Key Points
-
Bone homeostasis is dependent on the concerted actions of bone-building osteoblasts and bone-degrading osteoclasts — a process called bone remodelling.
-
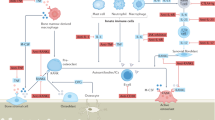
Two of the major factors that induce osteoblast differentiation and activation are: the signalling mediated by bone morphogenetic proteins via runt-related transcription factor 2; and the WNT–Frizzled–β-catenin pathway. Conversely, Dickkopf-related protein 1 (DKK1) and sclerostin inhibit osteoblast activation.
-
Major molecules involved in osteoclast differentiation and activation include macrophage colony-stimulating factor and receptor activator of nuclear factor-κB (RANK) as well as its ligand (RANKL).
-
Inflammation is associated with the overproduction of various cytokines, such as tumour necrosis factor, interleukin-1 (IL-1), IL-6 or IL-17. Their upregulation in the course of inflammation leads to excessive bone degradation mainly due to hyperactivation of osteoclasts, although some cytokines can also impair osteoblast function.
-
Many diseases lead to inflammatory bone loss, including inflammatory bowel disease, chronic obstructive lung disease, cystic fibrosis, periodontitis, rheumatoid arthritis and other inflammatory diseases.
-
Inflammatory bone loss is always systemic, and in some diseases — such as rheumatoid arthritis or periodontitis — it can also involve local bone.
-
Therapies interfering with inflammation also affect systemic inflammatory bone loss, primarily by reducing the effects of cytokines on osteoclast activation; however, many of these treatments will not fully control inflammation. Owing to this ongoing inflammatory activity (even at low levels), bone loss will continue to accrue and therefore also requires specific targeting of bone cells.
-
Bisphosphonates and denosumab are among the bone-targeting therapies that have been shown to be effective in treating inflammatory bone loss, but it is assumed that blockers of DKK1 and sclerostin — which are upregulated by cytokines and inhibit osteoblast repair mechanisms — are also likely to be effective.
Abstract
Bone is a tissue undergoing continuous building and degradation. This remodelling is a tightly regulated process that can be disturbed by many factors, particularly hormonal changes. Chronic inflammation can also perturb bone metabolism and promote increased bone loss. Inflammatory diseases can arise all over the body, including in the musculoskeletal system (for example, rheumatoid arthritis), the intestine (for example, inflammatory bowel disease), the oral cavity (for example, periodontitis) and the lung (for example, cystic fibrosis). Wherever inflammatory diseases occur, systemic effects on bone will ensue, as well as increased fracture risk. Here, we discuss the cellular and signalling pathways underlying, and strategies for therapeutically interfering with, the inflammatory loss of bone.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Sims, N. A. & Gooi, J. H. Bone remodeling: multiple cellular interactions required for coupling of bone formation and resorption. Semin. Cell Dev. Biol. 19, 444–451 (2008).
Bodine, P. V. & Komm, B. S. Wnt signaling and osteoblastogenesis. Rev. Endocr. Metab. Disord. 7, 33–39 (2006).
Yavropoulou, M. P. & Yovos, J. G. Osteoclastogenesis — current knowledge and future perspectives. J. Musculoskelet. Neuronal. Interact. 8, 204–216 (2008).
Eriksen, E. F. Cellular mechanisms of bone remodeling. Rev. Endocr. Metab. Disord. 11, 219–227 (2010).
Raisz, L. G. & Rodan, G. A. Pathogenesis of osteoporosis. Endocrinol. Metab. Clin. North Am. 32, 15–24 (2003).
Frenkel, B. et al. Regulation of adult bone turnover by sex steroids. J. Cell Physiol. 224, 305–310 (2010).
Mosekilde, L. Primary hyperparathyroidism and the skeleton. Clin. Endocrinol. (Oxf.) 69, 1–19 (2008).
Bliuc, D. et al. Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA 301, 513–521 (2009).
Johnell, O. et al. Mortality after osteoporotic fractures. Osteoporos. Int. 15, 38–42 (2004).
Grabowski, P. Physiology of bone. Endocr. Dev. 16, 32–48 (2009).
Holm, K. & Hedricks, C. Immobility and bone loss in the aging adult. Crit. Care Nurs. Q. 12, 46–51 (1989).
Michalakis, K., Peitsidis, P. & Ilias, I. Pregnancy- and lactation-associated osteoporosis: a narrative mini-review. Endocr. Regul. 45, 43–47 (2011).
Howe, T. E. et al. Exercise for preventing and treating osteoporosis in postmenopausal women. Cochrane Database. Syst. Rev. CD000333 (2011).
Sinaki, M. et al. The role of exercise in the treatment of osteoporosis. Curr. Osteoporos. Rep. 8, 138–144 (2010).
Mundy, G. R. Osteoporosis and inflammation. Nutr. Rev. 65, S147–S151 (2007).
Romas, E. & Gillespie, M. T. Inflammation-induced bone loss: can it be prevented? Rheum. Dis. Clin. North Am. 32, 759–773 (2006).
Smolen, J. S. et al. Radiographic changes in rheumatoid arthritis patients attaining different disease activity states with methotrexate monotherapy and infliximab plus methotrexate: the impacts of remission and TNF-blockade. Ann. Rheum. Dis. 68, 823–827 (2009).
Gaur, T. et al. Canonical WNT signaling promotes osteogenesis by directly stimulating Runx2 gene expression. J. Biol. Chem. 280, 33132–33140 (2005).
Derynck, R. & Zhang, Y. E. Smad-dependent and Smad-independent pathways in TGF-β family signalling. Nature 425, 577–584 (2003). This study provides an authoritative review of the signal transduction pathways mediated bythe transforming growth factor-β protein family, to which BMPs belong, which utilize SMADs as intracellular effectors of transcriptional regulation.
Qin, L. et al. Gene expression profiles and transcription factors involved in parathyroid hormone signaling in osteoblasts revealed by microarray and bioinformatics. J. Biol. Chem. 278, 19723–19731 (2003).
Bodine, P. V., Seestaller-Wehr, L., Kharode, Y. P., Bex, F. J. & Komm, B. S. Bone anabolic effects of parathyroid hormone are blunted by deletion of the Wnt antagonist secreted frizzled-related protein-1. J. Cell Physiol. 210, 352–357 (2007).
Gazzerro, E. & Canalis, E. Bone morphogenetic proteins and their antagonists. Rev. Endocr. Metab. Disord. 7, 51–65 (2006).
Franceschi, R. T. & Xiao, G. Regulation of the osteoblast-specific transcription factor, Runx2: responsiveness to multiple signal transduction pathways. J. Cell Biochem. 88, 446–454 (2003).
Zaidi, M. Skeletal remodeling in health and disease. Nature Med. 13, 791–801 (2007).
Lian, J. B. et al. Networks and hubs for the transcriptional control of osteoblastogenesis. Rev. Endocr. Metab. Disord. 7, 1–16 (2006).
Yamane, T. et al. Wnt signaling regulates hemopoiesis through stromal cells. J. Immunol. 167, 765–772 (2001).
Van Den Berg, D. J., Sharma, A. K., Bruno, E. & Hoffman, R. Role of members of the Wnt gene family in human hematopoiesis. Blood 92, 3189–3202 (1998).
van Amerongen, R. & Nusse, R. Towards an integrated view of Wnt signaling in development. Development 136, 3205–3214 (2009).
Fedi, P. et al. Isolation and biochemical characterization of the human Dkk-1 homologue, a novel inhibitor of mammalian Wnt signaling. J. Biol. Chem. 274, 19465–19472 (1999).
Kwack, M. H. et al. Dihydrotestosterone-inducible Dickkopf 1 from balding dermal papilla cells causes apoptosis in follicular keratinocytes. J. Invest. Dermatol. 128, 262–269 (2008).
Tian, E. et al. The role of the Wnt-signaling antagonist DKK1 in the development of osteolytic lesions in multiple myeloma. N. Engl. J. Med. 349, 2483–2494 (2003).
Diarra, D. et al. Dickkopf-1 is a master regulator of joint remodeling. Nature Med. 13, 156–163 (2007).
Mao, B. et al. Kremen proteins are Dickkopf receptors that regulate Wnt/β-catenin signalling. Nature 417, 664–667 (2002).
Veverka, V. et al. Characterization of the structural features and interactions of sclerostin: molecular insight into a key regulator of Wnt-mediated bone formation. J. Biol. Chem. 284, 10890–10900 (2009).
Romero, G. et al. Parathyroid hormone receptor directly interacts with Dishevelled to regulate β-catenin signaling and osteoclastogenesis. J. Biol. Chem. 285, 14756–14763 (2010).
Nakashima, K. et al. The novel zinc finger-containing transcription factor osterix is required for osteoblast differentiation and bone formation. Cell 108, 17–29 (2002). This publication provides the first description of the role of osterix in osteoblast differentiation.
Koga, T. et al. NFAT and osterix cooperatively regulate bone formation. Nature Med. 11, 880–885 (2005).
Chang, J. et al. Inhibition of osteoblastic bone formation by nuclear factor-κB. Nature Med. 15, 682–689 (2009).
Krum, S. A., Chang, J., Miranda-Carboni, G. & Wang, C. Y. Novel functions for NFκB: inhibition of bone formation. Nature Rev. Rheumatol. 6, 607–611 (2010).
Gooi, J. H. et al. Calcitonin impairs the anabolic effect of PTH in young rats and stimulates expression of sclerostin by osteocytes. Bone 46, 1486–1497 (2010).
Yasuda, H. et al. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc. Natl Acad. Sci. USA 95, 3597–3602 (1998). This was one of the initial studies describing RANKL and its pivotal osteoclastogenic role.
Murshed, M., Harmey, D., Millan, J. L., McKee, M. D. & Karsenty, G. Unique coexpression in osteoblasts of broadly expressed genes accounts for the spatial restriction of ECM mineralization to bone. Genes Dev. 19, 1093–1104 (2005).
Bonewald, L. F. Osteocytes as dynamic multifunctional cells. Ann. NY Acad. Sci. 1116, 281–290 (2007).
Bonewald, L. F. & Johnson, M. L. Osteocytes, mechanosensing and Wnt signaling. Bone 42, 606–615 (2008).
Schneider, P., Meier, M., Wepf, R. & Muller, R. Towards quantitative 3D imaging of the osteocyte lacuno–canalicular network. Bone 47, 848–858 (2010).
van Bezooijen, R. L., ten Dijke, P., Papapoulos, S. E. & Lowik, C. W. SOST/sclerostin, an osteocyte-derived negative regulator of bone formation. Cytokine Growth Factor Rev. 16, 319–327 (2005).
Murakami, M. et al. IL-6-induced homodimerization of gp130 and associated activation of a tyrosine kinase. Science 260, 1808–1810 (1993). This paper clarifies the complexity of IL-6R molecules and signalling.
Rose-John, S., Scheller, J., Elson, G. & Jones, S. A. Interleukin-6 biology is coordinated by membrane-bound and soluble receptors: role in inflammation and cancer. J. Leukoc. Biol. 80, 227–236 (2006).
Walker, E. C. et al. Oncostatin M promotes bone formation independently of resorption when signaling through leukemia inhibitory factor receptor in mice. J. Clin. Invest. 120, 582–592 (2010).
Malaval, L., Liu, F., Vernallis, A. B. & Aubin, J. E. GP130/OSMR is the only LIF/IL-6 family receptor complex to promote osteoblast differentiation of calvaria progenitors. J. Cell Physiol. 204, 585–593 (2005).
Sims, N. A. & Walsh, N. C. gp130 cytokines and bone remodelling in health and disease. BMB Rep. 43, 513–523 (2010).
Manolagas, S. C. Birth and death of bone cells: basic regulatory mechanisms and implications for the pathogenesis and treatment of osteoporosis. Endocr. Rev. 21, 115–137 (2000).
Hay, E., Lemonnier, J., Fromigue, O., Guenou, H. & Marie, P. J. Bone morphogenetic protein receptor IB signaling mediates apoptosis independently of differentiation in osteoblastic cells. J. Biol. Chem. 279, 1650–1658 (2004).
Bradford, P. G., Gerace, K. V., Roland, R. L. & Chrzan, B. G. Estrogen regulation of apoptosis in osteoblasts. Physiol. Behav. 99, 181–185 (2010).
Moriishi, T. et al. Overexpression of Bcl2 in osteoblasts inhibits osteoblast differentiation and induces osteocyte apoptosis. PLoS ONE 6, e27487 (2011).
de Vernejoul, M. C. & Kornak, U. Heritable sclerosing bone disorders: presentation and new molecular mechanisms. Ann. NY Acad. Sci. 1192, 269–277 (2010).
Li, X. et al. Targeted deletion of the sclerostin gene in mice results in increased bone formation and bone strength. J. Bone Miner. Res. 23, 860–869 (2008).
Teitelbaum, S. L. Bone resorption by osteoclasts. Science 289, 1504–1508 (2000).
Wagner, E. F. Bone development and inflammatory disease is regulated by AP-1 (Fos/Jun). Ann. Rheum. Dis. 69 (Suppl. 1), 86–88 (2010).
Schonthaler, H. B., Guinea-Viniegra, J. & Wagner, E. F. Targeting inflammation by modulating the Jun/AP-1 pathway. Ann. Rheum. Dis. 70 (Suppl. 1), 109–112 (2011).
Shaulian, E. & Karin, M. AP-1 as a regulator of cell life and death. Nature Cell Biol. 4, E131–E136 (2002).
Koga, T. et al. Costimulatory signals mediated by the ITAM motif cooperate with RANKL for bone homeostasis. Nature 428, 758–763 (2004).
Takayanagi, H. et al. Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev. Cell 3, 889–901 (2002).
Delaisse, J. M. et al. Matrix metalloproteinases (MMP) and cathepsin K contribute differently to osteoclastic activities. Microsc. Res. Tech. 61, 504–513 (2003).
Supanchart, C. & Kornak, U. Ion channels and transporters in osteoclasts. Arch. Biochem. Biophys. 473, 161–165 (2008).
Goldring, S. R., Roelke, M. S., Petrison, K. K. & Bhan, A. K. Human giant cell tumors of bone identification and characterization of cell types. J. Clin. Invest. 79, 483–491 (1987).
Dougall, W. C. et al. RANK is essential for osteoclast and lymph node development. Genes Dev. 13, 2412–2424 (1999).
Hofbauer, L. C. et al. The roles of osteoprotegerin and osteoprotegerin ligand in the paracrine regulation of bone resorption. J. Bone Miner. Res. 15, 2–12 (2000).
Wiktor-Jedrzejczak, W. et al. Total absence of colony-stimulating factor 1 in the macrophage-deficient osteopetrotic (op/op) mouse. Proc. Natl Acad. Sci. USA 87, 4828–4832 (1990).
Grigoriadis, A. E. et al. c-Fos: a key regulator of osteoclast-macrophage lineage determination and bone remodeling. Science 266, 443–448 (1994). This study reveals the essential role of the transcription factor AP1, and particularly its component FOS, in osteoclastogenesis.
Binder, N. B. et al. Estrogen-dependent and C-C chemokine receptor-2-dependent pathways determine osteoclast behavior in osteoporosis. Nature Med. 15, 417–424 (2009).
Kim, M. S., Day, C. J. & Morrison, N. A. MCP-1 is induced by receptor activator of nuclear factor-κB ligand, promotes human osteoclast fusion, and rescues granulocyte macrophage colony-stimulating factor suppression of osteoclast formation. J. Biol. Chem. 280, 16163–16169 (2005).
Kong, Y. Y. et al. Activated T cells regulate bone loss and joint destruction in adjuvant arthritis through osteoprotegerin ligand. Nature 402, 304–309 (1999). This publication links the adaptive immune system to osteoclast activation and inflammatory bone destruction.
Roggia, C. et al. Up-regulation of TNF-producing T cells in the bone marrow: a key mechanism by which estrogen deficiency induces bone loss in vivo. Proc. Natl Acad. Sci. USA 98, 13960–13965 (2001). This study provides evidence of the role of TNF in osteoporosis induced by oestrogen deficiency.
Kapinas, K. & Delany, A. M. MicroRNA biogenesis and regulation of bone remodeling. Arthritis Res. Ther. 13, 220 (2011).
Sugatani, T., Vacher, J. & Hruska, K. A. A microRNA expression signature of osteoclastogenesis. Blood 117, 3648–3657 (2011).
Kapinas, K., Kessler, C., Ricks, T., Gronowicz, G. & Delany, A. M. miR-29 modulates Wnt signaling in human osteoblasts through a positive feedback loop. J. Biol. Chem. 285, 25221–25231 (2010).
Sugatani, T. & Hruska, K. A. Impaired micro-RNA pathways diminish osteoclast differentiation and function. J. Biol. Chem. 284, 4667–4678 (2009).
Gough, A. K., Lilley, J., Eyre, S., Holder R. L. & Emery P. Generalised bone loss in patients with early rheumatoid arthritis. Lancet 344, 23–27 (1994).
Romas, E. Bone loss in inflammatory arthritis: mechanisms and therapeutic approaches with bisphosphonates. Best Pract. Res. Clin. Rheumatol. 19, 1065–1079 (2005).
Roldan, J. F., del, Rincón, I. & Escalante, A. Loss of cortical bone from the metacarpal diaphysis in patients with rheumatoid arthritis: independent effects of systemic inflammation and glucocorticoids. J. Rheumatol. 33, 508–516 (2006).
Gravallese, E. M. et al. Identification of cell types responsible for bone resorption in rheumatoid arthritis and juvenile rheumatoid arthritis. Am. J. Pathol. 152, 943–951 (1998). This study reveals the role of synovial-derived osteoclasts in local bone damage (erosions) in patients with rheumatoid arthritis.
Garcia-Carrasco, M. et al. Osteoporosis in patients with systemic lupus erythematosus. Isr. Med. Assoc. J. 11, 486–491 (2009).
Grisar, J. et al. Ankylosing spondylitis, psoriatic arthritis, and reactive arthritis show increased bone resorption, but differ with regard to bone formation. J. Rheumatol. 29, 1430–1436 (2002).
Ali, T., Lam, D., Bronze, M. S. & Humphrey, M. B. Osteoporosis in inflammatory bowel disease. Am. J. Med. 122, 599–604 (2009).
Paganelli, M. et al. Inflammation is the main determinant of low bone mineral density in pediatric inflammatory bowel disease. Inflamm. Bowel Dis. 13, 416–423 (2007).
Bianchi, M. L. & Bardella, M. T. Bone in celiac disease. Osteoporos. Int. 19, 1705–1716 (2008).
Cashman, K. D. Altered bone metabolism in inflammatory disease: role for nutrition. Proc. Nutr. Soc. 67, 196–205 (2008).
Shead, E. F., Haworth, C. S., Barker, H., Bilton, D. & Compston, J. E. Osteoclast function, bone turnover and inflammatory cytokines during infective exacerbations of cystic fibrosis. J. Cyst. Fibros. 9, 93–98 (2010).
Dam, T. T., Harrison, S., Fink, H. A., Ramsdell, J. & Barrett-Connor, E. Bone mineral density and fractures in older men with chronic obstructive pulmonary disease or asthma. Osteoporos. Int. 21, 1341–1349 (2010).
Yoshihara, A., Seida, Y., Hanada, N. & Miyazaki, H. A longitudinal study of the relationship between periodontal disease and bone mineral density in community-dwelling older adults. J. Clin. Periodontol. 31, 680–684 (2004).
Redlich, K. et al. Repair of local bone erosions and reversal of systemic bone loss upon therapy with anti-tumor necrosis factor in combination with osteoprotegerin or parathyroid hormone in tumor necrosis factor-mediated arthritis. Am. J. Pathol. 164, 543–555 (2004).
Lin, C. L., Moniz, C., Chambers, T. J. & Chow, J. W. Colitis causes bone loss in rats through suppression of bone formation. Gastroenterology 111, 1263–1271 (1996).
Mattila, K. J., Valle, M. S., Nieminen, M. S., Valtonen, V. V. & Hietaniemi, K. L. Dental infections and coronary atherosclerosis. Atherosclerosis 103, 205–211 (1993).
Reddy, M. S. Oral osteoporosis: is there an association between periodontitis and osteoporosis? Compend. Contin. Educ. Dent. 23, 21–28 (2002).
Kawai, T. et al. B and T lymphocytes are the primary sources of RANKL in the bone resorptive lesion of periodontal disease. Am. J. Pathol. 169, 987–998 (2006).
Teng, Y. T. et al. Functional human T-cell immunity and osteoprotegerin ligand control alveolar bone destruction in periodontal infection. J. Clin. Invest. 106, R59–R67 (2000).
Korn, T., Bettelli, E., Oukka, M. & Kuchroo, V. K. IL-17 and Th17 cells. Annu. Rev. Immunol. 27, 485–517 (2009).
Hundorfean, G., Neurath, M. F. & Mudter, J. Functional relevance of T helper 17 (Th17) cells and the IL-17 cytokine family in inflammatory bowel disease. Inflamm. Bowel Dis. 18, 180–186 (2012).
Wong, P. K. et al. Interleukin-6 modulates production of T lymphocyte-derived cytokines in antigen-induced arthritis and drives inflammation-induced osteoclastogenesis. Arthritis Rheum. 54, 158–168 (2006). This study provides evidence of the role of IL-6 and associated IL-17 production on osteoclast generation.
Dinarello, C. A. Interleukin-1 and interleukin-1 antagonism. Blood 77, 1627–1652 (1991).
Tracey K. J. & Cerami, A. Tumor necrosis factor: a pleiotropic cytokine and therapeutic target. Annu. Rev. Med. 45, 491–503 (1994).
Dinarello, C. A. et al. Tumor necrosis factor (cachectin) is an endogeneous pyrogen and induces production of interleukin 1. J. Exp. Med. 163, 1433–1450 (1986).
Naka, T., Nishimoto, N. & Kishimoto, T. The paradigm of IL-6: from basic science to medicine. Arthritis Res. 4 (Suppl. 3), 233–242 (2002).
Kishimoto, T. IL-6: from its discovery to clinical applications. Int. Immunol. 22, 347–352 (2010).
Wallach, D. et al. Tumor necrosis factor receptor and Fas signaling mechanisms. Annu. Rev. Immunol. 17, 331–367 (1999).
Goeddel, D. V. Signal transduction by tumor necrosis factor: the Parker B. Francis Lectureship. Chest 116 (Suppl. 1), 69–73 (1999).
Sethi, G., Sung, B. & Aggarwal, B. B. TNF: a master switch for inflammation to cancer. Front. Biosci. 13, 5094–5107 (2008).
Ikejima, T., Okusawa, S., Ghezzi, P., van der Meer, J. W. & Dinarello, C. A. Interleukin-1 induces tumor necrosis factor (TNF) in human peripheral blood mononuclear cells in vitro and a circulating TNF-like activity in rabbits. J. Infect. Dis. 162, 215–223 (1990).
Legendre, F., Bogdanowicz, P., Boumediene, K. & Pujol, J. P. Role of interleukin 6 (IL-6)/IL-6R-induced signal tranducers and activators of transcription and mitogen-activated protein kinase/extracellular signal-related kinase in upregulation of matrix metalloproteinase and ADAMTS gene expression in articular chondrocytes. J. Rheumatol. 32, 1307–1316 (2005).
Rowan, A. D. et al. Synergistic effects of glycoprotein 130 binding cytokines in combination with interleukin-1 on cartilage collagen breakdown. Arthritis Rheum. 44, 1620–1632 (2001).
Shingu, M. et al. The effects of cytokines on metalloproteinase inhibitors (TIMP) and collagenase production by human chondrocytes and TIMP production by synovial cells and endothelial cells. Clin. Exp. Immunol. 94, 145–149 (1993).
Solis-Herruzo, J. A. et al. Interleukin-6 increases rat metalloproteinase-13 gene expression through stimulation of activator protein 1 transcription factor in cultured fibroblasts. J. Biol. Chem. 274, 30919–30926 (1999).
Richards, C., Gauldie, J. & Baumann, H. Cytokine control of acute phase protein expression. Eur. Cytokine Netw. 2, 89–98 (1991).
Dinarello, C. A. Interleukin-1 and the pathogenesis of the acute-phase response. N. Engl. J. Med. 311, 1413–1418 (1984).
Kushner, I. Regulation of the acute phase response by cytokines. Perspect. Biol. Med. 36, 611–622 (1993).
Andus, T., Geiger, T., Hirano, T., Kishimoto, T. & Heinrich, P. C. Action of recombinant human interleukin 6, interleukin 1β and tumor necrosis factor α on the mRNA induction of acute-phase proteins. Eur. J. Immunol. 18, 739–746 (1988).
Zhang, Y. H., Lin, J. X. & Vilcek, J. Interleukin-6 induction by tumor necrosis factor and interleukin-1 in human fibroblasts involves activation of a nuclear factor binding to a kappa B-like sequence. Mol. Cell Biol. 10, 3818–3823 (1990).
Everaerdt, B., Brouckaert, P. & Fiers, W. Recombinant IL-1 receptor antagonist protects against TNF-induced lethality in mice. J. Immunol. 152, 5041–5049 (1994).
Devlin, R. D., Reddy, S. V., Savino, R., Ciliberto, G. & Roodman G. D. IL-6 mediates the effects of IL-1 or TNF, but not PTHrP or 1,25(OH)2D3, on osteoclast-like cell formation in normal human bone marrow cultures. J. Bone Miner. Res. 13, 393–399 (1998).
Ma, T. et al. Human interleukin-1-induced murine osteoclastogenesis is dependent on RANKL, but independent of TNF-α. Cytokine 26, 138–144 (2004).
Lee, Z. H. et al. IL-1α stimulation of osteoclast survival through the PI 3-kinase/Akt and ERK pathways. J. Biochem. 131, 161–166 (2002).
Kobayashi, K. et al. Tumor necrosis factor α stimulates osteoclast differentiation by a mechanism independent of the ODF/RANKL–RANK interaction. J. Exp. Med. 191, 275–286 (2000).
Kotake, S. et al. Interleukin-6 and soluble interleukin-6 receptors in the synovial fluids from rheumatoid arthritis patients are responsible for osteoclast-like cell formation. J. Bone Miner. Res. 11, 88–95 1996.
De Benedetti, F. et al. Impaired skeletal development in interleukin-6-transgenic mice: a model for the impact of chronic inflammation on the growing skeletal system. Arthritis Rheum. 54, 3551–3563 (2006).
Lam, J. et al. TNF-α induces osteoclastogenesis by direct stimulation of macrophages exposed to permissive levels of RANK ligand. J. Clin. Invest. 106, 1481–1488 (2000). This report reveals the role of the pro-inflammatory cytokine TNF in osteoclastogenesis.
Abu-Amer, Y., Ross, F. P., Edwards, J. & Teitelbaum S. L. Lipopolysaccharide-stimulated osteoclastogenesis is mediated by tumor necrosis factor via its p55 receptor. J. Clin. Invest. 100, 1557–1565 (1997).
Bluml, S. et al. Antiinflammatory effects of tumor necrosis factor on hematopoietic cells in a murine model of erosive arthritis. Arthritis Rheum. 62, 1608–1619 (2010). This study dissects the osteoclastogenic effects of TNF signals, showing that these effects are mediated via activation of TNF receptor 1 rather than TNF receptor 2, and that the latter may have protective effects.
Zhang, Y. H., Heulsmann, A., Tondravi, M. M., Mukherjee, A. & Abu-Amer, Y. Tumor necrosis factor-α (TNF) stimulates RANKL-induced osteoclastogenesis via coupling of TNF type 1 receptor and RANK signaling pathways. J. Biol. Chem. 276, 563–568 (2001).
Ravetch, J. V. & Bolland, S. IgG Fc receptors. Annu. Rev. Immunol. 19, 275–290 (2001).
Zou, W. et al. Syk, c-Src, the αvβ3 integrin, and ITAM immunoreceptors, in concert, regulate osteoclastic bone resorption. J. Cell Biol. 176, 877–888 (2007).
Vignery, A. Macrophage fusion: the making of osteoclasts and giant cells. J. Exp. Med. 202, 337–340 (2005).
Zvaifler, N. J. Rheumatoid synovitis. An extravascular immune complex disease. Arthritis Rheum. 17, 297–305 (1974).
Weissmann, G. Rheumatoid arthritis and systemic lupus erythematosus as immune complex diseases. Bull. NYU Hosp. Jt Dis. 67, 251–253 (2009).
Brown, E. E., Edberg, J. C. & Kimberly, R. P. Fc receptor genes and the systemic lupus erythematosus diathesis. Autoimmunity 40, 567–581 (2007).
Mocsai, A. et al. The immunomodulatory adapter proteins DAP12 and Fc receptor γ-chain (FcRγ) regulate development of functional osteoclasts through the Syk tyrosine kinase. Proc. Natl Acad. Sci. USA 101, 6158–6163 (2004).
Scott, D. L., Symmons, D. P., Coulton, B. L. & Popert, A. J. Long-term outcome of treating rheumatoid arthritis: results after 20 years. Lancet 1, 1108–1111 (1987).
Aringer, M. & Smolen, J. S. Therapeutic blockade of TNF in patients with SLE — promising or crazy? Autoimmunity Rev. 18 May 2011 (doi:10.1016/j.autrev.2011.05.001.2011).
Sokolove, J., Zhao, X., Chandra, P. E. & Robinson, W. H. Immune complexes containing citrullinated fibrinogen costimulate macrophages via Toll-like receptor 4 and Fcγ receptor. Arthritis Rheum. 63, 53–62 (2011).
Guerne, P. A., Carson, D. A. & Lotz, M. IL-6 production by human articular chondrocytes. Modulation of its synthesis by cytokines, growth factors, and hormones in vitro. J. Immunol. 144, 499–505 (1990).
van Gool, J., van Vugt, H., Helle, M. & Aarden, L. A. The relation among stress, adrenalin, interleukin 6 and acute phase proteins in the rat. Clin. Immunol. Immunopathol. 57, 200–210 (1990).
Chrousos, G. P. The hypothalamic–pituitary–adrenal axis and immune-mediated inflammation. N. Engl. J. Med. 332, 1351–1362 (1995).
Perlstein, R. S., Whitnall, M. H., Abrams, J. S., Mougey, E. H. & Neta, R. Synergistic roles of interleukin-6, interleukin-1, and tumor necrosis factor in the adrenocorticotropin response to bacterial lipopolysaccharide in vivo. Endocrinology 132, 946–952 (1993).
Rachon, D., Mysliwska, J., Suchecka-Rachon, K., Wieckiewicz, J. & Mysliwski, A. Effects of oestrogen deprivation on interleukin-6 production by peripheral blood mononuclear cells of postmenopausal women. J. Endocrinol. 172, 387–395 (2002).
Poli, V. et al. Interleukin-6 deficient mice are protected from bone loss caused by estrogen depletion. EMBO J. 13, 1189–1196 (1994).
Girasole, G., Passeri, G., Jilka, R. L. & Manolagas, S. C. Interleukin-11: a new cytokine critical for osteoclast development. J. Clin. Invest. 93, 1516–1524 (1994).
Okamoto, H. et al. The synovial expression and serum levels of interleukin-6, interleukin-11, leukemia inhibitory factor, and oncostatin M in rheumatoid arthritis. Arthritis Rheum. 40, 1096–1105 (1997).
Sasai, M. et al. Delayed onset and reduced severity of collagen-induced arthritis in interleukin-6-deficient mice. Arthritis Rheum. 42, 1635–1643 (1999).
Gilbert, L. et al. Expression of the osteoblast differentiation factor RUNX2 (Cbfa1/AML3/Pebp2αA) is inhibited by tumor necrosis factor-α. J. Biol. Chem. 277, 2695–2701 (2002).
Gilbert, L. C., Rubin, J. & Nanes, M. S. The p55 TNF receptor mediates TNF inhibition of osteoblast differentiation independently of apoptosis. Am. J. Physiol. Endocrinol. Metab. 288, E1011–E1018 (2005).
Kaneki, H. et al. Tumor necrosis factor promotes Runx2 degradation through up-regulation of Smurf1 and Smurf2 in osteoblasts. J. Biol. Chem. 281, 4326–4333 (2006).
Abbas, S., Zhang, Y. H., Clohisy, J. C. & Abu-Amer, Y. Tumor necrosis factor-α inhibits pre-osteoblast differentiation through its type-1 receptor. Cytokine 22, 33–41 (2003).
Mukai, T. et al. TNF-α inhibits BMP-induced osteoblast differentiation through activating SAPK/JNK signaling. Biochem. Biophys. Res. Commun. 356, 1004–1010 (2007).
Ding, J. et al. TNF-α and IL-1β inhibit RUNX2 and collagen expression but increase alkaline phosphatase activity and mineralization in human mesenchymal stem cells. Life Sci. 84, 499–504 (2009).
Hughes, F. J. & Howells, G. L. Interleukin-6 inhibits bone formation in vitro. Bone Miner. 21, 21–28 (1993).
Hughes, F. J. & Howells, G. L. Interleukin-11 inhibits bone formation in vitro. Calcif. Tissue Int. 53, 362–364 (1993).
Li, X. et al. Sclerostin binds to LRP5/6 and antagonizes canonical Wnt signaling. J. Biol. Chem. 280, 19883–19887 (2005).
Winkler, D. G. et al. Osteocyte control of bone formation via sclerostin, a novel BMP antagonist. EMBO J. 22, 6267–6276 (2003).
Mason, J. J. & Williams, B. O. SOST and DKK: antagonists of LRP family signaling as targets for treating bone disease. J. Osteoporos. 2010, 460120 (2010).
Viswanathan, A. & Sylvester, F. A. Chronic pediatric inflammatory diseases: effects on bone. Rev. Endocr. Metab. Disord. 9, 107–122 (2008).
Capriles, V. D., Martini, L. A. & Areas, J. A. Metabolic osteopathy in celiac disease: importance of a gluten-free diet. Nutr. Rev. 67, 599–606 (2009).
Payne, J. B. & Golub, L. M. Using tetracyclines to treat osteoporotic/osteopenic bone loss: from the basic science laboratory to the clinic. Pharmacol. Res. 63, 121–129 (2011).
Elborn, J. S. How can we prevent multisystem complications of cystic fibrosis? Semin. Respir. Crit. Care Med. 28, 303–311 (2007).
Haugeberg, G., Conaghan, P. G., Quinn, M. & Emery, P. Bone loss in patients with active early rheumatoid arthritis: infliximab and methotrexate compared with methotrexate treatment alone. Explorative analysis from a 12-month randomised, double-blind, placebo-controlled study. Ann. Rheum. Dis. 68, 1898–1901 (2009).
Strand, V. & Simon, L. S. Low dose glucocorticoids in early rheumatoid arthritis. Clin. Exp. Rheumatol. 21 (Suppl. 31), 186–190 (2003).
Kim, H. J. et al. Glucocorticoids suppress bone formation via the osteoclast. J. Clin. Invest. 116, 2152–2160 (2006).
Hoes, J. N. et al. EULAR evidence-based recommendations on the management of systemic glucocorticoid therapy in rheumatic diseases. Ann. Rheum. Dis. 66, 1560–1567 (2007).
Teitelbaum, S. L., Seton, M. P. & Saag, K. G. Should bisphosphonates be used for long-term treatment of glucocorticoid-induced osteoporosis? Arthritis Rheum. 63, 325–328 (2011).
Lange, U., Teichmann, J., Muller-Ladner, U. & Strunk, J. Increase in bone mineral density of patients with rheumatoid arthritis treated with anti-TNF-α antibody: a prospective open-label pilot study. Rheumatology (Oxford) 44, 1546–1548 (2005).
Bernstein, M., Irwin, S. & Greenberg, G. R. Maintenance infliximab treatment is associated with improved bone mineral density in Crohn's disease. Am. J. Gastroenterol. 100, 2031–2035 (2005).
Veerappan, S. G., O'Morain, C. A., Daly, J. S. & Ryan, B. M. Review article: the effects of antitumour necrosis factor-a on bone metabolism in inflammatory bowel disease. Aliment. Pharmacol. Ther. 33, 1261–1272 (2011).
Kremer, J. M. et al. Tocilizumab inhibits structural joint damage in rheumatoid arthritis patients with inadequate responses to methotrexate: results from the double-blind treatment phase of a randomized placebo-controlled trial of tocilizumab safety and prevention of structural joint damage at one year. Arthritis Rheum. 63, 609–621 (2011).
Ito, H. et al. A pilot randomized trial of a human anti-interleukin-6 receptor monoclonal antibody in active Crohn's disease. Gastroenterology 126, 989–996 (2004).
Hsu, B., Zhou, B., Smolen, J. S. & Weinblatt, M. Proof-of-concept for CNTO 136, a human anti-interleukin-6 monoclonal antibody, in a multicenter, randomized, double-blind, placebo-controlled, Phase 2 study in patients with active rheumatoid arthritis despite methotrexate therapy. Ann. Rheum. Dis. 70 (Suppl. 3), 459 (2011).
Mease, P. et al. Inhibition of IL-6 with ALD518 improves disease activity in rheumatoid arthritis in a randomized, double-blind, placebo-controlled, dose ranging Phase 2 clinical trial. Ann. Rheum. Dis. 69 (Suppl. 3), 98 (2011).
Hickling, M., Golor, G., Juillon, A., Shaw, S. & Kretsos, K. Safety and pharmacokinetics of CDP6038, an anti-IL-6 monoclonal antibody, administered by subcutaneous injection and intravenous infusion to healthy male volunteers: a Phase 1 study. Ann. Rheum. Dis. 70 (Suppl. 3), 471 (2011).
Nam, J. L. et al. Current evidence for the management of rheumatoid arthritis with biological disease-modifying antirheumatic drugs: a systematic literature review informing the EULAR recommendations for the management of RA. Ann. Rheum. Dis. 69, 976–986 (2010).
Baltzer, A. W. et al. Gene therapy for osteoporosis: evaluation in a murine ovariectomy model. Gene Ther. 8, 1770–1776 (2001).
Genovese, M. et al. Secukinumab (ain457) showed a rapid decrease of disease activity in patients with active rheumatoid arthritis including those with high inflammatory burden. Ann. Rheum. Dis. 70 (Suppl. 3), 472 (2011).
Bluml, S. et al. Essential role of microRNA-155 in the pathogenesis of autoimmune arthritis in mice. Arthritis Rheum. 63, 1281–1288 (2011).
Yang, M. & Mattes, J. Discovery, biology and therapeutic potential of RNA interference, microRNA and antagomirs. Pharmacol. Ther. 117, 94–104 (2008).
Smolen J. S. & Steiner, G. Therapeutic strategies for rheumatoid arthritis. Nature Rev. Drug Discov. 2, 473–488 (2003).
Westhovens, R. et al. Clinical efficacy and safety of abatacept in methotrexate-naive patients with early rheumatoid arthritis and poor prognostic factors. Ann. Rheum. Dis. 68, 1870–1877 (2009).
Tak, P. P. et al. Inhibition of joint damage and improved clinical outcomes with rituximab plus methotrexate in early active rheumatoid arthritis: the IMAGE trial. Ann. Rheum. Dis. 70, 39–46 (2011).
Hein, G. et al. Influence of rituximab on markers of bone remodeling in patients with rheumatoid arthritis: a prospective open-label pilot study. Rheumatol Int. 31, 269–272 (2011).
van der Heijde, D. et al. Tofacitinib (CP-690,550), an oral Janus kinase inhibitor, in combination with methotrexate reduced the progression of structural damage in patients with rheumatoid arthritis: a 24-month Phase 3 study. [meeting abstract] Arthritis Rheum. 63 (Suppl. 10), 2592 (2011).
Weinblatt, M. E. et al. An oral spleen tyrosine kinase (Syk) inhibitor for rheumatoid arthritis. N. Engl. J. Med. 363, 1303–1312 (2010).
Massey, D. C., Bredin, F. & Parkes, M. Use of sirolimus (rapamycin) to treat refractory Crohn's disease. Gut 57, 1294–1296 (2008).
Cejka, D. et al. Mammalian target of rapamycin signaling is crucial for joint destruction in experimental arthritis and is activated in osteoclasts from patients with rheumatoid arthritis. Arthritis Rheum. 62, 2294–2302 (2010).
Westenfeld, R. et al. Impact of sirolimus, tacrolimus and mycophenolate mofetil on osteoclastogenesis —implications for post-transplantation bone disease. Nephrol. Dial. Transplant. 12, 4115–4123 (2011).
Dobashi, Y., Watanabe, Y., Miwa, C., Suzuki, S. & Koyama, S. Mammalian target of rapamycin: a central node of complex signaling cascades. Int. J. Clin. Exp. Pathol. 4, 476–495 (2011).
Markman, B., Dienstmann, R. & Tabernero, J. Targeting the PI3K/Akt/mTOR pathway — beyond rapalogs. Oncotarget 1, 530–543 (2010).
Kim, J. E. & Chen, J. Regulation of peroxisome proliferator-activated receptor-γ activity by mammalian target of rapamycin and amino acids in adipogenesis. Diabetes 53, 2748–2756 (2004).
Wan, Y. PPARγ in bone homeostasis. Trends Endocrinol. Metab. 21, 722–728 (2010).
Glantschnig, H., Fisher, J. E., Wesolowski, G., Rodan, G. A. & Reszka, A. A. M-CSF, TNFα and RANK ligand promote osteoclast survival by signaling through mTOR/S6 kinase. Cell Death. Differ. 10, 1165–1177 (2003).
Garrett, I. R. et al. Selective inhibitors of the osteoblast proteasome stimulate bone formation in vivo and in vitro. J. Clin. Invest. 111, 1771–1782 (2003).
Chen, D., Frezza, M., Schmitt, S., Kanwar, J. & Dou, P. Bortezomib as the first proteasome inhibitor anticancer drug: current status and future perspectives. Curr. Cancer Drug Targets 11, 239–253 (2011).
von Metzler, I. et al. Bortezomib inhibits human osteoclastogenesis. Leukemia 21, 2025–2034 (2007).
Zavrski, I. et al. Proteasome inhibitors abrogate osteoclast differentiation and osteoclast function. Biochem. Biophys. Res. Commun. 333, 200–205 (2005).
Hongming, H. & Jian, H. Bortezomib inhibits maturation and function of osteoclasts from PBMCs of patients with multiple myeloma by downregulating TRAF6. Leuk. Res. 33, 115–122 (2009).
Schmidt, N. et al. Targeting the proteasome: partial inhibition of the proteasome by bortezomib or deletion of the immunosubunit LMP7 attenuates experimental colitis. Gut 59, 896–906 (2010).
Lee, S. W., Kim, J. H., Park, Y. B. & Lee, S. K. Bortezomib attenuates murine collagen-induced arthritis. Ann. Rheum. Dis. 68, 1761–1767 (2009).
Yannaki, E. et al. The proteasome inhibitor bortezomib drastically affects inflammation and bone disease in adjuvant-induced arthritis in rats. Arthritis Rheum. 62, 3277–3288 (2010).
Russell, R. G. Bisphosphonates: the first 40 years. Bone 49, 2–19 (2011).
Lin, C. L., Moniz, C. & Chow, J. W. Treatment with fluoride or bisphosphonates prevents bone loss associated with colitis in the rat. Calcif. Tissue Int. 67, 373–377 (2000).
Eggelmeijer, F. et al. Increased bone mass with pamidronate treatment in rheumatoid arthritis. Results of a three-year randomized, double-blind trial. Arthritis Rheum. 39, 396–402 (1996).
Kwak, H. B. et al. Risedronate directly inhibits osteoclast differentiation and inflammatory bone loss. Biol. Pharm. Bull. 32, 1193–1198 (2009).
Watts, N. B. & Diab, D. L. Long-term use of bisphosphonates in osteoporosis. J. Clin. Endocrinol. Metab. 95, 1555–1565 (2010).
McClung, M. R. et al. Denosumab in postmenopausal women with low bone mineral density. N. Engl. J. Med. 354, 821–831 (2006).
Cummings, S. R. et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N. Engl. J. Med. 361, 756–765 (2009).
Cohen, S. B. et al. Denosumab treatment effects on structural damage, bone mineral density, and bone turnover in rheumatoid arthritis: A twelve-month, multicenter, randomized, double-blind, placebo-controlled, Phase II clinical trial. Arthritis Rheum. 58, 1299–1309 (2008). The results from this trial provide proof of concept that selectively targeting osteoclasts prevents local inflammatory bone loss but does not affect inflammation or cartilage damage.
Dore, R. K. et al. Effects of denosumab on bone mineral density and bone turnover in patients with rheumatoid arthritis receiving concurrent glucocorticoids or bisphosphonates. Ann. Rheum. Dis. 69, 872–875 (2010).
Adler, R. A. & Gill, R. S. Clinical utility of denosumab for treatment of bone loss in men and women. Clin. Interv. Aging 6, 119–124 (2011).
Saad, F. et al. Incidence, risk factors, and outcomes of osteonecrosis of the jaw: integrated analysis from three blinded active-controlled Phase III trials in cancer patients with bone metastases. Ann. Oncol. 10 Oct 2011 (doi:10.1093/annonc/mdr435).
Takahata, M., Awad, H. A., O'Keefe, R. J., Bukata, S. V. & Schwarz, E. M. Endogenous tissue engineering: PTH therapy for skeletal repair. Cell Tissue Res. 31 May 2011 (doi:10.1007/s00441-011-1188-4).
Vincent, C. et al. Pro-inflammatory cytokines TNF-related weak inducer of apoptosis (TWEAK) and TNFα induce the mitogen-activated protein kinase (MAPK)-dependent expression of sclerostin in human osteoblasts. J. Bone Miner. Res. 24, 1434–1449 (2009).
Heiland, G. R. et al. Neutralisation of Dkk-1 protects from systemic bone loss during inflammation and reduces sclerostin expression. Ann. Rheum. Dis. 69, 2152–2159 (2010). This study reveals the efficacy of blocking DKK1 on inflammatory bone loss in an experimental model.
Betts, A. M. et al. The application of target information and preclinical pharmacokinetic/pharmacodynamic modeling in predicting clinical doses of a Dickkopf-1 antibody for osteoporosis. J. Pharmacol. Exp. Ther. 333, 2–13 (2010).
Ominsky, M. S. et al. Inhibition of sclerostin by monoclonal antibody enhances bone healing and improves bone density and strength of nonfractured bones. J. Bone Miner. Res. 26, 1012–1021 (2011).
Tian, X. et al. Treatment with a sclerostin antibody increases cancellous bone formation and bone mass regardless of marrow composition in adult female rats. Bone 47, 529–533 (2010).
Li, X. et al. Sclerostin antibody treatment increases bone formation, bone mass, and bone strength in a rat model of postmenopausal osteoporosis. J. Bone Miner. Res. 24, 578–588 (2009).
Rickels, M. R., Zhang, X., Mumm, S. & Whyte, M. P. Oropharyngeal skeletal disease accompanying high bone mass and novel LRP5 mutation. J. Bone Miner. Res. 20, 878–885 (2005).
Zeckey, C., Hildebrand, F., Frink, M. & Krettek, C. Heterotopic ossifications following implant surgery — epidemiology, therapeutical approaches and current concepts. Semin. Immunopathol. 33, 273–286 (2011).
Boraiah, S., Paul, O., Hawkes, D., Wickham, M. & Lorich, D. G. Complications of recombinant human BMP-2 for treating complex tibial plateau fractures: a preliminary report. Clin. Orthop. Relat. Res. 467, 3257–3262 (2009).
Balemans, W., Cleiren, E., Siebers, U., Horst, J. & Van Hul, W. A generalized skeletal hyperostosis in two siblings caused by a novel mutation in the SOST gene. Bone 36, 943–947 (2005).
Hoff, M. et al. Adalimumab reduces hand bone loss in rheumatoid arthritis independent of clinical response: subanalysis of the PREMIER study. BMC Musculoskelet. Disord. 12, 54 (2011).
Eklund, S. A. & Burt, B. A. Risk factors for total tooth loss in the United States; longitudinal analysis of national data. J. Public Health Dent. 54, 5–14 (1994).
Gladman, D. D. et al. Consensus on a core set of domains for psoriatic arthritis. J. Rheumatol. 34, 1167–1170 (2007).
van der Heijde, D. et al. How to report radiographic data in randomized clinical trials in rheumatoid arthritis: guidelines from a roundtable discussion. Arthritis Rheum. 47, 215–218 (2002).
Smolen, J. S., Aletaha, D., Koeller, M., Weisman, M. & Emery, P. New therapies for the treatment of rheumatoid arthritis. Lancet 370, 1861–1874 (2007).
Smolen, J. S. et al. Evidence of radiographic benefit of infliximab plus methotrexate in rheumatoid arthritis patients who had no clinical improvement: a detailed subanalysis of the ATTRACT trial. Arthritis Rheum. 52, 1020–1030 (2005).
Smolen, J. S. et al. The need for prognosticators in rheumatoid arthritis. Biological and clinical markers — where are we now? Arthritis Res. Ther. 10, 208 (2008).
Smolen, J. S., Martinez-Avila, J. C. & Aletaha, D. Tocilizumab inhibits progression of joint damage in rheumatoid arthritis irrespective of its antiinflammatory effects: disassociation of the link between inflammation and destruction. Ann. Rheum. Dis. 25 Nov 2011 (doi:10.1136/annrheumdis-2011-200395).
Mattes, J., Yang, M. & Foster, P. S. Regulation of microRNA by antagomirs: a new class of pharmacological antagonists for the specific regulation of gene function? Am. J. Respir. Cell. Mol. Biol. 36, 8–12 (2007).
Sims, N. A. et al. Targeting osteoclasts with zoledronic acid prevents bone destruction in collagen-induced arthritis. Arthritis Rheum. 50, 2338–2346 (2004).
Herrak, P. et al. Zoledronic acid protects against local and systemic bone loss in tumor necrosis factor-mediated arthritis. Arthritis Rheum. 50, 2327–2337 (2004).
Jarrett, S. J. et al. Preliminary evidence for a structural benefit of the new bisphosphonate zoledronic acid in early rheumatoid arthritis. Arthritis Rheum. 54, 1410–1414 (2006).
Schett, G. et al. Osteoprotegerin protects against generalized bone loss in tumor necrosis factor-transgenic mice. Arthritis Rheum. 48, 2042–2051 (2003).
Schett, G. et al. Additive bone-protective effects of anabolic treatment when used in conjunction with RANKL and tumor necrosis factor inhibition in two rat arthritis models. Arthritis Rheum. 52, 1604–1611 (2005).
Aletaha, D., Funovits, J. & Smolen, J. S. Physical disability in rheumatoid arthritis is associated with cartilage damage rather than bone destruction. Ann. Rheum. Dis. 70, 733–739 (2011).
van der Heijde, D. et al. Expert agreement confirms that negative changes in hand and foot radiographs are a surrogate for repair in patients with rheumatoid arthritis. Arthritis Res. Ther. 9, R62 (2007).
Machold, K. P. et al. Very recent onset arthritis — clinical, laboratory and radiological findings during the first year of disease. J. Rheumatol. 29, 2278–2287 (2002).
Plant, M. J., Jones, P. W., Saklatvala, J., Ollier, W. E. & Dawes, P. T. Patterns of radiological progression in rheumatoid arthritis: results of an 8 year prospective study. J. Rheumatol. 25, 417–426 (1998).
Van der Heijde, D. M. Joint erosions and patients with early rheumatoid arthritis. Br. J. Rheumatol. 34,(Suppl. 2), 74–78 (1995).
Redlich, K. et al. Tumor necrosis factor α-mediated joint destruction is inhibited by targeting osteoclasts with osteoprotegerin. Arthritis Rheum. 46, 785–792 (2002).
Muller-Ladner, U. et al. Synovial fibroblasts of patients with rheumatoid arthritis attach to and invade normal human cartilage when engrafted into SCID mice. Am. J. Pathol. 149, 1607–1615 (1996).
Colucci, S. et al. Lymphocytes and synovial fluid fibroblasts support osteoclastogenesis through RANKL, TNFα, and IL-7 in an in vitro model derived from human psoriatic arthritis. J. Pathol. 212, 47–55 (2007).
Redlich, K. et al. Osteoclasts are essential for TNF-α-mediated joint destruction. J. Clin. Invest. 110, 1419–1427 (2002).
Partsch, G. et al. Highly increased levels of tumor necrosis factor-α and other proinflammatory cytokines in psoriatic arthritis synovial fluid. J. Rheumatol. 24, 518–523 (1997).
Gortz, B. et al. Arthritis induces lymphocytic bone marrow inflammation and endosteal bone formation. J. Bone Miner. Res. 19, 990–998 (2004).
Jimenez-Boj, E. et al. Bone erosions and bone marrow edema as defined by magnetic resonance imaging reflect true bone marrow inflammation in rheumatoid arthritis. Arthritis Rheum. 56, 1118–1124 (2007).
Arnett, F. C. et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 31, 315–324 (1988).
Shimizu, S., Shiozawa, S., Shiozawa, K., Imura, S. & Fujita, T. Quantitative histologic studies on the pathogenesis of periarticular osteoporosis in rheumatoid arthritis. Arthritis Rheum. 28, 25–31 (1985).
Mansson, B., Gulfe, A., Geborek, P., Heinegard, D. & Saxne, T. Release of cartilage and bone macromolecules into synovial fluid: differences between psoriatic arthritis and rheumatoid arthritis. Ann. Rheum. Dis. 60, 27–31 (2001).
Furie, R. et al. A Phase III, randomized, placebo-controlled study of belimumab, a monoclonal antibody that inhibits B lymphocyte stimulator, in patients with systemic lupus erythematosus. Arthritis Rheum. 63, 3918–3930 (2011).
Hsu, B., Sheng, S., Smolen, J. & Weinblatt, M. Results from a 2-part, proof-of-concept, dose-ranging, randomized, double-blind, placebo-controlled, Phase 2 study of sirukumab, a human anti-interleukin-6 monoclonal antibody, in active rheumatoid arthritis patients despite methotrexate therapy. [meeting abstract] Arthritis Rheum. 63 (Suppl. 10), 2631 (2011).
Griffiths, C. E. et al. Comparison of ustekinumab and etanercept for moderate-to-severe psoriasis. N. Engl. J. Med. 362, 118–128 (2010).
Taylor, P. C. et al. Ofatumumab, a fully human anti-CD20 monoclonal antibody, in biological-naive, rheumatoid arthritis patients with an inadequate response to methotrexate: a randomised, double-blind, placebo-controlled clinical trial. Ann. Rheum. Dis. 70, 2119–2125 (2011).
van Vollenhoven, R. F., Kinnman, N., Vincent, E., Wax, S. & Bathon, J. Atacicept in patients with rheumatoid arthritis and an inadequate response to methotrexate: results of a Phase II, randomized, placebo-controlled trial. Arthritis Rheum. 63, 1782–1792 (2011).
Hartung, H. P. & Kieseier, B. C. Atacicept: targeting B cells in multiple sclerosis. Ther. Adv. Neurol. Disord. 3, 205–216 (2010).
Fulciniti, M. et al. Anti-DKK1 mAb (BHQ880) as a potential therapeutic agent for multiple myeloma. Blood 114, 371–379 (2009).
Cusick, T. et al. Odanacatib treatment increases hip bone mass and cortical thickness by preserving endocortical bone formation and stimulating periosteal bone formation in the ovariectomized adult rhesus monkey. J Bone Miner. Res. 23 Nov 2011 (doi:10.1002/jbmr.1477).
Eisman, J. A. et al. Odanacatib in the treatment of postmenopausal women with low bone mineral density: three-year continued therapy and resolution of effect. J. Bone Miner. Res. 26, 242–251 (2011).
Acknowledgements
The authors would like to thank B. Niederreiter for expert support.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Josef Smolen has received grant support and/or speaking or consulting fees from Abbott, Amgen, Bristol-Myers Squibb, GlaxoSmithKline, Janssen, Lilly, Merck Sharp & Dohme, Pfizer, Roche, Sandoz and UCB Pharma, and has been a principal investigator in clinical trials at Abbott, Janssen, Pfizer, Roche, Sandoz and UCB Pharma.
Kurt Redlich has received grant support and/or speaking or consulting fees from Abbott, Bristol-Myers Squibb, Amgen, GlaxoSmithKline, Janssen, Merck Sharp & Dohme, Pfizer, Roche and UCB Pharma.
Related links
Glossary
- Receptor activator of NF-κB ligand
-
(RANKL). The ligand for receptor activator of NF-κB (RANK). RANKL is expressed on osteoblasts and other mesenchymal cells, as well as on non-mesenchymal cells such as T- and B lymphocytes. It is also secreted as a soluble molecule. Furthermore, mesenchymal cells can produce osteoprotegerin, which is a decoy receptor of RANKL that prevents the binding of RANKL to RANK. A monoclonal antibody against RANKL, denosumab, is licensed for the treatment of osteoporosis.
- Glycoprotein 130
-
(gp130). A transmembrane protein forming the signalling subunit of the interleukin-6 (IL-6) receptor family. When IL-6 or other IL-6 family members bind to their specific receptors (α-chain), they can interact with the gp130 protein (β-chain). Subsequently, this triplet dimerizes to form a hexamer. The gp130 co-receptor activates signal transduction via Janus kinases.
- Sclerosteosis
-
A rare, hereditary bone disease in which bones grow abnormally to produce a high bone mass phenotype; it is due to mutations in the gene encoding sclerostin, which is produced primarily by osteocytes and inhibits osteoblast function.
- Receptor activator of NF-κB
-
(RANK). A transmembrane protein belonging to the tumour necrosis factor receptor family and expressed on the surface of cells of haematopoietic origin. RANK is the pivotal cytokine receptor for osteoclastogenesis and leads to the activation of the transcription factor activator protein 1, which involves mainly heterodimers of the JUN and FOS families of proteins.
- MicroRNAs
-
(miRNAs). Short ribonucleic acid molecules that comprise about 22 nucleotides; they bind to complementary sequences of mRNA molecules, leading to translational repression or degradation of the mRNA. Cellular functions can be inhibited or activated by miRNAs.
- T helper 17
-
(TH17). A recently recognized, unique T helper (TH) cell subset that distinguishes itself from the traditional TH1 and TH2 subsets by producing interleukin-17 (IL-17). TH17 cells appear to have a pivotal role in the pathogenesis of chronic inflammatory and autoimmune diseases, and IL-17 is a strong inducer of RANKL and can thus activate osteoclastogenesis.
- Bone mineral density
-
(BMD). An estimate of the amount of mineral matter, expressed as g per cm2 of bone. Its extent is expressed as a t-score (the number of standard deviations above or below the mean of gender- and ethnicity-matched healthy adults of 30 years of age) and z-score (the same as above but matched for the same age). A t-score lower than −2.5 standard deviations reflects osteoporosis.
- Proteasome
-
A very large intracellular protein complex that enzymatically degrades damaged proteins but is also involved in the regulation of intracellular proteins. Proteasomes degrade bone morphogenetic proteins (BMPs), and proteasome inhibitors may therefore enhance the ability of BMPs to activate osteoblasts.
- Exostoses
-
Bony overgrowths on a bony surface, frequently seen as spurs. This abnormal or exaggerated bone formation can occur in the context of an inflammatory response that involves bone, and may cause pain or discomfort.
Rights and permissions
About this article
Cite this article
Redlich, K., Smolen, J. Inflammatory bone loss: pathogenesis and therapeutic intervention. Nat Rev Drug Discov 11, 234–250 (2012). https://doi.org/10.1038/nrd3669
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd3669
This article is cited by
-
Surface characteristics and in vitro biocompatibility of titanium preserved in a vitamin C-containing saline storage solution
Journal of Materials Science: Materials in Medicine (2024)
-
Rspo2 exacerbates rheumatoid arthritis by targeting aggressive phenotype of fibroblast-like synoviocytes and disrupting chondrocyte homeostasis via Wnt/β-catenin pathway
Arthritis Research & Therapy (2023)
-
Associations between blood inflammatory markers and bone mineral density and strength in the femoral neck: findings from the MIDUS II study
Scientific Reports (2023)
-
Is Helicobacter pylori infection associated with osteoporosis? a systematic review and meta-analysis
Journal of Bone and Mineral Metabolism (2023)
-
Inflammatory Processes Affecting Bone Health and Repair
Current Osteoporosis Reports (2023)