Key Points
-
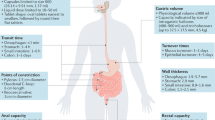
Transdermal delivery of drugs, proteins and other bioactive molecules is an attractive alternative for compounds that cannot be administered orally due to degradation in the gastrointestinal tract and liver. As a result, transdermal patches exist in the United States to administer twelve different drugs or drug combinations to treat a variety of indications.
-
Although it would be beneficial to deliver more drugs from patches, the skin's outer layer of stratum corneum provides a barrier to transport that prevents transdermal delivery of most compounds at therapeutic levels. A broad variety of chemical enhancers has been used to increase drug penetration across skin, but their efficacy has been limited by associated skin irritation and toxicity.
-
Electrical and acoustical energy have been used to increase transdermal delivery. Iontophoresis, involving increased transport across skin mediated primarily by electrophoretic migration, is used in a few FDA-approved products and is poised to have increased impact. Additional research has shown that short, high-voltage pulses causing electroporation can increase transdermal transport not only by electrophoretic movement, but also through short-lived nanometre pores created within the skin. Also of commercial interest, ultrasound and acoustic shock waves have been shown to increase skin permeability by a mechanism believed to involve transient disruption of skin nanostructure.
-
Skin can also be disrupted on the micron scale to increase permeability. Arrays of microscopic needles inserted painlessly into skin have been shown to increase skin permeability for the delivery of small drugs, macromolecules and microparticles, which has stimulated increasing industrial activity. Jet injectors, thermal ablation methods and very small hypodermic needles are also the subject of renewed and on-going commercial interest.
-
After an initial period of development that led to passive systems like the nicotine patch, transdermal drug delivery is experiencing a resurgence of activity using active enhancement methods base on nano- and micro-scale disruption of skin structure. Synergistic combinations of these methods could provide still better results.
Abstract
The past twenty five years have seen an explosion in the creation and discovery of new medicinal agents. Related innovations in drug delivery systems have not only enabled the successful implementation of many of these novel pharmaceuticals, but have also permitted the development of new medical treatments with existing drugs. The creation of transdermal delivery systems has been one of the most important of these innovations, offering a number of advantages over the oral route. In this article, we discuss the already significant impact this field has made on the administration of various pharmaceuticals; explore limitations of the current technology; and discuss methods under exploration for overcoming these limitations and the challenges ahead.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Rein, H. Experimental electroendosmotic studies on living human skin. Z. Biol. 81, 124– (1924).
Blank, I. H. Penetration of low-molecular-weight alcohols into skin. I. Effect of concentration of alcohol and type of vehicle. J. Invest. Dermatol. 43, 415–420 (1964).
Scheuplein, R. J. Mechanism of percutaneous adsorption I. Routes of penetration and the influence of solubility. J. Invest. Dermatol. 45, 334–345 (1965).
Scheuplein, R. J. & Blank, I. H. Permeability of the skin. Physiol. Rev. 51, 702–747 (1971).
Michaels, A. S., Chandrasekaran, S. K. & Shaw, J. E. Drug permeation through human skin: theory and in vitro experimental measurement. AIChE J. 21, 985–996 (1975). Rigorous modelling of transdermal transport that provided the foundation for subsequent models.
Sifton, D. W. (ed.) Physicians' Desk Reference (Thomson PDR, Montvale, 2003).
Cramer, M. P. & Saks, S. R. Translating safety, efficacy and compliance into economic value for controlled release dosage forms. Pharmacoeconomics 5, 482–504 (1994).
Henningfield, J. E. Nicotine medications for smoking cessation. N. Engl. J. Med. 333, 1196–1203 (1995).
Zaffaroni, A. Overview and evolution of therapeutic systems. Ann. NY Acad. Sci. 618, 405–421 (1991).
Wertz, P. W. & Downing, D. T. in Transdermal Drug Delivery: Developmental Issues and Research Intiatives (eds Hadgraft, J. & Guy, R. H.) 1–17 (Marcel Dekker, 1989).
Champion, R. H., Burton, J. L., Burns, D. A. & Breathnach, S. M. (eds). Textbook of Dermatology (Blackwell Science, London, 1998).
Elias, P. M. & Feingold, K. R. Coordinate regulation of epidermal differentiation and barrier homeostasis. Skin Pharmacol. Appl. Skin Physiol. 14 (Suppl. 1), 28–34 (2001).
Marrink, S. J. & Berendsen, J. C. Permeation process of small molecules across lipid membranes studied by molecular dynamics simulations. J. Phys. Chem. 100, 16729–16738 (1996).
Peck, K. D., Ghanem, A. H. & Higuchi, W. I. Hindered diffusion of polar molecules through and effective pore radii estimates of intact and ethanol treated human epidermal membrane. Pharm. Res. 11, 1306–1314 (1994).
Mitragotri, S. Modeling skin permeability to hydrophilic and hydrophobic solutes based on four permeation pathways. J. Control. Release 86, 69–92 (2003).
Fiserova-Bergerova, V., Pierce, J. T. & Droz, P. O. Dermal absoprtion potential of industrial chemicals: criteria for skin notation. Am. J. Ind. Med. 17, 617–635 (1990).
McKone, T. E. & Howard, R. A. Estimating dermal uptake of non-ionic organic chemicals from water and soil: I. Unified fugacity-based models for risk assessment. Risk Analysis 12, 543–557 (1992).
Potts, R. O. & Guy, R. H. Predicting skin permeability. Pharm. Res. 9, 663–669 (1992). Broadly predictive semi-empirical model of skin permeability.
Moss, G. P., Dearden, J. C., Patel, H. & Cronin, M. T. D. Quantitative structure–permeability relationships (QSPRs) for percutaneous absorption. Toxicol. In Vitro 16, 299–317 (2002).
Anderson, B. D. & Raykar, P. V. Solute structure-permeability relationships in human stratum corneum. J. Invest. Dermatol. 93, 280–286 (1989).
Cleek, R. L. & Bunge, A. L. A new method for estiamting dermal absorption from chemical exposure. 1. General approach. Pharm. Res. 10, 497–506 (1993).
Abraham, M. H., Martins, F. & Mitchell, R. C. Algorithms for skin permeability using hydrogen bond descriptors: the problem of steroids. J. Pharm. Pharmac. 49, 858–865 (1997).
Buchwald, P. & Bodor, N. A simple, predictive, structure-based skin permeability model. J. Pharm. Pharmacol. 53, 1087–1098 (2001).
Barrat, M. D. Quantitative structure–activity relationships for skin permeability. Toxicol. In Vitro 9, 27–37 (1995).
Pugh, W. J., Hadgraft, J. & Roberts, M. S. in Dermal Absorption and Toxicity Assessment (eds Roberts, M. S. & Walters, K.) 245–268 (Marcek Dekker, New York, 1998).
Lim, C. W., Fujiwara, S., Yamashita, F. & Hashida, M. Prediction of human skin permeability using a combination of molecular orbital calculations and artificial neural network. Biol. Pharm. Bull. 25, 361–366 (2002).
Frasch, H. F. A Random-walk model for skin permeation. Risk. Anal. 22, 265–276 (2002).
Kasting, G. B., Smith, R. L. & Cooper, E. R. Effect of lipid solubility and molecular size on percutaneous absorption. Pharmacol. Skin 1, 138–153 (1987).
French, E., Potton, C. & Walters, K. in Pharmaceutical Skin Penetration Enhnacement (eds Walters, K. & Hadgraft, J.) 113–144 (Marcel Dekker, New York, 1993).
Kanikkannan, N., Kanimalla, K., Lamba, S. S. & Singh, M. Structure–activity relationship of chemical penetration enhancers in transdermal drug delivery. Curr. Med. Chem. 7, 593–608 (2000).
Lashmar, U. T., Hadgraft, J. & Thomas, N. Topical application of penetration enhancers to the skin of nude mice: a histophatholgical study. J. Pharm. Pharmacol. 41, 118–122 (1989).
Takanashi, Y., Higashiyama, K., Komiya, H., Takayama, K. & Nagai, T. Thiomenthol derivatives as novel percutaneous absorption enhancers. Drug Dev. Ind. Pharm. 25, 89–94 (1999).
Akimoto, T. & Nagase, Y. Novel transdermal drug penetration enhancer: synthesis and enhancing effect of alkyldisiloxane comounds containign glucopyranosyl group. J. Control. Rel. 88, 243–252 (2003).
McVary, K. T., Polepalle, S., Riggi, S. & Pelham, R. W. Topical prostaglandin E1 SEPA gel for the treatment of erectile dysfunction. J. Urol. 162, 726–730 (1999).
Williams, A. C. & Barry, B. W. Skin absorption enhancers. Crit. Rev. Ther. Drug Carrier Syst. 9, 305–353 (1992).
Finin, B. C. & Morgan, T. M. Transdermal penetration enhancers: applications, limitations, and potential. J. Pharm. Sci. 88, 955–958 (1999).
Ongpipattnakul, B., Burnette, R. R., Potts, R. O. & Francoeur, M. L. Evidence that oleic acid exists in a separate phase within stratum corneum lipids. Pharm. Res. 8, 350–354 (1991).
Cevc, G. Transfersomes, liposomes and other lipid suspensions on the skin: permeation enhancement, vesicle penetration, and transdermal drug delivery. Crit. Rev. Ther. Drug Carrier Syst. 13, 257–388 (1996). Overview of lipid-based systems for transdermal delivery.
Karande, P., Jain, A. & Mitragotri, S. Discovery of transdermal penetration enhancers by high-throughput screening. Nature Biotechnol. 4 Jan 2004 (doi:10.1038/nbt928). Study establishing high-throughput screening methods for transdermal formulations.
Costello, C. T. & Jeske, A. H. Iontophoresis: applications in transdermal medication delivery. Phys. Ther. 75, 554–563 (1995).
Warwick, W. J. et al. Evaluation of a cystic fibrosis screening system incorporating a miniature sweat stimulator and disposable chloride sensor. Clin. Chem. 32, 850–853 (1986).
Hölzle, E. & Alberti, N. Long-term efficacy and side effects of tap water iontophoresis of palmoplantar hyperhidrosis — the usefulness of home therapy. Dermatologica 175, 126–135 (1987).
Miller, K. A., Balakrishnan, G., Eichbauer, G. & Betley, K. 1% lidocaine injection, EMLA cream, or 'numby stuff' for topical analgesia associated with peripheral intravenous cannulation. AANA J. 69, 185–187 (2001).
Tamada, J. et al. Noninvasive glucose monitoring: comprehensive clinical results. Cygnus Research Team. JAMA 282, 1839–1844 (1999).
Ledger, P. W. Skin biological issues in electrically enhanced transdermal delivery. Adv. Drug Deliv. Rev. 9, 289–307 (1992). Overview of biological issues associated with transdermal delivery, especially in iontophoresis.
Banga, A. K. Electrically-Assisted Transdermal and Topical Drug Delivery (Taylor & Francis, London, 1998).
Amsden, B. G. & Goosen, M. F. A. Transdermal delivery of peptide and protein drugs: an overview. AIChE J. 41, 1972–1997 (1995). Overview of challenges associated with transdermal delivery of macromolecules, especially in iontophoresis.
Pikal, M. J. The role of electroosmotic flow in transdermal iontophoresis. Adv. Drug Deliv. Rev. 46, 281–305 (2001).
Higuchi, W. I., Li, S. K., Ghanem, A. H., Zhu, H. & Song, Y. Mechanistic aspects of iontophoresis in human epidermal membrane. J. Control. Release 62, 13–23 (1999).
Prausnitz, M. R. The effects of electric current applied to the skin: a review for transdermal drug delivery. Adv. Drug Deliv. Rev. 18, 395–425 (1996).
Chizmadzhev, Y. A. et al. Electrical properties of skin at moderate voltages: contribution of appendegeal macropores. Biophys. J. 74, 843–856 (1998).
Prausnitz, M. R., Bose, V. G., Langer, R. & Weaver, J. C. Electroporation of mammalian skin: a mechanism to enhance transdermal drug delivery. Proc. Natl Acad. Sci. USA 90, 10504–10508 (1993). Study that established skin electroporation for transdermal delivery.
Prausnitz, M. R. A practical assessment of transdermal drug delivery by skin electroporation. Adv. Drug Deliv. Rev. 35, 61–76 (1999).
Weaver, J. C., Vaughan, T. E. & Chizmadzhev, Y. Theory of electrical creation of aqueous pathways across skin transport barriers. Adv. Drug Deliv. Rev. 35, 21–40 (1999).
Prausnitz, M. R., Pliquett, U. & Vanbever, R. in Electrochemotherapy, Electrogenetherapy, and Transdermal Drug Delivery (eds Jaroszeski, M. J., Heller, R. & Gilbert, R.) 213–245 (Humana Press, Totowa, 2000).
Vanbever, R. & Preat, V. In vivo efficacy and safety of skin electroporation. Adv. Drug Deliv. Rev. 35, 77–88 (1999).
Wallace, M. S. et al. Topical delivery of lidocaine in healthy volunteers by electroporation, electroincorporation, or iontophoresis: an evaluation of skin anesthesia. Reg. Anesth. Pain Med. 26, 229–238 (2001).
Prausnitz, M. R., Edelman, E. R., Gimm, J. A., Langer, R. & Weaver, J. C. Transdermal delivery of heparin by skin electroporation. Bio/Technology 13, 1205–1209 (1995).
Sen, A., Daly, M. E. & Hui, S. W. Transdermal insulin delivery using lipid enhanced electroporation. Biochim. Biophys. Acta 1564, 5–8 (2002).
Misra, A., Ganga, S. & Upadhyay, P. Needle-free, non-adjuvanted skin immunization by electroporation-enhanced transdermal delivery of diphtheria toxoid and a candidate peptide vaccine against hepatitis B virus. Vaccine 18, 517–523 (1999).
Zewert, T. E., Pliquett, U. F., Langer, R. & Weaver, J. C. Transdermal transport of DNA antisense oligonucleotides by electroporation. Biochem. Biophys. Res. Com. 212, 286–292 (1995).
Zhang, L., Li, L., Hofmann, G. A. & Hoffman, R. M. Depth-targeted efficient gene delivery and expression in the skin by pulsed electric fields: an approach to gene therapy of skin aging and other diseases. Biochem. Biophys. Res. Comm. 220, 633–636 (1996).
Hofmann, G. A., Rustrum, W. V. & Suder, K. S. Electro-incorporation of microcarriers as a method for the transdermal delivery of large molecules. Bioelectrochem. Bioenerg. 38, 209–222 (1995).
Prausnitz, M. R., Pliquett, U., Langer, R. & Weaver, J. C. Rapid temporal control of transdermal drug delivery by electroporation. Pharm. Res. 11, 1834–1837 (1994).
Heller, R., Gilbert, R. & Jaroszeski, M. J. Clinical applications of electrochemotherapy. Adv. Drug Deliv. Rev. 35, 119–130 (1999).
Pliquett, U. Mechanistic studies of molecular transdermal transport due to skin electroporation. Adv. Drug Deliv. Rev. 35, 41–60 (1999).
Prausnitz, M. R. et al. Imaging of transport pathways across human stratum corneum during high-voltage and low-voltage electrical exposures. J. Pharm. Sci. 85, 1363–1370 (1996).
Fellinger, K. & Schmidt, J. Klinik and therapies des chromischen gelenkreumatismus. Maudrich Vienna, Austria 549–552 (1954).
Tyle, P. & Agrawala, P. Drug delivery by phonophoresis. Pharm. Res. 6, 355–361 (1989).
Merino, G., Kalia, Y. N. & Guy, R. H. Ultrasound-enhanced transdermal transport. J. Pharm. Sci. 92, 1125–1137 (2003).
Mitragotri, S., Blankschtein, D. & Langer, R. Ultrasound-mediated transdermal protein delivery. Science 269, 850–853 (1995). Study demonstrating transdermal protein delivery using ultrasound.
Mitragotri, S. & Kost, J. Transdermal delivery of heparin and low-molecular weight heparin using low-frequency ultrasound. Pharm. Res. 18, 1151–1156 (2000).
Tachibana, K. Transdermal delivery of insulin to alloxan-diabetc rabits by ultrasound exposure. Pharm. Res. 9, 952–954 (1992).
Mitragotri, S. & Kost, J. Low-frequency sonophoresis: a non-invasive method for drug delivery and diagnostics. Biotech. Progress 16, 488–492 (2000).
Kost, J., Mitragotri, S., Gabbay, R., Pishko, M. & Langer, R. Transdermal extraction of glucose and other analytes using ultrasound. Nature Med. 6, 347–350 (2000).
Mitragotri, S., Edwards, D. A., Blankschtein, D. & Langer, R. A mechanistic study of ultrasonically enhanced transdermal drug delivery. J. Pharm. Sci. 84, 697–706 (1995).
Mitragotri, S., Blankschtein, D. & Langer, R. in Encyclopedia of Pharmaceutical Technology. (ed. Boylan, J.) 103–122 (Marcel Dekker, New York, 1996).
Menon, G. K., Kollias, N. & Doukas, A. G. Ultrastructural evidence of stratum corneum permeabilization induced by photomechanical waves. J. Invest. Dermatol. 121, 104–109 (2003).
Lee, S., McAuliffe, D. J., Mulholland, S. E. & Doukas, A. G. Photomechanical transdermal delivery of insulin in vivo. Lasers Surg. Med. 28, 282–285 (2001).
McAllister, D. V., Allen, M. G. & Prausnitz, M. R. Microfabricated microneedles for gene and drug delivery. Annu. Rev. Biomed. Eng. 2, 289–313 (2000).
Henry, S., McAllister, D., Allen, M. G. & Prausnitz, M. R. Microfabricated microneedles: a novel method to increase transdermal drug delivery. J. Pharm. Sci. 87, 922–925 (1998).
McAllister, D. V. et al. Microfabricated needles for transdermal delivery of macromolecules and nanoparticles: fabrication methods and transport studies. Proc. Natl Acad. Sci. USA 100, 13755–13760 (2003). Study describing fabrication methods and transdermal delivery using microneedles.
Lin, W. et al. Transdermal delivery of antisense oligonucleotides with microprojection patch (Macroflux) technology. Pharm. Res. 18, 1789–1793 (2001).
Martanto, W. et al. Transdermal delivery of insulin using microneedles in vivo. Proc. Intl. Symp. Control. Rel. Bioact. Mater. A666 (2003).
Cormier, M. & Daddona, P. E. in Modified-Release Drug Delivery Technology (eds Rathbone, M. J., Hadgraft, J. & Roberts, M. S.) 589–598 (Marcel Dekker, New York, 2003).
Matriano, J. A. et al. Macroflux microprojection array patch technology: a new and efficient approach for intracutaneous immunization. Pharm. Res. 19, 63–70 (2002).
Mikszta, J. A. et al. Improved genetic immunization via micromechanical disruption of skin-barrier function and targeted epidermal delivery. Nature Med. 8, 415–419 (2002).
Kaushik, S. et al. Lack of pain associated with microfabricated microneedles. Anesth. Analg. 92, 502–504 (2001).
Gerstel, M. S. & Place, V. A. Drug delivery device. US Patent 3,964,482 (1976).
Sintov, A. C. et al. Radiofrequency-driven skin microchanneling as a new way for electrically assisted transdermal delivery of hydrophilic drugs. J. Control. Release 89, 311–320 (2003).
Bramson, J. et al. Enabling topical immunization via microporation: a novel method for pain-free and needle-free delivery of adenovirus-based vaccines. Gene Ther. 10, 251–260 (2003).
Gebhart, S. et al. Glucose sensing in transdermal body fluid collected under continuous vacuum pressure via micropores in the stratum corneum. Diabetes Technol. Ther. 5, 159–166 (2003).
Hingson, R. A. & Figge, F. H. A survey of the development of jet injection in parenteral therapy. Curr. Res. Anesth. Analg. 31, 361–366 (1952).
Bremseth, D. L. & Pass, F. Delivery of insulin by jet injection: recent observations. Diabetes Technol. Ther. 3, 225–232 (2001). Evaluation of jet injection for transdermal delivery.
Burkoth, T. L. et al. Transdermal and transmucosal powdered drug delivery. Crit. Rev. Ther. Drug Carrier Syst. 16, 331–384 (1999).
Mikkelsen Lynch, P. et al. A pharmacokinetic and tolerability evaluation of two continuous subcutaneous infusion systems compared to an oral controlled-release morphine. J. Pain Symptom Manage. 19, 348–356 (2000).
Mitragotri, S. Synergistic effect of enhancers for transdermal drug delivery. Pharm. Res. 17, 1354–1359 (2000). Overview of enhancer combinations for transdermal delivery.
Srinivasan, V., Su, M. H., Higuchi, W. I. & Behl, C. R. Iontophoresis of polypeptides: effect of ethanol pretreatment of human skin. J. Pharm. Sci. 79, 588–591 (1990).
Oh, S. Y., Jeong, S. Y., Park, T. G. & Lee, J. H. Enhanced transdermal delivery of AZT (Zidovudine) using iontophoresis and penetration enhancer. J. Control. Release 51, 161–168 (1998).
Wearley, L. & Chien, Y. W. Enhancement of the in vitro skin permeability of azidothymidine (AZT) via iontophoresis and chemical enhancers. Pharm. Res. 7, 34–40 (1990).
Ganga, S., Ramarao, J. & Singh, J. Effect of azone on the iontophoretic transdermal delivery of metoprolol tartarate through human epidermis in vitro. J. Control. Rel. 42, 57–64 (1996).
Bhatia, K. S., Gao, S. & Singh, J. Effetct of penetration enhancers and iontophoresis on the FT-IR spectroscopy and LHRH permeability. J. Control. Rel. 47, 81–89 (1997).
Johnson, M. E., Mitragotri, S., Patel, A., Blankschtein, D. & Langer, R. Synergistic effect of ultrasound and chemical enhancers on transdermal drug delivery. J. Pharm. Sci. 85, 670–679 (1996).
Mitragotri, S. et al. Synergistic effect of ultrasound and sodium lauryl sulfate on transdermal drug delivery. J. Pharm. Sci. 89, 892–900 (2000).
Le, L., Kost, J. & Mitragotri, S. Combined effect of low-frequency ultrasound and iontophoresis: applications for transdermal heparin delivery. Pharm. Res. 17, 1151–1154 (2000).
Vanbever, R., Prausnitz, M. R. & Preat, V. Macromolecules as novel transdermal transport enhancers for skin electroporation. Pharm. Res. 14, 638–644 (1997).
Zewert, T. E., Pliquett, U. F., Vanbever, R., Langer, R. & Weaver, J. C. Creation of transdermal pathways for macromolecule transport by skin electroporation and a low toxicity, pathway-enlarging molecule. Bioelectrochem. Bioenerg. 49, 11–20 (1999).
Bommanon, D. B., Tamada, J., Leung, L. & Potts, R. O. Effects of electroporation on transdermal iontophoretic delivery of leutinizing hormone releasing hormone. Pharm. Res. 11, 1809–1814 (1994).
Benowitz, N. L. Drug therapy. Pharmacologic aspects of cigarette smoking and nicotine addition. N. Engl. J. Med. 319, 1318–1330 (1988).
Centers Disease Control and Prevention. Cigarette smoking among adults — United States, 1988. MMWR Morb. Mortal. Wkly Rep. 40, 757–759, 765 (1991).
Gora, M. L. Nicotine transdermal systems. Ann. Pharmacother. 27, 742–750 (1993).
Cordoba-Diaz, M. et al. Validation protocol of an automated in-line flow-through diffusion equipment for in vitro permeation studies. J. Control. Release 69, 357–367 (2000).
Moody, R. P. Automated In Vitro Dermal Absorption (AIVDA): predicting skin permeation of atrazine with finite and infinite (swimming/bathing) exposure models. Toxicol. In Vitro 14, 467–474 (2000).
Karande, P. & Mitragotri, S. Dependence of skin permeability on contact area. Pharm. Res. 20, 257–263 (2003).
Cima, M., Chen, H. & Gyory, J. R. System and method for optimizing tissue barrier transfer of compounds. WO 02/06518 (2002).
Pritchard, J. F. et al. Making better drugs: decision gates in non-clinical drug development. Nature Rev. Drug Discov. 2, 542–553 (2003).
Leduc, S. Introduction of medicinal substances into the depth of tissues by electric current. Ann. d'Electrobiol. 3, 545–560 (1900).
Tachibana, K. & Tachibana, S. Transdermal delivery of insulin by ultrasonic vibration. J. Pharm. Pharmocol. 43, 270–271 (1991).
Lee, S., McAuliffe, D. J., Flotte, T. J., Kollias, N. & Doukas, A. G. Photomechanical transcutaneous delivery of macromolecules. J. Invest. Dermatol. 111, 925–929 (1998).
Acknowledgements
We thank R. Gale for helpful discussions. This work was supported in part by National Institutes of Health grants.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
M.R.P. owns shares in BioValve Technologies. S.M. owns shares and is an advisor or consultant to Sontra Medical Corp. and fqubed. R.L. owns shares and is an advisor to Sontra Medical Corp. and TransForm Pharmaceuticals. All authors are inventors on patents on the subject of transdermal delivery that are owned by their current or former academic or industrial employers.
Related links
Glossary
- TRANSDERMAL DELIVERY
-
The movement of compounds across the stratum corneum and into systemic circulation.
- STRATUM CORNEUM
-
The outer layer of epidermis, consisting of several layers of corneocytes in a lipid-rich matrix.
- EPIDERMIS
-
The outer, epithelial portion of the skin.
- CORNEOCYTE
-
The non-living, keratin-filled squamous cell of the stratum corneum.
- DESQUAMATED
-
Shed from the surface of the skin.
- SURFACTANT
-
A molecule typically containing separate hydrophilic and hydrophobic domains that reduces surface tension of water.
- OLEIC ACID
-
A carboxylic acid with a linear chain of 18 carbon atoms and one double bond (C18H34O2).
- TOPICAL DELIVERY
-
The movement of compounds across the stratum corneum and locally into the skin.
- IONTOPHORESIS
-
The movement of molecules across the skin or other tissue under the influence of an electric field.
- HYPERHIDROSIS
-
Excessive sweating, especially of the hands and feet.
- ELECTROPHORESIS
-
The migration of molecules with a net charge under the influence of an electric field.
- ELECTRO-OSMOSIS
-
The movement in an electric field of liquid within a porous medium having a fixed net charge.
- ELECTROPORATION
-
The formation of aqueous pathways across a lipid bilayer by a pulsed electric field.
- ULTRASOUND
-
A sound (that is, pressure) wave at a frequency greater than 20 kHz.
- SONOPHORESIS
-
The movement of molecules across the skin or other tissue under the influence of an acoustic field.
- CAVITATION
-
The formation of gaseous bubbles within a liquid by ultrasound or other mechanical forces.
- MICRONEEDLE
-
A needle of micrometre dimensions usually fabricated using techniques derived from the microelectronics industry.
- LANGERHANS' CELL
-
Dendritic clear cells in the epidermis believed to be antigen fixing and processing cells of monocytic origin.
- THERMAL PORATION
-
The formation of aqueous pathways across stratum corneum by the application of pulsed heat.
- JET INJECTION
-
The high-velocity penetration into or across the skin of liquid droplets (or solid particles) often containing a drug.
Rights and permissions
About this article
Cite this article
Prausnitz, M., Mitragotri, S. & Langer, R. Current status and future potential of transdermal drug delivery. Nat Rev Drug Discov 3, 115–124 (2004). https://doi.org/10.1038/nrd1304
Issue Date:
DOI: https://doi.org/10.1038/nrd1304
This article is cited by
-
Mesoporous silica coated spicules for photodynamic therapy of metastatic melanoma
Journal of Nanobiotechnology (2024)
-
Non-invasive transdermal delivery of biomacromolecules with fluorocarbon-modified chitosan for melanoma immunotherapy and viral vaccines
Nature Communications (2024)
-
Fabrication and characterization of coated microneedle patches based on PEGDA for transdermal administration of metformin
Drug Delivery and Translational Research (2024)
-
Nanocrystals and nanosuspensions: an exploration from classic formulations to advanced drug delivery systems
Drug Delivery and Translational Research (2024)
-
AgNPs-Modified Polylactic Acid Microneedles: Preparation and In Vivo/In Vitro Antimicrobial Studies
Pharmaceutical Research (2024)