Abstract
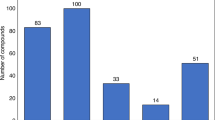
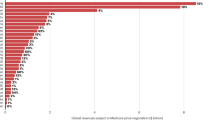
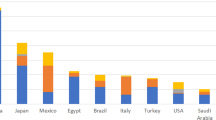
The development of 'follow on' or 'me too' drugs — generally defined as a drug with a similar chemical structure or the same mechanism of action as a drug that is already marketed — has attracted contrasting views. Some have argued that follow-on drugs often provide useful alternative or enhanced therapeutic options for particular patients or patient subpopulations, as well as introducing price competition. Others, however, consider that the development of such drugs is duplicative and that the resources needed would be better directed elsewhere. Implicit in some of this criticism is the notion that the development of me-too drugs is undertaken after a first-in-class drug has made it to market and proved commercially successful. In this Perspective, using analysis of development and patent filing histories of entrants to new drug classes in the past five decades, we provide new evidence that the development of multiple new drugs in a given class is better characterized as a race, rather than the imitation of successful products.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Angell, M. The Truth About the Drug Companies: How They Deceive Us and What to Do About It (Random House, New York, 2004).
Hollis, A. Comment on “The economics of follow-on drug research and development: trends in entry rates and the timing of development”. Pharmacoeconomics 3, 1187–1192 (2005).
Werthheimer, A., Levy, R. & O'Connor, T. Too many drugs? The clinical and economic value of incremental innovations. Res. Hum. Cap. Dev. 14, 77–118 (2001).
Lu, Z. J. & Comanor, W. S. Strategic pricing of new pharmaceuticals. Rev. Econ. Stat. 80, 108–118 (2008).
Towse, A. & Leighton, T. in Risk and Return in the Pharmaceutical Industry (eds Sussex, J. & Marchant, N.) 91–105 (Office of Health Economics, London, 1999).
DiMasi, J. A. Price trends for prescription pharmaceuticals: 1995–1999. US Department of Health and Human Services website [online], (2000).
Lee, T. H. “Me-too” products — friend or foe? N. Engl. J. Med. 350, 211–212 (2004).
DiMasi, J. A. & Pacquette, C. The economics of follow-on drug research and development: trends in the entry rates and timing of development. Pharmacoeconomics 22, 1–14 (2004).
Kemp, B. A. in Drug Development and Marketing (ed. Helms, R. B.) (American Enterprise Institute, Washington DC, 1975).
Kettler, H. E. Competition Through Innovation, Innovation Through Competition. (Office of Health Economics, London, 1998).
Newhouse, J. P., Seiguer, E. & Frank, R. G. Was Part D a giveaway to the pharmaceutical industry? Inquiry 44, 15–25 (2007).
DiMasi, J. A. New drug development in the United States from 1963 to 1999. Clin. Pharmacol. Ther. 69, 286–296 (2001).
Scherer, F. M. Time-cost tradeoffs in uncertain empirical research projects. Nav. Res. Logisitc. Q. 13, 71–82 (1966).
Comanor, W. S. & Scherer, F. M. Memorandum on the proposed acquisition by Pfizer of Wyeth. The American Antitrust Institute website [online], (2009).
Wastila, L. J., Ulcickas, M. E. & Lasagna, L. The World Health Organization's essential drug list. J. Clin. Res. Drug Dev. 3, 105–113 (1989).
Grabowski, H. G., Vernon, J. & DiMasi, J. A. Returns on research and development for 1990s new drug introductions. Pharmacoeconomics 20, 11–29 (2002).
DiMasi, J. A., Hansen, R. W. & Grabowski, H. G. The price of innovation: new estimates of drug development costs. J. Health Econ. 22, 151–185 (2003).
Acknowledgements
This project was supported, in part, by a grant from the Pharmaceutical Research and Manufacturers of America (PhRMA). The research, writing and analysis for this article was conducted independently by the authors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The Tufts Center for the Study of Drug Development is partially funded by unrestricted grants from pharmaceutical companies, contract research organizations, trade associations, niche providers and other corporate interests. The principal investigator, J.A.D., has consulted for the pharmaceutical industry and served as an expert witness in litigation involving pharmaceutical firms.
Supplementary information
Supplementary information Table S1
First-in-class* new drugs, second entrants‡ and time to competitive entry (PDF 410 kb)
Rights and permissions
About this article
Cite this article
DiMasi, J., Faden, L. Competitiveness in follow-on drug R&D: a race or imitation?. Nat Rev Drug Discov 10, 23–27 (2011). https://doi.org/10.1038/nrd3296
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd3296
This article is cited by
-
How the new European regulation on medical devices will affect innovation
Nature Biomedical Engineering (2020)
-
Competitive pricing within pharmaceutical classes: evidence on “follow-on” drugs in Germany 1993–2008
The European Journal of Health Economics (2015)