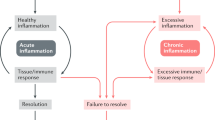
Key Points
-
Much of the current treatment for chronic inflammatory diseases is based on the inhibition of mediators that drive acute inflammation.
-
This strategy has never fully cured inflammation-based disease processes, serving only to deal with the symptoms of the conditions. Invariably, established therapeutics also inhibited essential physiological processes, resulting in unsavoury side effects or, in some recent advancements, leaving the patient open to opportunistic infection. So, an improvement in the efficacy of existing medicines with greater tolerability is welcome.
-
In addition to factors that drive inflammation, we are now beginning to understand that there are a whole range of mediators, receptors and intracellular signalling events that are important and have an active role in orchestrating acute inflammatory resolution.
-
We are beginning to realize that in addition to halting the trafficking of leukocytes to an inflamed site, inflammatory cells die in a highly specialized manner and are subsequently cleared in a way that is particularly conducive to the host's well-being. Successful resolution will limit tissue damage and, arguably, reduce the propensity for the development of chronic inflammation.
-
As pro-resolution mediators show potent anti-inflammatory effects in vitro and show great promise in the treatment of experimental inflammatory diseases in vivo, perhaps we can now take a closer look at what switches off acute inflammation normally and develop drugs based on their mode of action to treat chronic, non-resolving inflammatory diseases. It is hoped that novel compounds based on the mechanism of action of a given pro-resolving pathway will be modulatory in nature, with fewer side effects compared to existing anti-inflammatories.
Abstract
Treatment of inflammatory diseases today is largely based on interrupting the synthesis or action of mediators that drive the host's response to injury. Non-steroidal anti-inflammatories, steroids and antihistamines, for instance, were developed on this basis. Although such small-molecule inhibitors have provided the main treatment for inflammatory arthropathies and asthma, they are not without their shortcomings. This review offers an alternative approach to the development of novel therapeutics based on the endogenous mediators and mechanisms that switch off acute inflammation and bring about its resolution. It is thought that this strategy will open up new avenues for the future management of inflammation-based diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Florey, H. W. General Pathology (Lloyd–Luke Ltd, London, 1970). Provides a fantastic historical perspective of inflammation research as well as detailed accounts of some of the classic experiments that have contributed to our current understanding of the inflammatory response.
Cotran, R. S., Kumar, V. & Collins, T. (eds) in Robbins Pathologic Basis of Disease Vol. 6 50–88 (W. B. Saunders Company, Philadelphia, 1999).
Cotran, R. S., Kumar, V. & Collins, T. (eds) in Robbins Pathologic Basis of Disease Vol. 6 89–112 (W. B. Saunders Company, Philadelphia, 1999).
Dumonde, D. C. & Glynn, L. E. The production of arthritis in rabbits by an immunological reaction to fibrin. Br. J. Exp. Pathol. 43, 373 (1962).
Dumonde, D. C. & Glynn, L. E. The reaction of guinea-pigs to autologous and heterologous fibrin implants. J. Pathol. Bacteriol. 90, 649–657 (1965).
Weissmann, G., Becher, G., Wiedermann, G. & Bernheimer, A. W. Studies on lysozymes. VII. Acute and chronic arthritis produced by intra-articular injections of streptolysin 'S' in rabbits. Am. J. Pathol. 46, 129 (1965).
Willoughby, D. A. & Ryan, G. B. Evidence for a possible endogenous antigen in chronic inflammation. J. Pathol. 101, 233–239 (1970).
Recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis: 2001 update. American College of Rheumatology Ad Hoc Committee on Glucocoritcoid-Induced Osteoporosis. Arthritis Rheum. 44, 1496–1503 (2001).
Beer, H. D., Fassler, R. & Werner, S. Glucocorticoid-regulated gene expression during cutaneous wound repair. Vitam. Horm. 59, 217–239 (2000).
Mukherjee, D. Selective cyclooxygenase-2 (COX-2) inhibitors and potential risk of cardiovascular events. Biochem. Pharmacol. 63, 817–821 (2002).
Antoni, C. & Braun, J. Side effects of anti-TNF therapy: current knowledge. Clin. Exp. Rheumatol. 20, S152–S157 (2002).
Urade, Y. & Hayaishi, O. Prostaglandin D synthase: structure and function. Vitam. Horm. 58, 89–120 (2000).
Straus, D. S. & Glass, C. K. Cyclopentenone prostaglandins: new insights on biological activities and cellular targets. Med. Res. Rev. 21, 185–210 (2001). This is a comprehensive review of cyclopentenone prostaglandins that covers their synthesis, structure and role in pathophysiology.
Jiang, C., Ting, A. T. & Seed, B. PPAR-gamma agonists inhibit production of monocyte inflammatory cytokines. Nature 391, 82–86 (1998).
Ricote, M., Li, A. C., Willson, T. M., Kelly, C. J. & Glass, C. K. The peroxisome proliferator-activated receptor-gamma is a negative regulator of macrophage activation. Nature 391, 79–82 (1998). References 14 and 15 report on the receptor-mediated actions of the cyclopentenone prostaglandins that lead to a suppression of inflammation.
Nosjean, O. & Boutin, J. A. Natural ligands of PPARgamma: are prostaglandin J(2) derivatives really playing the part? Cell Signal. 14, 573–583 (2002).
Straus, D. S. et al. 15-deoxy-delta 12,14-prostaglandin J2 inhibits multiple steps in the NF-kappa B signaling pathway. Proc. Natl Acad. Sci. USA 97, 4844–4849 (2000).
Perez-Sala, D., Cernuda-Morollon, E. & Canada, F. J. Molecular basis for the direct inhibition of AP-1 DNA binding by 15-deoxy-Delta 12,14-prostaglandin J2. J. Biol. Chem. 278, 51251–51260 (2003).
Chen, C. W., Chang, Y. H., Tsi, C. J. & Lin, W. W. Inhibition of IFN-gamma-mediated inducible nitric oxide synthase induction by the peroxisome proliferator-activated receptor gamma agonist, 15-deoxy-delta 12,14-prostaglandin J2, involves inhibition of the upstream Janus kinase/STAT1 signaling pathway. J. Immunol. 171, 979–988 (2003).
Maggi, L. B. Jr et al. Anti-inflammatory actions of 15-deoxy-delta 12,14-prostaglandin J2 and troglitazone: evidence for heat shock-dependent and-independent inhibition of cytokine-induced inducible nitric oxide synthase expression. Diabetes 49, 346–355 (2000).
Petrova, T. V., Akama, K. T. & Van Eldik, L. J. Cyclopentenone prostaglandins suppress activation of microglia: down-regulation of inducible nitric-oxide synthase by 15-deoxy-Delta12,14-prostaglandin J2. Proc. Natl Acad. Sci. USA 96, 4668–4673 (1999).
Bernardo, A., Levi, G. & Minghetti, L. Role of the peroxisome proliferator-activated receptor-gamma (PPAR-gamma) and its natural ligand 15-deoxy-Delta12, 14-prostaglandin J2 in the regulation of microglial functions. Eur. J. Neurosci. 12, 2215–2223 (2000).
Gosset, P. et al. Prostaglandin D2 affects the maturation of human monocyte-derived dendritic cells: consequence on the polarization of naive Th cells. J. Immunol. 170, 4943–4952 (2003).
Jackson, S. M. et al. Peroxisome proliferator-activated receptor activators target human endothelial cells to inhibit leukocyte-endothelial cell interaction. Arterioscler. Thromb. Vasc. Biol. 19, 2094–2104 (1999).
Pasceri, V., Wu, H. D., Willerson, J. T. & Yeh, E. T. Modulation of vascular inflammation in vitro and in vivo by peroxisome proliferator-activated receptor-gamma activators. Circulation 101, 235–238 (2000).
Zhang, X., Wang, J. M., Gong, W. H., Mukaida, N. & Young, H. A. Differential regulation of chemokine gene expression by 15-deoxy-delta 12,14 prostaglandin J2. J. Immunol. 166, 7104–7111 (2001).
Zernecke, A., Erl, W., Fraemohs, L., Lietz, M. & Weber, C. Suppression of endothelial adhesion molecule up-regulation with cyclopentenone prostaglandins is dissociated from IkappaB-alpha kinase inhibition and cell death induction. FASEB J. 17, 1099–1101 (2003).
Gilroy, D. W., Tomlinson, A. & Willoughby, D. A. Differential effects of inhibitors of cyclooxygenase (cyclooxygenase 1 and cyclooxygenase 2) in acute inflammation. Eur. J. Pharmacol. 355, 211–217 (1998).
Gilroy, D. W. et al. Inducible cyclooxygenase may have anti-inflammatory properties. Nature Med. 5, 698–701 (1999). This paper originally described the synthesis of anti-inflammatory eicosanoids during acute inflammatory resolution, thereby highlighting that the host actively manufactures soluble mediators to switch off acute inflammation and limit tissue damage.
Gilroy, D. W., Newson, J., Sawmynaden, P., Willoughby, D. A. & Croxtall, J. D. A novel role for phospholipase A2 isoforms in the checkpoint control of acute inflammation. FASEB J. 18, 489–498 (2004).
Gilroy, D. W. et al. Inducible cyclooxygenase-derived 15-deoxy(Delta)12-14PGJ2 brings about acute inflammatory resolution in rat pleurisy by inducing neutrophil and macrophage apoptosis. FASEB J. 17, 2269–2271 (2003).
Ward, C. et al. Prostaglandin D2 and its metabolites induce caspase-dependent granulocyte apoptosis that is mediated via inhibition of I kappa B alpha degradation using a peroxisome proliferator-activated receptor-gamma-independent mechanism. J. Immunol. 168, 6232–6243 (2002).
Castrillo, A. et al. Potentiation of protein kinase C zeta activity by 15-deoxy-delta(12,14)-prostaglandin J(2) induces an imbalance between mitogen-activated protein kinases and NF-kappaB that promotes apoptosis in macrophages. Mol. Cell Biol. 23, 1196–1208 (2003).
Li, L. et al. 15-deoxy-delta 12,14-prostaglandin J2 induces apoptosis of human hepatic myofibroblasts. A pathway involving oxidative stress independently of peroxisome-proliferator-activated receptors. J. Biol. Chem. 276, 38152–38158 (2001).
Reference deleted in proof.
Setoguchi, K. et al. Peroxisome proliferator-activated receptor-gamma haploinsufficiency enhances B cell proliferative responses and exacerbates experimentally induced arthritis. J. Clin. Invest. 108, 1667–1675 (2001).
Ando, M. et al. Retrovirally introduced prostaglandin D2 synthase suppresses lung injury induced by bleomycin. Am. J. Respir. Cell. Mol. Biol. 28, 582–591 (2003).
Cuzzocrea, S. et al. The cyclopentenone prostaglandin 15-deoxy-delta(12,14)-PGJ2 attenuates the development of colon injury caused by dinitrobenzene sulphonic acid in the rat. Br. J. Pharmacol. 138, 678–688 (2003).
Chatterjee, P. K. et al. The cyclopentenone prostaglandin 15-deoxy-Delta(12,14)-prostaglandin J(2) ameliorates ischemic acute renal failure. Cardiovasc. Res. 61, 630–643 (2004).
Kawahito, Y. et al. 15-deoxy-delta(12,14)-PGJ(2) induces synoviocyte apoptosis and suppresses adjuvant-induced arthritis in rats. J. Clin. Invest. 106, 189–197 (2000).
Diab, A. et al. Peroxisome proliferator-activated receptor-gamma agonist 15-deoxy-delta(12,14)-prostaglandin J(2) ameliorates experimental autoimmune encephalomyelitis. J. Immunol. 168, 2508–2515 (2002).
Matsuoka, T. et al. Prostaglandin D2 as a mediator of allergic asthma. Science 287, 2013–2017 (2000).
Honda, K. et al. Prostaglandin D2 reinforces TH2 type inflammatory responses of airways to low-dose antigen through bronchial expression of macrophage-derived chemokine. J. Exp. Med. 198, 533–543 (2003).
Lee, T. S., Tsai, H. L. & Chau, L. Y. Induction of heme oxygenase-1 expression in murine macrophages is essential for the anti-inflammatory effect of low dose 15-deoxy-delta 12,14-prostaglandin J2. J. Biol. Chem. 278, 19325–19330 (2003).
Ryter, S. W., Otterbein, L. E., Morse, D. & Choi, A. M. Heme oxygenase/carbon monoxide signaling pathways: regulation and functional significance. Mol. Cell. Biochem. 234–235, 249–263 (2002).
Willis, D. M. A., Frederick, R. & Willoughby, D. A. Heme oxygenase: a novel target for the modulation of the inflammatory response. Nature Med. 2, 87–90 (1996).
Graca-Souza, A. V., Arruda, M. A., de Freitas, M. S., Barja-Fidalgo, C. & Oliveira, P. L. Neutrophil activation by heme: implications for inflammatory processes. Blood 99, 4160–4165 (2002).
Alcaraz, M. J., Fernandez, P. & Guillen, M. I. Anti-inflammatory actions of the heme oxygenase-1 pathway. Curr. Pharm. Des. 9, 2541–2551 (2003).
Sato, K. et al. Carbon monoxide generated by heme oxygenase-1 suppresses the rejection of mouse-to-rat cardiac transplants. J. Immunol. 166, 4185–4194 (2001).
Hancock, W. W., Buelow, R., Sayegh, M. H. & Turka, L. A. Antibody-induced transplant arteriosclerosis is prevented by graft expression of anti-oxidant and anti-apoptotic genes. Nature Med. 4, 1392–1396 (1998).
Woo, J., Iyer, S., Mori, N. & Buelow, R. Alleviation of graft-versus-host disease after conditioning with cobalt-protoporphyrin, an inducer of heme oxygenase-1. Transplantation 69, 623–633 (2000).
Levy, B. D., Clish, C. B., Schmidt, B., Gronert, K. & Serhan, C. N. Lipid mediator class switching during acute inflammation: signals in resolution. Nature Immunol. 2, 612–619 (2001). In addition to reference 25, this paper reinforces the concept of lipid mediator class switching from pro-inflammatory at the onset of acute inflammation to pro-resolving at resolution.
Papayianni, A., Serhan, C. N. & Brady, H. R. Lipoxin A4 and B4 inhibit leukotriene-stimulated interactions of human neutrophils and endothelial cells. J. Immunol. 156, 2264–2272 (1996).
Serhan, C. N., Takano, T., Clish, C. B., Gronert, K. & Petasis, N. Aspirin-triggered 15-epi-lipoxin A4 and novel lipoxin B4 stable analogs inhibit neutrophil-mediated changes in vascular permeability. Adv. Exp. Med. Biol. 469, 287–293 (1999).
Maddox, J. F. & Serhan, C. N. Lipoxin A4 and B4 are potent stimuli for human monocyte migration and adhesion: selective inactivation by dehydrogenation and reduction. J. Exp. Med. 183, 137–146 (1996).
Maddox, J. F. et al. Lipoxin A4 stable analogs are potent mimetics that stimulate human monocytes and THP-1 cells via a G-protein-linked lipoxin A4 receptor. J. Biol. Chem. 272, 6972–6978 (1997).
Jozsef, L., Zouki, C., Petasis, N. A., Serhan, C. N. & Filep, J. G. Lipoxin A4 and aspirin-triggered 15-epi-lipoxin A4 inhibit peroxynitrite formation, NF-kappa B and AP-1 activation, and IL-8 gene expression in human leukocytes. Proc. Natl Acad. Sci. USA 99, 13266–13271 (2002).
Bandeira-Melo, C. et al. Cyclooxygenase-2-derived prostaglandin E2 and lipoxin A4 accelerate resolution of allergic edema in Angiostrongylus costaricensis-infected rats: relationship with concurrent eosinophilia. J. Immunol. 164, 1029–1036 (2000).
Godson, C. et al. Cutting edge: lipoxins rapidly stimulate nonphlogistic phagocytosis of apoptotic neutrophils by monocyte-derived macrophages. J. Immunol. 164, 1663–1667 (2000).
Mitchell, S. et al. Lipoxins, aspirin-triggered epi-lipoxins, lipoxin stable analogues, and the resolution of inflammation: stimulation of macrophage phagocytosis of apoptotic neutrophils in vivo. J. Am. Soc. Nephrol. 13, 2497–2507 (2002).
Brink, C. et al. International Union of Pharmacology XXXVII. Nomenclature for leukotriene and lipoxin receptors. Pharmacol. Rev. 55, 195–227 (2003).
Bandeira-Melo, C. et al. Cutting edge: lipoxin (LX) A4 and aspirin-triggered 15-epi-LXA4 block allergen-induced eosinophil trafficking. J. Immunol. 164, 2267–2271 (2000).
Serhan, C. N. et al. Novel functional sets of lipid-derived mediators with antiinflammatory actions generated from omega-3 fatty acids via cyclooxygenase 2-nonsteroidal antiinflammatory drugs and transcellular processing. J. Exp. Med. 192, 1197–1204 (2000).
Serhan, C. N. et al. Resolvins: a family of bioactive products of omega-3 fatty acid transformation circuits initiated by aspirin treatment that counter proinflammation signals. J. Exp. Med. 196, 1025–1037 (2002). References 63 and 64 report on the role of aspirin-triggered lipid mediator metabolites of polyunsaturated fatty acids and their spontaneous synthesis during the resolving phase of acute inflammation.
Hong, S., Gronert, K., Devchand, P. R., Moussignac, R. L. & Serhan, C. N. Novel docosatrienes and 17S-resolvins generated from docosahexaenoic acid in murine brain, human blood and glial cells. Autacoids in anti-inflammation. J. Biol. Chem. 278, 14677–14687 (2003).
Brezinski, D. A., Nesto, R. W. & Serhan, C. N. Angioplasty triggers intracoronary leukotrienes and lipoxin A4. Impact of aspirin therapy. Circulation 86, 56–63 (1992).
Van Dyke, T. E. & Serhan, C. N. Resolution of inflammation: a new paradigm for the pathogenesis of periodontal diseases. J. Dent. Res. 82, 82–90 (2003).
Claria, J. et al. Altered biosynthesis of leukotrienes and lipoxins and host defense disorders in patients with cirrhosis and ascites. Gastroenterology 115, 147–156 (1998).
Levy, B. D. et al. Multi-pronged inhibition of airway hyper-responsiveness and inflammation by lipoxin A(4). Nature Med. 8, 1018–1023 (2002).
Munger, K. A. et al. Transfection of rat kidney with human 15-lipoxygenase suppresses inflammation and preserves function in experimental glomerulonephritis. Proc. Natl Acad. Sci. USA 96, 13375–13380 (1999).
Leonard, M. O. et al. 15-Epi-16-(para-fluorophenoxy)-lipoxin A(4)-methyl ester, a synthetic analogue of 15-epi-lipoxin A(4), is protective in experimental ischemic acute renal failure. J. Am. Soc. Nephrol. 13, 1657–1662 (2002).
Schottelius, A. J. et al. An aspirin-triggered lipoxin A4 stable analog displays a unique topical anti-inflammatory profile. J. Immunol. 169, 7063–7070 (2002).
Souza, M. H., de Lima, O. M., Zamuner, S. R., Fiorucci, S. & Wallace, J. L. Gastritis increases resistance to aspirin-induced mucosal injury via COX-2-mediated lipoxin synthesis. Am. J. Physiol. Gastrointest. Liver Physiol. 285, G54–G61 (2003).
Hughes, J. et al. Neutrophil fate in experimental glomerular capillary injury in the rat. Emigration exceeds in situ clearance by apoptosis. Am. J. Pathol. 150, 223–234 (1997).
Martin, C. et al. Chemokines acting via CXCR2 and CXCR4 control the release of neutrophils from the bone marrow and their return following senescence. Immunity 19, 583–593 (2003).
Bellingan, G. J., Caldwell, H., Howie, S. E., Dransfield, I. & Haslett, C. In vivo fate of the inflammatory macrophage during the resolution of inflammation: inflammatory macrophages do not die locally, but emigrate to the draining lymph nodes. J. Immunol. 157, 2577–2585 (1996).
Bellingan, G. J. et al. Adhesion molecule-dependent mechanisms regulate the rate of macrophage clearance during the resolution of peritoneal inflammation. J. Exp. Med. 196, 1515–1521 (2002). References 76 and 77 show comprehensively that in resolving peritonitis, the fate of macrophage clearance is through lymphatic drainage.
Savill, J. & Fadok, V. Corpse clearance defines the meaning of cell death. Nature 407, 784–788 (2000). A concise review highlighting the concept of apoptosis and phagocytosis and their relevance to inflammatory disease processes.
Nicholson, D. W. From bench to clinic with apoptosis-based therapeutic agents. Nature 407, 810–816 (2000).
Brunner, T. & Mueller, C. Apoptosis in disease: about shortage and excess. Essays Biochem. 39, 119–130 (2003).
Fadok, V. A., Bratton, D. L. & Henson, P. M. Phagocyte receptors for apoptotic cells: recognition, uptake, and consequences. J. Clin. Invest. 108, 957–962 (2001).
Savill, J., Dransfield, I., Gregory, C. & Haslett, C. A blast from the past: clearance of apoptotic cells regulates immune responses. Nature Rev. Immunol. 2, 965–975 (2002).
Ward, C., Dransfield, I., Chilvers, E. R., Haslett, C. & Rossi, A. Pharmacological manipulation of granulocyte apoptosis: potential therapeutic targets. Trends Pharmacol. Sci. 20, 503–509 (1999).
Giles, K. M., Hart, S. P., Haslett, C., Rossi, A. G. & Dransfield, I. An appetite for apoptotic cells? Controversies and challenges. Br. J. Haematol. 109, 1–12 (2000).
Rossi, A. G. et al. Regulation of macrophage phagocytosis of apoptotic cells by cAMP. J. Immunol. 160, 3562–3568 (1998).
Liu, Y. et al. Glucocorticoids promote nonphlogistic phagocytosis of apoptotic leukocytes. J. Immunol. 162, 3639–3646 (1999).
Giles, K. M. et al. Glucocorticoid augmentation of macrophage capacity for phagocytosis of apoptotic cells is associated with reduced p130Cas expression, loss of paxillin/pyk2 phosphorylation, and high levels of active Rac. J. Immunol. 167, 976–986 (2001).
Ren, Y. & Savill, J. Proinflammatory cytokines potentiate thrombospondin-mediated phagocytosis of neutrophils undergoing apoptosis. J. Immunol. 154, 2366–2374 (1995).
McCutcheon, J. C. et al. Regulation of macrophage phagocytosis of apoptotic neutrophils by adhesion to fibronectin. J. Leukoc. Biol. 64, 600–607 (1998).
Hart, S. P., Dougherty, G. J., Haslett, C. & Dransfield, I. CD44 regulates phagocytosis of apoptotic neutrophil granulocytes, but not apoptotic lymphocytes, by human macrophages. J. Immunol. 159, 919–925 (1997).
Teder, P. et al. Resolution of lung inflammation by CD44. Science 296, 155–158 (2002).
Woolley, K. L. et al. Eosinophil apoptosis and the resolution of airway inflammation in asthma. Am. J. Respir. Crit. Care Med. 154, 237–243 (1996).
Murphy, F. J., Seery, L. T. & Hayes, I. Therapeutic approaches to the modulation of apoptosis. Essays Biochem. 39, 131–153 (2003).
Degterev, A., Boyce, M. & Yuan, J. A decade of caspases. Oncogene 22, 8543–8567 (2003).
Earnshaw, W. C., Martins, L. M. & Kaufmann, S. H. Mammalian caspases: structure, activation, substrates, and functions during apoptosis. Annu. Rev. Biochem. 68, 383–424 (1999).
Graczyk, P. P. Caspase inhibitors as anti-inflammatory and antiapoptotic agents. Prog. Med. Chem. 39, 1–72 (2002).
Iwata, A., Harlan, J. M., Vedder, N. B. & Winn, R. K. The caspase inhibitor z-VAD is more effective than CD18 adhesion blockade in reducing muscle ischemia-reperfusion injury: implication for clinical trials. Blood 100, 2077–2080 (2002).
Natori, S., Higuchi, H., Contreras, P. & Gores, G. J. The caspase inhibitor IDN-6556 prevents caspase activation and apoptosis in sinusoidal endothelial cells during liver preservation injury. Liver Transpl. 9, 278–284 (2003).
Hoglen, N. C. et al. Characterization of IDN-6556: a liver-targeted caspase inhibitor. J. Pharmacol. Exp. Ther. 23 Jan 2004 [epub ahead of print].
Valentino, K. L., Gutierrez, M., Sanchez, R., Winship, M. J. & Shapiro, D. A. First clinical trial of a novel caspase inhibitor: anti-apoptotic caspase inhibitor, IDN-6556, improves liver enzymes. Int. J. Clin. Pharmacol. Ther. 41, 441–449 (2003).
Karin, M. New twists in gene regulation by glucocorticoid receptor: is DNA binding dispensable? Cell 93, 487–490 (1998).
Ghosh, S., May, M. J. & Kopp, E. B. NF-kappa B and Rel proteins: evolutionarily conserved mediators of immune responses. Annu. Rev. Immunol. 16, 225–260 (1998).
Khoshnan, A. et al. The NF-kappa B cascade is important in Bcl-xL expression and for the anti-apoptotic effects of the CD28 receptor in primary human CD4+ lymphocytes. J. Immunol. 165, 1743–1754 (2000).
Ward, C. et al. NF-kappaB activation is a critical regulator of human granulocyte apoptosis in vitro. J. Biol. Chem. 274, 4309–4318 (1999).
Lawrence, T., Gilroy, D. W., Colville-Nash, P. R. & Willoughby, D. A. Possible new role for NF-kappaB in the resolution of inflammation. Nature Med. 7, 1291–1297 (2001). Shows an important role for NF-κB in the resolution of acute inflammation that underpins a novel protective role for this otherwise pro-inflammatory factor.
Ishikawa, H. et al. Chronic inflammation and susceptibility to bacterial infections in mice lacking the polypeptide (p)105 precursor (NF-kappaB1) but expressing p50. J. Exp. Med. 187, 985–996 (1998).
Bohuslav, J. et al. Regulation of an essential innate immune response by the p50 subunit of NF-kappaB. J. Clin. Invest. 102, 1645–1652 (1998).
Chen, L. W. et al. The two faces of IKK and NF-kappaB inhibition: prevention of systemic inflammation but increased local injury following intestinal ischemia-reperfusion. Nature Med. 9, 575–581 (2003).
Munck, A., Guyre, P. M. & Holbrook, N. J. Physiological functions of glucocorticoids in stress and their relation to pharmacological actions. Endocr. Rev. 5, 25–44 (1984).
Flower, R. J., Parente, L., Persico, P. & Salmon, J. A. A comparison of the acute inflammatory response in adrenalectomised and sham-operated rats. Br. J. Pharmacol. 87, 57–62 (1986).
Perretti, M., Becherucci, C., Scapigliati, G. & Parente, L. The effect of adrenalectomy on interleukin-1 release in vitro and in vivo. Br. J. Pharmacol. 98, 1137–1142 (1989).
Adcock, I. M. Molecular mechanisms of glucocorticosteroid actions. Pulm. Pharmacol. Ther. 13, 115–126 (2000).
Auphan, N., DiDonato, J. A., Rosette, C., Helmberg, A. & Karin, M. Immunosuppression by glucocorticoids: inhibition of NF-kappa B activity through induction of I kappa B synthesis. Science 270, 286–290 (1995).
Buttgereit, F., Burmester, G. R. & Brand, M. D. Bioenergetics of immune functions: fundamental and therapeutic aspects. Immunol. Today 21, 192–199 (2000).
Nissen, R. M. & Yamamoto, K. R. The glucocorticoid receptor inhibits NFkappaB by interfering with serine-2 phosphorylation of the RNA polymerase II carboxy-terminal domain. Genes Dev. 14, 2314–2329 (2000).
Flower, R. J. Eleventh Gaddum memorial lecture. Lipocortin and the mechanism of action of the glucocorticoids. Br. J. Pharmacol. 94, 987–1015 (1988).
Croxtall, J. D. & Flower, R. J. Lipocortin 1 mediates dexamethasone-induced growth arrest of the A549 lung adenocarcinoma cell line. Proc. Natl Acad. Sci. USA 89, 3571–3575 (1992).
De Caterina, R. et al. Macrophage-specific eicosanoid synthesis inhibition and lipocortin-1 induction by glucocorticoids. J. Appl. Physiol. 75, 2368–2375 (1993).
Mancuso, F., Flower, R. J. & Perretti, M. Leukocyte transmigration, but not rolling or adhesion, is selectively inhibited by dexamethasone in the hamster post-capillary venule. Involvement of endogenous lipocortin 1. J. Immunol. 155, 377–386 (1995).
Perretti, M., Ahluwalia, A., Harris, J. G., Goulding, N. J. & Flower, R. J. Lipocortin-1 fragments inhibit neutrophil accumulation and neutrophil-dependent edema in the mouse. A qualitative comparison with an anti-CD11b monoclonal antibody. J. Immunol. 151, 4306–4314 (1993).
Gerke, V. & Moss, S. E. Annexins: from structure to function. Physiol. Rev. 82, 331–371 (2002).
Perretti, M. et al. Endogenous lipid- and peptide-derived anti-inflammatory pathways generated with glucocorticoid and aspirin treatment activate the lipoxin A4 receptor. Nature Med. 8, 1296–1302 (2002). Demonstrates, for the first time, the biochemical and functional link between endogenous and exogenous annexin-1 and ALX. Experiments of binding, immunoprecipitation and in vivo analyses of PMN recruitment support this novel idea.
Le, Y., Murphy, P. M. & Wang, J. M. Formyl-peptide receptors revisited. Trends Immunol. 23, 541–548 (2002).
Perretti, M. & Gavins, F. N. Annexin 1: an endogenous anti-inflammatory protein. News Physiol. Sci. 18, 60–64 (2003).
Perretti, M. et al. Acute inflammatory response in the mouse: exacerbation by immunoneutralization of lipocortin 1. Br. J. Pharmacol. 117, 1145–1154 (1996).
Hannon, R. et al. Aberrant inflammation and resistance to glucocorticoids in annexin 1−/− mouse. FASEB J. 17, 253–255 (2003).
Yang, Y. H. et al. Modulation of inflammation and response to dexamethasone by Annexin 1 in antigen-induced arthritis. Arthritis Rheum. 50, 976–984 (2004). References 126 and 127 were the first studies with the newly generated annexin-1-null mice to confirm the crucial, not redundant, role that this anti-inflammatory protein plays in prolonging the acute inflammatory response as well as in exacerbating joint inflammation in a model of arthritis.
Perretti, M. The annexin 1 receptor(s): is the plot unravelling? Trends Pharmacol. Sci. 24, 574–579 (2003). A concise review highlighting the opportunities that derive from the identification of the molecular targets that are responsible for annexin-1 actions.
Hasko, G. & Cronstein, B. N. Adenosine: an endogenous regulator of innate immunity. Trends Immunol. 25, 33–39 (2004).
Sitkovsky, M. V. Use of the A(2A) adenosine receptor as a physiological immunosuppressor and to engineer inflammation in vivo. Biochem. Pharmacol. 65, 493–501 (2003).
Cronstein, B. N., Eberle, M. A., Gruber, H. E. & Levin, R. I. Methotrexate inhibits neutrophil function by stimulating adenosine release from connective tissue cells. Proc. Natl Acad. Sci. USA 88, 2441–2445 (1991).
Cronstein, B. N., Naime, D. & Ostad, E. The antiinflammatory mechanism of methotrexate. Increased adenosine release at inflamed sites diminishes leukocyte accumulation in an in vivo model of inflammation. J. Clin. Invest. 92, 2675–2682 (1993).
Montesinos, M. C. et al. Adenosine A2A or A3 receptors are required for inhibition of inflammation by methotrexate and its analog MX-68. Arthritis Rheum. 48, 240–247 (2003).
Xaus, J. et al. IFN-gamma up-regulates the A2B adenosine receptor expression in macrophages: a mechanism of macrophage deactivation. J. Immunol. 162, 3607–3614 (1999).
Hasko, G., Deitch, E. A., Szabo, C., Nemeth, Z. H. & Vizi, E. S. Adenosine: a potential mediator of immunosuppression in multiple organ failure. Curr. Opin. Pharmacol. 2, 440–444 (2002).
Blackburn, M. R. Too much of a good thing: adenosine overload in adenosine-deaminase-deficient mice. Trends Pharmacol. Sci. 24, 66–70 (2003).
Catania, A., Gatti, S., Colombo, G. & Lipton, J. M. Targeting melanocortin receptors as a novel strategy to control inflammation. Pharmacol. Rev. 56, 1–29 (2004).
Fadok, V. A., Bratton, D. L., Frasch, S. C., Warner, M. L. & Henson, P. M. The role of phosphatidylserine in recognition of apoptotic cells by phagocytes. Cell Death Differ. 5, 551–562 (1998).
Arur, S. et al. Annexin I is an endogenous ligand that mediates apoptotic cell engulfment. Dev. Cell 4, 587–598 (2003).
Huynh, M. L., Fadok, V. A. & Henson, P. M. Phosphatidylserine-dependent ingestion of apoptotic cells promotes TGF-beta1 secretion and the resolution of inflammation. J. Clin. Invest. 109, 41–50 (2002).
Blom, A. B., van Lent, P. L., Holthuysen, A. E. & van den Berg, W. B. Skewed balances in regulation of stimulating and inhibitory FC gamma receptors on macrophages of CIA sensitive mice. Inflamm. Res. 50 (Suppl. 3), S155 (2001).
Edenius, C., Haeggstrom, J. & Lindgren, J. A. Transcellular conversion of endogenous arachidonic acid to lipoxins in mixed human platelet-granulocyte suspensions. Biochem. Biophys. Res. Commun. 157, 801–807 (1988).
Fiore, S. & Serhan, C. N. Formation of lipoxins and leukotrienes during receptor-mediated interactions of human platelets and recombinant human granulocyte/macrophage colony-stimulating factor-primed neutrophils. J. Exp. Med. 172, 1451–1457 (1990).
Serhan, C. N., Hamberg, M. & Samuelsson, B. Lipoxins: novel series of biologically active compounds formed from arachidonic acid in human leukocytes. Proc. Natl Acad. Sci. USA 81, 5335–5339 (1984).
Claria, J. & Serhan, C. N. Aspirin triggers previously undescribed bioactive eicosanoids by human endothelial cell-leukocyte interactions. Proc. Natl Acad. Sci. USA 92, 9475–9479 (1995).
McLoughlin, R. M. et al. Interplay between IFN-gamma and IL-6 signaling governs neutrophil trafficking and apoptosis during acute inflammation. J. Clin. Invest. 112, 598–607 (2003).
Hurst, S. M. et al. Il-6 and its soluble receptor orchestrate a temporal switch in the pattern of leukocyte recruitment seen during acute inflammation. Immunity 14, 705–714 (2001).
Rossi, A. et al. Anti-inflammatory cyclopentenone prostaglandins are direct inhibitors of IkappaB kinase. Nature 403, 103–108 (2000).
Castrillo, A., Diaz-Guerra, M. J., Hortelano, S., Martin-Sanz, P. & Bosca, L. Inhibition of IkappaB kinase and IkappaB phosphorylation by 15-deoxy-delta(12,14)-prostaglandin J(2) in activated murine macrophages. Mol. Cell. Biol. 20, 1692–1698 (2000).
Cernuda-Morollon, E., Pineda-Molina, E., Canada, F. J. & Perez-Sala, D. 15-Deoxy-delta 12,14-prostaglandin J2 inhibition of NF-kappaB-DNA binding through covalent modification of the p50 subunit. J. Biol. Chem. 276, 35530–35536 (2001). References 148–150 show that the cyclopentenone prostaglandins have potent inhibitory signalling effects by directly binding to and altering NF-κB activation in addition to their modes of action discussed in references 14 and 15.
Bell-Parikh, L. C. et al. Biosynthesis of 15-deoxy delta12,14–PGJ2 and the ligation of PPARgamma. J. Clin. Invest. 112, 945–955 (2003).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
DATABASES
LocusLink
OMIM
Glossary
- VASCULAR LEAKAGE
-
The process of the escape of plasma and plasma proteins, along with white blood cells, from the vessel is known as exudation. This inflammatory exudate accounts for an increase in the volume of interstitial fluid (oedema) and tissue swelling at the local site of injury.
- NON-PHLOGISTIC
-
Non-inflammatory. This term is used to describe the clearance of leukocytes in a manner that does not elicit an inflammatory response.
- NF-κB
-
(Nuclear factor of κB). A group of sequence-specific transcription factors that are best known as key regulators of the innate and adaptive inflammatory responses, cell survival and ancogenesis. In mammals, NF-κB consists of five structurally related and functionally conserved proteins: RELA (p65), RELB, c-REL, NF-κB1 (p105 and p50) and NF-κB2 (p100 and p52).
- PEROXISOME PROLIFERATOR-ACTIVATED RECEPTORS
-
(PPARs). Members of the nuclear receptor family that regulate the transcription of genes involved in lipid and lipoprotein metabolism, glucose and energy homeostasis, as well as cellular differentiation. They consist of three isotypes: alpha (NR1C1), gamma (NR1C3) and beta/delta (NRC1C2), with a differential tissue distribution.
- SIGNAL TRANSDUCERS AND ACTIVATORS OF TRANSCRIPTION
-
Signal transducers and activators of transcription (STAT) proteins are intracellular effector molecules of cytokine-modulated signalling.
- POLYMORPHONUCLEAR LEUKOCYTES
-
(PMNs). Discovered by Paul Ehrlich who used contemporary fixing and staining techniques to identify the lobulated nucleus and the granules that typify cells that we now classify as eosinophils, basophils and neutrophils. In the context of this review, PMNs refer to neutrophils.
- DELAYED-TYPE HYPERSENSITIVITY
-
(DTH). A delayed-type hypersensitivity or type IV hypersensitivity is mediated by T lymphocytes and not by antibody–antigen complexes (arthus or type III hypersensitivity). Typically, this response occurs 24–72 hours after the sensitized host is exposed to the offending antigen. For example, a DTH reaction might be set up experimentally by sensitizing to methylated bovine serum albumin in Freund's complete adjuvant and challenging 12 days later with the same antigen.
- TH1
-
The TH1/TH2 hypothesis came to prominence in the late 1980s, indicating that mouse T-helper (TH) cells broadly express differing cytokine profiles. Whereas T-helper 1 (TH1) cells secrete IFN-γ and TNF-α; TH2 cells secrete IL-4, IL-5 and IL-13. In addition, TH3 and regulatory CD25+CD4+ T cells exist that produce TGF-β and IL-10, respectively.
- BILIVERDIN and BILIRUBIN
-
Haem catabolites with anti-inflammatory properties due to scavenging of free radicals.
- AIR-POUCH MODEL
-
The inflation of an air pouch on the dorsum of mice or rats allows for the introduction of inflammatory stimuli to examine either innate or adaptive immune responses.
- cAMP
-
(Cyclic adenosine monophosphate). A second messenger system with inflammo-modulatory properties.
Rights and permissions
About this article
Cite this article
Gilroy, D., Lawrence, T., Perretti, M. et al. Inflammatory Resolution: new opportunities for drug discovery. Nat Rev Drug Discov 3, 401–416 (2004). https://doi.org/10.1038/nrd1383
Issue Date:
DOI: https://doi.org/10.1038/nrd1383
This article is cited by
-
Targeting disease with benzoxazoles: a comprehensive review of recent developments
Medicinal Chemistry Research (2024)
-
Evaluation of antibacterial, antioxidant, and anti-inflammatory properties of GC/MS characterized methanol leaf extract of Terminalia superba (Combretaceae, Engl. & Diels)
Future Journal of Pharmaceutical Sciences (2023)
-
Analysis of AT7519 as a pro-resolution compound in an acetaminophen-induced mouse model of acute inflammation by UPLC-MS/MS
Journal of Inflammation (2023)
-
Inflammation and tumor progression: signaling pathways and targeted intervention
Signal Transduction and Targeted Therapy (2021)
-
Prostaglandin A1 Decreases the Phosphorylation of Tau by Activating Protein Phosphatase 2A via a Michael Addition Mechanism at Cysteine 377
Molecular Neurobiology (2021)