Key Points
-
Most drug discovery efforts aimed at type 2 diabetes target insulin action in peripheral tissues (muscle, fat and liver).
-
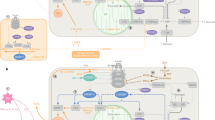
Recently, data have emerged that suggest the central nervous system (CNS) senses and integrates information from a host of neural, hormonal and nutrient signals. These signals are generated in response to the ingestion of food and regulate glucose output by the liver and glucose uptake by peripheral tissues.
-
CNS administration of insulin, leptin, glucagon-like peptide 1, glucose and long-chain fatty acids have all been shown to regulate liver glucose production, glucose uptake by skeletal muscle and/or insulin secretion.
-
AMP-activated protein kinase and mammalian target of rapamycin are important hypothalamic fuel sensors that regulate energy and possibly glucose homeostasis.
-
Before the CNS can be pharmacologically targeted for the treatment of diabetes, several obstacles must be overcome, including: differential peripheral and central actions on glucose and/or energy homeostasis; the blood–brain barrier limiting CNS exposure to circulating substances; and the fact that the key regions of the CNS that regulate homeostasis are deep in the midbrain and not easily studied in human subjects.
-
There are various strategies that have the potential to overcome these obstacles, including pharmacological developments that take advantage of the physical properties of the blood–brain barrier to allow specific delivery of drugs to the CNS, and targeting components of the fuel-sensing pathways downstream of hormones and nutrients that are specific to the CNS.
-
Given the overlap between the circuits that regulate energy and glucose homeostasis, it is reasonable to propose that part of the link between obesity and diabetes involves dysregulation of these common CNS circuits. Targeting these circuits presents new avenues for the development of more effective therapies that could produce both weight loss and improvements in glucose regulation.
Abstract
Research on the role of peripheral organs in the regulation of glucose homeostasis has led to the development of various monotherapies that aim to improve glucose uptake and insulin action in these organs for the treatment of type 2 diabetes. It is now clear that the central nervous system (CNS) also plays an important part in orchestrating appropriate glucose metabolism, with accumulating evidence linking dysregulated CNS circuits to the failure of normal glucoregulatory mechanisms. There is evidence that there is substantial overlap between the CNS circuits that regulate energy balance and those that regulate glucose levels, suggesting that their dysregulation could link obesity and diabetes. These findings present new targets for therapies that may be capable of both inducing weight loss and improving glucose regulation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Unger, R. H. The milieu interieur and the islets of Langerhans. Diabetologia 20, 1–11 (1981).
Bernard, C. Leçons de physiologie expérimentale appliqués à la médecine (Eds Baillère et Fils, Paris, 1854). Claude Bernard's thesis in 1854 introduced the idea that glucose levels were regulated by CNS actions at the liver (the source of glucose) and at the skeletal muscle (a consumer of glucose).
Sandoval, D. A. & Davis, S. N. Metabolic consequences of exercise-associated autonomic failure. Exerc. Sport Sci. Rev. 34, 72–76 (2006).
Biggers, D. W. et al. Role of brain in counterregulation of insulin-induced hypoglycemia in dogs. Diabetes 38, 7–16 (1989).
Lam, T. K., Gutierrez-Juarez, R., Pocai, A. & Rossetti, L. Regulation of blood glucose by hypothalamic pyruvate metabolism. Science 309, 943–947 (2005). This article showed that the CNS, specifically the mediobasal hypothalamus, responds to increased glucose availability to regulate hepatic glucose production.
Lam, T. K. et al. Hypothalamic sensing of circulating fatty acids is required for glucose homeostasis. Nature Med. 11, 320–327 (2005).
Lam, T. K., Schwartz, G. J. & Rossetti, L. Hypothalamic sensing of fatty acids. Nature Neurosci. 8, 579–584 (2005).
Pocai, A. et al. Hypothalamic KATP channels control hepatic glucose production. Nature 434, 1026–1031 (2005).
Pocai, A. et al. Central leptin acutely reverses diet-induced hepatic insulin resistance. Diabetes 54, 3182–3189 (2005).
Obici, S., Feng, Z., Arduini, A., Conti, R. & Rossetti, L. Inhibition of hypothalamic carnitine palmitoyltransferase-1 decreases food intake and glucose production. Nature Med. 9, 756–761 (2003).
Obici, S., Zhang, B. B., Karkanias, G. & Rossetti, L. Hypothalamic insulin signaling is required for inhibition of glucose production. Nature Med. 8, 1376–1382 (2002).
Obici, S., Feng, Z., Karkanias, G., Baskin, D. G. & Rossetti, L. Decreasing hypothalamic insulin receptors causes hyperphagia and insulin resistance in rats. Nature Neurosci. 5, 566–572 (2002). This article showed that the insulin receptors located in the arcuate nucleus of the hypothalamus were important for regulation of hepatic glucose production.
Obici, S. et al. Central administration of oleic acid inhibits glucose production and food intake. Diabetes 51, 271–275 (2002). In this work, the authors found that LCFAs serve as a signal to the CNS that leads to reduction of both hepatic glucose production and food intake.
Obici, S. et al. Central melanocortin receptors regulate insulin action. J. Clin. Invest. 108, 1079–1085 (2001).
Sandoval, D. A., Bagnol, D., Woods, S. C., D'Alessio, D. A. & Seeley, R. J. Arcuate glucagon-like peptide 1 receptors regulate glucose homeostasis but not food intake. Diabetes 57, 2046–2054 (2008). This article established that the incretin GLP1 acts on the CNS, specifically in the arcuate nucleus of the hypothalamus (as well as the periphery), to improve glucose homeostasis at the three major glucoregulatory organs (liver, skeletal muscle and pancreas).
Pocai, A. et al. Restoration of hypothalamic lipid sensing normalizes energy and glucose homeostasis in overfed rats. J. Clin. Invest. 116, 1081–1091 (2006).
Ono, H. et al. Activation of hypothalamic S6 kinase mediates diet-induced hepatic insulin resistance in rats. J. Clin. Invest. 118, 2959–2968 (2008).
Morrison, C. D., Morton, G. J., Niswender, K. D., Gelling, R. W. & Schwartz, M. W. Leptin inhibits hypothalamic Npy and Agrp gene expression via a mechanism that requires phosphatidylinositol 3-OH-kinase signaling. Am. J. Physiol. Endocrinol. Metab. 289, E1051–E1057 (2005).
Coppari, R. et al. The hypothalamic arcuate nucleus: a key site for mediating leptin's effects on glucose homeostasis and locomotor activity. Cell Metab. 1, 63–72 (2005).
Cherrington, A. D. Banting Lecture 1997. Control of glucose uptake and release by the liver in vivo. Diabetes 48, 1198–1214 (1999).
McIntyre, N., Holsworth, D. C. & Turner, D. S. New interpretation of oral glucose tolerance. Lancet 2, 20–21 (1964).
Kreymann, B., Ghatei, M. A., Williams, G. & Bloom, S. R. Glucagon-like peptide-1 7–36: a physiological incretin in man. Lancet 2, 1300–1303 (1987).
Goke, R., Fehmann, H. & Goke, B. Glucagon-like peptide-1 (7–36) amide is a new incretin/enterogastrone candidate. J. Clin. Invest. 135, 135–144 (1991).
Drucker, D. J. Glucagon and the glucagon-like peptides. Pancreas 5, 484–488 (1990).
Cohen, A. & Horton, E. S. Progress in the treatment of type 2 diabetes: new pharmacologic approaches to improve glycemic control. Curr. Med. Res. Opin. 23, 905–917 (2007).
Williams-Herman, D. et al. Efficacy and safety of initial combination therapy with sitagliptin and metformin in patients with type 2 diabetes: a 54-week study. Curr. Med. Res. Opin. 25, 569–583 (2009).
Williams, B. et al. Gastric emptying, glucose responses, and insulin secretion after a liquid test meal: effects of exogenous glucagon-like peptide 1 (GLP-1)-(7–36) amide in type 2 diabetic patients. J. Clin. Endocrinol. Metab. 81, 327–332 (1996).
Schirra, J. et al. Endogenous glucagon-like peptide 1 controls endocrine pancreatic secretion and antro-pyloro-duodenal motility in humans. Gut 55, 243–251 (2006).
von Mering, J. & Minkowski, O. Diabetes mellitus nach pankreas extirpation Arch. Exp. Pathol. Pharmacol. 26, 371 (1889).
Banting, F. G. & Best, C. H. Pancreatic extracts. 1922. J. Lab. Clin. Med. 115, 254–272 (1990).
Unger, R. H. The Banting Memorial Lecture 1975. Diabetes and the alpha cell. Diabetes 25, 136–151 (1976).
Bernard, C. Recherches sur une nouvelle fonction du foie. Thèse presentée a la Faculté des Sciences de Paris. (L. Martinet, Paris, 1853)
Fukushima, M., Tokunaga, K., Lupien, J., Kemnitz, J. W. & Bray, G. A. Dynamic and static phases of obesity following lesions in PVN and VMH. Am. J. Physiol. 253, R523–R529 (1987).
Berthoud, H.-R. & Jeanrenaud, B. Acute hyperinsulinemia and its reversal by vagotomy after lesions of the ventromedial hypothalamus in anesthetized rats. Endocrinology 105, 146–151 (1979).
Inoue, S., Bray, G. A. & Mullen, Y. S. Transplantation of pancreatic β-cells prevents development of hypothalamic obesity in rats. Am. J. Physiol. 235, E266–E271 (1978).
Wallis, M. G., Appleby, G. J., Youd, J. M., Clark, M. G. & Penschow, J. D. Reduced glycogen phosphorylase activity in denervated hindlimb muscles of rat is related to muscle atrophy and fibre type. Life Sci. 64, 221–228 (1999).
Bertelli, D. F. et al. Reversal of denervation-induced insulin resistance by SHIP2 protein synthesis blockade. Am. J. Physiol. Endocrinol. Metab. 284, 679–687 (2003).
Shimazu, T. & Amakawa, A. Regulation of glycogen metabolism in liver by the autonomic nervous system. II. Neural control of glycogen biosynthesis. Biochim. Biophys. Acta 165, 335–348 (1968).
Seeley, R. J. & Woods, S. C. Monitoring of stored and available fuel by the CNS: implications for obesity. Nature Rev. Neurosci. 4, 901–909 (2003).
Schwartz, M. W., Woods, S. C., Porte, D. J., Seeley, R. J. & Baskin, D. G. Central nervous system control of food intake. Nature 404, 661–671 (2000).
Haque, M. S. et al. Role of the sympathetic nervous system and insulin in enhancing glucose uptake in peripheral tissues after intrahypothalamic injection of leptin in rats. Diabetes 48, 1706–1712 (1999).
Minokoshi, Y., Okano, Y. & Shimazu, T. Regulatory mechanism of the ventromedial hypothalamus in enhancing glucose uptake in skeletal muscle. Brain Res. 649, 343–347 (1994).
Schwartz, M. W. et al. Kinetics and specificity of insulin uptake from plasma into cerebrospinal fluid. Am. J. Physiol. 259, 378–383 (1990).
Edgerton, D. S. et al. Insulin's direct effects on the liver dominate the control of hepatic glucose production. J. Clin. Invest. 116, 521–527 (2006).
Konner, A. C. et al. Insulin action in AgRP-expressing neurons is required for suppression of hepatic glucose production. Cell Metab. 5, 438–449 (2007).
Benoit, S. C. et al. The catabolic action of insulin in the brain is mediated by melanocortins. J. Neurosci. 22, 9048–9052 (2002).
Garcia-Echeverria, C. & Sellers, W. R. Drug discovery approaches targeting the PI3K/Akt pathway in cancer. Oncogene 27, 5511–5526 (2008).
Taniguchi, C. M. et al. The p85α regulatory subunit of phosphoinositide 3-kinase potentiates c-Jun N-terminal kinase-mediated insulin resistance. Mol. Cell. Biol. 27, 2830–2840 (2007).
Niswender, K. D. et al. Insulin activation of phosphatidylinositol 3-kinase in the hypothalamic arcuate nucleus: a key mediator of insulin-induced anorexia. Diabetes 52, 227–231 (2003).
Minokoshi, Y. et al. AMP-kinase regulates food intake by responding to hormonal and nutrient signals in the hypothalamus. Nature 428, 569–574 (2004). This article established that AMPK is important in fuel sensing in the hypothalamus and is regulated by changes in leptin.
Banks, W. A. et al. Leptin enters the brain by a saturable system independent of insulin. Peptides 17, 305–311 (1996).
Liu, L. et al. Intracerebroventricular leptin regulates hepatic but not peripheral glucose fluxes. J. Biol. Chem. 273, 31160–31167 (1998).
Gutierrez-Juarez, R., Obici, S. & Rossetti, L. Melanocortin-independent effects of leptin on hepatic glucose fluxes. J. Biol. Chem. 279, 49704–49715 (2004).
Morton, G. J. et al. Leptin regulates insulin sensitivity via phosphatidylinositol-3-OH kinase signaling in mediobasal hypothalamic neurons. Cell Metab. 2, 411–420 (2005).
Buettner, C. et al. Critical role of STAT3 in leptin's metabolic actions. Cell Metab. 4, 49–60 (2006).
Bates, S. H. et al. STAT3 signalling is required for leptin regulation of energy balance but not reproduction. Nature 421, 856–859 (2003).
Plum, L. et al. Enhanced PIP3 signaling in POMC neurons causes KATP channel activation and leads to diet-sensitive obesity. J. Clin. Invest. 116, 1886–1901 (2006).
Cota, D. et al. Hypothalamic mTOR signaling regulates food intake. Science 312, 927–930 (2006). This article established that hypothalamic mTOR also acts as a fuel sensor in the hypothalamus and is regulated by changes in leucine and leptin.
Dennis, P. B. et al. Mammalian TOR: a homeostatic ATP sensor. Science 294, 1102–1105 (2001).
Andersson, U. et al. AMP-activated protein kinase plays a role in the control of food intake. J. Biol. Chem. 279, 12005–12008 (2004).
Considine, R. V. et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N. Engl. J. Med. 334, 292–295 (1996).
Tulipano, G., Vergoni, A. V., Soldi, D., Muller, E. E. & Cocchi, D. Characterization of the resistance to the anorectic and endocrine effects of leptin in obesity-prone and obesity-resistant rats fed a high-fat diet. J. Endocrinol. 183, 289–298 (2004).
Ozcan, L. et al. Endoplasmic reticulum stress plays a central role in development of leptin resistance. Cell Metab. 9, 35–51 (2009).
Zhang, X. et al. Hypothalamic IKKβ/NF-κB and ER stress link overnutrition to energy imbalance and obesity. Cell 135, 61–73 (2008).
Merchenthaler, I., Lane, M. & Shughrue, P. Distribution of pre-pro-glucagon and glucagon-like peptide-1 receptor messenger RNAs in the rat central nervous system. J. Comp. Neurol. 403, 261–280 (1999).
Perrin, C., Knauf, C. & Burcelin, R. Intracerebroventricular infusion of glucose, insulin, and the adenosine monophosphate-activated kinase activator, 5-aminoimidazole-4-carboxamide-1-β-D-ribofuranoside, controls muscle glycogen synthesis. Endocrinology 145, 4025–4033 (2004).
Villanueva-Penacarrillo, M. L. et al. Glucagon-like peptide-1 binding to rat hepatic membranes. J. Endocrinol. 146, 183–189 (1995).
Wheeler, M. B. et al. Functional expression of the rat glucagon-like peptide-I receptor, evidence for coupling to both adenylyl cyclase and phospholipase-C. Endocrinology 133, 57–62 (1993).
Bullock, B. P., Heller, R. S. & Habener, J. F. Tissue distribution of messenger ribonucleic acid encoding the rat glucagon-like peptide-1 receptor. Endocrinology 137, 2968–2978 (1996).
Dunphy, J. L., Taylor, R. G. & Fuller, P. J. Tissue distribution of rat glucagon receptor and GLP-1 receptor gene expression. Mol. Cell. Endocrinol. 141, 179–186 (1998).
Prigeon, R. L., Quddusi, S., Paty, B. & D'Alessio, D. A. Suppression of glucose production by GLP-1 independent of islet hormones: a novel extrapancreatic effect. Am. J. Physiol. Endocrinol. Metab. 285, 701–707 (2003).
Ionut, V., Hucking, K., Liberty, I. F. & Bergman, R. N. Synergistic effect of portal glucose and glucagon-like peptide-1 to lower systemic glucose and stimulate counter-regulatory hormones. Diabetologia 48, 967–975 (2005).
Dardevet, D. et al. Insulin secretion-independent effects of GLP-1 on canine liver glucose metabolism do not involve portal vein GLP-1 receptors. Am. J. Physiol. Gastrointest. Liver Physiol. 289, 806–814 (2005).
Vahl, T. P. et al. Glucagon-like peptide-1 (GLP-1) receptors expressed on nerve terminals in the portal vein mediate the effects of endogenous GLP-1 on glucose tolerance in rats. Endocrinology 148, 4965–4973 (2007).
Nakagawa, A. et al. Receptor gene expression of glucagon-like peptide-1, but not glucose-dependent insulinotropic polypeptide, in rat nodose ganglion cells. Auton. Neurosci. 110, 36–43 (2004).
Knauf, C. et al. Brain glucagon-like peptide-1 increases insulin secretion and muscle insulin resistance to favor hepatic glycogen storage. J. Clin. Invest. 115, 3554–3563 (2005).
Ayala, J. E. et al. The glucagon-like peptide-1 receptor regulates endogenous glucose production and muscle glucose uptake independent of its incretin action. Endocrinology 150, 1155–1164 (2008).
Woltman, T. & Reidelberger, R. Effects of duodenal and distal ileal infusions of glucose and oleic acid on meal patterns in rats. Am. J. Physiol. 269, R7–R14 (1995).
Zhou, S. Y., Lu, Y. X. & Owyang, C. Gastric relaxation induced by hyperglycemia is mediated by vagal afferent pathways in the rat. Am. J. Physiol. Gastrointest. Liver Physiol. 294, 1158–1164 (2008).
Kang, L. et al. Glucokinase is a critical regulator of ventromedial hypothalamic neuronal glucosensing. Diabetes 55, 412–420 (2006).
Levin, B. E., Dunn-Meynell, A. A. & Routh, V. H. Brain glucosensing and the KATP channel. Nature Neurosci. 4, 459–460 (2001).
Levin, B. E. Neuronal glucose sensing: still a physiological orphan? Cell. Metab. 6, 252–254 (2007).
Sokoloff, L. et al. The [14C]deoxyglucose method for the measurement of local cerebral glucose utilization: theory, procedure, and normal values in the conscious and anesthetized albino rat. J. Neurochem. 28, 897–916 (1977).
Clegg, D. J., Wortman, M. D., Benoit, S. C., McOsker, C. C. & Seeley, R. J. Comparison of central and peripheral administration of C75 on food intake, body weight, and conditioned taste aversion. Diabetes 51, 3196–3201 (2002).
Wortman, M. D., Clegg, D. J., D'Alessio, D., Woods, S. C. & Seeley, R. J. C75 inhibits food intake by increasing CNS glucose metabolism. Nature Med. 9, 483–485 (2003).
Loftus, T. M. et al. Reduced food intake and body weight in mice treated with fatty acid synthase inhibitors. Science 288, 2299–2300 (2000).
Hu, Z., Cha, S. H., van Haasteren, G., Wang, J. & Lane, M. D. Effect of centrally administered C75, a fatty acid synthase inhibitor, on ghrelin secretion and its downstream effects. Proc. Natl Acad. Sci. USA 102, 3972–3977 (2005).
Proulx, K., Cota, D., Woods, S. C. & Seeley, R. J. Fatty acid synthase inhibitors modulate energy balance via mammalian target of rapamycin complex 1 signaling in the central nervous system. Diabetes 57, 3231–3238 (2008).
Shimokawa, T., Kumar, M. V. & Lane, M. D. Effect of a fatty acid synthase inhibitor on food intake and expression of hypothalamic neuropeptides. Proc. Natl Acad. Sci. USA 99, 66–71 (2002).
He, W., Lam, T. K., Obici, S. & Rossetti, L. Molecular disruption of hypothalamic nutrient sensing induces obesity. Nature Neurosci. 9, 227–233 (2006).
Wolfgang, M. J. & Lane, M. D. The role of hypothalamic malonyl-CoA in energy homeostasis. J. Biol. Chem. 281, 37265–37269 (2006). The data presented in this article provided support for the hypothesis that hypothalamic malonyl CoA is an intermediary in the control of energy homeostasis.
Ronnett, G. V., Kim, E. K., Landree, L. E. & Tu, Y. Fatty acid metabolism as a target for obesity treatment. Physiol. Behav. 85, 25–35 (2005).
Cruciani-Guglielmacci, C. et al. Beta oxidation in the brain is required for the effects of non-esterified fatty acids on glucose-induced insulin secretion in rats. Diabetologia 47, 2032–2038 (2004).
Benani, A. et al. Role for mitochondrial reactive oxygen species in brain lipid sensing: redox regulation of food intake. Diabetes 56, 152–160 (2007).
Greenberg, D., Smith, G. P. & Gibbs, J. Intraduodenal infusions of fats elicit satiety in sham-feeding rats. Am. J. Physiol. 259, R110–R118 (1990).
Matzinger, D. et al. The role of long chain fatty acids in regulating food intake and cholecystokinin release in humans. Gut 46, 688–693 (2000).
Wang, P. Y. et al. Upper intestinal lipids trigger a gut–brain–liver axis to regulate glucose production. Nature 452, 1012–1016 (2008).
Hardie, D. G., Corton, J., Ching, Y. P., Davies, S. P. & Hawley, S. Regulation of lipid metabolism by the AMP-activated protein kinase. Biochem. Soc. Trans. 25, 1229–1231 (1997).
Thomas, G. The S6 kinase signaling pathway in the control of development and growth. Biol. Res. 35, 305–313 (2002).
Lee, K., Li, B., Xi, X., Suh, Y. & Martin, R. J. Role of neuronal energy status in the regulation of adenosine 5′-monophosphate-activated protein kinase, orexigenic neuropeptides expression, and feeding behavior. Endocrinology 146, 3–10 (2005).
McCrimmon, R. J. et al. Activation of AMP-activated protein kinase within the ventromedial hypothalamus amplifies counterregulatory hormone responses in rats with defective counterregulation. Diabetes 55, 1755–1760 (2006).
Han, S. M. et al. Hypothalamic AMP-activated protein kinase mediates counter-regulatory responses to hypoglycaemia in rats. Diabetologia 48, 2170–2178 (2005).
Davis, M. R. & Shamoon, H. Counterregulatory adaptation to recurrent hypoglycemia in normal humans. J. Clin. Endocrinol. Metab. 73, 995–1001 (1991).
Heller, S. R. & Cryer, P. E. Reduced neuroendocrine and symptomatic responses to subsequent hypoglycemia after one episode of hypoglycemia in nondiabetic humans. Diabetes 40, 223–226 (1991).
Alquier, T., Kawashima, J., Tsuji, Y. & Kahn, B. B. Role of hypothalamic adenosine 5′-monophosphate-activated protein kinase in the impaired counterregulatory response induced by repetitive neuroglucopenia. Endocrinology 148, 1367–1375 (2007).
Lindsley, J. E. & Rutter, J. Nutrient sensing and metabolic decisions. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 139, 543–559 (2004).
Shaw, R. J. & Cantley, L. C. Ras, PI3K and mTOR signalling controls tumour cell growth. Nature 441, 424–430 (2006).
Wullschleger, S., Loewith, R. & Hall, M. N. TOR signaling in growth and metabolism. Cell 124, 471–484 (2006).
Cota, D., Matter, E. K., Woods, S. C. & Seeley, R. J. The role of hypothalamic mammalian target of rapamycin complex 1 signaling in diet-induced obesity. J. Neurosci. 28, 7202–7208 (2008).
Claret, M. et al. AMPK is essential for energy homeostasis regulation and glucose sensing by POMC and AgRP neurons. J. Clin. Invest. 117, 2325–2336 (2007).
Dunn-Meynell, A. A., Rawson, N. E. & Levin, B. E. Distribution and phenotype of neurons containing the ATP-sensitive K+ channel in rat brain. Brain Res. 814, 41–54 (1998).
Wang, R. et al. The regulation of glucose-excited neurons in the hypothalamic arcuate nucleus by glucose and feeding-relevant peptides. Diabetes 53, 1959–1965 (2004).
Mirshamsi, S. et al. Leptin and insulin stimulation of signalling pathways in arcuate nucleus neurones: PI3K dependent actin reorganization and KATP channel activation. BMC Neurosci. 5, 54 (2004).
Parton, L. E. et al. Glucose sensing by POMC neurons regulates glucose homeostasis and is impaired in obesity. Nature 449, 228–232 (2007).
Gyte, A., Pritchard, L. E., Jones, H. B., Brennand, J. C. & White, A. Reduced expression of the KATP channel subunit, Kir6.2, is associated with decreased expression of neuropeptide Y and agouti-related protein in the hypothalami of Zucker diabetic fatty rats. J. Neuroendocrinol. 19, 941–951 (2007).
Meguid, M. M., Fetissov, S. O., Blaha, V. & Yang, Z. J. Dopamine and serotonin VMN release is related to feeding status in obese and lean Zucker rats. Neuroreport 11, 2069–2072 (2000).
Lieberman, J. A. et al. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N. Engl. J. Med. 353, 1209–1223 (2005).
Ader, M. et al. Metabolic dysregulation with atypical antipsychotics occurs in the absence of underlying disease: a placebo-controlled study of olanzapine and risperidone in dogs. Diabetes 54, 862–871 (2005).
Houseknecht, K. L. et al. Acute effects of atypical antipsychotics on whole-body insulin resistance in rats: implications for adverse metabolic effects. Neuropsychopharmacology 32, 289–297 (2007).
Chintoh, A. F. et al. Insulin resistance and decreased glucose-stimulated insulin secretion after acute olanzapine administration. J. Clin. Psychopharmacol. 28, 494–499 (2008).
Cincotta, A. H., Tozzo, E. & Scislowski, P. W. D. Bromocriptine/SKF38393 treatment ameliorates obesity and associated metabolic dysfunctions in obese (ob/ob) mice. Life Sci. 61, 951 (1997).
Nonogaki, K., Strack, A. M., Dallman, M. F. & Tecott, L. H. Leptin-independent hyperphagia and type 2 diabetes in mice with a mutated serotonin 5-HT2C receptor gene. Nature Med. 4, 1152–1156 (1998).
Hakansson, M. L., Brown, H., Ghilardi, N., Skoda, R. C. & Meister, B. Leptin receptor immunoreactivity in chemically defined target neurons of the hypothalamus. J. Neurosci. 18, 559–572 (1998).
Horvath, T. L., Naftolin, F. & Leranth, C. Beta-endorphin innervation of dopamine neurons in the rat hypothalamus: a light and electron microscopic double immunostaining study. Endocrinology 131, 1547–1555 (1992).
Zhou, L. et al. Serotonin 2C receptor agonists improve type 2 diabetes via melanocortin-4 receptor signaling pathways. Cell. Metab. 6, 398–405 (2007).
Xu, Y. et al. 5-HT2CRs expressed by pro-opiomelanocortin neurons regulate energy homeostasis. Neuron 60, 582–589 (2008). This article demonstrated that expression of the serotonin type 2C receptor in proopiomelanocortin neurons is sufficient to mediate the effects of serotonergic compounds on food intake and thus illustrated the importance of the serotonergic melanocortin circuitry in the long-term regulation of energy balance.
Garfield, A. S. & Heisler, L. K. Pharmacological targeting of the serotonergic system for the treatment of obesity. J. Physiol. 587, 49–60 (2008).
Woods, S. C., Lotter, E. C., McKay, L. D. & Porte, D. Jr. Chronic intracerebroventricular infusion of insulin reduces food intake and body weight of baboons. Nature 282, 503–505 (1979).
Air, E. L. et al. Small molecule insulin mimetics reduce food intake and body weight and prevent development of obesity. Nature Med. 8, 179–183 (2002).
Kahn, S. E. et al. Glycemic durability of rosiglitazone, metformin, or glyburide monotherapy. N. Engl. J. Med. 355, 2427–2443 (2006).
McConell, G. K. et al. Differential attenuation of AMPK activation during acute exercise following exercise training or AICAR treatment. J. Appl. Physiol. 105, 1422–1427 (2008).
Um, S. H. et al. Absence of S6K1 protects against age- and diet-induced obesity while enhancing insulin sensitivity. Nature 431, 200–205 (2004).
Khamzina, L., Veilleux, A., Bergeron, S. & Marette, A. Increased activation of the mammalian target of rapamycin pathway in liver and skeletal muscle of obese rats: possible involvement in obesity-linked insulin resistance. Endocrinology 146, 1473–1481 (2005).
Narkar, V. A. et al. AMPK and PPARδ agonists are exercise mimetics. Cell 134, 405–415 (2008).
Anderson, K. A. et al. Hypothalamic CAMKK2 contributes to the regulation of energy balance. Cell. Metab. 7, 377–388 (2008). The data presented in this article illustrated that CAMKK2 has a regulatory role in AMPK activation specifically in the hypothalamus and that inhibition of CAMKK2 could be a pharmacological target for weight loss.
Banks, W. A. The blood–brain barrier as a cause of obesity. Curr. Pharm. Des. 14, 1606–1614 (2008).
Banks, W. A., DiPalma, C. R. & Farrell, C. L. Impaired transport of leptin across the blood–brain barrier in obesity. Peptides 20, 1341–1345 (1999).
Israel, P. A. et al. Effect of diet-induced obesity and experimental hyperinsulinemia on insulin uptake into CSF of the rat. Brain Res. Bull. 30, 571–575 (1993).
Stein, L. J. et al. Reduced effect of experimental peripheral hyperinsulinemia to elevate cerebrospinal fluid insulin concentrations of obese Zucker rats. Endocrinology 121, 1611–1615 (1987).
Patel, M. M., Goyal, B. R., Bhadada, S. V., Bhatt, J. S. & Amin, A. F. Getting into the brain: approaches to enhance brain drug delivery. CNS Drugs 23, 35–58 (2009).
Bodor, N. Redox drug delivery systems for targeting drugs to the brain. Ann. NY Acad. Sci. 507, 289–306 (1987).
Brewster, M. E. et al. Effect of molecular manipulation on the estrogenic activity of a brain-targeting estradiol chemical delivery system. J. Med. Chem. 37, 4237–4244 (1994).
Kreuter, J. Nanoparticulate systems for brain delivery of drugs. Adv. Drug Deliv. Rev. 47, 65–81 (2001).
Giussani, C. et al. Local intracerebral delivery of endogenous inhibitors by osmotic minipumps effectively suppresses glioma growth in vivo. Cancer Res. 63, 2499–2505 (2003).
Gill, S. S. et al. Direct brain infusion of glial cell line-derived neurotrophic factor in Parkinson disease. Nature Med. 9, 589–595 (2003).
Rosenstock, J. et al. Two-year pulmonary safety and efficacy of inhaled human insulin (Exubera) in adult patients with type 2 diabetes. Diabetes Care 31, 1723–1728 (2008).
Tataranni, P. A. & DelParigi, A. Functional neuroimaging: a new generation of human brain studies in obesity research. Obes. Rev. 4, 229–238 (2003).
Woods, S. C., Schwartz, M. W., Baskin, D. G. & Seeley, R. J. Food intake and the regulation of body weight. Annu. Rev. Psychol. 51, 255–277 (2000).
Stumvoll, M. et al. Uptake and release of glucose by the human kidney. Postabsorptive rates and responses to epinephrine. J. Clin. Invest. 96, 2528–2533 (1995).
Troy, S. et al. Intestinal gluconeogenesis is a key factor for early metabolic changes after gastric bypass but not after gastric lap-band in mice. Cell. Metab. 8, 201–211 (2008).
Aftab Guy, D., Sandoval, D., Richardson, M. A., Tate, D. & Davis, S. N. Effects of glycemic control on target organ responses to epinephrine in type 1 diabetes. Am. J. Physiol. Endocrinol. Metab. 289, E258–E265 (2005).
Niswender, K. D. et al. Intracellular signalling: key enzyme in leptin-induced anorexia. Nature 413, 794–795 (2001).
Hill, J. W. et al. Acute effects of leptin require PI3K signaling in hypothalamic proopiomelanocortin neurons in mice. J. Clin. Invest. 118, 1796–1805 (2008).
Donahey, J. C., van Dijk, G., Woods, S. C. & Seeley, R. J. Intraventricular GLP-1 reduces short- but not long-term food intake or body weight in lean and obese rats. Brain Res. 779, 75–83 (1998).
Cota, D., Proulx, K. & Seeley, R. J. The role of CNS fuel sensing in energy and glucose regulation. Gastroenterology 132, 2158–2168 (2007).
Acknowledgements
The authors acknowledge grant support from the National Institutes of Health National Institute of Diabetes and Digestive and Kidney Diseases (DK075365 to D.A.S. and DK54080, DK54890 and DK56863 to R.J.S.) and the American Diabetes Association (support to S.O.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Randy J. Seeley is on the scientific advisory boards for eli Lilly and Company, ethicon endoSurgery, Johnson & Johnson and Zafgen, and has received research support from amylin and ethicon endoSurgery.
Related links
Glossary
- Postprandial
-
Relating to the time period immediately after a meal.
- Incretin
-
A hormone that is secreted from the gut in response to a meal and stimulates insulin secretion.
- Glucagon-like peptide 1
-
A hormone secreted from the L-cells of the distal intestine that acts as an incretin.
- Exenatide
-
A long-acting glucagon-like peptide 1 analogue that is used to treat type 2 diabetes. Its known effects include stimulation of insulin secretion and modest weight loss.
- Blood–brain barrier
-
An endothelial layer composed of tight junctions that limit the ability of substances to freely diffuse into the brain. The blood–brain barrier functions to protect the central nervous system from the blood.
- Gluconeogenesis
-
The metabolic process that takes place in the kidneys, liver and gut, whereby glucose is made from non-glucose precursors such as glycerol, lactate and amino acids.
- KATP channel
-
A potassium channel located on the cell membranes of many tissues, including the pancreas and the brain, that is opened or closed by changes in ATP levels.
- AMP-activated protein kinase
-
(AMPK). A cellular fuel sensor that is activated in response to low ATP (energy) levels. Activation of AMPK stimulates the metabolic breakdown of fuel to generate ATP and restore the energy levels of the cell. When AMPK is activated in the central nervous system (CNS), it is a signal of low energy status in the whole organism and the CNS responds by stimulating food intake to restore energy needs.
- Leptin
-
A hormone secreted by adipose tissue that is thought to serve as a signal of adiposity to the central nervous system.
- Saturable transport system
-
A system to transport a molecule across the plasma membrane involving a specific transmembrane carrier, for which the rate of uptake decreases as the concentration of the molecule in the plasma increases.
- Glycogenolysis
-
The process of breaking down stored glycogen to make glucose.
- Mammalian target of rapamycin
-
(mTOR). A cellular fuel sensor that is activated in response to high ATP levels. Activation of mTOR serves to stimulate pathways that are involved in protein biosynthesis. When mTOR is activated in the central nervous system (CNS), the CNS responds to this signal of nutrient excess in the whole organism by inhibiting food intake.
- Endoplasmic reticulum stress
-
Stress in the endoplasmic reticulum (where protein and sterol synthesis and protein folding take place) results when the influx of unfolded proteins exceeds the ability of the endoplasmic reticulum to fold the proteins.
- Hyperinsulinaemic euglycaemic clamp
-
The gold standard for assessing insulin sensitivity. It involves a primed continuous infusion of insulin and a variable rate of infusion of exogenous glucose that is determined by the glucose level at that time point, with the goal of maintaining glucose at basal levels.
- Glucose rate of appearance
-
The rate of glucose appearance in the plasma. Glucose enters the plasma primarily through the gastrointestinal tract and the liver. In the context of euglycaemic clamp conditions, glucose rate of appearance corresponds to liver glucose output.
- Sulphonylurea receptor
-
The regulatory subunit of the KATP channel. This receptor is targeted clinically to increase insulin secretion in patients with type 2 diabetes.
Rights and permissions
About this article
Cite this article
Sandoval, D., Obici, S. & Seeley, R. Targeting the CNS to treat type 2 diabetes. Nat Rev Drug Discov 8, 386–398 (2009). https://doi.org/10.1038/nrd2874
Issue Date:
DOI: https://doi.org/10.1038/nrd2874
This article is cited by
-
Hepatic glycogen participates in the regulation of hypothalamic pAkt/Akt ratio in high-sugar/high-fat diet-induced obesity
Metabolic Brain Disease (2022)
-
Stimulation of the hepatoportal nerve plexus with focused ultrasound restores glucose homoeostasis in diabetic mice, rats and swine
Nature Biomedical Engineering (2022)
-
Comorbid brain disorders associated with diabetes: therapeutic potentials of prebiotics, probiotics and herbal drugs
Translational Medicine Communications (2019)
-
The blood–brain barrier as an endocrine tissue
Nature Reviews Endocrinology (2019)
-
Mechanisms of inflammatory responses and development of insulin resistance: how are they interlinked?
Journal of Biomedical Science (2016)