenorhabditis elegans. Planta Med. 75, 216–221 (2009).
Zhang, L., Jie, G., Zhang, J. & Zhao, B. Significant longevity-extending effects of EGCG on Caenorhabditis elegans under stress. Free Radic. Biol. Med. 46, 414–421 (2009).
Niu, Y. et al. The phytochemical, EGCG, extends lifespan by reducing liver and kidney function damage and improving age-associated inflammation and oxidative stress in healthy rats. Aging Cell 12, 1041–1049 (2013).
Rezai-Zadeh, K. et al. Green tea epigallocatechin-3-gallate (EGCG) reduces β-amyloid mediated cognitive impairment and modulates tau pathology in Alzheimer transgenic mice. Brain Res. 1214, 177–187 (2008).
Choi, Y. B., Kim, Y. I., Lee, K. S., Kim, B. S. & Kim, D. J. Protective effect of epigallocatechin gallate on brain damage after transient middle cerebral artery occlusion in rats. Brain Res. 1019, 47–54 (2004).
Koh, S.-H. et al. The effect of epigallocatechin gallate on suppressing disease progression of ALS model mice. Neurosci. Lett. 395, 103–107 (2006).
Zhou, J. et al. Epigallocatechin-3-gallate (EGCG), a green tea polyphenol, stimulates hepatic autophagy and lipid clearance. PLoS ONE 9, e87161 (2014).
Gupta, S. C., Kismali, G. & Aggarwal, B. B. Curcumin, a component of turmeric: from farm to pharmacy. BioFactors 39, 2–13 (2013).
Kim, H.-S., Quon, M. J. & Kim, J.-A. New insights into the mechanisms of polyphenols beyond antioxidant properties; lessons from the green tea polyphenol, epigallocatechin 3-gallate. Redox Biol. 2, 187–195 (2014).
Morselli, E. et al. Spermidine and resveratrol induce autophagy by distinct pathways converging on the acetylproteome. J. Cell Biol. 192, 615–629 (2011).
Bauer, M. A. et al. Spermidine promotes mating and fertilization efficiency in model organisms. Cell Cycle 12, 346–352 (2013).
Soda, K. et al. Long-term oral polyamine intake increases blood polyamine concentrations. J. Nutr. Sci. Vitaminol. 55, 361–366 (2009).
Gupta, V. K. et al. Restoring polyamines protects from age-induced memory impairment in an autophagy-dependent manner. Nature Neurosci. 16, 1453–1460 (2013).
Matsumoto, M., Kurihara, S., Kibe, R., Ashida, H. & Benno, Y. Longevity in mice is promoted by probiotic-induced suppression of colonic senescence dependent on upregulation of gut bacterial polyamine production. PLoS ONE 6, e23652 (2011).
Kibe, R. et al. Upregulation of colonic luminal polyamines produced by intestinal microbiota delays senescence in mice. Sci. Rep. 4, 4548 (2014).
Soda, K., Kano, Y., Chiba, F., Koizumi, K. & Miyaki, Y. Increased polyamine intake inhibits age-associated alteration in global DNA methylation and 1,2-dimethylhydrazine-induced tumorigenesis. PLoS ONE 8, e64357 (2013).
Goodwin, A. C. et al. Polyamine catabolism contributes to enterotoxigenic Bacteroides fragilis-induced colon tumorigenesis. Proc. Natl Acad. Sci. USA 108, 15354–15359 (2011).
LaRocca, T. J., Gioscia-Ryan, R. A., Hearon Jr., C. M. & Seals, D. R. The autophagy enhancer spermidine reverses arterial aging. Mech. Ageing Dev. 134, 314–320 (2013).
Paul, S. & Kang, S. C. Natural polyamine inhibits mouse skin inflammation and macrophage activation. Inflamm. Res. 62, 681–688 (2013).
Soda, K., Dobashi, Y., Kano, Y., Tsujinaka, S. & Konishi, F. Polyamine-rich food decreases age-associated pathology and mortality in aged mice. Exp. Gerontol. 44, 727–732 (2009).
Harrison, D. E. et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 460, 392–395 (2009).
Miller, R. A. et al. An Aging Interventions Testing Program: study design and interim report. Aging Cell 6, 565–575 (2007).
Nadon, N. L. et al. Design of aging intervention studies: the NIA interventions testing program. Age 30, 187–199 (2008).
Lee, I. H. & Finkel, T. Regulation of autophagy by the p300 acetyltransferase. J. Biol. Chem. 284, 6322–6328 (2009).
Nakagawa, Y. et al. p300 plays a critical role in maintaining cardiac mitochondrial function and cell survival in postnatal hearts. Circ. Res. 105, 746–754 (2009).
Mackeh, R. et al. Reactive oxygen species, AMP-activated protein kinase and the transcription cofactor p300 regulate α-tubulin acetyltransferase-1 (αTAT-1/MEC-17)-dependent microtubule hyperacetylation during cell stress. J. Biol. Chem. 289, 11816–11828 (2014).
Sebti, S. et al. BAT3 modulates p300-dependent acetylation of p53 and autophagy-related protein 7 (ATG7) during autophagy. Proc. Natl Acad. Sci. USA 111, 4115–4120 (2014).
Devipriya, B. & Kumaradhas, P. Probing the effect of intermolecular interaction and understanding the electrostatic moments of anacardic acid in the active site of p300 enzyme via DFT and charge density analysis. J. Mol. Graph. Model. 34, 57–66 (2012).
Seong, Y.-A., Shin, P.-G., Yoon, J.-S., Yadunandam, A. K. & Kim, G.-D. Induction of the endoplasmic reticulum stress and autophagy in human lung carcinoma a549 cells by anacardic acid. Cell Biochem. Biophys. 68, 369–377 (2014).
Hemshekhar, M., Sebastin Santhosh, M., Kemparaju, K. & Girish, K. S. Emerging roles of anacardic acid and its derivatives: a pharmacological overview. Bas. Clin. Pharmacol. Toxicol. 110, 122–132 (2011).
Lin, S.-Y. et al. GSK3-TIP60-ULK1 signaling pathway links growth factor deprivation to autophagy. Science 336, 477–481 (2012).
Balasubramanyam, K. et al. Polyisoprenylated benzophenone, garcinol, a natural histone acetyltransferase inhibitor, represses chromatin transcription and alters global gene expression. J. Biol. Chem. 279, 33716–33726 (2004).
Chen, X. et al. Chemoprevention of 7,12-dimethylbenz[a]anthracene (DMBA)-induced hamster cheek pouch carcinogenesis by a 5-lipoxygenase inhibitor, garcinol. Nutr. Cancer 64, 1211–1218 (2012).
Masullo, M. et al. Direct interaction of garcinol and related polyisoprenylated benzophenones of Garcinia cambogia fruits with the transcription factor STAT1 as a likely mechanism of their inhibitory effect on cytokine signaling pathways. J. Nat. Prod. 77, 543–549 (2014).
Quideau, S., Deffieux, D., Douat-Casassus, C. & Pouységu, L. Plant polyphenols: chemical properties, biological activities, and synthesis. Angew. Chem. Int. Ed. Engl. 50, 586–621 (2011).
Bowers, E. M. et al. Virtual ligand screening of the p300/CBP histone acetyltransferase: identification of a selective small molecule inhibitor. Chem. Biol. 17, 471–482 (2010).
Liu, Y. et al. Inhibition of p300 impairs FOXP3+ T regulatory cell function and promotes antitumor immunity. Nature Med. 19, 1173–1177 (2013).
Webster, B. R. et al. Restricted mitochondrial protein acetylation initiates mitochondrial autophagy. J. Cell Sci. 126, 4843–4849 (2013).
Johnson, S. C. et al. mTOR inhibition alleviates mitochondrial disease in a mouse model of Leigh syndrome. Science 342, 1524–1528 (2013).
Hamaï, A. & Codogno, P. New targets for acetylation in autophagy. Sci. Signal. 5, e29 (2012).
Lin, Y. et al. Functional dissection of lysine deacetylases reveals that HDAC1 and p300 regulate AMPK. Nature 482, 251–255 (2012).
Houtkooper, R. H. & Auwerx, J. Exploring the therapeutic space around NAD+. J. Cell Biol. 199, 205–209 (2012).
Lee, I. H. et al. A role for the NAD-dependent deacetylase SIRT1 in the regulation of autophagy. Proc. Natl Acad. Sci. 105, 3374 (2008).
Ou, X., Lee, M. R., Huang, X., Messina-Graham, S. & Broxmeyer, H. E. SIRT1 positively regulates autophagy and mitochondria function in embryonic stem cells under oxidative stress. Stem Cells 32, 1183–1194 (2014).
Kuma, A. et al. The role of autophagy during the early neonatal starvation period. Nature 432, 1032–1036 (2004).
Boily, G. et al. SIRT1 regulates energy metabolism and response to caloric restriction in mice. PLoS ONE 3, e1759 (2008).
Guarente, L. Calorie restriction and sirtuins revisited. Genes Dev. 27, 2072–2085 (2013).
Mercken, E. M. et al. SIRT1 but not its increased expression is essential for lifespan extension in caloric-restricted mice. Aging Cell 13, 193–196 (2014).
Someya, S. et al. SIRT3 mediates reduction of oxidative damage and prevention of age-related hearing loss under caloric restriction. Cell 143, 802–812 (2010).
Kanfi, Y. et al. The sirtuin SIRT6 regulates lifespan in male mice. Nature 483, 218–221 (2012).
Fontana, L., Weiss, E. P., Villareal, D. T., Klein, S. & Holloszy, J. O. Long-term effects of calorie or protein restriction on serum IGF-1 and IGFBP-3 concentration in humans. Aging Cell 7, 681–687 (2008).
Hubbard, B. P. et al. Evidence for a common mechanism of SIRT1 regulation by allosteric activators. Science 339, 1216–1219 (2013).
Park, S.-J. et al. Resveratrol ameliorates aging-related metabolic phenotypes by inhibiting cAMP phosphodiesterases. Cell 148, 421–433 (2012).
Baur, J. A. et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature 444, 337–342 (2006).
Jimenez-Gomez, Y. et al. Resveratrol improves adipose insulin signaling and reduces the inflammatory response in adipose tissue of rhesus monkeys on high-fat, high-sugar diet. Cell. Metab. 18, 533–545 (2013).
Pearson, K. J. et al. Resveratrol delays age-related deterioration and mimics transcriptional aspects of dietary restriction without extending life span. Cell. Metab. 8, 157–168 (2008).
Liu, K., Zhou, R., Wang, B. & Mi, M.-T. Effect of resveratrol on glucose control and insulin sensitivity: a meta-analysis of 11 randomized controlled trials. Am. J. Clin. Nutr. 99, 1510–1519 (2014).
Goh, K. P. et al. Effects of resveratrol in patients with type 2 diabetes mellitus on skeletal muscle SIRT1 expression and energy expenditure. Int. J. Sport Nutr. Exerc. Metab. 24, 2–13 (2014).
Fiori, J. L. et al. Resveratrol prevents β-cell dedifferentiation in nonhuman primates given a high-fat/high-sugar diet. Diabetes 62, 3500–3513 (2013).
Yoshino, J. et al. Resveratrol supplementation does not improve metabolic function in nonobese women with normal glucose tolerance. Cell. Metab. 16, 658–664 (2012).
Soare, A., Weiss, E. P., Holloszy, J. O. & Fontana, L. Multiple dietary supplements do not affect metabolic and cardio-vascular health. Aging 6, 149–157 (2013).
Milne, J. C. et al. Small molecule activators of SIRT1 as therapeutics for the treatment of type 2 diabetes. Nature 450, 712–716 (2007).
Minor, R. K. et al. SRT1720 improves survival and healthspan of obese mice. Sci. Rep. 1, 70 (2011).
Mitchell, S. J. et al. The SIRT1 activator SRT1720 extends lifespan and improves health of mice fed a standard diet. Cell Rep. 6, 836–843 (2014).
Amat, R. et al. SIRT1 controls the transcription of the peroxisome proliferator-activated receptor-γ co-activator-1α (PGC1α) gene in skeletal muscle through the PGC1α autoregulatory loop and interaction with MyoD. J. Biol. Chem. 284, 21872–21880 (2009).
Sinclair, D. A. & Guarente, L. Small-molecule allosteric activators of sirtuins. Annu. Rev. Pharmacol. Toxicol. 54, 363–380 (2014).
Mouchiroud, L. et al. The NAD+/sirtuin pathway modulates longevity through activation of mitochondrial UPR and FOXO signaling. Cell 154, 430–441 (2013).
Ashrafian, H., Horowitz, J. D. & Frenneaux, M. P. Perhexiline. Cardiovasc. Drug Rev. 25, 76–97 (2007).
Bordone, L. et al. SIRT1 transgenic mice show phenotypes resembling calorie restriction. Aging Cell 6, 759–767 (2007).
Price, N. L. et al. SIRT1 is required for AMPK activation and the beneficial effects of resveratrol on mitochondrial function. Cell. Metab. 15, 675–690 (2012).
Herranz, D. et al. Sirt1 improves healthy ageing and protects from metabolic syndrome-associated cancer. Nature Commun. 1, 3 (2010).
Gao, P. et al. Overexpression of SIRT1 in vascular smooth muscle cells attenuates angiotensin II-induced vascular remodeling and hypertension in mice. J. Mol. Med. 92, 347–357 (2014).
Timmers, S. et al. Calorie restriction-like effects of 30 days of resveratrol supplementation on energy metabolism and metabolic profile in obese humans. Cell. Metab. 14, 612–622 (2011).
Lagouge, M. et al. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC1α. Cell 127, 1109–1122 (2006).
Blum, C. A. et al. SIRT1 modulation as a novel approach to the treatment of diseases of aging. J. Med. Chem. 54, 417–432 (2011).
Yao, T. P. et al. Gene dosage-dependent embryonic development and proliferation defects in mice lacking the transcriptional integrator p300. Cell 93, 361–372 (1998).
Bedford, D. C. et al. Disrupting the CH1 domain structure in the acetyltransferases CBP and p300 results in lean mice with increased metabolic control. Cell. Metab. 14, 219–230 (2011).
Mielgo-Ayuso, J. et al. Effects of dietary supplementation with epigallocatechin-3-gallate on weight loss, energy homeostasis, cardiometabolic risk factors and liver function in obese women: randomised, double-blind, placebo-controlled clinical trial. Br. J. Nutr. 111, 1263–1271 (2014).
Mantelingu, K. et al. Specific inhibition of p300-HAT alters global gene expression and represses HIV replication. Chem. Biol. 14, 645–657 (2007).
Eliseeva, E. D., Valkov, V., Jung, M. & Jung, M. O. Characterization of novel inhibitors of histone acetyltransferases. Mol. Cancer Ther. 6, 2391–2398 (2007).
Levine, B. & Kroemer, G. Autophagy in the pathogenesis of disease. Cell 132, 27–42 (2008).
Mariño, G., Niso-Santano, M., Baehrecke, E. H. & Kroemer, G. Self-consumption: the interplay of autophagy and apoptosis. Nature Rev. Mol. Cell. Biol. 15, 81–94 (2014).
López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. The hallmarks of aging. Cell 153, 1194–1217 (2013).
Mariño, G. et al. Longevity-relevant regulation of autophagy at the level of the acetylproteome. Autophagy 7, 647–649 (2011).
Holmqvist, P.-H. & Mannervik, M. Genomic occupancy of the transcriptional co-activators p300 and CBP. Transcription 4, 18–23 (2013).
Contreras, A. U. et al. Deacetylation of p53 induces autophagy by suppressing Bmf expression. J. Cell Biol. 201, 427–437 (2013).
Füllgrabe, J. et al. The histone H4 lysine 16 acetyltransferase hMOF regulates the outcome of autophagy. Nature 500, 468–471 (2013).
Ghosh, H. S., McBurney, M. & Robbins, P. D. SIRT1 negatively regulates the mammalian target of rapamycin. PLoS ONE 5, e9199 (2010).
Miller, R. A. et al. Rapamycin-mediated lifespan increase in mice is dose and sex dependent and metabolically distinct from dietary restriction. Aging Cell 13, 468–477 (2014).
Anisimov, V. N. et al. Rapamycin increases lifespan and inhibits spontaneous tumorigenesis in inbred female mice. Cell Cycle 10, 4230–4236 (2011).
Neff, F. et al. Rapamycin extends murine lifespan but has limited effects on aging. J. Clin. Invest. 123, 3272–3291 (2013).
Lamming, D. W. et al. Rapamycin-induced insulin resistance is mediated by mTORC2 loss and uncoupled from longevity. Science 335, 1638–1643 (2012).
Wilkinson, J. E. et al. Rapamycin slows aging in mice. Aging Cell 11, 675–682 (2012).
Delvecchio, M., Gaucher, J., Aguilar-Gurrieri, C., Ortega, E. & Panne, D. Structure of the p300 catalytic core and implications for chromatin targeting and HAT regulation. Nature Struct. Mol. Biol. 20, 1040–1046 (2013).
Dominy, J., Puigserver, P. & Cantó, C. In vivo measurement of the acetylation state of sirtuin substrates as a proxy for sirtuin activity. Methods Mol. Biol. 1077, 217–237 (2013).
Kepp, O., Galluzzi, L., Lipinski, M., Yuan, J. & Kroemer, G. Cell death assays for drug discovery. Nature Rev. Drug Discov. 10, 221–237 (2011).
Coschigano, K. T., Clemmons, D., Bellush, L. L. & Kopchick, J. J. Assessment of growth parameters and life span of GHR/BP gene-disrupted mice. Endocrinology 141, 2608–2613 (2000).
Zhou, Y. et al. A mammalian model for Laron syndrome produced by targeted disruption of the mouse growth hormone receptor/binding protein gene (the Laron mouse). Proc. Natl Acad. Sci. USA 94, 13215–13220 (1997).
Arum, O., Bonkowski, M. S., Rocha, J. S. & Bartke, A. The growth hormone receptor gene-disrupted mouse fails to respond to an intermittent fasting diet. Aging Cell 8, 756–760 (2009).
Schiaffino, S. & Mammucari, C. Regulation of skeletal muscle growth by the IGF1–Akt/PKB pathway: insights from genetic models. Skelet. Muscle 1, 4 (2011).
Florez-McClure, M. L., Hohsfield, L. A., Fonte, G., Bealor, M. T. & Link, C. D. Decreased insulin-receptor signaling promotes the autophagic degradation of β-amyloid peptide in C. elegans. Autophagy 3, 569–580 (2007).
Longo, V. D. Linking sirtuins, IGFI signaling, and starvation. Exp. Gerontol. 44, 70–74 (2009).
Yuan, R. et al. Aging in inbred strains of mice: study design and interim report on median lifespans and circulating IGF1 levels. Aging Cell 8, 277–287 (2009).
Levine, M. E. et al. Low protein intake is associated with a major reduction in IGF1, cancer, and overall mortality in the 65 and younger but not older population. Cell. Metab. 19, 407–417 (2014).
Liu, Y. et al. Autosis is a Na+,K+-ATPase-regulated form of cell death triggered by autophagy-inducing peptides, starvation, and hypoxia-ischemia. Proc. Natl Acad. Sci. USA 110, 20364–20371 (2013).
Bricker, D. K. et al. A mitochondrial pyruvate carrier required for pyruvate uptake in yeast, Drosophila, and humans. Science 337, 96–100 (2012).
Phan, T. T. et al. Multi-centre experience on the use of perhexiline in chronic heart failure and refractory angina: old drug, new hope. Eur. J. Heart Fail. 11, 881–886 (2009).
Abozguia, K. et al. Metabolic modulator perhexiline corrects energy deficiency and improves exercise capacity in symptomatic hypertrophic cardiomyopathy. Circulation 122, 1562–1569 (2010).
Guay, C., Madiraju, S. R. M., Aumais, A., Joly, E. & Prentki, M. A role for ATP-citrate lyase, malic enzyme, and pyruvate/citrate cycling in glucose-induced insulin secretion. J. Biol. Chem. 282, 35657–35665 (2007).
Cappello, A. R. et al. The mitochondrial citrate carrier (CIC) is present and regulates insulin secretion by human male gamete. Endocrinology 153, 1743–1754 (2012).
Asghar, M. et al. Super CitriMax (HCA-SX) attenuates increases in oxidative stress, inflammation, insulin resistance, and body weight in developing obese Zucker rats. Mol. Cell. Biochem. 304, 93–99 (2007).
Hanai, J.-I., Doro, N., Seth, P. & Sukhatme, V. P. ATP citrate lyase knockdown impacts cancer stem cells in vitro. Cell Death Dis. 4, e696 (2013).
Zhao, Y., Huang, Z.-J., Rahman, M., Luo, Q. & Thorlacius, H. Radicicol, an HSP90 inhibitor, inhibits intestinal inflammation and leakage in abdominal sepsis. J. Surg. Res. 182, 312–318 (2013).
He, Y. et al. Radicicol, a heat shock protein 90 inhibitor, inhibits differentiation and adipogenesis in 3T3-L1 preadipocytes. Biochem. Biophys. Res. Commun. 436, 169–174 (2013).
Sonoda, H. et al. The protective effect of radicicol against renal ischemia — reperfusion injury in mice. J. Pharmacol. Sci. 112, 242–246 (2010).
Conte, T. C. et al. Radicicol improves regeneration of skeletal muscle previously damaged by crotoxin in mice. Toxicon 52, 146–155 (2008).
Griffin, T. M., Valdez, T. V. & Mestril, R. Radicicol activates heat shock protein expression and cardioprotection in neonatal rat cardiomyocytes. Am. J. Physiol. Heart Circ. Physiol. 287, H1081–H1088 (2004).
Hatzivassiliou, G. et al. ATP citrate lyase inhibition can suppress tumor cell growth. Cancer Cell 8, 311–321 (2005).
Pearce, N. J. et al. The role of ATP citrate-lyase in the metabolic regulation of plasma lipids. Hypolipidaemic effects of SB-204990, a lactone prodrug of the potent ATP citrate-lyase inhibitor SB-201076. Biochem. J. 334, 113–119 (1998).
Stuart, S. D. et al. A strategically designed small molecule attacks α-ketoglutarate dehydrogenase in tumor cells through a redox process. Cancer Metab. 2, 4 (2014).
Zachar, Z. et al. Non-redox-active lipoate derivates disrupt cancer cell mitochondrial metabolism and are potent anticancer agents in vivo. J. Mol. Med. 89, 1137–1148 (2011).
Lee, K. C. et al. Formation and anti-tumor activity of uncommon in vitro and in vivo metabolites of CPI-613, a novel anti-tumor compound that selectively alters tumor energy metabolism. Drug Metab. Lett. 5, 163–182 (2011).
Kang, S.-K., Cha, S.-H. & Jeon, H.-G. Curcumin-induced histone hypoacetylation enhances caspase-3-dependent glioma cell death and neurogenesis of neural progenitor cells. Stem Cells Dev. 15, 165–174 (2006).
Sunagawa, Y. et al. A natural p300-specific histone acetyltransferase inhibitor, curcumin, in addition to angiotensin-converting enzyme inhibitor, exerts beneficial effects on left ventricular systolic function after myocardial infarction in rats. Circ. J. 75, 2151–2159 (2011).
Morimoto, T. et al. The dietary compound curcumin inhibits p300 histone acetyltransferase activity and prevents heart failure in rats. J. Clin. Invest. 118, 868–878 (2008).
Marcu, M. G. et al. Curcumin is an inhibitor of p300 histone acetylatransferase. Med. Chem. 2, 169–174 (2006).
Balasubramanyam, K. et al. Curcumin, a novel p300/CREB-binding protein-specific inhibitor of acetyltransferase, represses the acetylation of histone/nonhistone proteins and histone acetyltransferase-dependent chromatin transcription. J. Biol. Chem. 279, 51163–51171 (2004).
Bimonte, S. et al. Curcumin inhibits tumor growth and angiogenesis in an orthotopic mouse model of human pancreatic cancer. BioMed Res. Int. 2013, 810423 (2013).
Huang, M. T. et al. Inhibitory effects of dietary curcumin on forestomach, duodenal, and colon carcinogenesis in mice. Cancer Res. 54, 5841–5847 (1994).
Rao, C. V., Rivenson, A., Simi, B. & Reddy, B. S. Chemoprevention of colon cancer by dietary curcumin. Ann. NY Acad. Sci. 768, 201–204 (1995).
Sharma, R. A. et al. Pharmacodynamic and pharmacokinetic study of oral Curcuma extract in patients with colorectal cancer. Clin. Cancer Res. 7, 1894–1900 (2001).
Seo, K.-I. et al. Effect of curcumin supplementation on blood glucose, plasma insulin, and glucose homeostasis related enzyme activities in diabetic db/db mice. Mol. Nutr. Food Res. 52, 995–1004 (2008).
Liao, V. H.-C. et al. Curcumin-mediated lifespan extension in Caenorhabditis elegans. Mech. Ageing Dev. 132, 480–487 (2011).
Suckow, B. K. & Suckow, M. A. Lifespan extension by the antioxidant curcumin in Drosophila melanogaster. Int. J. Biomed. Sci. 2, 402–405 (2006).
Ryu, M.-J. et al. Natural derivatives of curcumin attenuate the Wnt/β-catenin pathway through down-regulation of the transcriptional coactivator p300. Biochem. Biophys. Res. Commun. 377, 1304–1308 (2008).
Shakibaei, M. et al. Curcumin enhances the effect of chemotherapy against colorectal cancer cells by inhibition of NF-κB and Src protein kinase signaling pathways. PLoS ONE 8, e57218 (2013).
Kubota, M. et al. Preventive effects of curcumin on the development of azoxymethane-induced colonic preneoplastic lesions in male C57BL/KsJ–db/db obese mice. Nutr. Cancer 64, 72–79 (2012).
Jang, H.-J., Ridgeway, S. D. & Kim, J.-A. Effects of the green tea polyphenol epigallocatechin-3-gallate on high-fat diet-induced insulin resistance and endothelial dysfunction. Am. J. Physiol. Endocrinol. Metab. 305, E1444–E1451 (2013).
Chen, Y.-K. et al. Effects of green tea polyphenol (–)-epigallocatechin-3-gallate on newly developed high-fat/Western-style diet-induced obesity and metabolic syndrome in mice. J. Agr. Food Chem. 59, 11862–11871 (2011).
Rajendran, P., Ho, E., Williams, D. E. & Dashwood, R. H. Dietary phytochemicals, HDAC inhibition, and DNA damage/repair defects in cancer cells. Clin. Epigenet. 3, 4 (2011).
Wu, Y. et al. Anacardic acid (6-pentadecylsalicylic acid) inhibits tumor angiogenesis by targeting Src/FAK/Rho GTPases signaling pathway. J. Pharmacol. Exp. Ther. 339, 403–411 (2011).
Li, F. et al. Garcinol, a polyisoprenylated benzophenone modulates multiple proinflammatory signaling cascades leading to the suppression of growth and survival of head and neck carcinoma. Cancer Prev. Res. 6, 843–854 (2013).
Arif, M. et al. Mechanism of p300 specific histone acetyltransferase inhibition by small molecules. J. Med. Chem. 52, 267–277 (2009).
Mai, A. et al. Small-molecule inhibitors of histone acetyltransferase activity: identification and biological properties. J. Med. Chem. 49, 6897–6907 (2006).
Chimenti, F. et al. A novel histone acetyltransferase inhibitor modulating GCN5 network: cyclopentylidene-[4-(4′-chlorophenyl)thiazol-2-yl)hydrazone. J. Med. Chem. 52, 530–536 (2009).
Jara, J. A. et al. Antiproliferative and uncoupling effects of delocalized, lipophilic, cationic gallic acid derivatives on cancer cell lines. Validation in vivo in singenic mice. J. Med. Chem. 57, 2440–2454 (2014).
Lu, Y. et al. Gallic acid suppresses cell viability, proliferation, invasion and angiogenesis in human glioma cells. Eur. J. Pharmacol. 641, 102–107 (2010).
Kim, M.-J. et al. Gallic acid, a histone acetyltransferase inhibitor, suppresses β-amyloid neurotoxicity by inhibiting microglial-mediated neuroinflammation. Mol. Nutr. Food Res. 55, 1798–1808 (2011).
Lissa, D. et al. Resveratrol and aspirin eliminate tetraploid cells for anticancer chemoprevention. Proc. Natl Acad. Sci. USA 111, 3020–3025 (2014).
Lu, S.-P., Kato, M. & Lin, S.-J. Assimilation of endogenous nicotinamide riboside is essential for calorie restriction-mediated life span extension in Saccharomyces cerevisiae. J. Biol. Chem. 284, 17110–17119 (2009).
Belenky, P. et al. Nicotinamide riboside promotes SIR2 silencing and extends lifespan via NRK and URH1/PNP1/MEU1 pathways to NAD+. Cell 129, 473–484 (2007).
Dong, J. et al. Quercetin reduces obesity-associated ATM infiltration and inflammation in mice: a mechanism including AMPKα1/SIRT1. J. Lipid Res. 55, 363–374 (2014).
Pratheeshkumar, P. et al. Quercetin inhibits angiogenesis mediated human prostate tumor growth by targeting VEGFR2 regulated AKT/mTOR/P70S6K signaling pathways. PLoS ONE 7, e47516 (2012).
Angst, E. et al. The flavonoid quercetin inhibits pancreatic cancer growth in vitro and in vivo. Pancreas 42, 223–229 (2013).
Yum, S. et al. Piceatannol, a hydroxystilbene natural product, stabilizes HIF1α protein by inhibiting HIF prolyl hydroxylase. Eur. J. Pharmacol. 699, 124–131 (2013).
Kinoshita, Y. et al. Effect of long-term piceatannol treatment on eNOS levels in cultured endothelial cells. Biochem. Biophys. Res. Commun. 430, 1164–1168 (2013).
Kwon, J. Y. et al. Piceatannol, natural polyphenolic stilbene, inhibits adipogenesis via modulation of mitotic clonal expansion and insulin receptor-dependent insulin signaling in early phase of differentiation. J. Biol. Chem. 287, 11566–11578 (2012).
Minakawa, M., Miura, Y. & Yagasaki, K. Piceatannol, a resveratrol derivative, promotes glucose uptake through glucose transporter 4 translocation to plasma membrane in L6 myocytes and suppresses blood glucose levels in type 2 diabetic model db/db mice. Biochem. Biophys. Res. Commun. 422, 469–475 (2012).
Acknowledgements
The authors are supported by the Ligue contre le Cancer (équipe labellisée), Agence National de la Recherche, Association pour la Recherche sur le Cancer, Cancéropôle Ile-de-France, Institut National du Cancer (INCa), Fondation Bettencourt-Schueller, Fondation de France, Fondation pour la Recherche Médicale, the European Commission (ArtForce), the European Research Council, the LabEx Immuno-Oncology, the SIRIC Stratified Oncology Cell DNA Repair and Tumor Immune Elimination (Socrate), Cancer Research and Personalized Medicine (Carpem) and the Paris Alliance of Cancer Research Institutes. T.E. is a recipient of an APART (Austrian Programme for Advanced Research and Technology) fellowship of the Austrian Academy of Sciences at the Institute of Molecular Biosciences, University of Graz, Austria. F.M. is supported by the Austrian Science Fund FWF (grants LIPOTOX, I1000, P23490-B12 and P24381-B20).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
FURTHER INFORMATION
Glossary
- Autophagy
-
A neologism (from the Greek 'auto' (self) and 'phagein' (to eat)) that describes the capacity of cells to sequester portions of their cytoplasm and to subject them to lysosomal degradation.
- Caloric restriction
-
A dietary regimen that is based on low calorie intake without malnutrition.
- Caloric restriction mimetics
-
Pharmaceutical agents that induce the same biochemical alterations as does caloric restriction.
- Healthspan
-
The length of time that an individual is in optimal health.
- Hyperinsulinaemic–euglycaemic clamp studies
-
A physiological test used on whole animals to measure insulin-stimulated glucose uptake by all tissues of the body, hence measuring insulin sensitivity or resistance.
- Mitophagy
-
Specific autophagic removal of mitochondria, which are usually dysfunctional.
- Rapamycin
-
A macrolide antibiotic produced by the bacteria Streptomyces hygroscopicus that inhibits a negative regulator of autophagy, namely the mechanistic target of mammalian target of rapamycin complex 1 (mTORC1).
Rights and permissions
About this article
Cite this article
Madeo, F., Pietrocola, F., Eisenberg, T. et al. Caloric restriction mimetics: towards a molecular definition. Nat Rev Drug Discov 13, 727–740 (2014). https://doi.org/10.1038/nrd4391
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd4391
This article is cited by
-
Adipose tissue macrophages: implications for obesity-associated cancer
Military Medical Research (2023)
-
Combinatorial interventions in aging
Nature Aging (2023)
-
Morusin and mulberrin extend the lifespans of yeast and C. elegans via suppressing nutrient-sensing pathways
GeroScience (2023)
-
Molecular mechanism of caloric restriction mimetics-mediated neuroprotection of age-related neurodegenerative diseases: an emerging therapeutic approach
Biogerontology (2023)
-
Caloric restriction-mimetics for the reduction of heart failure risk in aging heart: with consideration of gender-related differences
Military Medical Research (2022)