Key Points
-
The high attrition rate of compounds entering clinical testing as potential anticancer drugs indicates a need for better methods to predict efficacy before testing in humans.
-
The poor correlation between therapeutic activity of compounds tested in xenograft mouse models and their efficacy in humans does not necessarily mean that more faithful genetically engineered mouse models (GEMMs) will be of limited use in drug development.
-
Indeed, a major untapped solution could lie in the use of refined GEMMs of human cancer that are capable of facilitating the identification of the right target, the right drug and the right patients.
-
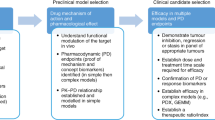
The attributes of a 'well-designed' GEMM include moderate penetrance and short latency of single tumours, engineered alleles that are representative of the human disease, and simplicity in colony management and technical use.
-
There are several important applications for GEMMs in anticancer drug development, including target validation, assessment of tumour response, investigation of pharmacodynamic markers of drug action, modelling resistance and understanding toxicity, which are discussed in this article.
Abstract
Deficiencies in the standard preclinical methods for evaluating potential anticancer drugs,such as xenograft mouse models, have been highlighted as a key obstacle in the translation of the major advances in basic cancer research into meaningful clinical benefits. In this article, we discuss the established uses and limitations of xenograft mouse models for cancer drug development, and then describe the opportunities and challenges in the application of novel genetically engineered mouse models that more faithfully mimic the genetic and biological evolution of human cancers. Greater use of such models in target validation, assessment of tumour response, investigation of pharmacodynamic markers of drug action, modelling resistance and understanding toxicity has the potential to markedly improve the success of cancer drug development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Leaf, C. Why we're losing the war on cancer: and how to win it. Fortune 149, 77–92 (2004).
National Cancer Institute. Cancer Trends Progress Report [online] (2005).
Weiss, A. J. et al. Phase II study of 5-azacytidine in solid tumors. Cancer Treat. Rep. 61, 55–58 (1977).
Lomen, P. L., Khilanani, P. & Kessel, D. Phase I study using combination of hydroxyurea and 5-azacytidine (NSC-102816). Neoplasma 27, 101–106 (1980).
Lomen, P. L., Baker, L. H., Neil, G. L. & Samson, M. K. Phase I study of 5-azacytidine (NSC-102816) using 24-hour continuous infusion for 5 days. Cancer Chemother. Rep. 59, 1123–1126 (1975).
Silverman, L. R. et al. Randomized controlled trial of azacitidine in patients with the myelodysplastic syndrome: a study of the cancer and leukemia group B. J. Clin. Oncol. 20, 2429–2440 (2002).
Kola, I. & Landis, J. Can the pharmaceutical industry reduce attrition rates? Nature Rev. Drug Discov. 3, 711–715 (2004).
Horrobin, D. F. Are large clinical trials in rapidly lethal diseases usually unethical? Lancet 361, 695–697 (2003).
Decoster, G., Stein, G. & Holdener, E. E. Responses and toxic deaths in phase I clinical trials. Ann. Oncol. 1, 175–181 (1990).
Roberts, T. G. et al. Trends in the risks and benefits to patients with cancer participating in phase 1 clinical trials. JAMA 292, 2130–2140 (2004). References 9 and 10 illustrate a significant problem with Phase I trials (namely a ∼4% response rate) carried out using empirically discovered would-be chemotherapeutics. We believe that using better preclinical models would improve this low level of clinical benefit, allowing for more efficient and ethical drug discovery.
Voskoglou-Nomikos, T., Pater, J. L. & Seymour, L. Clinical predictive value of the in vitro cell line, human xenograft, and mouse allograft preclinical cancer models. Clin. Cancer Res. 9, 4227–4239 (2003).
Johnson, J. I. et al. Relationships between drug activity in NCI preclinical in vitro and in vivo models and early clinical trials. Br. J. Cancer 84, 1424–1431 (2001).
Reya, T., Morrison, S. J., Clarke, M. F. & Weissman, I. L. Stem cells, cancer, and cancer stem cells. Nature 414, 105–111 (2001).
Mellinghoff, I. K. et al. Molecular determinants of the response of glioblastomas to EGFR kinase inhibitors. N. Engl. J. Med. 353, 2012–2024 (2005).
Sarraf, P. et al. Differentiation and reversal of malignant changes in colon cancer through PPARγ. Nature Med. 4, 1046–1052 (1998).
Kulke, M. H. et al. A phase II study of troglitazone, an activator of the PPARγ receptor, in patients with chemotherapy-resistant metastatic colorectal cancer. Cancer J. 8, 395–399 (2002).
Saez, E. et al. Activators of the nuclear receptor PPARγ enhance colon polyp formation. Nature Med. 4, 1058–1061 (1998).
Boehm, T., Folkman, J., Browder, T. & O'Reilly, M. S. Antiangiogenic therapy of experimental cancer does not induce acquired drug resistance. Nature 390, 404–407 (1997).
O'Reilly, M. S., Holmgren, L., Chen, C. & Folkman, J. Angiostatin induces and sustains dormancy of human primary tumors in mice. Nature Med. 2, 689–692 (1996).
Holmgren, L., O'Reilly, M. S. & Folkman, J. Dormancy of micrometastases: balanced proliferation and apoptosis in the presence of angiogenesis suppression. Nature Med. 1, 149–153 (1995).
Hansma, A. H. et al. Recombinant human endostatin administered as a 28-day continuous intravenous infusion, followed by daily subcutaneous injections: a phase I and pharmacokinetic study in patients with advanced cancer. Ann. Oncol. 16, 1695–1701 (2005).
Twombly, R. First clinical trials of endostatin yield lukewarm results. J. Natl Cancer Inst. 94, 1520–1521 (2002).
Soff, G. A. et al. In vivo generation of angiostatin isoforms by administration of a plasminogen activator and a free sulfhydryl donor: a phase I study of an angiostatic cocktail of tissue plasminogen activator and mesna. Clin. Cancer Res. 11, 6218–6225 (2005).
Thomas, J. P. et al. Phase I pharmacokinetic and pharmacodynamic study of recombinant human endostatin in patients with advanced solid tumors. J. Clin. Oncol. 21, 223–231 (2003).
Davis, D. W. et al. Quantitative analysis of biomarkers defines an optimal biological dose for recombinant human endostatin in primary human tumors. Clin. Cancer Res. 10, 33–42 (2004).
Sausville, E. A. & Burger, A. M. Contributions of human tumor xenografts to anticancer drug development. Cancer Res. 66, 3351–3354 (2006).
Thompson, J., Stewart, C. F. & Houghton, P. J. Animal models for studying the action of topoisomerase I targeted drugs. Biochim. Biophys. Acta 1400, 301–319 (1998).
Peterson, J. K. & Houghton, P. J. Integrating pharmacology and in vivo cancer models in preclinical and clinical drug development. Eur. J. Cancer 40, 837–844 (2004).
Okami, K. et al. Analysis of PTEN/MMAC1 alterations in aerodigestive tract tumors. Cancer Res. 58, 509–511 (1998).
Meyer, W. H. et al. Development and characterization of pediatric osteosarcoma xenografts. Cancer Res. 50, 2781–2785 (1990).
Furman, W. L. et al. Direct translation of a protracted irinotecan schedule from a xenograft model to a phase I trial in children. J. Clin. Oncol. 17, 1815–1824 (1999).
Sun, B., Chen, M., Hawks, C. L., Pereira-Smith, O. M. & Hornsby, P. J. The minimal set of genetic alterations required for conversion of primary human fibroblasts to cancer cells in the subrenal capsule assay. Neoplasia 7, 585–593 (2005).
Sun, B., Chen, M., Hawks, C., Hornsby, P. J. & Wang, X. Tumorigenic study on hepatocytes coexpressing SV40 with Ras. Mol. Carcinog. 45, 213–219 (2006).
Bachoo, R. M. et al. Epidermal growth factor receptor and Ink4a/Arf. Convergent mechanisms governing terminal differentiation and transformation along the neural stem cell to astrocyte axis. Cancer Cell 1, 269–277 (2002).
Yang, J. et al. Twist, a master regulator of morphogenesis, plays an essential role in tumor metastasis. Cell 117, 927–939 (2004).
Tassone, P. et al. Combination therapy with interleukin-6 receptor superantagonist Sant7 and dexamethasone induces antitumor effects in a novel SCID-hu in vivo model of human multiple myeloma. Clin. Cancer Res. 11, 4251–4258 (2005).
Mitsiades, C. S. et al. Fluorescence imaging of multiple myeloma cells in a clinically relevant SCID/NOD in vivo model: biologic and clinical implications. Cancer Res. 63, 6689–6696 (2003).
Bardeesy, N. et al. Both p16Ink4a and the p19Arf–p53 pathway constrain progression of pancreatic adenocarcinoma in the mouse. Proc. Natl Acad. Sci. USA 103, 5947–5952 (2006).
Sharpless, N. E., Kannan, K., Xu, J., Bosenberg, M. W. & Chin, L. Both products of the mouse Ink4a/Arf locus suppress melanoma formation in vivo. Oncogene 22, 5055–5059 (2003).
Chin, L. et al. Cooperative effects of INK4a and ras in melanoma susceptibility in vivo. Genes Dev. 11, 2822–2834 (1997).
Bardeesy, N. et al. Dual inactivation of RB and p53 pathways in RAS-induced melanomas. Mol. Cell. Biol. 21, 2144–2153 (2001).
Castresana, J. S. et al. Lack of allelic deletion and point mutation as mechanisms of p53 activation in human malignant melanoma. Int. J. Cancer 55, 562–565 (1993).
Albino, A. P. et al. Mutation and expression of the p53 gene in human malignant melanoma. Melanoma Res. 4, 35–45 (1994).
Lubbe, J., Reichel, M., Burg, G. & Kleihues, P. Absence of p53 gene mutations in cutaneous melanoma. J. Invest. Dermatol. 102, 819–821 (1994).
Rhim, K. J. et al. Aberrant expression of p53 gene product in malignant melanoma. J. Korean Med. Sci. 9, 376–381 (1994).
Kamb, A. et al. A cell cycle regulator potentially involved in genesis of many tumor types. Science 264, 436–440 (1994).
Hussussian, C. J. et al. Germline p16 mutations in familial melanoma. Nature Genet. 8, 15–21 (1994).
Koh, J., Enders, G. H., Dynlacht, B. D. & Harlow, E. Tumour-derived p16 alleles encoding proteins defective in cell-cycle inhibition. Nature 375, 506–510 (1995).
Flores, J. F. et al. Loss of the p16INK4a and p15INK4b genes, as well as neighboring 9p21 markers, in sporadic melanoma. Cancer Res. 56, 5023–5032 (1996).
Weissleder, R. Scaling down imaging: molecular mapping of cancer in mice. Nature Rev. Cancer 2, 11–18 (2002).
Graves, E. E., Weissleder, R. & Ntziachristos, V. Fluorescence molecular imaging of small animal tumor models. Curr. Mol. Med. 4, 419–430 (2004).
Sotillo, R. et al. Cooperation between Cdk4 and p27kip1 in tumor development: a preclinical model to evaluate cell cycle inhibitors with therapeutic activity. Cancer Res. 65, 3846–3852 (2005).
Stewart, T. A., Pattengale, P. K. & Leder, P. Spontaneous mammary adenocarcinomas in transgenic mice that carry and express MTV/myc fusion genes. Cell 38, 627–637 (1984).
Quaife, C. J., Pinkert, C. A., Ornitz, D. M., Palmiter, R. D. & Brinster, R. L. Pancreatic neoplasia induced by ras expression in acinar cells of transgenic mice. Cell 48, 1023–1034 (1987).
Brinster, R. L. et al. Transgenic mice harboring SV40 T-antigen genes develop characteristic brain tumors. Cell 37, 367–379 (1984).
Hanahan, D. Heritable formation of pancreatic β-cell tumours in transgenic mice expressing recombinant insulin/simian virus 40 oncogenes. Nature 315, 115–122 (1985).
Adams, J. M. & Cory, S. Transgenic models of tumor development. Science 254, 1161–1167 (1991).
Jacks, T. et al. Effects of an Rb mutation in the mouse. Nature 359, 295–300 (1992).
Jacks, T. et al. Tumor spectrum analysis in p53-mutant mice. Curr. Biol. 4, 1–7 (1994).
Donehower, L. A. et al. Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 356, 215–221 (1992).
Van Dyke, T. & Jacks, T. Cancer modeling in the modern era: progress and challenges. Cell 108, 135–144 (2002).
Chin, L. et al. Essential role for oncogenic Ras in tumour maintenance. Nature 400, 468–472 (1999).
Felsher, D. W. & Bishop, J. M. Reversible tumorigenesis by MYC in hematopoietic lineages. Mol. Cell 4, 199–207 (1999). References 62 and 63 are classic papers that articulate and prove the concept of the importance of oncogenes, such as MYC and RAS , in tumour maintenance as opposed to tumour progression. Since this research it has become well recognized that establishing the role of a particular gene in tumour maintenance is a crucial step in target validation.
D'Cruz, C. M. et al. c-MYC induces mammary tumorigenesis by means of a preferred pathway involving spontaneous Kras2 mutations. Nature Med. 7, 235–239 (2001).
Politi, K. et al. Lung adenocarcinomas induced in mice by mutant EGF receptors found in human lung cancers respond to a tyrosine kinase inhibitor or to down-regulation of the receptors. Genes Dev. 20, 1496–1510 (2006).
Fisher, G. H. et al. Induction and apoptotic regression of lung adenocarcinomas by regulation of a K-Ras transgene in the presence and absence of tumor suppressor genes. Genes Dev. 15, 3249–3262 (2001).
Ji, H. et al. The impact of human EGFR kinase domain mutations on lung tumorigenesis and in vivo sensitivity to EGFR-targeted therapies. Cancer Cell 9, 485–495 (2006).
Boxer, R. B., Jang, J. W., Sintasath, L. & Chodosh, L. A. Lack of sustained regression of c-MYC-induced mammary adenocarcinomas following brief or prolonged MYC inactivation. Cancer Cell 6, 577–586 (2004).
Shachaf, C. M. et al. MYC inactivation uncovers pluripotent differentiation and tumour dormancy in hepatocellular cancer. Nature 431, 1112–1117 (2004).
Berthet, C., Aleem, E., Coppola, V., Tessarollo, L. & Kaldis, P. Cdk2 knockout mice are viable. Curr. Biol. 13, 1775–1785 (2003).
Ortega, S. et al. Cyclin-dependent kinase 2 is essential for meiosis but not for mitotic cell division in mice. Nature Genet. 35, 25–31 (2003).
Martin, A. et al. Cdk2 is dispensable for cell cycle inhibition and tumor suppression mediated by p27Kip1 and p21Cip1. Cancer Cell 7, 591–598 (2005).
Aleem, E., Kiyokawa, H. & Kaldis, P. Cdc2–cyclin E complexes regulate the G1/S phase transition. Nature Cell Biol. 7, 831–836 (2005).
Yu, Q. et al. Requirement for CDK4 kinase function in breast cancer. Cancer Cell 9, 23–32 (2006).
Landis, M. W., Pawlyk, B. S., Li, T., Sicinski, P. & Hinds, P. W. Cyclin D1-dependent kinase activity in murine development and mammary tumorigenesis. Cancer Cell 9, 13–22 (2006).
Toogood, P. L. et al. Discovery of a potent and selective inhibitor of cyclin-dependent kinase 4/6. J. Med. Chem. 48, 2388–2406 (2005).
Lowe, S. W., Ruley, H. E., Jacks, T. & Housman, D. E. p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 74, 957–967 (1993). One of the first GEMM studies to have major implications for the development of human chemotherapeutics. This work showed that the response to cytotoxics in certain malignant cells requires p53 function, establishing a key mechanism of chemotherapy resistance.
Omer, C. A. et al. Mouse mammary tumor virus-Ki-rasB transgenic mice develop mammary carcinomas that can be growth-inhibited by a farnesyl:protein transferase inhibitor. Cancer Res. 60, 2680–2688 (2000). An important early GEMM study showing that FTI efficacy does not correlate with k-Ras mutation. The importance of this work was not fully appreciated until after a large number of human clinical trials were completed in which FTIs failed to demonstrate activity in tumours harbouring mutant k-RAS.
Bergers, G., Song, S., Meyer-Morse, N., Bergsland, E. & Hanahan, D. Benefits of targeting both pericytes and endothelial cells in the tumor vasculature with kinase inhibitors. J. Clin. Invest. 111, 1287–1295 (2003).
Pietras, K. & Hanahan, D. A multitargeted, metronomic, and maximum-tolerated dose 'chemo-switch' regimen is antiangiogenic, producing objective responses and survival benefit in a mouse model of cancer. J. Clin. Oncol. 23, 939–952 (2005).
Yilmaz, O. H. et al. Pten dependence distinguishes haematopoietic stem cells from leukaemia-initiating cells. Nature 441, 475–482 (2006).
Wendel, H. G. et al. Survival signalling by Akt and eIF4E in oncogenesis and cancer therapy. Nature 428, 332–337 (2004).
Bergers, G., Javaherian, K., Lo, K. M., Folkman, J. & Hanahan, D. Effects of angiogenesis inhibitors on multistage carcinogenesis in mice. Science 284, 808–812 (1999).
Casanovas, O., Hicklin, D. J., Bergers, G. & Hanahan, D. Drug resistance by evasion of antiangiogenic targeting of VEGF signaling in late-stage pancreatic islet tumors. Cancer Cell 8, 299–309 (2005). A clever study showing how GEMMs can be used to tackle a difficult problem in drug discovery: how to combine and sequence novel anticancer agents.
Zhang, Z. et al. Farnesyltransferase inhibitors are potent lung cancer chemopreventive agents in A/J mice with a dominant-negative p53 and/or heterozygous deletion of Ink4a/Arf. Oncogene 22, 6257–6265 (2003).
Boolbol, S. K. et al. Cyclooxygenase-2 overexpression and tumor formation are blocked by sulindac in a murine model of familial adenomatous polyposis. Cancer Res. 56, 2556–2560 (1996).
Opitz, O. G. et al. A mouse model of human oral-esophageal cancer. J. Clin. Invest. 110, 761–769 (2002).
Jacoby, R. F., Seibert, K., Cole, C. E., Kelloff, G. & Lubet, R. A. The cyclooxygenase-2 inhibitor celecoxib is a potent preventive and therapeutic agent in the min mouse model of adenomatous polyposis. Cancer Res. 60, 5040–5044 (2000).
Laird, P. W. et al. Suppression of intestinal neoplasia by DNA hypomethylation. Cell 81, 197–205 (1995).
McCabe, M. T. et al. Inhibition of DNA methyltransferase activity prevents tumorigenesis in a mouse model of prostate cancer. Cancer Res. 66, 385–392 (2006).
Pao, W. et al. Acquired resistance of lung adenocarcinomas to gefitinib or erlotinib is associated with a second mutation in the EGFR kinase domain. PLoS Med. 2, e73 (2005).
Shah, N. P. et al. Multiple BCR-ABL kinase domain mutations confer polyclonal resistance to the tyrosine kinase inhibitor imatinib (STI571) in chronic phase and blast crisis chronic myeloid leukemia. Cancer Cell 2, 117–125 (2002).
Roumiantsev, S. et al. Clinical resistance to the kinase inhibitor STI-571 in chronic myeloid leukemia by mutation of Tyr-253 in the Abl kinase domain P-loop. Proc. Natl Acad. Sci. USA 99, 10700–10705 (2002).
Gorre, M. E. et al. Clinical resistance to STI-571 cancer therapy caused by BCR-ABL gene mutation or amplification. Science 293, 876–880 (2001).
Maggi, A. & Ciana, P. Reporter mice and drug discovery and development. Nature Rev. Drug Discov. 4, 249–255 (2005).
Crone, S. A. et al. ErbB2 is essential in the prevention of dilated cardiomyopathy. Nature Med. 8, 459–465 (2002). An excellent example of how a modern, tissue-specific GEMM can be used to understand unexpected toxicity of a novel agent (in this case, a HER2/ neu antibody).
Artandi, S. E. et al. Telomere dysfunction promotes non-reciprocal translocations and epithelial cancers in mice. Nature 406, 641–645 (2000).
Blaug, S., Chien, C. & Shuster, M. J. Managing innovation: university–industry partnerships and the licensing of the Harvard mouse. Nature Biotechnol. 22, 761–764 (2004).
Marshall, E. Intellectual property. DuPont ups ante on use of Harvard's OncoMouse. Science 296, 1212 (2002).
Maebius, S. B. & Wegner, H. C. Merck V. Integra: the impact of a broader 'safe harbor' exemption on nanobiotechnology. Nanotechnol. Law Business 2, 1–6 (2005).
Nickerson, C. Canada court blocks Harvard bid to patent research mouse. Boston Globe (Boston) A20 6 December (2002).
Check, E. Canada stops Harvard's oncomouse in its tracks. Nature 420, 593 (2002).
Jaffe, S. Ongoing battle over transgenic mice. The Scientist 18, 46 (2004).
Threadgill, D. W. et al. Targeted disruption of mouse EGF receptor: effect of genetic background on mutant phenotype. Science 269, 230–234 (1995).
Tsutsui, T. et al. Targeted disruption of CDK4 delays cell cycle entry with enhanced p27Kip1 activity. Mol. Cell. Biol. 19, 7011–7019 (1999).
Rane, S. G. et al. Loss of Cdk4 expression causes insulin-deficient diabetes and Cdk4 activation results in β-islet cell hyperplasia. Nature Genet. 22, 44–52 (1999).
Little, C. C. & Cloudman, A. M. The occurrence of a dominant spotting mutation in the house mouse. Proc. Natl Acad. Sci. USA 23, 535–537 (1937).
Acknowledgements
We thank P. Nisen, K.-K. Wong, K. Anderson, D. Hanahan, D. Frost, G. Gordon, A. Shoemaker and S. Mellis for stimulating discussions and critical reading of the manuscript. R. A. D. is an ACS Research Professor and an Ellison Medical Foundation Senior Scholar. This work was supported by grants from the Sidney Kimmel Foundation for Cancer Research (N.E.S.), the Ellison Medical Foundation (N.E.S. and R.A.D.) and the National Institutes of Health. R.A.D. is supported by the LeBow Fund to Cure Myeloma and the Robert A. and Renee E. Belfer Foundation Institute for Innovative Cancer Science.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
R.A.D. is a director, co-founder and scientific advisor of AVEO Pharmaceuticals, Inc., which develops and uses mouse models of human cancer, including GEMMs and xenotransplants; R.A.D. also serves on the cancer scientific advisory council of Abbott Pharmaceuticals. The views expressed by R.A.D. are his own and do not reflect those of the management of AVEO or Abbott Pharmaceuticals.
Related links
Glossary
- Xenograft model
-
Xenograft mouse models of cancer are created by injecting homogeneous human tumour cell lines into immunodeficient (for example, severe combined immunodeficiency) mice.
- RECIST Criteria
-
Response Evaluation Criteria in Solid Tumors are standardized, radiographic criteria for determining tumour response or progression in human clinical trials of cancer therapeutics.
- CRE recombinase
-
A phage enzyme that is used in murine genetic engineering. Expression of the enzyme causes selective excision of all genetic material between two LoxP sites. Murine strains can be engineered with LoxP sites flanking a gene of interest, stop codon and so on, and expression of CRE in these strains allows for tissue-specific and inducible changes in gene function.
- Tet-regulation
-
The ability to regulate gene expression by feeding engineered murine strains tetracycline analogues (for example, doxycycline). Mice are engineered to contain a transgene of interest that is either induced (tet-ON) or repressed (tet-OFF) in the presence of doxycycline. These strains allow for the study of tissue-specific and inducible changes in gene expression. An advantage of tet-regulation is that the gene of interest can be serially induced and repressed by withdrawing and adding doxycycline to the animal's drinking water.
- Oncogene addiction
-
The notion that cancer cells strictly require the activity of certain mutant oncogenes (for example, BCR–ABL in chronic myelogenous leukaemia), and therefore these oncogenes are required for tumour maintenance and are desirable therapeutic targets.
- Metronomic therapy
-
Continuous or frequent treatment with low doses of cancer therapeutics, often given in a schedule-dependent manner with other methods of therapy. The goal is to inhibit an important cancer-relevant process (for example, angiogenesis) with minimal toxicity.
Rights and permissions
About this article
Cite this article
Sharpless, N., DePinho, R. The mighty mouse: genetically engineered mouse models in cancer drug development. Nat Rev Drug Discov 5, 741–754 (2006). https://doi.org/10.1038/nrd2110
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd2110
This article is cited by
-
Promising preclinical patient-derived organoid (PDO) and xenograft (PDX) models in upper gastrointestinal cancers: progress and challenges
BMC Cancer (2023)
-
Evaluation of antioxidant, anti-inflammatory, anticancer activities and molecular docking of Moringa oleifera seed oil extract against experimental model of Ehrlich ascites carcinoma in Swiss female albino mice
BMC Complementary Medicine and Therapies (2023)
-
Factors impacting the efficacy of the in-situ vaccine with CpG and OX40 agonist
Cancer Immunology, Immunotherapy (2023)
-
Modelling acute myeloid leukemia (AML): What’s new? A transition from the classical to the modern
Drug Delivery and Translational Research (2023)
-
The development of in vitro organotypic 3D vulvar models to study tumor-stroma interaction and drug efficacy
Cellular Oncology (2023)