Abstract
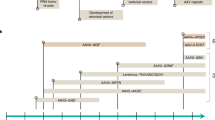
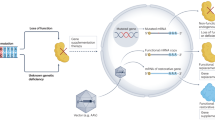
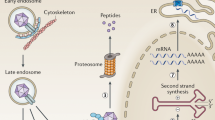
Adeno-associated viral (AAV) vectors are a rapidly emerging gene therapy platform for the treatment of neurological diseases. In preclinical studies, transgenes encoding therapeutic proteins, microRNAs, antibodies or gene-editing machinery have been successfully delivered to the central nervous system with natural or engineered viral capsids via various routes of administration. Importantly, initial clinical studies have demonstrated encouraging safety and efficacy in diseases such as Parkinson disease and spinal muscular atrophy, as well as durability of transgene expression. Here, we discuss key considerations and challenges in the future design and development of therapeutic AAV vectors, highlighting the most promising targets and recent clinical advances.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
12 September 2018
Details related to the copyright permissions for the images shown in Figure 3 have been added to the figure legend.
References
Mendell, J. R. et al. Single-dose gene-replacement therapy for spinal muscular atrophy. N. Engl. J. Med. 377, 1713–1722 (2017). This seminal clinical study demonstrates the safety and early efficacy of the first intravenously administered AAV-based therapy designed to correct the SMN deficiency in the CNS.
Dominguez, E. et al. Intravenous scAAV9 delivery of a codon-optimized SMN1 sequence rescues SMA mice. Hum. Mol. Genet. 20, 681–693 (2011).
Foust, K. D. et al. Rescue of the spinal muscular atrophy phenotype in a mouse model by early postnatal delivery of SMN. Nat. Biotechnol. 28, 271–274 (2010).
Thomsen, G. M. et al. Delayed disease onset and extended survival in the SOD1G93A rat model of amyotrophic lateral sclerosis after suppression of mutant SOD1 in the motor cortex. J. Neurosci. 34, 15587–15600 (2014).
Borel, F. et al. Therapeutic rAAVrh10 mediated SOD1 silencing in adult SOD1(G93A) mice and nonhuman primates. Hum. Gene Ther. 27, 19–31 (2016).
Foust, K. D. et al. Therapeutic AAV9-mediated suppression of mutant SOD1 slows disease progression and extends survival in models of inherited ALS. Mol. Ther. 21, 2148–2159 (2013).
McBride, J. L. et al. Artificial miRNAs mitigate shRNA-mediated toxicity in the brain: implications for the therapeutic development of RNAi. Proc. Natl Acad. Sci. USA 105, 5868–5873 (2008).
Stanek, L. M. et al. Silencing mutant huntingtin by adeno-associated virus-mediated RNA interference ameliorates disease manifestations in the YAC128 mouse model of Huntington's disease. Hum. Gene Ther. 25, 461–474 (2014).
Boudreau, R. L. et al. Nonallele-specific silencing of mutant and wild-type huntingtin demonstrates therapeutic efficacy in Huntington's disease mice. Mol. Ther. 17, 1053–1063 (2009).
Hocquemiller, M., Giersch, L., Audrain, M., Parker, S. & Cartier, N. Adeno-associated virus-based gene therapy for CNS diseases. Hum. Gene Ther. 27, 478–496 (2016).
Katz, M. L. et al. AAV gene transfer delays disease onset in a TPP1-deficient canine model of the late infantile form of Batten disease. Sci. Transl Med. 7, 313ra180 (2015).
Rosenberg, J. B. et al. Comparative efficacy and safety of multiple routes of direct CNS administration of adeno-associated virus gene transfer vector serotype rh.10 expressing the human arylsulfatase A cDNA to nonhuman primates. Hum. Gene Ther. Clin. Dev. 25, 164–177 (2014).
Fu, H., Dirosario, J., Killedar, S., Zaraspe, K. & McCarty, D. M. Correction of neurological disease of mucopolysaccharidosis IIIB in adult mice by rAAV9 trans-blood-brain barrier gene delivery. Mol. Ther. 19, 1025–1033 (2011).
Weismann, C. M. et al. Systemic AAV9 gene transfer in adult GM1 gangliosidosis mice reduces lysosomal storage in CNS and extends lifespan. Hum. Mol. Genet. 24, 4353–4364 (2015).
Bailey, R. M., Armao, D., Nagabhushan Kalburgi, S. & Gray, S. J. Development of intrathecal AAV9 gene therapy for giant axonal neuropathy. Mol. Ther. Methods Clin. Dev. 9, 160–171 (2018).
Haurigot, V. et al. Whole body correction of mucopolysaccharidosis IIIA by intracerebrospinal fluid gene therapy. J. Clin. Invest. 123, 3254–3271 (2013).
Garg, S. K. et al. Systemic delivery of MeCP2 rescues behavioral and cellular deficits in female mouse models of Rett syndrome. J. Neurosci. 33, 13612–13620 (2013).
Gadalla, K. K. et al. Improved survival and reduced phenotypic severity following AAV9/MECP2 gene transfer to neonatal and juvenile male Mecp2 knockout mice. Mol. Ther. 21, 18–30 (2013).
Gadalla, K. K. E. et al. Development of a novel AAV gene therapy cassette with improved safety features and efficacy in a mouse model of Rett syndrome. Mol. Ther. Methods Clin. Dev. 5, 180–190 (2017).
Valori, C. F. et al. Systemic delivery of scAAV9 expressing SMN prolongs survival in a model of spinal muscular atrophy. Sci. Transl Med. 2, 35ra42 (2010).
Gerard, C. et al. An AAV9 coding for frataxin clearly improved the symptoms and prolonged the life of Friedreich ataxia mouse models. Mol. Ther. Methods Clin. Dev. 1, 14044 (2014).
Liu, W. et al. Vectored intracerebral immunization with the anti-tau monoclonal antibody PHF1 markedly reduces tau pathology in mutant tau transgenic mice. J. Neurosci. 36, 12425–12435 (2016).
Ising, C. et al. AAV-mediated expression of anti-tau scFvs decreases tau accumulation in a mouse model of tauopathy. J. Exp. Med. 214, 1227–1238 (2017).
Swiech, L. et al. In vivo interrogation of gene function in the mammalian brain using CRISPR-Cas9. Nat. Biotechnol. 33, 102–106 (2015).
Suzuki, K. et al. In vivo genome editing via CRISPR/Cas9 mediated homology-independent targeted integration. Nature 540, 144–149 (2016).
Davidson, B. L. et al. Recombinant adeno-associated virus type 2, 4, and 5 vectors: transduction of variant cell types and regions in the mammalian central nervous system. Proc. Natl Acad. Sci. USA 97, 3428–3432 (2000).
Klein, R. L., Dayton, R. D., Tatom, J. B., Henderson, K. M. & Henning, P. P. AAV8, 9, Rh10, Rh43 vector gene transfer in the rat brain: effects of serotype, promoter and purification method. Mol. Ther. 16, 89–96 (2008).
Hutson, T. H., Verhaagen, J., Yáñez-Muñoz, R. J. & Moon, L. D. F. Corticospinal tract transduction: a comparison of seven adeno-associated viral vector serotypes and a non-integrating lentiviral vector. Gene Ther. 19, 49–60 (2012).
Bartlett, J. S., Samulski, R. J. & McCown, T. J. Selective and rapid uptake of adeno-associated virus type 2 in brain. Hum. Gene Ther. 9, 1181–1186 (1998).
Burger, C. et al. Recombinant AAV viral vectors pseudotyped with viral capsids from serotypes 1, 2, and 5 display differential efficiency and cell tropism after delivery to different regions of the central nervous system. Mol. Ther. 10, 302–317 (2004).
Passini, M. A. Intracranial delivery of CLN2 reduces brain pathology in a mouse model of classical late infantile neuronal ceroid lipofuscinosis. J. Neurosci. 26, 1334–1342 (2006).
Passini, M. et al. Intraventricular brain injection of adeno-associated virus type 1 (AAV1) in neonatal mice results in complementary patterns of neuronal transduction to AAV2 and total long-term correction of storage lesions in the brains of β-glucuronidase-deficient mice. J. Virol. 77, 7034 (2003).
Federici, T. et al. Robust spinal motor neuron transduction following intrathecal delivery of AAV9 in pigs. Gene Ther. 19, 852–859 (2012).
Cearley, C. N. et al. Expanded repertoire of AAV vector serotypes mediate unique patterns of transduction in mouse brain. Mol. Ther. 16, 1710–1718 (2008).
Liu, G., Martins, I. H., Chiorini, J. A. & Davidson, B. L. Adeno-associated virus type 4 (AAV4) targets ependyma and astrocytes in the subventricular zone and RMS. Gene Ther. 12, 1503–1508 (2005).
Taymans, J.-M. et al. Comparative analysis of adeno-associated viral vector serotypes 1, 2, 5, 7, and 8 in mouse brain. Hum. Gene Ther. 18, 195–206 (2007).
Samaranch, L. et al. Strong cortical and spinal cord transduction after AAV7 and AAV9 delivery into the CSF of non-human primates. Hum. Gene Ther. 24, 526–532 (2013).
Vite, C. H., Passini, M. A., Haskins, M. E. & Wolfe, J. H. Adeno-associated virus vector-mediated transduction in the cat brain. Gene Ther. 10, 1874–1881 (2003).
Richardson, R. M. et al. T2 imaging in monitoring of intraparenchymal real-time convection-enhanced delivery. Neurosurgery 69, 154–163; discussion 163 (2011).
Passini, M. A. et al. Translational fidelity of intrathecal delivery of self-complementary AAV9-survival motor neuron 1 for spinal muscular atrophy. Hum. Gene Ther. 25, 619–630 (2014).
Miyanohara, A. et al. Potent spinal parenchymal AAV9-mediated gene delivery by subpial injection in adult rats and pigs. Mol. Ther. Methods Clin. Dev. 3, 16046 (2016).
Gibson, L. A. et al. Adeno-associated viral gene therapy using PHP.B:NPC1 ameliorates disease phenotype in mouse model of Niemann-Pick C1 disease. Mol. Ther. 25, 1–363 (2017).
Morabito, G. et al. AAV-PHP.B-mediated global-scale expression in the mouse nervous system enables GBA1 gene therapy for wide protection from synucleinopathy. Mol. Ther. 25, 2727–2742 (2017).
Balazs, A. B. et al. Antibody-based protection against HIV infection by vectored immunoprophylaxis. Nature 481, 81–84 (2011).
Balazs, A. B., Bloom, J. D., Hong, C. M., Rao, D. S. & Baltimore, D. Broad protection against influenza infection by vectored immunoprophylaxis in mice. Nat. Biotechnol. 31, 647–652 (2013).
Deverman, B. E. et al. Cre-dependent selection yields AAV variants for widespread gene transfer to the adult brain. Nat. Biotechnol. 34, 204–209 (2016). A novel, cell-type-specific, functional in vivo selection system enables the identification of several engineered AAV capsids that provide up to 40-fold greater CNS transduction than the previous standard, AAV9, after IV administration in adult mice.
Ravina, B. et al. Intraputaminal AADC gene therapy for advanced Parkinson's disease: interim results of a phase 1b Trial [abstract]. Hum. Gene Ther. 28, OR12 (2017).
Hwu, W. L. et al. Gene therapy for aromatic L-amino acid decarboxylase deficiency. Sci. Transl Med. 4, 134ra61 (2012).
Mittermeyer, G. et al. Long-term evaluation of a phase 1 study of AADC gene therapy for Parkinson's disease. Hum. Gene Ther. 23, 377–381 (2012).
Sehara, Y. et al. Persistent expression of dopamine-synthesizing enzymes 15 years after gene transfer in a primate model of Parkinson's disease. Hum. Gene Ther. Clin. Dev. 28, 74–79 (2017).
Grieger, J. C., Soltys, S. M. & Samulski, R. J. Production of recombinant adeno-associated virus vectors using suspension HEK293 cells and continuous harvest of vector from the culture media for GMP FIX and FLT1 clinical vector. Mol. Ther. 24, 287–297 (2016).
Kotin, R. M. & Snyder, R. O. Manufacturing clinical grade recombinant adeno-associated virus using invertebrate cell lines. Hum. Gene Ther. 28, 350–360 (2017).
Cartier, N. et al. Hematopoietic stem cell gene therapy with a lentiviral vector in X-linked adrenoleukodystrophy. Science 326, 818–823 (2009).
Cartier, N. et al. Lentiviral hematopoietic cell gene therapy for X-linked adrenoleukodystrophy. Methods Enzymol. 507, 187–198 (2012).
Petrs-Silva, H. et al. High-efficiency transduction of the mouse retina by tyrosine-mutant AAV serotype vectors. Mol. Ther. 17, 463–471 (2009).
Lisowski, L. et al. Selection and evaluation of clinically relevant AAV variants in a xenograft liver model. Nature 506, 382–386 (2014).
Atchison, R. W., Casto, B. C. & Hammon, W. M. Adenovirus-associated defective virus particles. Science 149, 754–756 (1965). The first identification of small viruses now known as AAVs as contaminants of adenovirus preparations.
Hoggan, M. D., Blacklow, N. R. & Rowe, W. P. Studies of small DNA viruses found in various adenovirus preparations: physical, biological, and immunological characteristics. Proc. Natl Acad. Sci. USA 55, 1467–1474 (1966).
Parks, W. P., Green, M., Pina, M. & Melnick, J. L. Physicochemical characterization of adeno-associated satellite virus type 4 and its nucleic acid. J. Virol. 1, 980–987 (1967).
Chiorini, J. A., Yang, L., Liu, Y., Safer, B. & Kotin, R. M. Cloning of adeno-associated virus type 4 (AAV4) and generation of recombinant AAV4 particles. J. Virol. 71, 6823–6833 (1997).
Handa, A., Muramatsu, S., Qiu, J., Mizukami, H. & Brown, K. E. Adeno-associated virus (AAV)-3-based vectors transduce haematopoietic cells not susceptible to transduction with AAV-2-based vectors. J. Gen. Virol. 81, 2077–2084 (2000).
Gao, G. et al. Clades of adeno-associated viruses are widely disseminated in human tissues. J. Virol. 78, 6381–6388 (2004).
Gao, G. et al. Adeno-associated viruses undergo substantial evolution in primates during natural infections. Proc. Natl Acad. Sci. USA 100, 6081–6086 (2003).
Sabatino, D. E. et al. Identification of mouse AAV capsid-specific CD8+ T cell epitopes. Mol. Ther. 12, 1023–1033 (2005).
Arbetman, A. E. et al. Novel caprine adeno-associated virus (AAV) capsid (AAV-Go.1) is closely related to the primate AAV-5 and has unique tropism and neutralization properties. J. Virol. 79, 15238–15245 (2005).
Markakis, E. A. et al. Comparative transduction efficiency of AAV vector serotypes 1–6 in the substantia nigra and striatum of the primate brain. Mol. Ther. 18, 588–593 (2010).
Bevan, A. K. et al. Systemic gene delivery in large species for targeting spinal cord, brain, and peripheral tissues for pediatric disorders. Mol. Ther. 19, 1971–1980 (2011).
Gray, S. J., Nagabhushan Kalburgi, S., McCown, T. J. & Jude Samulski, R. Global CNS gene delivery and evasion of anti-AAV-neutralizing antibodies by intrathecal AAV administration in non-human primates. Gene Ther. 20, 450–459 (2013).
Samaranch, L. et al. Adeno-associated virus serotype 9 transduction in the central nervous system of nonhuman primates. Hum. Gene Ther. 23, 382–389 (2012).
Hinderer, C. et al. Evaluation of intrathecal routes of administration for adeno-associated viral vectors in large animals. Hum. Gene Ther. 29, 15–24 (2018).
Hinderer, C. et al. Widespread gene transfer in the central nervous system of cynomolgus macaques following delivery of AAV9 into the cisterna magna. Mol. Ther. Methods Clin. Dev. 1, 14051 (2014).
Foust, K. D. et al. Intravascular AAV9 preferentially targets neonatal neurons and adult astrocytes. Nat. Biotechnol. 27, 59–65 (2009). This report is one of two studies to first demonstrate that AAV vectors can cross the BBB and provide widespread gene expression throughout the neonatal and adult CNS; this study focuses on the mouse.
Duque, S. et al. Intravenous administration of self-complementary AAV9 enables transgene delivery to adult motor neurons. Mol. Ther. 17, 1187–1196 (2009). This report is one of two studies to first demonstrate that AAV vectors can cross the BBB and provide widespread gene expression throughout the neonatal and adult CNS; this study includes the cat.
Wang, D. B. et al. Expansive gene transfer in the rat CNS rapidly produces amyotrophic lateral sclerosis relevant sequelae when TDP-43 is overexpressed. Mol. Ther. 18, 2064–2074 (2010).
Gray, S. J. et al. Preclinical differences of intravascular AAV9 delivery to neurons and glia: a comparative study of adult mice and nonhuman primates. Mol. Ther. 19, 1058–1069 (2011). This study highlights the species-specific tropism differences between mouse and NHPs that may complicate translational efforts of engineered AAV capsids.
Samaranch, L. et al. AAV9 transduction in the central nervous system of non-human primates. Hum. Gene Ther. 23, 382–389 (2012).
Huang, L. Y., Halder, S. & Agbandje-McKenna, M. Parvovirus glycan interactions. Curr. Opin. Virol. 7, 108–118 (2014).
Masamizu, Y. et al. Local and retrograde gene transfer into primate neuronal pathways via adeno-associated virus serotype 8 and 9. Neuroscience 193, 249–258 (2011).
Huang, L. Y. et al. Characterization of the adeno-associated virus 1 and 6 sialic acid binding site. J. Virol. 90, 5219–5230 (2016).
Kern, A. et al. Identification of a heparin-binding motif on adeno-associated virus type 2 capsids. J. Virol. 77, 11072–11081 (2003).
Opie, S. R. et al. Identification of amino acid residues in the capsid proteins of adeno-associated virus type 2 that contribute to heparan sulfate proteoglycan binding. J. Virol. 77, 6995–7006 (2003).
Wu, Z. et al. Single amino acid changes can influence titer, heparin binding, and tissue tropism in different adeno-associated virus serotypes. J. Virol. 80, 11393–11397 (2006).
Shen, S., Bryant, K. D., Brown, S. M., Randell, S. H. & Asokan, A. Terminal N-linked galactose is the primary receptor for adeno-associated virus 9. J. Biol. Chem. 286, 13532–13540 (2011).
Bell, C. L., Gurda, B. L., Van Vliet, K., Agbandje-Mckenna, M. & Wilson, J. M. Identification of the galactose binding domain of the AAV9 capsid. J. Virol. 86, 7326–7333 (2012).
Murlidharan, G., Corriher, T., Ghashghaei, H. T. & Asokan, A. Unique glycan signatures regulate adeno-associated virus tropism in the developing brain. J. Virol. 89, 3976–3987 (2015).
Adachi, K., Enoki, T., Kawano, Y., Veraz, M. & Nakai, H. Drawing a high-resolution functional map of adeno-associated virus capsid by massively parallel sequencing. Nat. Commun. 5, 3075 (2014). In this tour de force study, the authors use alanine-scanning mutagenesis and AAV genome barcoding to map onto the linear AAV capsid sequence key functional features, including glycan binding, circulating half-life and liver and other organ transduction.
Albright, B. H. et al. Mapping the structural determinants required for AAVrh.10 transport across the blood-brain barrier. Mol. Ther. 26, 510–523 (2018).
Grimm, D. et al. In vitro and in vivo gene therapy vector evolution via multispecies interbreeding and retargeting of adeno-associated viruses. J. Virol. 82, 5887–5911 (2008).
Li, W. et al. Engineering and selection of shuffled AAV genomes: a new strategy for producing targeted biological nanoparticles. Mol. Ther. 16, 1252–1260 (2008).
Perabo, L. et al. Combinatorial engineering of a gene therapy vector: directed evolution of adeno-associated virus. J. Gene Med. 8, 155–162 (2006).
Excoffon, K. J. D. A. et al. Directed evolution of adeno-associated virus to an infectious respiratory virus. Proc. Natl Acad. Sci. USA 106, 3865–3870 (2009).
Maheshri, N., Koerber, J. T., Kaspar, B. K. & Schaffer, D. V. Directed evolution of adeno-associated virus yields enhanced gene delivery vectors. Nat. Biotechnol. 24, 198–204 (2006).
Muller, O. J. et al. Random peptide libraries displayed on adeno-associated virus to select for targeted gene therapy vectors. Nat. Biotechnol. 21, 1040–1046 (2003).
Tervo, D. G. et al. A designer AAV variant permits efficient retrograde access to projection neurons. Neuron 92, 372–382 (2016).
Dalkara, D. et al. In vivo-directed evolution of a new adeno-associated virus for therapeutic outer retinal gene delivery from the vitreous. Sci. Transl Med. 5, 189ra76 (2013).
Korbelin, J. et al. A brain microvasculature endothelial cell-specific viral vector with the potential to treat neurovascular and neurological diseases. EMBO Mol. Med. 8, 609–625 (2016).
Koerber, J. T., Maheshri, N., Kaspar, B. K. & Schaffer, D. V. Construction of diverse adeno-associated viral libraries for directed evolution of enhanced gene delivery vehicles. Nat. Protoc. 1, 701–706 (2006).
Pulicherla, N. et al. Engineering liver-detargeted AAV9 vectors for cardiac and musculoskeletal gene transfer. Mol. Ther. 19, 1070–1078 (2011).
Ojala, D. S. et al. In vivo selection of a computationally designed SCHEMA AAV library yields a novel variant for infection of adult neural stem cells in the SVZ. Mol. Ther. 26, 304–319 (2018).
Chan, K. Y. et al. Engineered AAVs for efficient noninvasive gene delivery to the central and peripheral nervous systems. Nat. Neurosci. 20, 1172–1179 (2017).
Gao, Y., Geng, L., Chen, V. P. & Brimijoin, S. Therapeutic delivery of butyrylcholinesterase by brain-wide viral gene transfer to mice. Molecules 22, E1145 (2017).
Allen, W. E. et al. Global representations of goal-directed behavior in distinct cell types of mouse neocortex. Neuron 94, 891–907.e6 (2017).
Jackson, K. L., Dayton, R. D., Deverman, B. E. & Klein, R. L. Better targeting, better efficiency for wide-scale neuronal transduction with the synapsin promoter and AAV-PHP.B. Front. Mol. Neurosci. 9, 116 (2016).
Matsuzaki, Y. et al. Intravenous administration of the adeno-associated virus-PHP.B capsid fails to upregulate transduction efficiency in the marmoset brain. Neurosci. Lett. 665, 182–188 (2018).
Hordeaux, J. et al. The neurotropic properties of AAV-PHP.B are limited to C57BL/6J mice. Mol. Ther. 26, 664–668 (2018).
Samaranch, L. et al. AAV9-mediated expression of a non-self protein in nonhuman primate central nervous system triggers widespread neuroinflammation driven by antigen-presenting cell transduction. Mol. Ther. 22, 329–337 (2014). This study highlights the risk of expressing the GFP transgene in the CNS with inflammation and immune responses as well as ataxia after direct CNS delivery in NHPs.
Sah, D. et al. Translation of intravenous delivery of AAV gene therapy for the treatment of CNS diseases [abstract]. Hum. Gene Ther. 28, P209 (2017).
Sah, D. et al. Safety and increased transduction efficiency in the adult nonhuman primate central nervous system with intravenous delivery of two novel adeno-associated virus capsids [abstract 661]. Mol. Ther. (2018).
Ferrari, F. K., Samulski, T., Shenk, T. & Samulski, R. J. Second-strand synthesis is a rate-limiting step for efficient transduction by recombinant adeno-associated virus vectors. J. Virol. 70, 3227–3234 (1996).
Chen, Y. H., Claflin, K., Geoghegan, J. C. & Davidson, B. L. Sialic acid deposition impairs the utility of AAV9, but not peptide-modified AAVs for brain gene therapy in a mouse model of lysosomal storage disease. Mol. Ther. 20, 1393–1399 (2012).
Lawlor, P. A., Bland, R. J., Mouravlev, A., Young, D. & During, M. J. Efficient gene delivery and selective transduction of glial cells in the mammalian brain by AAV serotypes isolated from nonhuman primates. Mol. Ther. 17, 1692–1702 (2009).
von Jonquieres, G. et al. Glial promoter selectivity following AAV-delivery to the immature brain. PLoS ONE 8, e65646 (2013).
Dirren, E. et al. Intracerebroventricular injection of adeno-associated virus 6 and 9 vectors for cell type-specific transgene expression in the spinal cord. Hum. Gene Ther. 25, 109–120 (2014).
Zolotukhin, S. et al. Recombinant adeno-associated virus purification using novel methods improves infectious titer and yield. Gene Ther. 6, 973–985 (1999). This paper describes a simple and universally applicable purification protocol for AAV comprising an iodixanol step gradient, which has become a commonly used alternative to caesium chloride gradients.
Perdomini, M. et al. Prevention and reversal of severe mitochondrial cardiomyopathy by gene therapy in a mouse model of Friedreich's ataxia. Nat. Med. 20, 542–547 (2014).
Miniarikova, J. et al. Design, characterization, and lead selection of therapeutic miRNAs targeting huntingtin for development of gene therapy for Huntington's disease. Mol. Ther. Nucleic Acids 5, e297 (2016).
Monteys, A. M., Ebanks, S. A., Keiser, M. S. & Davidson, B. L. CRISPR/Cas9 editing of the mutant Huntingtin allele in vitro and in vivo. Mol. Ther. 25, 12–23 (2017).
McPhee, S. W. et al. Immune responses to AAV in a phase I study for Canavan disease. J. Gene Med. 8, 577–588 (2006).
Leone, P. et al. Long-term follow-up after gene therapy for canavan disease. Sci. Transl Med. 4, 165ra163 (2012).
Eberling, J. L. et al. Results from a phase I safety trial of hAADC gene therapy for Parkinson disease. Neurology 70, 1980–1983 (2008). This article presents the initial results from one of the first CNS-targeted gene therapy trials and highlights the safety of IPa AAV delivery and AADC transgene expression in patients with PD.
LeWitt, P. A. et al. AAV2-GAD gene therapy for advanced Parkinson's disease: a double-blind, sham-surgery controlled, randomised trial. Lancet Neurol. 10, 309–319 (2011).
Bartus, R. T. et al. Issues regarding gene therapy products for Parkinson's disease: the development of CERE-120 (AAV-NTN) as one reference point. Parkinsonism Relat. Disord. 13 (Suppl. 3), S469–S477 (2007).
Limberis, M. P. et al. Intranasal antibody gene transfer in mice and ferrets elicits broad protection against pandemic influenza. Sci. Transl Med. 5, 187ra72 (2013).
Huang, X. et al. Genome editing abrogates angiogenesis in vivo. Nat. Commun. 8, 112 (2017).
Yu, W. et al. Nrl knockdown by AAV-delivered CRISPR/Cas9 prevents retinal degeneration in mice. Nat. Commun. 8, 14716 (2017).
Laoharawee, K. et al. Dose-dependent prevention of metabolic and neurologic disease in murine MPS II by ZFN-mediated in vivo genome editing. Mol. Ther. 26, 1127–1136 (2018).
Zeitler, B. J. et al. Designed zinc finger protein transcription factors for single-gene regulation throughout the central nervous system [abstract 949]. Mol. Ther. (2018).
Charlesworth, C. T. et al. Identification of pre-existing adaptive immunity to Cas9 proteins in humans. Preprint at bioRxiv https://doi.org/10.1101/243345 (2018).
van Dyck, C. H. Anti-amyloid-beta monoclonal antibodies for Alzheimer's disease: pitfalls and promise. Biol. Psychiatry 83, 311–319 (2018).
Paul, S. M. Therapeutic antibodies for brain disorders. Sci. Transl Med. 3, 84ps20 (2011).
Chavez, A. et al. Highly efficient Cas9-mediated transcriptional programming. Nat. Methods 12, 326–328 (2015).
Hilton, I. B. et al. Epigenome editing by a CRISPR-Cas9-based acetyltransferase activates genes from promoters and enhancers. Nat. Biotechnol. 33, 510–517 (2015).
Pawluk, A. et al. Naturally occurring off-switches for CRISPR-Cas9. Cell 167, 1829–1838.e9 (2016).
Kleinstiver, B. P. et al. High-fidelity CRISPR-Cas9 nucleases with no detectable genome-wide off-target effects. Nature 529, 490–495 (2016).
Zetsche, B. et al. Multiplex gene editing by CRISPR-Cpf1 using a single crRNA array. Nat. Biotechnol. 35, 31–34 (2017).
Chen, J. S. et al. Enhanced proofreading governs CRISPR-Cas9 targeting accuracy. Nature 550, 407–410 (2017).
Gao, L. et al. Engineered Cpf1 variants with altered PAM specificities. Nat. Biotechnol. 35, 789–792 (2017).
Cox, D. B. T. et al. RNA editing with CRISPR-Cas13. Science 358, 1019–1027 (2017).
Abudayyeh, O. O. et al. C2c2 is a single-component programmable RNA-guided RNA-targeting CRISPR effector. Science 353, aaf5573 (2016).
Li, X. et al. Base editing with a Cpf1-cytidine deaminase fusion. Nat. Biotechnol. 36, 324–327 (2018).
Komor, A. C., Kim, Y. B., Packer, M. S., Zuris, J. A. & Liu, D. R. Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Nature 533, 420–424 (2016).
Gaudelli, N. M. et al. Programmable base editing of A*T to G*C in genomic DNA without DNA cleavage. Nature 551, 464–471 (2017).
Harmatz, P. et al. Update on phase 1/2 clinical trials for MPS I and MPS II using ZFN-mediated in vivo genome editing. Mol. Genet. Metab. 123, S59–S60 (2018).
Cullen, B. R. RNAi the natural way. Nat. Genet. 37, 1163–1165 (2005).
Keiser, M. S., Geoghegan, J. C., Boudreau, R. L., Lennox, K. A. & Davidson, B. L. RNAi or overexpression: alternative therapies for spinocerebellar ataxia type 1. Neurobiol. Dis. 56, 6–13 (2013).
Rodriguez-Lebron, E. et al. Silencing mutant ATXN3 expression resolves molecular phenotypes in SCA3 transgenic mice. Mol. Ther. 21, 1909–1918 (2013).
Tardieu, M. et al. Intracerebral administration of adeno-associated viral vector serotype rh.10 carrying human SGSH and SUMF1 cDNAs in children with mucopolysaccharidosis type IIIA disease: results of a phase I/II trial. Hum. Gene Ther. 25, 506–516 (2014).
Lee, Y., Messing, A., Su, M. & Brenner, M. GFAP promoter elements required for region-specific and astrocyte-specific expression. Glia 56, 481–493 (2008).
Dimidschstein, J. et al. A viral strategy for targeting and manipulating interneurons across vertebrate species. Nat. Neurosci. 19, 1743–1749 (2016).
de Leeuw, C. N. et al. rAAV-compatible MiniPromoters for restricted expression in the brain and eye. Mol. Brain 9, 52 (2016).
Gray, S. J. et al. Optimizing promoters for recombinant adeno-associated virus-mediated gene expression in the peripheral and central nervous system using self-complementary vectors. Hum. Gene Ther. 22, 1143–1153 (2011).
Tornoe, J., Kusk, P., Johansen, T. E. & Jensen, P. R. Generation of a synthetic mammalian promoter library by modification of sequences spacing transcription factor binding sites. Gene 297, 21–32 (2002).
Adriaansen, J. et al. Reduction of arthritis following intra-articular administration of an adeno-associated virus serotype 5 expressing a disease-inducible TNF-blocking agent. Ann. Rheum. Dis. 66, 1143–1150 (2007).
Geurts, J. et al. Application of a disease-regulated promoter is a safer mode of local IL-4 gene therapy for arthritis. Gene Ther. 14, 1632–1638 (2007).
Chtarto, A. et al. An adeno-associated virus-based intracellular sensor of pathological nuclear factor-kappaB activation for disease-inducible gene transfer. PLoS ONE 8, e53156 (2013).
Bockstael, O. et al. Intracisternal delivery of NFkappaB-inducible scAAV2/9 reveals locoregional neuroinflammation induced by systemic kainic acid treatment. Front. Mol. Neurosci. 7, 92 (2014).
Shen, F. et al. Adeno-associated viral vector-mediated hypoxia-regulated VEGF gene transfer promotes angiogenesis following focal cerebral ischemia in mice. Gene Ther. 15, 30–39 (2008).
Huang, M. T. & Gorman, C. M. Intervening sequences increase efficiency of RNA 3′ processing and accumulation of cytoplasmic RNA. Nucleic Acids Res. 18, 937–947 (1990).
Lacy-Hulbert, A. et al. Interruption of coding sequences by heterologous introns can enhance the functional expression of recombinant genes. Gene Ther. 8, 649–653 (2001).
Choi, J. H. et al. Optimization of AAV expression cassettes to improve packaging capacity and transgene expression in neurons. Mol. Brain 7, 17 (2014).
Xie, J. et al. MicroRNA-regulated, systemically delivered rAAV9: a step closer to CNS-restricted transgene expression. Mol. Ther. 19, 526–535 (2010).
Majowicz, A. et al. Mir-142-3p target sequences reduce transgene-directed immunogenicity following intramuscular adeno-associated virus 1 vector-mediated gene delivery. J. Gene Med. 15, 219–232 (2013).
Ferreira, J. P., Overton, K. W. & Wang, C. L. Tuning gene expression with synthetic upstream open reading frames. Proc. Natl Acad. Sci. USA 110, 11284–11289 (2013).
Ward, N. J. et al. Codon optimization of human factor VIII cDNAs leads to high-level expression. Blood 117, 798–807 (2011).
Radcliffe, P. A. et al. Analysis of factor VIII mediated suppression of lentiviral vector titres. Gene Ther. 15, 289–297 (2008).
Weinberg, M. S., Samulski, R. J. & McCown, T. J. Adeno-associated virus (AAV) gene therapy for neurological disease. Neuropharmacology 69, 82–88 (2013).
Hadaczek, P. et al. Widespread AAV1- and AAV2-mediated transgene expression in the nonhuman primate brain: implications for Huntington's disease. Mol. Ther. Methods Clin. Dev. 3, 16037 (2016).
Kells, A. P. et al. Efficient gene therapy-based method for the delivery of therapeutics to primate cortex. Proc. Natl Acad. Sci. USA 106, 2407–2411 (2009).
Cearley, C. N. & Wolfe, J. H. A single injection of an adeno-associated virus vector into nuclei with divergent connections results in widespread vector distribution in the brain and global correction of a neurogenetic disease. J. Neurosci. 27, 9928–9940 (2007).
Castle, M. J., Gershenson, Z. T., Giles, A. R., Holzbaur, E. L. & Wolfe, J. H. Adeno-associated virus serotypes 1, 8, and 9 share conserved mechanisms for anterograde and retrograde axonal transport. Hum. Gene Ther. 25, 705–720 (2014).
Green, F. et al. Axonal transport of AAV9 in nonhuman primate brain. Gene Ther. 23, 520–526 (2016).
Zerah, M. et al. Intracerebral gene therapy using AAVrh.10-hARSA recombinant vector to treat patients with early-onset forms of metachromatic leukodystrophy: preclinical feasibility and safety assessments in nonhuman primates. Hum. Gene Ther. Clin. Dev. 26, 113–124 (2015).
Tardieu, M. et al. Intracerebral gene therapy in children with mucopolysaccharidosis type IIIB syndrome: an uncontrolled phase 1/2 clinical trial. Lancet Neurol. 16, 712–720 (2017).
Lieberman, D. M., Laske, D. W., Morrison, P. F., Bankiewicz, K. S. & Oldfield, E. H. Convection-enhanced distribution of large molecules in gray matter during interstitial drug infusion. J. Neurosurg. 82, 1021–1029 (1995).
Kanaan, N. M. et al. Rationally engineered AAV capsids improve transduction and volumetric spread in the CNS. Mol. Ther. Nucleic Acids 8, 184–197 (2017).
Hinderer, C. et al. Intrathecal gene therapy corrects CNS pathology in a feline model of mucopolysaccharidosis I. Mol. Ther. 22, 2018–2027 (2014).
Zhang, H. et al. Several rAAV vectors efficiently cross the blood–brain barrier and transduce neurons and astrocytes in the neonatal mouse central nervous system. Mol. Ther. 19, 1440–1448 (2011).
Yang, B. et al. Global CNS transduction of adult mice by intravenously delivered rAAVrh.8 and rAAVrh.10 and nonhuman primates by rAAVrh.10. Mol. Ther. 22, 1299–1309 (2014).
Varadi, K. et al. Novel random peptide libraries displayed on AAV serotype 9 for selection of endothelial cell-directed gene transfer vectors. Gene Ther. 19, 800–809 (2012).
Hinderer, C. et al. Severe toxicity in nonhuman primates and piglets following high-dose intravenous administration of an adeno-associated virus vector expressing human SMN. Hum. Gene Ther. 29, 285–298 (2018).
Kondratov, O. et al. Direct head-to-head evaluation of recombinant adeno-associated viral vectors manufactured in human versus insect cells. Mol. Ther. 25, 2661–2675 (2017).
Adamson-Small, L. et al. A scalable method for the production of high-titer and high-quality adeno-associated type 9 vectors using the HSV platform. Mol. Ther. Methods Clin. Dev. 3, 16031 (2016).
Conway, J. E. et al. High-titer recombinant adeno-associated virus production utilizing a recombinant herpes simplex virus type I vector expressing AAV-2 Rep and Cap. Gene Ther. 6, 986–993 (1999).
Clément, N., Knop, D. R. & Byrne, B. J. Large-scale adeno-associated viral vector production using a herpesvirus-based system enables manufacturing for clinical studies. Hum. Gene Ther. 20, 796–806 (2009).
Liu, X., Voulgaropoulou, F., Chen, R., Johnson, P. R. & Clark, K. R. Selective Rep-Cap gene amplification as a mechanism for high-titer recombinant AAV production from stable cell lines. Mol. Ther. 2, 394–403 (2000).
Clark, K. R., Voulgaropoulou, F. & Johnson, P. R. A stable cell line carrying adenovirus-inducible rep and cap genes allows for infectivity titration of adeno-associated virus vectors. Gene Ther. 3, 1124–1132 (1996).
Clark, K. R., Voulgaropoulou, F., Fraley, D. M. & Johnson, P. R. Cell lines for the production of recombinant adeno-associated virus. Hum. Gene Ther. 6, 1329–1341 (1995).
Dunbar, C. E. et al. Gene therapy comes of age. Science 359, eaan4672 (2018).
Zhao, L. et al. Intracerebral adeno-associated virus gene delivery of apolipoprotein E2 markedly reduces brain amyloid pathology in Alzheimer's disease mouse models. Neurobiol. Aging 44, 159–172 (2016).
Bartus, R. T. & Johnson, E. M. Jr. Clinical tests of neurotrophic factors for human neurodegenerative diseases, part 1: where have we been and what have we learned? Neurobiol. Dis. 97, 156–168 (2017).
Glass, J. D. et al. Transplantation of spinal cord-derived neural stem cells for ALS: analysis of phase 1 and 2 trials. Neurology 87, 392–400 (2016).
Li, C. et al. Neutralizing antibodies against adeno-associated virus examined prospectively in pediatric patients with hemophilia. Gene Ther. 19, 288–294 (2012).
Halbert, C. L. et al. Prevalence of neutralizing antibodies against adeno-associated virus (AAV) types 2, 5, and 6 in cystic fibrosis and normal populations: implications for gene therapy using AAV vectors. Hum. Gene Ther. 17, 440–447 (2006).
Calcedo, R. et al. Adeno-associated virus antibody profiles in newborns, children, and adolescents. Clin. Vaccine Immunol. 18, 1586–1588 (2011).
Lugtenberg, D. et al. Chromosomal copy number changes in patients with non-syndromic X linked mental retardation detected by array CGH. J. Med. Genet. 43, 362–370 (2006).
Hanchard, N. A. et al. A partial MECP2 duplication in a mildly affected adult male: a putative role for the 3' untranslated region in the MECP2 duplication phenotype. BMC Med. Genet. 13, 71 (2012).
Friez, M. J. et al. Recurrent infections, hypotonia, and mental retardation caused by duplication of MECP2 and adjacent region in Xq28. Pediatrics 118, e1687-95 (2006).
Van Esch, H. et al. Duplication of the MECP2 region is a frequent cause of severe mental retardation and progressive neurological symptoms in males. Am. J. Hum. Genet. 77, 442–453 (2005).
Meins, M. et al. Submicroscopic duplication in Xq28 causes increased expression of the MECP2 gene in a boy with severe mental retardation and features of Rett syndrome. J. Med. Genet. 42, e12 (2005).
Ariani, F. et al. Real-time quantitative PCR as a routine method for screening large rearrangements in Rett syndrome: report of one case of MECP2 deletion and one case of MECP2 duplication. Hum. Mutat. 24, 172–177 (2004).
Na, E. S. et al. A mouse model for MeCP2 duplication syndrome: MeCP2 overexpression impairs learning and memory and synaptic transmission. J. Neurosci. 32, 3109–3117 (2012).
Nasir, J. et al. Targeted disruption of the Huntington's disease gene results in embryonic lethality and behavioral and morphological changes in heterozygotes. Cell 81, 811–823 (1995).
White, J. K. et al. Huntingtin is required for neurogenesis and is not impaired by the Huntington's disease CAG expansion. Nat. Genet. 17, 404–410 (1997).
McBride, J. L. et al. Preclinical safety of RNAi-mediated HTT suppression in the rhesus macaque as a potential therapy for Huntington's disease. Mol. Ther. 19, 2152–2162 (2011).
Benatar, M. et al. Randomized, double-blind, placebo-controlled trial of arimoclomol in rapidly progressive SOD1 ALS. Neurology 90, e565–e574 (2018).
Ross, C. A. et al. Huntington disease: natural history, biomarkers and prospects for therapeutics. Nat. Rev. Neurol. 10, 204–216 (2014).
Bateman, R. J. et al. Clinical and biomarker changes in dominantly inherited Alzheimer's disease. N. Engl. J. Med. 367, 795–804 (2012).
Kolb, S. J. et al. Natural history of infantile-onset spinal muscular atrophy. Ann. Neurol. 82, 883–891 (2017).
Finkel, R. S. et al. Nusinersen versus sham control in infantile-onset spinal muscular atrophy. N. Engl. J. Med. 377, 1723–1732 (2017).
Summerford, C., Bartlett, J. S. & Samulski, R. J. AlphaVbeta5 integrin: a co-receptor for adeno-associated virus type 2 infection. Nat. Med. 5, 78–82 (1999).
Ling, C. et al. Human hepatocyte growth factor receptor is a cellular coreceptor for adeno-associated virus serotype 3. Hum. Gene Ther. 21, 1741–1747 (2010).
Di Pasquale, G. et al. Identification of PDGFR as a receptor for AAV-5 transduction. Nat. Med. 9, 1306–1312 (2003).
Akache, B. et al. The 37/67-kilodalton laminin receptor is a receptor for adeno-associated virus serotypes 8, 2, 3, and 9. J. Virol. 80, 9831–9836 (2006).
Dudek, A. M. et al. An alternate route for adeno-associated virus entry independent of AAVR. J. Virol. 92, e02213-17 (2018).
Pillay, S. et al. AAV serotypes have distinctive interactions with domains of the cellular receptor AAVR. J. Virol. 91, e00391-17 (2017).
Bleker, S., Sonntag, F. & Kleinschmidt, J. A. Mutational analysis of narrow pores at the fivefold symmetry axes of adeno-associated virus type 2 capsids reveals a dual role in genome packaging and activation of phospholipase A2 activity. J. Virol. 79, 2528–2540 (2005).
Grieger, J. C., Johnson, J. S., Gurda-Whitaker, B., Agbandje-Mckenna, M. & Samulski, R. J. Surface-exposed adeno-associated virus Vp1-NLS capsid fusion protein rescues infectivity of noninfectious wild-type Vp2/Vp3 and Vp3-only capsids but not that of fivefold pore mutant virions. J. Virol. 81, 7833–7843 (2007).
Grieger, J. C., Snowdy, S. & Samulski, R. J. Separate basic region motifs within the adeno-associated virus capsid proteins are essential for infectivity and assembly. J. Virol. 80, 5199–5210 (2006).
Salganik, M. et al. Adeno-associated virus capsid proteins may play a role in transcription and second-strand synthesis of recombinant genomes. J. Virol. 88, 1071–1079 (2014).
Aydemir, F. et al. Mutants at the 2-fold interface of adeno-associated virus type 2 (AAV2) structural proteins suggest a role in viral transcription for AAV capsids. J. Virol. 90, 7196–7204 (2016).
Rabinowitz, J. E. et al. Cross-packaging of a single adeno-associated virus (AAV) type 2 vector genome into multiple AAV serotypes enables transduction with broad specificity. J. Virol. 76, 791–801 (2002).
Dong, J. Y., Fan, P. D. & Frizzell, R. A. Quantitative analysis of the packaging capacity of recombinant adeno-associated virus. Hum. Gene Ther. 7, 2101–2112 (1996).
Wu, Z., Yang, H. & Colosi, P. Effect of genome size on AAV vector packaging. Mol. Ther. 18, 80–86 (2010).
Miao, C. H. et al. Nonrandom transduction of recombinant adeno-associated virus vectors in mouse hepatocytes in vivo: cell cycling does not influence hepatocyte transduction. J. Virol. 74, 3793–3803 (2000).
Hirata, R. K. & Russell, D. W. Design and packaging of adeno-associated virus gene targeting vectors. J. Virol. 74, 4612–4620 (2000).
McCarty, D. M. et al. Adeno-associated virus terminal repeat (TR) mutant generates self-complementary vectors to overcome the rate-limiting step to transduction in vivo. Gene Ther. 10, 2112–2118 (2003). This paper presents the first study to describe the now commonly used self-complementary AAV vectors that improve the rate and efficiency of transduction by overcoming the requirement for second-strand synthesis.
Nathwani, A. C. et al. Long-term safety and efficacy following systemic administration of a self-complementary AAV vector encoding human FIX pseudotyped with serotype 5 and 8 capsid proteins. Mol. Ther. 19, 876–885 (2011).
Valles, F. et al. Qualitative imaging of adeno-associated virus serotype 2-human aromatic L-amino acid decarboxylase gene therapy in a phase I study for the treatment of Parkinson disease. Neurosurgery 67, 1377–1385 (2010).
Bankiewicz, K. S. et al. AAV viral vector delivery to the brain by shape-conforming MR-guided infusions. J. Control. Release 240, 434–442 (2016).
Shi, Y. et al. ApoE4 markedly exacerbates tau-mediated neurodegeneration in a mouse model of tauopathy. Nature 549, 523–527 (2017).
Liao, F. et al. Targeting of nonlipidated, aggregated apoE with antibodies inhibits amyloid accumulation. J. Clin. Invest. 128, 2144–2155 (2018).
Dodart, J.-C. et al. Gene delivery of human apolipoprotein E alters brain Abeta burden in a mouse model of Alzheimer's disease. Proc. Natl Acad. Sci. USA 102, 1211–1216 (2005).
Sevigny, J. et al. The antibody aducanumab reduces Abeta plaques in Alzheimer's disease. Nature 537, 50–56 (2016).
Chiang, A. C. A. et al. Combination anti-Abeta treatment maximizes cognitive recovery and rebalances mTOR signaling in APP mice. J. Exp. Med. 215, 1349–1364 (2018).
Arrant, A. E., Onyilo, V. C., Unger, D. E. & Roberson, E. D. Progranulin gene therapy improves lysosomal dysfunction and microglial pathology associated with frontotemporal dementia and neuronal ceroid lipofuscinosis. J. Neurosci. 38, 2341–2358 (2018).
Mis, M. S. C. et al. Development of therapeutics for C9ORF72 ALS/FTD-related disorders. Mol. Neurobiol. 54, 4466–4476 (2017).
Choudhury, S. R. et al. Widespread central nervous system gene transfer and silencing after systemic delivery of novel AAV-AS vector. Mol. Ther. 24, 726–735 (2016).
Muramatsu, S. et al. A phase I study of aromatic L-amino acid decarboxylase gene therapy for Parkinson's disease. Mol. Ther. 18, 1731–1735 (2010).
Rocha, E. M. et al. Glucocerebrosidase gene therapy prevents alpha-synucleinopathy of midbrain dopamine neurons. Neurobiol. Dis. 82, 495–503 (2015).
Bae, E. J. et al. Antibody-aided clearance of extracellular alpha-synuclein prevents cell-to-cell aggregate transmission. J. Neurosci. 32, 13454–13469 (2012).
Lee, J. S. & Lee, S. J. Mechanism of anti-alpha-synuclein immunotherapy. J. Mov. Disord. 9, 14–19 (2016).
Khodr, C. E. et al. An alpha-synuclein AAV gene silencing vector ameliorates a behavioral deficit in a rat model of Parkinson's disease, but displays toxicity in dopamine neurons. Brain Res. 1395, 94–107 (2011).
Zharikov, A. D. et al. shRNA targeting alpha-synuclein prevents neurodegeneration in a Parkinson's disease model. J. Clin. Invest. 125, 2721–2735 (2015).
Ramachandran, P. S., Boudreau, R. L., Schaefer, K. A., La Spada, A. R. & Davidson, B. L. Nonallele specific silencing of ataxin-7 improves disease phenotypes in a mouse model of SCA7. Mol. Ther. 22, 1635–1642 (2014).
Biferi, M. G. et al. A new AAV10-U7-mediated gene therapy prolongs survival and restores function in an ALS mouse model. Mol. Ther. 25, 2038–2052 (2017).
Towne, C., Setola, V., Schneider, B. L. & Aebischer, P. Neuroprotection by gene therapy targeting mutant SOD1 in individual pools of motor neurons does not translate into therapeutic benefit in fALS mice. Mol. Ther. 19, 274–283 (2011).
Wang, H. et al. Widespread spinal cord transduction by intrathecal injection of rAAV delivers efficacious RNAi therapy for amyotrophic lateral sclerosis. Hum. Mol. Genet. 23, 668–681 (2014).
Scotter, E. L., Chen, H. J. & Shaw, C. E. TDP-43 proteinopathy and ALS: insights into disease mechanisms and therapeutic targets. Neurotherapeutics 12, 352–363 (2015).
Cai, W. et al. shRNA mediated knockdown of Nav1.7 in rat dorsal root ganglion attenuates pain following burn injury. BMC Anesthesiol. 16, 59 (2016).
Acknowledgements
The authors thank their colleagues at Voyager Therapeutics for numerous discussions on adeno-associated viral (AAV) gene therapy for central nervous system (CNS) disorders and the Parkinson disease team and P. Larson for their work on the delivery of AAV2–aromatic-L-amino-acid decarboxylase (AADC) gene therapy in patients with Parkinson disease with intraparenchymal brain administration. The authors also thank E. Smith, W. Yen and M. Lawrence for assistance with the figures, tables and text, respectively. B.E.D. was supported by the Beckman Institute for the CLARITY, Optogenetics and Vector Engineering Research Center (CLOVER) at the California Institute of Technology, the Friedreich's Ataxia Research Alliance (FARA) and FARA Australasia and the CHDI Foundation and is currently supported by the Stanley Center for Psychiatric Research at Broad Institute. K.S.B. was supported by the Michael J. Fox Foundation. B.M.R., S.M.P. and D.W.Y.S. are currently employees of Voyager Therapeutics, a CNS gene therapy company working on AAV vectors for the treatment of severe neurological diseases.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare competing financial interests in the form of funding from Voyager Therapeutics, employment by Voyager Therapeutics and/or personal financial interests in Voyager Therapeutics.
Related links
RELATED LINKS
Glossary
- Capsid
-
The protein shell of the virus that protects the adeno-associated viral genome and mediates entry into and trafficking within the host cell.
- Intraparenchymal
-
(IPa). Direct delivery of an agent into the tissue of interest.
- Intrathecal
-
(IT). A route of access into the spinal cord cerebrospinal fluid via the space under the arachnoid membrane.
- Intracerebroventricular
-
(ICV). A route of access into the CSF via the cerebral ventricles (typically the lateral ventricle).
- Tropism
-
Specificity for a particular host tissue or cell type.
- Serotypes
-
Capsid variants or groups of capsids that have distinct neutralization properties.
- Intracisternal
-
A route of access into the CSF via the cerebellomedullary cistern.
- Dependoviruses
-
Genus of parvoviruses that are replication-incompetent in the absence of co-infection of the host cell with a second virus such as an adenovirus or HSV.
- Self-complementary AAV
-
An AAV genome that has been modified by elimination of the 5′ terminal resolution site and can fold into double-stranded DNA without the requirement for DNA synthesis.
- Convection enhanced delivery
-
(CED). Infusion of adeno-associated viral vectors or other molecules into the parenchyma under positive pressure to increase the distribution volume.
Rights and permissions
About this article
Cite this article
Deverman, B., Ravina, B., Bankiewicz, K. et al. Gene therapy for neurological disorders: progress and prospects. Nat Rev Drug Discov 17, 641–659 (2018). https://doi.org/10.1038/nrd.2018.110
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd.2018.110
This article is cited by
-
Continuous directed evolution of a compact CjCas9 variant with broad PAM compatibility
Nature Chemical Biology (2024)
-
Viral Vector-Based Gene Therapy for Epilepsy: What Does the Future Hold?
Molecular Diagnosis & Therapy (2024)
-
Recent developments in gene therapy research in India
Journal of Biosciences (2024)
-
AAV-based in vivo gene therapy for neurological disorders
Nature Reviews Drug Discovery (2023)
-
Structure-guided AAV capsid evolution strategies for enhanced CNS gene delivery
Nature Protocols (2023)