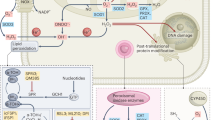
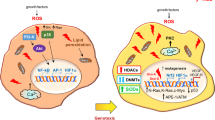
Key Points
-
The role of reactive oxygen species (ROS) in tumorigenesis is controversial, as they have been shown to have both tumour-promoting and tumour-inhibiting properties.
-
Although low to moderate ROS levels can be beneficial to cells by promoting proliferation pathways and DNA mutagenesis, high ROS levels can instead become detrimental and induce cell death.
-
Recent work has shown that multiple antioxidant pathways that inhibit ROS are upregulated during tumour initiation and progression.
-
These antioxidant pathways are composed of a multitude of both metabolic and non-metabolic enzymes, many of which can be targeted for inhibition.
-
Numerous standard chemotherapies are cytotoxic towards cancer cells owing to their ability to induce drastic increases in ROS levels.
-
The development of novel targeted therapies against antioxidant pathways may yield considerable benefits to the field of cancer treatment.
Abstract
The regulation of oxidative stress is an important factor in both tumour development and responses to anticancer therapies. Many signalling pathways that are linked to tumorigenesis can also regulate the metabolism of reactive oxygen species (ROS) through direct or indirect mechanisms. High ROS levels are generally detrimental to cells, and the redox status of cancer cells usually differs from that of normal cells. Because of metabolic and signalling aberrations, cancer cells exhibit elevated ROS levels. The observation that this is balanced by an increased antioxidant capacity suggests that high ROS levels may constitute a barrier to tumorigenesis. However, ROS can also promote tumour formation by inducing DNA mutations and pro-oncogenic signalling pathways. These contradictory effects have important implications for potential anticancer strategies that aim to modulate levels of ROS. In this Review, we address the controversial role of ROS in tumour development and in responses to anticancer therapies, and elaborate on the idea that targeting the antioxidant capacity of tumour cells can have a positive therapeutic impact.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Cairns, R. A., Harris, I. S. & Mak, T. W. Regulation of cancer cell metabolism. Nature Rev. Cancer 11, 85–95 (2011).
Diehn, M. et al. Association of reactive oxygen species levels and radioresistance in cancer stem cells. Nature 458, 780–783 (2009). This paper demonstrates that the mechanism by which cancer stem cells survive radiation (while the remaining tumour is eradicated) is through increased antioxidants and lower ROS levels.
Weinstein, I. B. & Joe, A. Oncogene addiction. Cancer Res. 68, 3077–3080 (2008).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Jones, R. G. & Thompson, C. B. Tumor suppressors and cell metabolism: a recipe for cancer growth. Genes Dev. 23, 537–548 (2009).
Finkel, T. Signal transduction by mitochondrial oxidants. J. Biol. Chem. 287, 4434–4440 (2012).
Handy, D. E. & Loscalzo, J. Redox regulation of mitochondrial function. Antioxid. Redox Signal. 16, 1323–1367 (2012).
Schrader, M. & Fahimi, H. D. Peroxisomes and oxidative stress. Biochim. Biophys. Acta 1763, 1755–1766 (2006).
Malhotra, J. D. & Kaufman, R. J. Endoplasmic reticulum stress and oxidative stress: a vicious cycle or a double-edged sword? Antioxid. Redox Signal. 9, 2277–2293 (2007).
Sena, L. A. & Chandel, N. S. Physiological roles of mitochondrial reactive oxygen species. Mol. Cell 48, 158–167 (2012).
Janssen-Heininger, Y. M. et al. Redox-based regulation of signal transduction: principles, pitfalls, and promises. Free Radic. Biol. Med. 45, 1–17 (2008).
Rhee, S. G. H2O2, a necessary evil for cell signaling. Science 312, 1882–1883 (2006).
Naik, E. & Dixit, V. M. Mitochondrial reactive oxygen species drive proinflammatory cytokine production. J. Exp. Med. 208, 417–420 (2011).
Gloire, G., Legrand-Poels, S. & Piette, J. NF-κB activation by reactive oxygen species: fifteen years later. Biochem. Pharmacol. 72, 1493–1505 (2006).
Oberley, L. W. Free radicals and diabetes. Free Radic. Biol. Med. 5, 113–124 (1988).
Trachootham, D., Alexandre, J. & Huang, P. Targeting cancer cells by ROS-mediated mechanisms: a radical therapeutic approach? Nature Rev. Drug Discov. 8, 579–591 (2009).
Ranjan, P. et al. Redox-dependent expression of cyclin D1 and cell proliferation by Nox1 in mouse lung epithelial cells. Antioxid. Redox Signal. 8, 1447–1459 (2006).
Martindale, J. L. & Holbrook, N. J. Cellular response to oxidative stress: signaling for suicide and survival. J. Cell. Physiol. 192, 1–15 (2002).
Leslie, N. R. et al. Redox regulation of PI 3-kinase signalling via inactivation of PTEN. EMBO J. 22, 5501–5510 (2003).
Xu, D., Rovira, I. I. & Finkel, T. Oxidants painting the cysteine chapel: redox regulation of PTPs. Dev. Cell 2, 251–252 (2002).
Harris, I. S. et al. PTPN12 promotes resistance to oxidative stress and supports tumorigenesis by regulating FOXO signaling. Oncogene http://dx.doi.org/10.1038/onc.2013.24 (2013).
Shi, X., Zhang, Y., Zheng, J. & Pan, J. Reactive oxygen species in cancer stem cells. Antioxid. Redox Signal. 16, 1215–1228 (2012).
Kim, H. M. et al. Increased CD13 expression reduces reactive oxygen species, promoting survival of liver cancer stem cells via an epithelial-mesenchymal transition-like phenomenon. Ann. Surg. Oncol. 19 (Suppl. 3), 539–548 (2012).
Schafer, Z. T. et al. Antioxidant and oncogene rescue of metabolic defects caused by loss of matrix attachment. Nature 461, 109–113 (2009). This study reported that, upon matrix detachment, cancer cells undergo an increase in ROS levels, which can be alleviated by oncogene expression.
Sporn, M. B. & Liby, K. T. NRF2 and cancer: the good, the bad and the importance of context. Nature Rev. Cancer 12, 564–571 (2012).
Taguchi, K., Motohashi, H. & Yamamoto, M. Molecular mechanisms of the Keap1-Nrf2 pathway in stress response and cancer evolution. Genes Cells 16, 123–140 (2011).
Meister, A. Selective modification of glutathione metabolism. Science 220, 472–477 (1983).
Sasaki, H. et al. Electrophile response element-mediated induction of the cystine/glutamate exchange transporter gene expression. J. Biol. Chem. 277, 44765–44771 (2002).
Mandal, P. K. et al. System x(c)- and thioredoxin reductase 1 cooperatively rescue glutathione deficiency. J. Biol. Chem. 285, 22244–22253 (2010).
Johansson, L., Gafvelin, G. & Arner, E. S. Selenocysteine in proteins — properties and biotechnological use. Biochim. Biophys. Acta 1726, 1–13 (2005).
Zhang, W. et al. Stromal control of cystine metabolism promotes cancer cell survival in chronic lymphocytic leukaemia. Nature Cell Biol. 14, 276–286 (2012). This paper identifies the functional role of CD44 in increasing cystine uptake and lowering ROS levels.
Ishimoto, T. et al. CD44 variant regulates redox status in cancer cells by stabilizing the xCT subunit of system xc− and thereby promotes tumor growth. Cancer Cell 19, 387–400 (2011).
McGrath-Morrow, S. et al. Nrf2 increases survival and attenuates alveolar growth inhibition in neonatal mice exposed to hyperoxia. Am. J. Physiol. Lung Cell. Mol. Physiol. 296, L565–L573 (2009).
Thimmulappa, R. K. et al. Identification of Nrf2-regulated genes induced by the chemopreventive agent sulforaphane by oligonucleotide microarray. Cancer Res. 62, 5196–5203 (2002).
Parsons, D. W. et al. An integrated genomic analysis of human glioblastoma multiforme. Science 321, 1807–1812 (2008).
Mardis, E. R. et al. Recurring mutations found by sequencing an acute myeloid leukemia genome. N. Engl. J. Med. 361, 1058–1066 (2009).
Yan, H. et al. IDH1 and IDH2 mutations in gliomas. N. Engl. J. Med. 360, 765–773 (2009).
Gross, S. et al. Cancer-associated metabolite 2-hydroxyglutarate accumulates in acute myelogenous leukemia with isocitrate dehydrogenase 1 and 2 mutations. J. Exp. Med. 207, 339–344 (2010).
Figueroa, M. E. et al. Leukemic IDH1 and IDH2 mutations result in a hypermethylation phenotype, disrupt TET2 function, and impair hematopoietic differentiation. Cancer Cell 18, 553–567 (2010).
Mitsuishi, Y. et al. Nrf2 redirects glucose and glutamine into anabolic pathways in metabolic reprogramming. Cancer Cell 22, 66–79 (2012).
Arner, E. S. & Holmgren, A. Physiological functions of thioredoxin and thioredoxin reductase. Eur. J. Biochem. 267, 6102–6109 (2000).
Chorley, B. N. et al. Identification of novel NRF2-regulated genes by ChIP-Seq: influence on retinoid X receptor alpha. Nucleic Acids Res. 40, 7416–7429 (2012).
Kim, Y. J. et al. Human prx1 gene is a target of Nrf2 and is up-regulated by hypoxia/reoxygenation: implication to tumor biology. Cancer Res. 67, 546–554 (2007).
Gutteridge, J. M. Iron promoters of the Fenton reaction and lipid peroxidation can be released from haemoglobin by peroxides. FEBS Lett. 201, 291–295 (1986).
Gozzelino, R., Jeney, V. & Soares, M. P. Mechanisms of cell protection by heme oxygenase-1. Annu. Rev. Pharmacol. Toxicol. 50, 323–354 (2010).
Alam, J. et al. Nrf2, a Cap'n'Collar transcription factor, regulates induction of the heme oxygenase-1 gene. J. Biol. Chem. 274, 26071–26078 (1999).
Orino, K. et al. Ferritin and the response to oxidative stress. Biochem. J. 357, 241–247 (2001).
Weinberg, E. D. The role of iron in cancer. Eur. J. Cancer Prev. 5, 19–36 (1996).
Kops, G. J. et al. Direct control of the Forkhead transcription factor AFX by protein kinase B. Nature 398, 630–634 (1999).
Brunet, A. et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96, 857–868 (1999).
Brunet, A. et al. Protein kinase SGK mediates survival signals by phosphorylating the forkhead transcription factor FKHRL1 (FOXO3a). Mol. Cell. Biol. 21, 952–965 (2001).
Greer, E. L. & Brunet, A. FOXO transcription factors at the interface between longevity and tumor suppression. Oncogene 24, 7410–7425 (2005).
Essers, M. A. et al. FOXO transcription factor activation by oxidative stress mediated by the small GTPase Ral and JNK. EMBO J. 23, 4802–4812 (2004).
Lehtinen, M. K. et al. A conserved MST-FOXO signaling pathway mediates oxidative-stress responses and extends life span. Cell 125, 987–1001 (2006).
Nemoto, S. & Finkel, T. Redox regulation of forkhead proteins through a p66shc-dependent signaling pathway. Science 295, 2450–2452 (2002).
Yalcin, S. et al. Foxo3 is essential for the regulation of ataxia telangiectasia mutated and oxidative stress-mediated homeostasis of hematopoietic stem cells. J. Biol. Chem. 283, 25692–25705 (2008).
Greer, E. L. et al. The energy sensor AMP-activated protein kinase directly regulates the mammalian FOXO3 transcription factor. J. Biol. Chem. 282, 30107–30119 (2007).
Cheng, Z. et al. Foxo1 integrates insulin signaling with mitochondrial function in the liver. Nature Med. 15, 1307–1311 (2009).
Mei, Y. et al. FOXO3a-dependent regulation of Pink1 (Park6) mediates survival signaling in response to cytokine deprivation. Proc. Natl Acad. Sci. USA 106, 5153–5158 (2009).
Martin, S. A., Hewish, M., Sims, D., Lord, C. J. & Ashworth, A. Parallel high throughput RNA interference screens identify PINK1 as a potential therapeutic target for the treatment of DNA mismatch repair deficient cancers. Cancer Res. 71, 1836–1848 (2011).
Nogueira, V. et al. Akt determines replicative senescence and oxidative or oncogenic premature senescence and sensitizes cells to oxidative apoptosis. Cancer Cell 14, 458–470 (2008). This paper reports the discovery that hyperactivation of the PI3K–AKT pathway can sensitize cancer cells to oxidative stress owing to the inactivation of FOXO factors and the resulting decrease in the expression of antioxidant enzymes.
Kops, G. J. et al. Forkhead transcription factor FOXO3a protects quiescent cells from oxidative stress. Nature 419, 316–321 (2002).
Woo, H. A., Bae, S. H., Park, S. & Rhee, S. G. Sestrin 2 is not a reductase for cysteine sulfinic acid of peroxiredoxins. Antioxid. Redox Signal. 11, 739–745 (2009).
Vousden, K. H. & Ryan, K. M. p53 and metabolism. Nature Rev. Cancer 9, 691–700 (2009).
Bensaad, K. et al. TIGAR, a p53-inducible regulator of glycolysis and apoptosis. Cell 126, 107–120 (2006). This paper identifies the role of the novel p53 gene TIGAR , which supports NADPH production by diverting metabolites into the PPP.
Suzuki, S. et al. Phosphate-activated glutaminase (GLS2), a p53-inducible regulator of glutamine metabolism and reactive oxygen species. Proc. Natl Acad. Sci. USA 107, 7461–7466 (2010).
Gao, P. et al. c-Myc suppression of miR-23a/b enhances mitochondrial glutaminase expression and glutamine metabolism. Nature 458, 762–765 (2009).
Chen, W. et al. Direct interaction between Nrf2 and p21(Cip1/WAF1) upregulates the Nrf2-mediated antioxidant response. Mol. Cell 34, 663–673 (2009).
Seoane, J., Le, H. V., Shen, L., Anderson, S. A. & Massague, J. Integration of Smad and forkhead pathways in the control of neuroepithelial and glioblastoma cell proliferation. Cell 117, 211–223 (2004).
Meiller, A. et al. p53-dependent stimulation of redox-related genes in the lymphoid organs of γ-irradiated mice — identification of haeme-oxygenase 1 as a direct p53 target gene. Nucleic Acids Res. 35, 6924–6934 (2007).
Pinnix, Z. K. et al. Ferroportin and iron regulation in breast cancer progression and prognosis. Sci. Transl. Med. 2, 43ra56 (2010).
Hayes, J. D., McMahon, M., Chowdhry, S. & Dinkova-Kostova, A. T. Cancer chemoprevention mechanisms mediated through the Keap1–Nrf2 pathway. Antioxid. Redox Signal. 13, 1713–1748 (2010).
Hu, R., Saw, C. L., Yu, R. & Kong, A. N. Regulation of NF-E2-related factor 2 signaling for cancer chemoprevention: antioxidant coupled with antiinflammatory. Antioxid. Redox Signal. 13, 1679–1698 (2010).
Frezza, C. et al. Haem oxygenase is synthetically lethal with the tumour suppressor fumarate hydratase. Nature 477, 225–228 (2011).
DeNicola, G. M. et al. Oncogene-induced Nrf2 transcription promotes ROS detoxification and tumorigenesis. Nature 475, 106–109 (2011). This study shows that the physiological expression of oncogenes can lead to decreased ROS levels through NRF2 antioxidant transcription.
Clements, C. M., McNally, R. S., Conti, B. J., Mak, T. W. & Ting, J. P. DJ-1, a cancer- and Parkinson's disease-associated protein, stabilizes the antioxidant transcriptional master regulator Nrf2. Proc. Natl Acad. Sci. USA 103, 15091–15096 (2006).
Kim, R. H. et al. DJ-1, a novel regulator of the tumor suppressor PTEN. Cancer Cell 7, 263–273 (2005).
Vasseur, S. et al. DJ-1/PARK7 is an important mediator of hypoxia-induced cellular responses. Proc. Natl Acad. Sci. USA 106, 1111–1116 (2009).
Vasseur, S. et al. Consequences of DJ-1 upregulation following p53 loss and cell transformation. Oncogene 31, 664–670 (2012).
Lee, J. & Wolfgang, M. J. Metabolomic profiling reveals a role for CPT1c in neuronal oxidative metabolism. BMC Biochem. 13, 23 (2012).
Zaugg, K. et al. Carnitine palmitoyltransferase 1C promotes cell survival and tumor growth under conditions of metabolic stress. Genes Dev. 25, 1041–1051 (2011).
Sanchez-Macedo, N. et al. Depletion of the novel p53-target gene carnitine palmitoyltransferase 1C delays tumor growth in the neurofibromatosis type I tumor model. Cell Death Differ. 20, 659–668 (2013).
Kim, Y. R. et al. Oncogenic NRF2 mutations in squamous cell carcinomas of oesophagus and skin. J. Pathol. 220, 446–451 (2010).
Shibata, T. et al. Cancer related mutations in NRF2 impair its recognition by Keap1-Cul3 E3 ligase and promote malignancy. Proc. Natl Acad. Sci. USA 105, 13568–13573 (2008).
Hayes, J. D. & McMahon, M. NRF2 and KEAP1 mutations: permanent activation of an adaptive response in cancer. Trends Biochem. Sci. 34, 176–188 (2009).
Tenbaum, S. P. et al. β-catenin confers resistance to PI3K and AKT inhibitors and subverts FOXO3a to promote metastasis in colon cancer. Nature Med. 18, 892–901 (2012).
Naka, K. et al. TGF-β–FOXO signalling maintains leukaemia-initiating cells in chronic myeloid leukaemia. Nature 463, 676–680 (2010).
Sykes, S. M. et al. AKT/FOXO signaling enforces reversible differentiation blockade in myeloid leukemias. Cell 146, 697–708 (2011).
Burgering, B. M. & Medema, R. H. Decisions on life and death: FOXO forkhead transcription factors are in command when PKB/Akt is off duty. J. Leukoc. Biol. 73, 689–701 (2003).
Olanich, M. E. & Barr, F. G. A call to ARMS: targeting the PAX3-FOXO1 gene in alveolar rhabdomyosarcoma. Expert Opin. Ther. Targets 17, 607–623 (2013).
del Peso, L., Gonzalez, V. M., Hernandez, R., Barr, F. G. & Nunez, G. Regulation of the forkhead transcription factor FKHR, but not the PAX3-FKHR fusion protein, by the serine/threonine kinase Akt. Oncogene 18, 7328–7333 (1999).
Li, B., Gordon, G. M., Du, C. H., Xu, J. & Du, W. Specific killing of Rb mutant cancer cells by inactivating TSC2. Cancer Cell 17, 469–480 (2010).
Jeon, S. M., Chandel, N. S. & Hay, N. AMPK regulates NADPH homeostasis to promote tumour cell survival during energy stress. Nature 485, 661–665 (2012).
Bae, I. et al. BRCA1 induces antioxidant gene expression and resistance to oxidative stress. Cancer Res. 64, 7893–7909 (2004).
Saha, T., Rih, J. K. & Rosen, E. M. BRCA1 down-regulates cellular levels of reactive oxygen species. FEBS Lett. 583, 1535–1543 (2009).
Gorrini, C. et al. BRCA1 interacts with Nrf2 to regulate antioxidant signaling and cell survival. J. Exp. Med. 210, 1529–1544 (2013).
Barzilai, A., Rotman, G. & Shiloh, Y. ATM deficiency and oxidative stress: a new dimension of defective response to DNA damage. DNA Repair 1, 3–25 (2002).
Ito, K. et al. Regulation of oxidative stress by ATM is required for self-renewal of haematopoietic stem cells. Nature 431, 997–1002 (2004).
Guo, Z., Kozlov, S., Lavin, M. F., Person, M. D. & Paull, T. T. ATM activation by oxidative stress. Science. 330, 517–521 (2010).
Alexander, A. et al. ATM signals to TSC2 in the cytoplasm to regulate mTORC1 in response to ROS. Proc. Natl Acad. Sci. USA 107, 4153–4158 (2010).
Adam, J. et al. Renal cyst formation in Fh1-deficient mice is independent of the Hif/Phd pathway: roles for fumarate in KEAP1 succination and Nrf2 signaling. Cancer Cell 20, 524–537 (2011). This paper demonstrates that NRF2 is stabilized upon loss of the tumour suppressor gene fumarate hydratase.
Ooi, A. et al. An antioxidant response phenotype shared between hereditary and sporadic type 2 papillary renal cell carcinoma. Cancer Cell 20, 511–523 (2011).
Sullivan, L. B. et al. The proto-oncometabolite fumarate binds glutathione to amplify ROS-dependent signaling. Mol. Cell 51, 236–248 (2013).
Mazurek, S., Boschek, C. B., Hugo, F. & Eigenbrodt, E. Pyruvate kinase type M2 and its role in tumor growth and spreading. Semin. Cancer Biol. 15, 300–308 (2005).
Christofk, H. R. et al. The M2 splice isoform of pyruvate kinase is important for cancer metabolism and tumour growth. Nature 452, 230–233 (2008).
Christofk, H. R., Vander Heiden, M. G., Wu, N., Asara, J. M. & Cantley, L. C. Pyruvate kinase M2 is a phosphotyrosine-binding protein. Nature 452, 181–186 (2008).
Vander Heiden, M. G. et al. Evidence for an alternative glycolytic pathway in rapidly proliferating cells. Science 329, 1492–1499 (2010).
Anastasiou, D. et al. Inhibition of pyruvate kinase M2 by reactive oxygen species contributes to antioxidant responses. Science 334, 1278–1283 (2011).
Locasale, J. W. et al. Phosphoglycerate dehydrogenase diverts glycolytic flux and contributes to oncogenesis. Nature Genet. 43, 869–874 (2011).
Possemato, R. et al. Functional genomics reveal that the serine synthesis pathway is essential in breast cancer. Nature 476, 346–350 (2011).
Jain, M. et al. Metabolite profiling identifies a key role for glycine in rapid cancer cell proliferation. Science 336, 1040–1044 (2012).
Chaneton, B. et al. Serine is a natural ligand and allosteric activator of pyruvate kinase M2. Nature 491, 458–462 (2012).
Goodison, S., Urquidi, V. & Tarin, D. CD44 cell adhesion molecules. Mol. Pathol. 52, 189–196 (1999).
Zoller, M. CD44: can a cancer-initiating cell profit from an abundantly expressed molecule? Nature Rev. Cancer 11, 254–267 (2011).
Blot, W. J. et al. Nutrition intervention trials in Linxian, China: supplementation with specific vitamin/mineral combinations, cancer incidence, and disease-specific mortality in the general population. J. Natl Cancer Inst. 85, 1483–1492 (1993).
Qiao, Y. L. et al. Total and cancer mortality after supplementation with vitamins and minerals: follow-up of the Linxian General Population Nutrition Intervention Trial. J. Natl Cancer Inst. 101, 507–518 (2009).
Zhang, W. et al. Vitamin intake and liver cancer risk: a report from two cohort studies in China. J. Natl Cancer Inst. 104, 1173–1181 (2012).
Hurst, R. et al. Selenium and prostate cancer: systematic review and meta-analysis. Am. J. Clin. Nutr. 96, 111–122 (2012).
Richman, E. L. & Chan, J. M. Selenium and prostate cancer: the puzzle isn't finished yet. Am. J. Clin. Nutr. 96, 1–2 (2012).
Klein, E. A. et al. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 306, 1549–1556 (2011). This was the first large study on the link between vitamin E supplementation and cancer risk, in contrast to early observations suggesting that vitamin E has a protective effect against cancer.
Su, Z. Y. et al. A perspective on dietary phytochemicals and cancer chemoprevention: oxidative stress, Nrf2, and epigenomics. Top. Curr. Chem. 329, 133–162 (2012).
Kim, Y. S., Farrar, W., Colburn, N. H. & Milner, J. A. Cancer stem cells: potential target for bioactive food components. J. Nutrit. Biochem. 23, 691–698 (2012).
Conklin, K. A. Chemotherapy-associated oxidative stress: impact on chemotherapeutic effectiveness. Integr. Cancer Ther. 3, 294–300 (2004).
Barrera, G. Oxidative stress and lipid peroxidation products in cancer progression and therapy. ISRN Oncol. 2012, 137289 (2012).
Santiago-Arteche, R. et al. Cancer chemotherapy reduces plasma total polyphenols and total antioxidants capacity in colorectal cancer patients. Mol. Biol. Rep. 39, 9355–9360 (2012).
Kaufmann, S. H. & Earnshaw, W. C. Induction of apoptosis by cancer chemotherapy. Exp. Cell Res. 256, 42–49 (2000).
Miller, W. H. et al. Mechanisms of action of arsenic trioxide. Cancer Res. 62, 3893–3903 (2002).
Yi, J. et al. The inherent cellular level of reactive oxygen species: one of the mechanisms determining apoptotic susceptibility of leukemic cells to arsenic trioxide. Apoptosis 7, 209–215 (2002).
Longley, D. B., Harkin, D. P. & Johnston, P. G. 5-fluorouracil: mechanisms of action and clinical strategies. Nature Rev. Cancer 3, 330–338 (2003).
Hwang, P. M. et al. Ferredoxin reductase affects p53-dependent, 5-fluorouracil-induced apoptosis in colorectal cancer cells. Nature Med. 7, 1111–1117 (2001).
Hwang, I. T. et al. Drug resistance to 5-FU linked to reactive oxygen species modulator 1. Biochem. Biophys. Res. Commun. 359, 304–310 (2007).
Zhang, Q. et al. Involvement of reactive oxygen species in 2-methoxyestradiol-induced apoptosis in human neuroblastoma cells. Cancer Lett. 313, 201–210 (2011).
Kachadourian, R. et al. 2-methoxyestradiol does not inhibit superoxide dismutase. Arch. Biochem. Biophys. 392, 349–353 (2001).
Lai, W. L. & Wong, N. S. ROS mediates 4HPR-induced posttranscriptional expression of the Gadd153 gene. Free Radic. Biol. Med. 38, 1585–1593 (2005).
Apraiz, A. et al. Dihydroceramide accumulation and reactive oxygen species are distinct and nonessential events in 4-HPR-mediated leukemia cell death. Biochem. Cell Biol. 90, 209–223 (2012).
Neckers, L. & Workman, P. Hsp90 molecular chaperone inhibitors: are we there yet? Clin. Cancer Res. 18, 64–76 (2012).
Hao, H. et al. HSP90 and its inhibitors. Oncol. Rep. 23, 1483–1492 (2010).
Scarbrough, P. M. et al. Simultaneous inhibition of glutathione- and thioredoxin-dependent metabolism is necessary to potentiate 17AAG-induced cancer cell killing via oxidative stress. Free Radic. Biol. Med. 52, 436–443 (2012).
De Raedt, T. et al. Exploiting cancer cell vulnerabilities to develop a combination therapy for Ras-driven tumors. Cancer Cell 20, 400–413 (2011).
Metzger-Filho, O. et al. Dissecting the heterogeneity of triple-negative breast cancer. J. Clin. Oncol. 30, 1879–1887 (2012).
Masaoka, A., Horton, J. K., Beard, W. A. & Wilson, S. H. DNA polymerase beta and PARP activities in base excision repair in living cells. DNA Repair 8, 1290–1299 (2009).
Luo, X. & Kraus, W. L. On PAR with PARP: cellular stress signaling through poly(ADP-ribose) and PARP-1. Genes Dev. 26, 417–432 (2012).
Berndtsson, M. et al. Acute apoptosis by cisplatin requires induction of reactive oxygen species but is not associated with damage to nuclear DNA. Int. J. Cancer 120, 175–180 (2007).
Kummar, S. et al. Advances in using PARP inhibitors to treat cancer. BMC Med. 10, 25 (2012).
Rottenberg, S. et al. High sensitivity of BRCA1-deficient mammary tumors to the PARP inhibitor AZD2281 alone and in combination with platinum drugs. Proc. Natl Acad. Sci. USA 105, 17079–17084 (2008). The discovery of PARP inhibitors have brought hope in the treatment of BRCA1-mutated cancers. This work shows the efficacy of PARP inhibitors in combination with platinum drugs.
Evers, B. et al. Selective inhibition of BRCA2-deficient mammary tumor cell growth by AZD2281 and cisplatin. Clin. Cancer Res. 14, 3916–3925 (2008).
Michels, J. et al. Cisplatin resistance associated with PARP hyperactivation. Cancer Res. 73, 2271–2280 (2013).
Michels, J. et al. Synergistic interaction between cisplatin and PARP inhibitors in non-small cell lung cancer. Cell Cycle 12, 877–883 (2013).
Yoshida, T., Goto, S., Kawakatsu, M., Urata, Y. & Li, T. S. Mitochondrial dysfunction, a probable cause of persistent oxidative stress after exposure to ionizing radiation. Free Radic. Res. 46, 147–153 (2012).
Wang, Y. et al. Total body irradiation causes residual bone marrow injury by induction of persistent oxidative stress in murine hematopoietic stem cells. Free Radic. Biol. Med. 48, 348–356 (2010).
Voorhees, P. M., Dees, E. C., O'Neil, B. & Orlowski, R. Z. The proteasome as a target for cancer therapy. Clin. Cancer Res. 9, 6316–6325 (2003).
Joazeiro, C. A., Anderson, K. C. & Hunter, T. Proteasome inhibitor drugs on the rise. Cancer Res. 66, 7840–7842 (2006).
Papa, L., Gomes, E. & Rockwell, P. Reactive oxygen species induced by proteasome inhibition in neuronal cells mediate mitochondrial dysfunction and a caspase-independent cell death. Apoptosis 12, 1389–1405 (2007).
Chen, Z. et al. Nuclear translocation of B-cell-specific transcription factor, BACH2, modulates ROS mediated cytotoxic responses in mantle cell lymphoma. PLoS ONE 8, e69126 (2013).
Kane, R. C. et al. Bortezomib for the treatment of mantle cell lymphoma. Clin. Cancer Res. 13, 5291–5294 (2007).
Denmeade, S. R. et al. Engineering a prostate-specific membrane antigen-activated tumor endothelial cell prodrug for cancer therapy. Sci. Transl. Med. 4, 140ra86 (2012).
Kardosh, A. et al. Aggravated endoplasmic reticulum stress as a basis for enhanced glioblastoma cell killing by bortezomib in combination with celecoxib or its non-coxib analogue, 2,5-dimethyl-celecoxib. Cancer Res. 68, 843–851 (2008).
Fribley, A., Zeng, Q. & Wang, C. Y. Proteasome inhibitor PS-341 induces apoptosis through induction of endoplasmic reticulum stress-reactive oxygen species in head and neck squamous cell carcinoma cells. Mol. Cell. Biol. 24, 9695–9704 (2004).
Tsutsumi, S. et al. Endoplasmic reticulum stress response is involved in nonsteroidal anti-inflammatory drug-induced apoptosis. Cell Death Differ. 11, 1009–1016 (2004).
Bernstein, W. B. & Dennis, P. A. Repositioning HIV protease inhibitors as cancer therapeutics. Curr. Opin. HIV AIDS 3, 666–675 (2008).
Tai, D. J. et al. Changes in intracellular redox status influence multidrug resistance in gastric adenocarcinoma cells. Exp. Ther. Med. 4, 291–296 (2012).
Ryu, C. S. et al. Elevation of cysteine consumption in tamoxifen-resistant MCF-7 cells. Biochem. Pharmacol. 85, 197–206 (2012).
Griffith, O. W. Mechanism of action, metabolism, and toxicity of buthionine sulfoximine and its higher homologs, potent inhibitors of glutathione synthesis. J. Biol. Chem. 257, 13704–13712 (1982).
Loganathan, S., Kandala, P. K., Gupta, P. & Srivastava, S. K. Inhibition of EGFR-AKT axis results in the suppression of ovarian tumors in vitro and in preclinical mouse model. PLoS ONE 7, e43577 (2012).
Trachootham, D. et al. Selective killing of oncogenically transformed cells through a ROS-mediated mechanism by β-phenylethyl isothiocyanate. Cancer Cell 10, 241–252 (2006).
Raj, L. et al. Selective killing of cancer cells by a small molecule targeting the stress response to ROS. Nature 475, 231–234 (2011).
Suarez-Almazor, M. E., Belseck, E., Shea, B., Wells, G. & Tugwell, P. Sulfasalazine for rheumatoid arthritis. Cochrane Database Syst Rev. 2009, CD000958 (2000).
Gout, P. W., Buckley, A. R., Simms, C. R. & Bruchovsky, N. Sulfasalazine, a potent suppressor of lymphoma growth by inhibition of the x(c)- cystine transporter: a new action for an old drug. Leukemia 15, 1633–1640 (2001).
Lo, M., Ling, V., Low, C., Wang, Y. Z. & Gout, P. W. Potential use of the anti-inflammatory drug, sulfasalazine, for targeted therapy of pancreatic cancer. Curr. Oncol. 17, 9–16 (2010).
Guan, J. et al. The xc- cystine/glutamate antiporter as a potential therapeutic target for small-cell lung cancer: use of sulfasalazine. Cancer Chemother. Pharmacol. 64, 463–472 (2009).
Montero, A. J. et al. Phase 2 study of neoadjuvant treatment with NOV-002 in combination with doxorubicin and cyclophosphamide followed by docetaxel in patients with HER-2 negative clinical stage II-IIIc breast cancer. Breast Cancer Res. Treat. 132, 215–223 (2012).
Townsend, D. M. et al. NOV-002, a glutathione disulfide mimetic, as a modulator of cellular redox balance. Cancer Res. 68, 2870–2877 (2008).
Sobhakumari, A. et al. Susceptibility of human head and neck cancer cells to combined inhibition of glutathione and thioredoxin metabolism. PLoS ONE 7, e48175 (2012).
Marzano, C. et al. Inhibition of thioredoxin reductase by auranofin induces apoptosis in cisplatin-resistant human ovarian cancer cells. Free Radic. Biol. Med. 42, 872–881 (2007).
Vaughn, A. E. & Deshmukh, M. Glucose metabolism inhibits apoptosis in neurons and cancer cells by redox inactivation of cytochrome c. Nature Cell Biol. 10, 1477–1483 (2008).
Polimeni, M. et al. Modulation of doxorubicin resistance by the glucose-6-phosphate dehydrogenase activity. Biochem. J. 439, 141–149 (2011).
Vander Heiden, M. G., Cantley, L. C. & Thompson, C. B. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324, 1029–1033 (2009).
Wang, J. B. et al. Targeting mitochondrial glutaminase activity inhibits oncogenic transformation. Cancer Cell 18, 207–219 (2010). This work underlines the importance of metabolic adaptation in cancer cells. It demonstrates that glutamine metabolism is crucial for cancer cell survival.
Le, A. et al. Glucose-independent glutamine metabolism via TCA cycling for proliferation and survival in B cells. Cell. Metab. 15, 110–121 (2012).
Reinert, R. B. et al. Role of glutamine depletion in directing tissue-specific nutrient stress responses to l-asparaginase. J. Biol. Chem. 281, 31222–31233 (2006).
Dolma, S., Lessnick, S. L., Hahn, W. C. & Stockwell, B. R. Identification of genotype-selective antitumor agents using synthetic lethal chemical screening in engineered human tumor cells. Cancer Cell 3, 285–296 (2003). This work demonstrates the potential of tailored therapeutic intervention against specific gene alterations in tumour cells.
Dixon, S. J. et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149, 1060–1072 (2012). This paper describes a new form of cell death that depends on iron.
Yagoda, N. et al. RAS–RAF–MEK-dependent oxidative cell death involving voltage-dependent anion channels. Nature 447, 864–868 (2007).
Shaw, A. T. et al. Selective killing of K-ras mutant cancer cells by small molecule inducers of oxidative stress. Proc. Natl Acad. Sci. USA 108, 8773–8778 (2011).
Dang, L. et al. Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature 462, 739–744 (2009). This study elucidates the presence of a novel metabolic pathway induced by a tumour-specific gene alteration. The finding offers the opportunity to develop a tailored anticancer therapy.
Sasaki, M. et al. D-2-hydroxyglutarate produced by mutant IDH1 perturbs collagen maturation and basement membrane function. Genes Dev. 26, 2038–2049 (2012).
Sasaki, M. et al. IDH1(R132H) mutation increases murine haematopoietic progenitors and alters epigenetics. Nature 488, 656–659 (2012).
Losman, J. A. et al. (R)-2-hydroxyglutarate is sufficient to promote leukemogenesis and its effects are reversible. Science 339, 1621–1625 (2013).
Greenlee, H., Hershman, D. L. & Jacobson, J. S. Use of antioxidant supplements during breast cancer treatment: a comprehensive review. Breast Cancer Res. Treat. 115, 437–452 (2009).
Misale, S. et al. Emergence of KRAS mutations and acquired resistance to anti-EGFR therapy in colorectal cancer. Nature 486, 532–536 (2012).
Bell, E. L. & Chandel, N. S. Mitochondrial oxygen sensing: regulation of hypoxia-inducible factor by mitochondrial generated reactive oxygen species. Essays Biochem. 43, 17–27 (2007).
Chandel, N. S. et al. Mitochondrial reactive oxygen species trigger hypoxia-induced transcription. Proc. Natl Acad. Sci. USA 95, 11715–11720 (1998).
Semenza, G. L. Oxygen sensing, homeostasis, and disease. N. Engl. J. Med. 365, 537–547 (2011).
Kincaid, M. M. & Cooper, A. A. ERADicate ER stress or die trying. Antioxid. Redox Signal. 9, 2373–2387 (2007).
Bravo, R. et al. Endoplasmic reticulum: ER stress regulates mitochondrial bioenergetics. Int. J. Biochem. Cell Biol. 44, 16–20 (2012).
Tanaka, H. et al. E2F1 and c-Myc potentiate apoptosis through inhibition of NF-κB activity that facilitates MnSOD-mediated ROS elimination. Mol. Cell 9, 1017–1029 (2002).
Irani, K. et al. Mitogenic signaling mediated by oxidants in Ras-transformed fibroblasts. Science 275, 1649–1652 (1997).
Lee, A. C. et al. Ras proteins induce senescence by altering the intracellular levels of reactive oxygen species. J. Biol. Chem. 274, 7936–7940 (1999).
Vafa, O. et al. c-Myc can induce DNA damage, increase reactive oxygen species, and mitigate p53 function: a mechanism for oncogene-induced genetic instability. Mol Cell. 9, 1031–1044 (2002).
Li, W. & Kong, A. N. Molecular mechanisms of Nrf2-mediated antioxidant response. Mol. Carcinog. 48, 91–104 (2009).
Meister, A. Glutathione deficiency produced by inhibition of its synthesis, and its reversal; applications in research and therapy. Pharmacol. Ther. 51, 155–194 (1991).
Murphy, M. P. Mitochondrial thiols in antioxidant protection and redox signaling: distinct roles for glutathionylation and other thiol modifications. Antioxid. Redox Signal. 16, 476–495 (2012).
Vurusaner, B., Poli, G. & Basaga, H. Tumor suppressor genes and ROS: complex networks of interactions. Free Radic. Biol. Med. 52, 7–18 (2012).
Bouayed, J. & Bohn, T. Exogenous antioxidants — double-edged swords in cellular redox state: health beneficial effects at physiologic doses versus deleterious effects at high doses. Oxid. Med. Cell Longev. 3, 228–237 (2010).
Wilson, J. X. Regulation of vitamin C transport. Annu. Rev. Nutr. 25, 105–125 (2005).
Brigelius-Flohe, R. & Traber, M. G. Vitamin E: function and metabolism. FASEB J. 13, 1145–1155 (1999).
Rayman, M. P. Selenium in cancer prevention: a review of the evidence and mechanism of action. Proc. Nutr. Soc. 64, 527–542 (2005).
Burton, G. W. & Ingold, K. U. β-carotene: an unusual type of lipid antioxidant. Science 224, 569–573 (1984).
Klaunig, J. E. & Kamendulis, L. M. The role of oxidative stress in carcinogenesis. Annu. Rev. Pharmacol. Toxicol. 44, 239–267 (2004).
Belfi, C. A., Chatterjee, S., Gosky, D. M., Berger, S. J. & Berger, N. A. Increased sensitivity of human colon cancer cells to DNA cross-linking agents after GRP78 up-regulation. Biochem. Biophys. Res. Commun. 257, 361–368 (1999).
Dufour, E. et al. Pancreatic tumor sensitivity to plasma L-asparagine starvation. Pancreas 41, 940–948 (2012).
Pieters, R. et al. L-asparaginase treatment in acute lymphoblastic leukemia: a focus on Erwinia asparaginase. Cancer 117, 238–249 (2011).
O'Dwyer, P. J. et al. Phase I trial of buthionine sulfoximine in combination with melphalan in patients with cancer. J. Clin. Oncol. 14, 249–256 (1996).
Lewis-Wambi, J. S. et al. Buthionine sulfoximine sensitizes antihormone-resistant human breast cancer cells to estrogen-induced apoptosis. Breast Cancer Res. 10, R104 (2008).
Zhu, J. et al. Using cyclooxygenase-2 inhibitors as molecular platforms to develop a new class of apoptosis-inducing agents. J. Natl Cancer Inst. 94, 1745–1757 (2002).
Gills, J. J. et al. Nelfinavir, a lead HIV protease inhibitor, is a broad-spectrum, anticancer agent that induces endoplasmic reticulum stress, autophagy, and apoptosis in vitro and in vivo. Clin. Cancer Res. 13, 5183–5194 (2007).
Simunek, T. et al. Anthracycline-induced cardiotoxicity: overview of studies examining the roles of oxidative stress and free cellular iron. Pharmacol. Rep. 61, 154–171 (2009).
The Alpha-Tocopherol Beta Carotene Cancer Prevention Study Group. The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. N. Engl. J. Med. 330, 1029–1035 (1994).
Omenn, G. S. et al. Effects of a combination of beta carotene and vitamin A on lung cancer and cardiovascular disease. N. Engl. J. Med. 334, 1150–1155 (1996).
Wang, F. et al. Targeted inhibition of mutant IDH2 in leukemia cells induces cellular differentiation. Science 340, 622–626 (2013).
Acknowledgements
The authors thank members of the Mak laboratory, specifically D. Cescon for his valuable input and M. Saunders for her scientific editing. The authors acknowledge support from Canadian Institutes of Health Research (CIHR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
T.W.M. owns stocks of Agios Pharmaceuticals, Inc.
Related links
DATABASES
Rights and permissions
About this article
Cite this article
Gorrini, C., Harris, I. & Mak, T. Modulation of oxidative stress as an anticancer strategy. Nat Rev Drug Discov 12, 931–947 (2013). https://doi.org/10.1038/nrd4002
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd4002
This article is cited by
-
Evaluation of serum thioredoxin as a hepatocellular carcinoma diagnostic marker
Egyptian Liver Journal (2024)
-
Oxidative stress promotes cytotoxicity in human cancer cell lines exposed to Escallonia spp. extracts
BMC Complementary Medicine and Therapies (2024)
-
Paraptosis: a non-classical paradigm of cell death for cancer therapy
Acta Pharmacologica Sinica (2024)
-
Toxoflavin analog D43 exerts antiproliferative effects on breast cancer by inducing ROS-mediated apoptosis and DNA damage
Scientific Reports (2024)
-
Association of serum and hair antioxidant minerals with an oxidative stress marker in relation with characteristics of healthy adults: a cross-sectional study
Scientific Reports (2024)