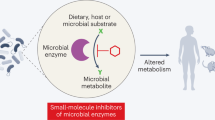
Abstract
The significant involvement of the gut microbiota in human health and disease suggests that manipulation of commensal microbial composition through combinations of antibiotics, probiotics and prebiotics could be a novel therapeutic approach. A systems perspective is needed to help understand the complex host–bacteria interactions and their association with pathophysiological phenotypes so that alterations in the composition of the gut microbiota in disease states can be reversed. In this article, we describe the therapeutic rationale and potential for targeting the gut microbiota, and discuss strategies and systems-oriented technologies for achieving this goal.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Nicholson, J. K., Holmes, E., Lindon, J. C. & Wilson, I. D. The challenges of modeling mammalian biocomplexity. Nature Biotech. 22, 1268–1274 (2004).
Gill, S. R. et al. Metagenomic analysis of the human distal gut microbiome. Science 312, 1355–1359 (2006).
Riesenfeld, C. S., Schloss, P. D. & Handelsman, J. Metagenomics: genomic analysis of microbial communities. Annu. Rev. Genet. 38, 525–552 (2004).
Dumas, M. E. et al. Metabolic profiling reveals a contribution of gut microbiota to fatty liver phenotype in insulin-resistant mice. Proc. Natl Acad. Sci. USA 103, 12511–12516 (2006).
Turnbaugh, P. J. et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444, 1027–1031 (2006).
Ley, R. E., Turnbaugh, P. J., Klein, S. & Gordon, J. I. Microbial ecology: human gut microbes associated with obesity. Nature 444, 1022–1023 (2006).
Brugman, S. et al. Antibiotic treatment partially protects against type 1 diabetes in the Bio-Breeding diabetes-prone rat. Is the gut flora involved in the development of type 1 diabetes? Diabetologia 49, 2105–2108 (2006).
Strober, W., Fuss, I. & Mannon, P. The fundamental basis of inflammatory bowel disease. J. Clin. Invest. 117, 514–521 (2007).
Chu, F. F. et al. Bacteria-induced intestinal cancer in mice with disrupted Gpx1 and Gpx2 genes. Cancer Res. 64, 962–968 (2004).
Guarner, F. & Malagelada, J. R. Gut flora in health and disease. Lancet 361, 512–519 (2003).
Xu, J. & Gordon, J. I. Honor thy symbionts. Proc. Natl Acad. Sci. USA 100, 10452–10459 (2003).
O'Hara, A. M. & Shanahan, F. The gut flora as a forgotten organ. EMBO Rep. 7, 688–693 (2006).
Nicholson, J. K. & Wilson, I. D. Understanding 'global' systems biology: metabonomics and the continuum of metabolism. Nature Rev. Drug Discov. 2, 668–676 (2003).
Grangette, C. et al. Enhanced antiinflammatory capacity of a Lactobacillus plantarum mutant synthesizing modified teichoic acids. Proc. Natl Acad. Sci. USA 102, 10321–10326 (2005).
Wakabayashi, C., Hasegawa, H., Murata, J. & Saiki, I. In vivo antimetastatic action of ginseng protopanaxadiol saponins is based on their intestinal bacterial metabolites after oral administration. Oncol. Res. 9, 411–417 (1997).
Ebbels, T. et al. Toxicity classification from metabonomic data using a density superposition approach: 'CLOUDS'. Anal. Chim. Acta 490, 109–122 (2003).
Backhed, F. et al. The gut microbiota as an environmental factor that regulates fat storage. Proc. Natl Acad. Sci. USA 101, 15718–15723 (2004).
Yamanaka, K., Ohba, H., Hasegawa, A., Sawamura, R. & Okada, S. Mutagenicity of dimethylated metabolites of inorganic arsenics. Chem. Pharm. Bull. (Tokyo) 37, 2753–2756 (1989).
Rizzo, R., Gulisano, M., Pavone, P., Fogliani, F. & Robertson, M. M. Increased antistreptococcal antibody titers and anti-basal ganglia antibodies in patients with Tourette syndrome: controlled cross-sectional study. J. Child. Neurol. 21, 747–753 (2006).
Mell, L. K., Davis, R. L. & Owens, D. Association between streptococcal infection and obsessive-compulsive disorder, Tourette's syndrome, and tic disorder. Pediatrics 116, 56–60 (2005).
Nicholson, J. K., Holmes, E. & Wilson, I. D. Gut microorganisms, mammalian metabolism and personalized health care. Nature Rev. Microbiol. 3, 431–438 (2005).
Arvola, T. et al. Prophylactic Lactobacillus GG reduces antibiotic-associated diarrhea in children with respiratory infections: a randomized study. Pediatrics 104, e64 (1999).
Mendall, M. A. & Kumar, D. Antibiotic use, childhood affluence and irritable bowel syndrome (IBS). Eur. J. Gastroenterol. Hepatol. 10, 59–62 (1998).
Wilcox, M. H. Gastrointestinal disorders and the critically ill. Clostridium difficile infection and pseudomembranous colitis. Best Pract. Res. Clin. Gastroenterol. 17, 475–493 (2003).
Jernberg, C., Lofmark, S., Edlund, C. & Jansson, J.K. Long-term ecological impacts of antibiotic administration on the human intestinal microbiota. ISME J. 1, 56–66 (2007).
Hadley, C. The infection connection. Helicobacter pylori is more than just the cause of gastric ulcers — it offers an unprecedented opportunity to study changes in human microecology and the nature of chronic disease. EMBO Rep. 7, 470–473 (2006).
Xu, J. et al. A genomic view of the human–Bacteroides thetaiotaomicron symbiosis. Science 299, 2074–2076 (2003).
Kelly, D. et al. Commensal anaerobic gut bacteria attenuate inflammation by regulating nuclear-cytoplasmic shuttling of PPAR-γ and RelA. Nature Immunol. 5, 104–112 (2004).
Samuel, B. S. et al. Genomic and metabolic adaptations of Methanobrevibacter smithii to the human gut. Proc. Natl Acad. Sci. USA 104, 10643–10648 (2007).
Tampakaki, A. P., Fadouloglou, V. E., Gazi, A. D., Panopoulos, N. J. & Kokkinidis, M. Conserved features of type III secretion. Cell. Microbiol. 6, 805–816 (2004).
Chan, F. K. et al. Eradication of Helicobacter pylori and risk of peptic ulcers in patients starting long-term treatment with non-steroidal anti-inflammatory drugs: a randomised trial. Lancet 359, 9–13 (2002).
Rachmilewitz, D. et al. Toll-like receptor 9 signaling mediates the anti-inflammatory effects of probiotics in murine experimental colitis. Gastroenterology 126, 520–528 (2004).
Rastall, R. A. et al. Modulation of the microbial ecology of the human colon by probiotics, prebiotics and synbiotics to enhance human health: an overview of enabling science and potential applications. FEMS Microbiol. Ecol. 52, 145–152 (2005).
Matsuzaki, T., Yamazaki, R., Hashimoto, S. & Yokokura, T. Antidiabetic effects of an oral administration of Lactobacillus casei in a non-insulin-dependent diabetes mellitus (NIDDM) model using KK-Ay mice. Endocr. J. 44, 357–365 (1997).
Matsuzaki, T. et al. Prevention of onset in an insulin-dependent diabetes mellitus model, NOD mice, by oral feeding of Lactobacillus casei. APMIS 105, 643–649 (1997).
Matsuzaki, T. et al. Effect of oral administration of Lactobacillus casei on alloxan-induced diabetes in mice. APMIS 105, 637–642 (1997).
Tabuchi, M. et al. Antidiabetic effect of Lactobacillus GG in streptozotocin-induced diabetic rats. Biosci. Biotechnol. Biochem. 67, 1421–1424 (2003).
Yadav, H., Jain, S. & Sinha, P. R. Antidiabetic effect of probiotic dahi containing Lactobacillus acidophilus and Lactobacillus casei in high fructose fed rats. Nutrition 23, 62–68 (2007).
Schlee, M. et al. Induction of human β-defensin 2 by the probiotic Escherichia coli Nissle 1917 is mediated through flagellin. Infect. Immun. 75, 2399–2407 (2007).
Gibson, G. R. & Roberfroid, M. B. Dietary modulation of the human colonic microbiota: introducing the concept of prebiotics. J. Nutr. 125, 1401–1412 (1995).
Chen, C. C. & Walker, W. A. Probiotics and prebiotics: role in clinical disease states. Adv. Pediatr. 52, 77–113 (2005).
Furrie, E. et al. Synbiotic therapy (Bifidobacterium longum/Synergy 1) initiates resolution of inflammation in patients with active ulcerative colitis: a randomised controlled pilot trial. Gut 54, 242–249 (2005).
Gunatilaka, A. A. Natural products from plant-associated microorganisms: distribution, structural diversity, bioactivity, and implications of their occurrence. J. Nat. Prod. 69, 509–526 (2006).
Dunlap, W. C. et al. Biomedicinals from the phytosymbionts of marine invertebrates: a molecular approach. Methods 42, 358–376 (2007).
Brindle, J. T. et al. Rapid and noninvasive diagnosis of the presence and severity of coronary heart disease using 1H-NMR-based metabonomics. Nature Med. 8, 1439–1444 (2002).
Qiu, Y. et al. Application of ethyl chloroformate derivatization for gas chromatography-mass spectrometry based metabonomic profiling. Anal. Chim. Acta 583, 277–283 (2007).
Wang, X. et al. Simultaneous determination of 17 ginsenosides in rat urine by ultra performance liquid chromatography-mass spectrometry with solid-phase extraction. Anal. Chim. Acta 594, 265–273 (2007).
Phipps, A. N., Stewart, J., Wright, B. & Wilson, I. D. Effect of diet on the urinary excretion of hippuric acid and other dietary-derived aromatics in rat. A complex interaction between diet, gut microflora and substrate specificity. Xenobiotica 28, 527–537 (1998).
Wang, Y. et al. Metabonomic investigations in mice infected with Schistosoma mansoni: an approach for biomarker identification. Proc. Natl Acad. Sci. USA 101, 12676–12681 (2004).
Clayton, T. A. et al. Pharmaco-metabonomic phenotyping and personalized drug treatment. Nature 440, 1073–1077 (2006).
Li, H. et al. Pharmacometabonomic phenotyping reveals different responses to xenobiotic intervention in rats. J. Proteome Res. 6, 1364–1370 (2007).
Williams, R. E., Eyton-Jones, H. W., Farnworth, M. J., Gallagher, R. & Provan, W. M. Effect of intestinal microflora on the urinary metabolic profile of rats: a 1H-nuclear magnetic resonance spectroscopy study. Xenobiotica 32, 783–794 (2002).
Handelsman, J. Metagenomics: application of genomics to uncultured microorganisms. Microbiol. Mol. Biol. Rev. 68, 669–685 (2004).
Eckburg, P. B. et al. Diversity of the human intestinal microbial flora. Science 308, 1635–1638 (2005).
Wilcks, A., van Hoek, A. H., Joosten, R. G., Jacobsen, B. B. & Aarts, H. J. Persistence of DNA studied in different ex vivo and in vivo rat models simulating the human gut situation. Food Chem. Toxicol. 42, 493–502 (2004).
Pang, X. et al. Inter-species transplantation of gut microbiota from human to pigs ISME J. 1, 156–162 (2007).
Malfertheiner, P. et al. Helicobacter pylori eradication and gastric ulcer healing — comparison of three pantoprazole-based triple therapies. Aliment. Pharmacol. Ther. 17, 1125–1135 (2003).
Surawicz, C. M. et al. Prevention of antibiotic-associated diarrhea by Saccharomyces boulardii: a prospective study. Gastroenterology 96, 981–988 (1989).
Surawicz, C. M. et al. The search for a better treatment for recurrent Clostridium difficile disease: use of high-dose vancomycin combined with Saccharomyces boulardii. Clin. Infect. Dis. 31, 1012–1017 (2000).
Cummings, J. H., Macfarlane, G. T. & Macfarlane, S. Intestinal bacteria and ulcerative colitis. Curr. Issues Intest. Microbiol. 4, 9–20 (2003).
Subramanian, S., Campbell, B. J. & Rhodes, J. M. Bacteria in the pathogenesis of inflammatory bowel disease. Curr. Opin. Infect. Dis. 19, 475–484 (2006).
Andoh, A. & Fujiyama, Y. Therapeutic approaches targeting intestinal microflora in inflammatory bowel disease. World J. Gastroenterol. 12, 4452–4460 (2006).
O'Mahony, L. et al. Probiotic impact on microbial flora, inflammation and tumour development in IL-10 knockout mice. Aliment. Pharmacol. Ther. 15, 1219–1225 (2001).
Singh, J. et al. Bifidobacterium longum, a lactic acid-producing intestinal bacterium inhibits colon cancer and modulates the intermediate biomarkers of colon carcinogenesis. Carcinogenesis 18, 833–841 (1997).
Dobbs, R. J. et al. Role of chronic infection and inflammation in the gastrointestinal tract in the etiology and pathogenesis of idiopathic parkinsonism. Part 1: eradication of Helicobacter in the cachexia of idiopathic parkinsonism. Helicobacter 10, 267–275 (2005).
Bjarnason, I. T. et al. Role of chronic infection and inflammation in the gastrointestinal tract in the etiology and pathogenesis of idiopathic parkinsonism. Part 2: response of facets of clinical idiopathic parkinsonism to Helicobacter pylori eradication. A randomized, double-blind, placebo-controlled efficacy study. Helicobacter 10, 276–287 (2005).
Weller, C. et al. Role of chronic infection and inflammation in the gastrointestinal tract in the etiology and pathogenesis of idiopathic parkinsonism. Part 3: predicted probability and gradients of severity of idiopathic parkinsonism based on H. pylori antibody profile. Helicobacter 10, 288–297 (2005).
Acknowledgements
This work was financially supported by the National Basic Research Program of China (2007CB914700) and the International Collaborative Project, Chinese Ministry of Science and Technology (2006DFA02700).
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
DATABASES
Entrez Genome Project
OMIM
FURTHER INFORMATION
Glossary
- Metabonomics
-
The quantitative measurement of the multivariate metabolic responses of multicellular systems to pathophysiological stimuli or genetic modification. It is an approach to understanding the global metabolic regulation of an organism and its commensal and symbiotic partners.
- Metagenomics
-
A culture-independent and sequence-based analysis of the collective microbial genomes contained in an environmental sample including soil, the oral cavity, faeces and aquatic habitats. The term is derived from the statistical concept of meta-analysis (the process of statistically combining separate analyses) and genomics (the comprehensive analysis of an organism's genetic material).
- Microbiome
-
The entire set of microbial species living in the human body.
- Pachinko model
-
A model for the illustration of xenobiotic metabolism in the host, in which the outcome of a xenobiotic is the result of a series of probabilistic interactions with endogenous and sym-endogenous elements.
- Pharmaco-metabonomics
-
The prediction of the outcome of a drug or xenobiotic intervention in an individual based on a mathematical model of pre-intervention metabolite signatures.
- Prebiotics
-
A non-digestible food ingredient that beneficially affects the host by selectively stimulating the growth and/or activity of one or a limited number of bacteria that can improve host health.
- Probiotics
-
Live microorganisms that, when administered in adequate amounts, confer a health benefit to the host.
- Type III secretion system
-
A complex protein secretion and delivery system used by many animal- and plant-interacting bacteria, especially Gram-negative pathogenic bacteria, for delivering effector proteins into eukaryotic cells.
Rights and permissions
About this article
Cite this article
Jia, W., Li, H., Zhao, L. et al. Gut microbiota: a potential new territory for drug targeting. Nat Rev Drug Discov 7, 123–129 (2008). https://doi.org/10.1038/nrd2505
Issue Date:
DOI: https://doi.org/10.1038/nrd2505
This article is cited by
-
Effects of prebiotic supplement on gut microbiota, drug bioavailability, and adverse effects in patients with colorectal cancer at different primary tumor locations receiving chemotherapy: study protocol for a randomized clinical trial
Trials (2023)
-
Bacterial metabolite butyrate in modulating sorafenib-targeted microRNAs to curtail its resistance in hepatocellular carcinoma
Journal of Cancer Research and Clinical Oncology (2023)
-
Machine learning for data integration in human gut microbiome
Microbial Cell Factories (2022)
-
Multi-compound and drug-combination pharmacokinetic research on Chinese herbal medicines
Acta Pharmacologica Sinica (2022)
-
Novel probiotic treatment of autism spectrum disorder associated social behavioral symptoms in two rodent models
Scientific Reports (2022)