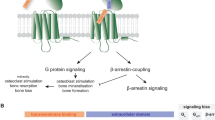
Key Points
-
G-protein-coupled receptors (GPCRs) are activated by a diverse range of ligands ranging from large proteins and proteases to small peptides, metabolites, neurotransmitters and ions. They are expressed on all cells in the body and play key parts in physiology and homeostasis. As such, GPCRs are one of the most important target classes for therapeutic drug discovery.
-
The development of drugs targeting GPCRs has therapeutic value across a wide range of diseases, including immune and inflammatory disorders, cancer and neurological and metabolic diseases. The progress made by targeting GPCRs with antibody-based therapeutics, as well as technical hurdles to overcome, are presented and discussed in this Review.
-
Advances in our understanding of GPCR biology in oncology, notably immuno-oncology, have made remarkable gains in recent years and facilitated further opportunities for strategic targeting either as stand-alone or combination therapies.
-
Many technical hurdles are still present but are not insurmountable given developments in stabilizing receptors using mutagenesis or additives as well as methods to overexpress receptors. These advances, combined with powerful high-throughput screening systems and an in-depth understanding of GPCR structure, biology and clinical relevance are enabling the knowledge necessary to overcome technical bottlenecks in GPCR antibody drug discovery and development.
-
It is also noticeable that the application of next-generation protein therapeutics, such as bispecifics and antibody–drug conjugates, to GPCR targets is gaining momentum.
-
Antibody therapeutics targeting C-C chemokine receptor type 4 (CCR4), CCR5 and calcitonin gene-related peptide (CGRP) are used as illustrative clinical case studies.
Abstract
G-protein-coupled receptors (GPCRs) are activated by a diverse range of ligands, from large proteins and proteases to small peptides, metabolites, neurotransmitters and ions. They are expressed on all cells in the body and have key roles in physiology and homeostasis. As such, GPCRs are one of the most important target classes for therapeutic drug discovery. The development of drugs targeting GPCRs has therapeutic value across a wide range of diseases, including cancer, immune and inflammatory disorders as well as neurological and metabolic diseases. The progress made by targeting GPCRs with antibody-based therapeutics, as well as technical hurdles to overcome, are presented and discussed in this Review. Antibody therapeutics targeting C-C chemokine receptor type 4 (CCR4), CCR5 and calcitonin gene-related peptide (CGRP) are used as illustrative clinical case studies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
01 September 2017
In the article, the clinical trial for the biparatopic CXCR2 nanobody was described as being for cancer instead of inflammation. This error has been corrected in the online version.
References
Lagerström, M. C. & Schiöth, H. B. Structural diversity of G protein-coupled receptors and significance for drug discovery. Nat. Rev. Drug Discov. 7, 339–357 (2008).
Zalewska, M., Siara, M. & Sajewicz, W. G protein-coupled receptors: abnormalities in signal transmission, disease states and pharmacotherapy. Acta Pol. Pharm. 71, 229–243 (2014).
Vassart, G. & Costagliola, S. G protein-coupled receptors: mutations and endocrine diseases. Nat. Rev. Endocrinol. 7, 362–372 (2011).
Thompson, M. D., Hendy, G. N., Percy, M. E., Bichet, D. G. & Cole, D. E. G protein-coupled receptor mutations and human genetic disease. Methods Mol. Biol. 1175, 153–187 (2014).
Overington, J. P., Al-Lazikani, B. & Hopkins, A. L. How many drug targets are there? Nat. Rev. Drug Discov. 5, 993–996 (2006). This is a key publication that reviews the drug target landscape.
Eglen, R. M. & Reisine, T. New insights into GPCR function: implications for HTS. Methods Mol. Biol. 552, 1–13 (2009).
Audet, M. & Bouvier, M. Restructuring G protein-coupled receptor activation. Cell 151, 14–23 (2012).
Smith, J. S. & Rajagopal, S. The β-arrestins: multifunctional regulators of G protein-coupled receptors. J. Biol. Chem. 291, 8969–8977 (2016).
Calebiro, D. et al. Persistent cAMP-signals triggered by internalized G-protein-coupled receptor. PLoS Biol. 7, e1000172 (2009).
Feinstein, T. N. et al. Retromer terminates the generation of cAMP by internalized PTH receptors. Nat. Chem. Biol. 7, 278–284 (2011).
Thomsen, A. R. et al. GPCR-G protein-β-arrestin super-complex mediates sustained G protein signalling. Cell 166, 907–919 (2016).
Wisler, J. W., Xiao, K., Thomsen, A. R. & Lefkowitz, R. J. Recent developments in biased agonism. Curr. Opin. Cell Biol. 27, 18–24 (2014).
Luttrell, L. M., Maudsley, S. & Bohn, L. M. Fulfilling the promise of “biased” G protein-coupled receptor agonism. Mol. Pharmacol. 88, 579–588 (2015).
Ullmer, C. et al. Functional monoclonal antibody acts as a biased agonist by inducing internalization of metabotropic glutamate receptor 7. Br. J. Pharmacol. 167, 1448–1466 (2012).
Coleman, J. L., Ngo, T. & Smith, N. J. The G protein-coupled receptor N-terminus and receptor signalling: N-tering a new era. Cell. Signal. 33, 1–9 (2017).
Barwell, J., Woolley, M. J., Wheatley, M., Conner, A. C. & Poyner, D. R. The role of the extracellular loops of the CGRP receptor, a family B GPCR. Biochem. Soc. Trans. 40, 433–437 (2012).
de Graaf, C., Foata, N., Engkvist, O. & Rognan, D. Molecular modelling of the second extracellular loop of G-protein coupled receptors and its implication on structure-based virtual screening. Proteins 71, 599–620 (2006).
Krishnan, A., Nijmeijer, S., de Graaf, C. & Schiöth, H. B. Classification, nomenclature and structural aspects of adhesion GPCRs. Handb. Exp. Pharmacol. 234, 15–41 (2016).
Santos, R. et al. A comprehensice map of molecular drug targets. Nat. Rev. Drug Discov. 16, 19–34 (2016).
Koglin, M. & Hutchings, C. J. Targeting G protein-coupled receptors with biologics for therapeutic use — part 2. BioProcess Int. 12, 62–65 (2014).
Congreve, M., Langmead, C. J., Mason, J. S. & Marshall, F. H. Progress in structure based drug design for G protein-coupled receptors. J. Med. Chem. 54, 4283–4311 (2011).
Leeson, P. D. & Springthorpe, B. The influence of drug-like concepts on decision-making in medicinal chemistry. Nat. Rev. Drug Discov. 6, 881–890 (2007).
Hutchings, C. J., Koglin, M. & Marshall, F. H. Therapeutic antibodies directed at G protein-coupled receptors. mAbs 2, 594–606 (2010). A review on the GPCR mAb pipeline in 2010 for comparison in progress and pipeline development.
Staus, D. P. et al. Regulation of β2-adrenergic receptor function by conformationally selective single-domain intrabodies. Mol. Pharmacol. 85, 472–481 (2014).
Webb, D. R., Handel, T. M., Kretz-Rommel, A. & Stevens, R. C. Opportunities for functional selectivity in GPCR antibodies. Biochem. Pharmacol. 85, 147–152 (2013).
BIO, Biomedtracker & Amplion. Clinical development success rates 2006–2015. BIO https://www.bio.org/sites/default/files/Clinical%20Development%20Success%20Rates%202006-2015%20-%20BIO,%20Biomedtracker,%20Amplion%202016.pdf
Koglin, M. & Hutchings, C. J. Targeting G protein-coupled receptors with biologics for therapeutic use — part 1. BioProcess Int. 12, 38–45 (2014).
Herr, D. R. in International Review of Cell and Molecular Biology Vol. 297 (ed. Kwang, W. J.) 45–81 (Academic Press, 2012).
Reichert, J. M. Antibodies to watch in 2015. mAbs 7, 1–8 (2015).
Ecker, D. M., Jones, S. D. & Levine, H. L. The therapeutic monoclonal antibody market. mAbs 7, 9–14 (2015).
Reichert, J. M. Antibodies to watch in 2017. mAbs 9, 167–181 (2016).
Reichert, J. M. Antibodies to watch update. The Antibody Society www.antibodysociety.org (updated 10 Jan 2017).
Jo, M. & Jung, S. T. Engineering therapeutic antibodies targeting G protein-coupled receptors. Exp. Mol. Med. 48, e207 (2016).
Wilkinson, T. C. Discovery of functional monoclonal antibodies targeting G protein-coupled receptors and ion channels. Biochem. Soc. Trans. 44, 831–837 (2016).
Tang, X. L., Wang, Y., Li, D. L., Luo, J. & Liu, M. Y. Orphan G protein-coupled receptors (GPCRs): biological functions and potential drug target. Acta Pharmacol. Sin. 33, 363–371 (2012).
Zhou, C. et al. G protein-coupled receptor GPR160 is associated with apoptosis and cell cycle arrest of prostate cancer cells. Oncotarget 7, 12823–12839 (2016).
Favara, D. M., Banham, A. H. & Harris, A. L. A review of ELTD1, a pro-angiogenic adhesion GPCR. Biochem. Soc. Trans. 42, 1658–1664 (2014).
Komatsu, H. Novel therapeutic GPCRs for psychiatric disorders. Int. J. Mol. Sci. 16, 14109–14121 (2015).
Leonard, S., Kinsella, G. K., Benetti, E. & Findlay, J. B. Regulating the effects of GPR21, a novel target for type 2 diabetes. Sci. Rep. 6, 27002 (2016).
Harrold, J. A. & Halford, J. C. Orphan G-protein-coupled receptors: strategies for identifying ligands and potential for use in eating disorders. Drugs R. D. 8, 287–299 (2007).
Valtcheva, N., Primorac, A., Jurisic, G., Hollmén, M. & Detmar, M. The orphan adhesion G protein-coupled receptor GPR97 regulates migration of lymphatic endothelial cells via the small GTPases RhoA and Cdc42. J. Biol. Chem. 288, 35736–35748 (2013).
O'Hayre, M. et al. The emerging mutational landscape of G proteins and G-protein-coupled receptors in cancer. Nat. Rev. Cancer 13, 412–424 (2013). This review provides an overview of GPCR mutations in the cancer landscape.
Bar-Shavit, R. et al. G protein-coupled receptors in cancer. Int. J. Mol. Sci. 17, E1320 (2016).
Han, X. Constitutively active chemokine CXC receptors. Adv. Pharmacol. 70, 265–301 (2014).
Ghanemi, A. Targeting G protein coupled receptor pathways as emerging molecule therapies. Saudi Pharm. J. 23, 115–129 (2015).
Zohn, I. E. et al. G2A is an oncogenic G protein-coupled receptor. Oncogene 19, 3866–3877 (2000).
Martin, C. B. et al. The thrombin receptor, PAR-1, causes transformation by activation of Rho-mediated signaling pathways. Oncogene 20, 1953–1963 (2001).
Montaner, S., Kufareva, I., Abagyan, R. & Gutkind, J. S. Molecular mechanisms deployed by virally encoded G protein-coupled receptors in human diseases. Annu. Rev. Pharmacol. Toxicol. 53, 331–354 (2013).
Willier, S., Butt, E. & Grunewald, T. G. Lysophosphatidic acid (LPA) signalling in cell migration and cancer invasion: a focussed review and analysis of LPA receptor gene expression on the basis of more than 1700 cancer microarrays. Biol. Cell 105, 317–333 (2013).
Reubi, J. C., Mäcke, H. R. & Krenning, E. P. Candidates for peptide receptor radiotherapy today and in the future. J. Nucl. Med. 46 (Suppl. 1), 67S–75S (2005).
Lewis, J. S. & Anderson, C. J. Radiometal-labeled somatostatin analogs for applications in cancer imaging and therapy. Methods Mol. Biol. 386, 227–240 (2007).
Dijksterhuis, J. P., Petersen, J. & Schulte, G. WNT/Frizzled signalling: receptor-ligand selectivity with focus on FZD-G protein signalling and its physiological relevance: IUPHAR review 3. Br. J. Pharmacol. 171, 1195–1209 (2014).
Tai, D. et al. Targeting the WNT signaling pathway in cancer therapeutics. Oncologist 20, 1189–1198 (2015).
Rimkus, T. K., Carpenter, R. L., Qasem, S., Chan, M. & Lo, H. W. Targeting the sonic hedgehog signaling pathway: review of smoothened and GLI inhibitors. Cancers (Basel) 8, E22 (2016).
Kubota, T., Michigami, T. & Ozono, K. Wnt signaling in bone metabolism. J. Bone Miner. Metab. 27, 265–271 (2009).
Sen, M., Chamorro, M., Reifert, J., Coor, M. & Carson, D. Blockade of Wnt-5A/Frizzled 5 signaling inhibits rheumatoid synoviocyte activation. Arthritis Rheum. 44, 772–781 (2001).
Nagayama, S. et al. Therapeutic potential of antibodies against FZD10, a cell-surface protein, for synovial sarcomas. Oncogene 24, 6201–6212 (2005).
Fukukawa, C. et al. Radioimmunotherapy of human synovial sarcoma using a monoclonal antibody against FZD10. Cancer Sci. 99, 432–440 (2008).
Highfill, S. L. et al. Disruption of CXCR2-mediated MDSC tumor trafficking enhances anti-PD1 efficacy. Sci. Transl Med. 6, 237ra67 (2014).
Steele, C. W. et al. CXCR2 inhibition profoundly suppresses metastases and augments immunotherapy in pancreatic ductal adenocarcinoma. Cancer Cell 29, 832–845 (2016).
Viola, A., Sarukhan, A., Bronte, V. & Molon, B. The pros and cons of chemokines in tumor immunology. Trends Immunol. 33, 496–504 (2012).
Nijmeier, S., Vischer, H. F. & Leurs, R. Adhesion GPCRs in immunology. Biochem. Pharmacol. 114, 88–102 (2016).
Stoveken, H. M., Hajduczok, A. G., Xu, L. & Tall, G. G. Adhesion G protein-coupled receptors are activated by exposure of a cryptic tethered agonist. Proc. Natl Acad. Sci. USA 112, 6194–6199 (2015).
Safaee, M. et al. CD97 is a multifunctional leukocyte receptor with distinct roles in human cancers (review). Int. J. Oncol. 43, 1343–1350 (2013).
Hsaio, C. C. et al. CD97 inhibits cell migration in human fibrosarcoma cells by modulating TIMP-2/MT1- MMP/MMP-2 activity — role of GPS autoproteolysis and functional cooperation between the N− and C-terminal fragments. FEBS J. 281, 4878–4891 (2014).
Hsiao, C. et al. The adhesion GPCR CD97/ADGRE5 inhibits apoptosis. Int. J. Biochem. Cell Biol. 65, 197–208 (2015).
Chidambaram, A., Fillmore, H. L., Van Meter, T. E., Dumur, C. I. & Broaddus, W. C. Novel report of expression and function of CD97 in malignant gliomas: correlation with Wilms tumor 1 expression and glioma cell invasiveness. J. Neurosurg. 116, 843–853 (2012).
Ward, Y. et al. LPA receptor heterodimerizes with CD97 to amplify LPA-initiated RHO-dependent signaling and invasion in prostate cancer cells. Cancer Res. 71, 7301–7311 (2011).
Ward, Y. et al. CD97 amplifies LPA receptor signaling and promotes thyroid cancer progression in a mouse model. Oncogene 32, 2726–2738 (2013).
Masiero, M. et al. A core human primary tumor angiogenesis signature identifies the endothelial orphan receptor ELTD1 as a key regulator of angiogenesis. Cancer Cell 24, 229–241 (2013).
Ziegler, J. et al. ELTD1, an effective anti-angiogenic target for gliomas: preclinical assessment in mouse GL261 and human G55 xenograft glioma models. Neuro Oncol. 19, 175–185 (2017).
Monk, K. R. et al. Adhesion G protein-coupled receptors: from in vitro pharmacology to in vivo mechanisms. Mol. Pharmacol. 88, 617–623 (2015).
Horuk, R. Chemokine receptor antagonists: overcoming developmental hurdles. Nat. Rev. Drug Discov. 8, 23–33 (2009).
Rothmeier, A. S. & Ruf, W. Protease-activated receptor 2 signaling in inflammation. Semin. Immunopathol. 34, 133–149 (2012).
Lee, S. E., Jeong, S. K. & Lee, S. H. Protease and protease-activated receptor-2 signaling in the pathogenesis of atopic dermatitis. Yonsei Med. J. 51, 808–822 (2010).
Vergnolle, N. Protease-activated receptors as drug targets in inflammation and pain. Pharmacol. Ther. 123, 292–309 (2009).
Wang, Y. et al. Multifunctional antibodies antibody agonists targeting glucagon-like-peptide-1, glucagon and glucose-dependent insulinotropic polypeptide receptors. Angew. Chem. Int. Ed. 55, 12475–12478 (2016).
Okamoto, H. et al. Glucagon receptor blockade with a human antibody normalizes blood glucose in diabetic mice and monkeys. Endocrinology 156, 2781–2794 (2015).
Hennen, S. et al. Structural insight into antibody-mediated antagonism of the glucagon-like peptide-1 receptor. Sci. Rep. 6, 26236 (2016).
Ravn, P. et al. Structural and pharmacological characterization of novel potent and selective monoclonal antibody antagonists of glucose-dependent insulinotropic polypeptide receptor. J. Biol. Chem. 288, 19760–19772 (2013).
Hara, T. et al. Role of free fatty acid receptors in the regulation of energy metabolism. Biochim. Biophys. Acta 1841, 1292–1300 (2014).
Miyamoto, J. et al. Nutritional signaling via free fatty acid receptors. Int. J. Mol. Sci. 17, 450 (2016).
Michel, M. C., Wieland, T. & Tsujimoto, G. How reliable are G-protein-coupled receptor antibodies? Naunyn Schmiedebergs Arch. Pharmacol. 379, 385–388 (2009).
Segala, E., Errey, J. C., Fiez-Vandal, C., Zhukov, A. & Cooke, R. M. Biosensor-based affinities and binding kinetics of small molecule antagonists to the adenosine A2A receptor reconstituted in HDL like particles. FEBS Lett. 589, 1399–1405 (2015).
Stenlund, P., Babcock, G. J., Sodroski, J. & Myszka, D. G. Capture and reconstitution of G protein-coupled receptors on a biosensor surface. Anal. Biochem. 316, 243–250 (2003).
Niwa, R. et al. Defucosylated chimeric anti-CC chemokine receptor 4 IgG1 with enhanced antibody-dependent cellular cytotoxicity shows potent therapeutic activity to T-cell leukemia and lymphoma. Cancer Res. 64, 2127–2133 (2004).
Shi, L. et al. Pharmacologic characterization of AMG 334, a potent and selective human monoclonal antibody against the calcitonin gene-related peptide receptor. J. Pharmacol. Exp. Ther. 356, 223–231 (2016).
Kuhne, M. R. et al. BMS-936564/MDX-1338: a fully human anti-CXCR4 antibody induces apoptosis in vitro and shows antitumor activity in vivo in hematologic malignancies. Clin. Cancer Res. 19, 357–366 (2013).
Kelley, L. P. & Kinsella, B. T. The role of N-linked glycosylation in determining the surface expression G protein interaction and effector coupling of the alpha (α) isoform of the human thromboxane A2 receptor. Biochim. Biophys. Acta 1621, 192–203 (2003).
Colvin, R. A., Campanella, G. S., Manice, L. A. & Luster, A. D. CXCR3 requires tyrosine sulfation for ligand binding and a second extracellular loop arginine residue for ligand-induced chemotaxis. Mol. Cell. Biol. 26, 5838–5849 (2006).
Rodriguez, M. C., Xie, Y. B., Wang, H., Collison, K. & Segaloff, D. L. Effects of truncations of the cytoplasmic tail of the luteinizing hormone/chorionic gonadotropin receptor on receptor-mediated hormone internalization. Mol. Endocrinol. 6, 327–336 (1992).
Dores, M. R. & Trejo, J. Ubiquitination of G protein-coupled receptors: functional implications and drug discovery. Mol. Pharmacol. 82, 563–570 (2012).
Kean, J., Bortolato, A., Hollenstein, K., Marshall, F. H. & Jazayeri, A. Conformational thermostabilisation of corticotropin releasing factor receptor 1. Sci. Rep. 5, 11954 (2015).
Scott, D. J., Kummer, L., Tremmel, D. & Plückthun, A. Stabilizing membrane proteins through protein engineering. Curr. Opin. Chem. Biol. 17, 427–435 (2013).
Scott, D. J., Kummer, L., Egloff, P., Bathgate, R. A. & Plückthun, A. Immprovding the apo-state detergent stability of NTS1 with CHESS for pharmacological and structural studies. Biochim. Biophys. Acta 1838, 2817–2824 (2014).
Schütz, M. et al. Directed evolution of G protein-coupled receptors in yeast for higher functional productions in eukaryotics expression hosts. Sci. Rep. 6, 21508 (2016).
Oswald, C. et al. Intracellular allosteric antagonism of the CCR9 receptor. Nature 540, 462–465 (2016).
Errey, J. C., Doré, A. S., Zhukov, A., Marshall, F. H. & Cooke, R. M. Purification of stabilized GPCRs for structural and biophysical analyses. Methods Mol. Biol. 1335, 1–15 (2015).
Hutchings, C. J. et al. Monoclonal anti-β1-adrenergic receptor antibodies activate G protein signaling in the absence of β-arrestin recruitment. mAbs 6, 246–261 (2014). This is the first proof-of-concept study for using stabilized GPCRs as antigens and for generating functional mAbs with diverse pharmacological properties.
Kleinschmidt, J. H. & Popot, J. L. Folding and stability of integral membrane proteins in amphipols. Arch. Biochem. Biophys. 564, 327–343 (2014).
Lee, S. C. & Pollock, N. L. Membrane proteins: is the future disc shaped? Biochem. Soc. Trans. 44, 1011–1018 (2016).
Wheatley, M. et al. GPCR-styrene maleic acid lipid particles (GPCR-SMALPs): their nature and potential. Biochem. Soc. Trans. 44, 619–623 (2016).
Denisov, I. G. & Sligar, S. G. Nanodiscs for structural and functional studies of membrane proteins. Nat. Struct. Mol. Biol. 23, 481–486 (2016).
Schuler, M. A., Denisov, I. G. & Sligar, S. G. Nanodiscs as a new tool to examine lipid-protein interactions. Methods Mol. Biol. 974, 415–433 (2013).
Frauenfeld, J. et al. A saposin-lipoprotein nanoparticle system for membrane proteins. Nat. Methods 13, 345–351 (2016).
Rues, R. B., Dötsch, V. & Bernhard, F. Co-translational formation and pharmacological characterization of beta1-adrenergic receptor/nanodisc complexes with different lipid environments. Biochim. Biophys. Acta 1858, 1306–1316 (2016).
Rasmussen, S. G. et al. Structure of a nanobody-stabilized active state of the β2 adrenoceptor. Nature 469, 175–180 (2011). This is an informative nexus of antibodies and GPCR structural biology.
Dunham, J. H. & Hall, R. A. Enhancement of the surface expression of G protein-coupled receptors. Trends Biotechnol. 27, 541–545 (2009).
Lundstrom, K. et al. Structural genomics on membrane proteins: comparison of more than 100 GPCRs in 3 expression systems. J. Struct. Funct. Genomics 7, 77–91 (2006).
Akermoun, M. et al. Characterization of 16 human G protein-coupled receptors expressed in baculovirus-infected insect cells. Protein Expr. Purif. 44, 65–74 (2005).
Butler, M. & Spearman, M. The choice of mammalian cell host and possibilities for glycosylation engineering. Curr. Opin. Biotechnol. 30, 107–112 (2014).
Johansson, T., Norris, T. & Peilot-Sjögren, H. Yellow fluorescent protein-based assay to measure GABAA channel activation and allosteric modulation in CHO-K1 cells. PLoS ONE 8, e59429 (2013).
Dukkipati, A., Park, H. H., Waghray, D., Fischer, S. & Garcia, K. C. BacMam system for high-level expression of recombinant soluble and membrane glycoproteins for structural studies. Protein Expr. Purif. 62, 160–170 (2008).
Delcayre, A. et al. Exosome Display technology: applications to the development of new diagnostics and therapeutics. Blood Cells Mol. Dis. 35, 158–168 (2005).
Banères, J. L., Popot, J. L. & Mouillac, B. New advances in production and functional folding of G-protein-coupled receptors. Trends Biotechnol. 29, 314–322 (2011).
Zheng, X. et al. Expression, stabilization and purification of membrane proteins via diverse protein synthesis systems and detergents involving cell-free associated with self-assembly peptide surfactants. Biotechnol. Adv. 32, 564–574 (2014).
Willis, S. et al. Virus-like particles as quantitative probes of membrane protein interactions. Biochemistry 47, 6988–6990 (2008).
Loisel, T. P. et al. Recovery of homogeneous and functional beta 2-adrenergic receptors from extracellular baculovirus particles. Nat. Biotechnol. 15, 1300–1304 (1997).
Hötzel, I. et al. Efficient production of antibodies against a mammalian integral membrane protein by phage display. Protein Eng. Des. Sel. 24, 679–689 (2011).
de Hoog, H. P. et al. Conformational antibody binding to a native, cell-free expressed GPCR in block copolymer membranes. PLoS ONE 9, e110847 (2014).
Chi, H., Wang, X., Li, J., Ren, H. & Huang, F. Chaperonin-enhanced Escherichia coli cell-free expression of functional CXCR4. J. Biotechnol. 231, 193–200 (2016).
Boshuizen, R. S. et al. A combination of in vitro techniques for efficient discovery of functional monoclonal antibodies against human CXC chemokine receptor-2 (CXCR2). mAbs 6, 1415–1424 (2014).
Giblin, P. et al. Fully human antibodies against the Protease-Activated Receptor-2 (PAR-2) with anti-inflammatory activity. Hum. Antibodies 20, 83–94 (2011).
Mumaw, M. M., de la Fuente, M., Noble, D. N. & Nieman, M. T. Targeting the anionic region of human protease-activated receptor 4 inhibits platelet aggregation and thrombosis without interfering with hemostasis. J. Thromb. Haemost. 12, 1331–1341 (2014).
Chain, B. M., Noursadeghi, M., Gardener, M., Tsang, J. & Wright, E. HIV blocking antibodies following immunisation with chimaeric peptides coding a short N-terminal sequence of the CCR5 receptor. Vaccine 26, 5752–5759 (2008).
Chain, B. et al. A linear epitope in the N-terminal domain of CCR5 and its interaction with antibody. PLoS ONE 10, e0128381 (2015).
Houimel, M. & Mazzucchelli, L. Identification of biologically active peptides that inhibit binding of human CXCL8 to its receptors from a random phage-epitope library. J. Leukoc. Biol. 85, 728–738 (2009).
Chambers, R. S. Antibody production using genetic immunization. Discov. Med. 3, 52–53 (2003).
Fujimoto, A. Enhancement of antibody responses to native G protein-coupled receptors using E.coli GroEL as a molecular adjuvant in DNA immunization. J. Immunol. Methods 375, 243–251 (2012).
Peyrassol, X. et al. Development by genetic immunization of monovalent antibodies (nanobodies) behaving as antagonists of the human ChemR23 receptor. J. Immunol. 196, 2893–2901 (2016).
Ebersbach, H. & Geisse, S. Antigen generation and display in therapeutic antibody drug discovery — a neglected by critical player. Biotechnol. J. 7, 1433–1443 (2012).
Liu, S., Wang, S. & Lu, S. DNA immunization as a technology platform for monoclonal antibody induction. Emerg. Microbes Infect. 5, e33 (2016).
van der Woning, B. et al. DNA immunization combined with scFv phage display identifies antagonistic GCGR specific antibodies and reveals new epitopes on the small extracellular loops. mAbs 8, 1126–1135 (2016).
Köhler, G. & Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 256, 495–497 (1975). Key publication that outlines the generation of hybridomas. The 1984 Nobel Prize in Physiology or Medicine was subsequently awarded jointly to N.K. Jerne, G.J.F. Köhler and C. Milstein 'for theories concerning the specificity in development and control of the immune system and the discovery of the principle for production of monoclonal antibodies'.
Galán, A. et al. Library-based display technologies: where do we stand? Mol. Biosyst. 12, 2342–2358 (2016).
Lonberg, N. Human monoclonal antibodies from transgenic mice. Handb. Exp. Pharmacol. 181, 69–97 (2008).
Macdonald, L. E. et al. Precise and in situ genetic immunization of 6 Mb of mouse immunoglobulin genes. Proc. Natl Acad. Sci. USA 111, 5147–5152 (2014).
Murphy, A. J. et al. Mice with megabase humanization of their immunoglobulin genes generate antibodies as efficiently as normal mice. Proc. Natl Acad. Sci. USA 111, 5153–5158 (2014).
Tomszak, F. et al. Selection of recombinant human antibodies. Adv. Exp. Med. Biol. 917, 23–54 (2016).
Doerner, A., Rhiel, L., Zielonka, S. & Kolmar, H. Therapeutic antibody engineering by high efficiency cell screening. FEBS Lett. 588, 278–287 (2014).
Mazsutis, L. et al. Single-cell analysis and sorting using droplet-based microfluidics. Nat. Protoc. 8, 870–891 (2013).
Hung, L. Y., Wang, C. H., Fu, C. Y., Gopinathan, P. & Lee, G. B. Microfluidics in the selection of affinity reagents for the detection of cancer: paving a way towards future diagnostics. Lab. Chip 16, 2759–2774 (2016).
Szekanecz, Z. & Koch, A. E. Successes and failures of chemokine-pathway targeting in rheumatoid arthritis. Nat. Rev. Rheumatol. 12, 5–13 (2016).
Schwickart, M. et al. Evaluation of assay interference and interpretation of CXCR4 in receptor occupancy results of preclinical study with MEDI3185, a fully human antibody to CXCR4. Cytometry B Clin. Cytom. 90, 209–219 (2016).
Quadrini, K. J. et al. Validation of a flow cytometry-based assay to assess C5aR receptor occupancy on neutrophils and monocytes for use in drug development. Cytometry B Clin. Cytom. 90, 177–190 (2016).
Mettler Izquierdo, S. et al. High-efficiency antibody discovery achieved with multiplexed microscopy. Microscopy (Oxf.) 65, 341–352 (2016).
Könitzer, J. D. et al. Generation of a highly diverse panel of antagonist chicken monoclonal antibodies against the GIP receptor. mAbs 9, 536–549 (2017).
Duvic, M., Evans, M. & Wang, C. Mogamulizumab for the treatment of cutaneous T-cell lymphoma: recent advances and clinical potential. Ther. Adv. Hematol. 7, 171–174 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01015560 (2015).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02455011 (2017).
Kashyap, M. K. et al. Ulocuplumab (BMS-936564 / MDX1338): a fully human anti-CXCR4 antibody induces cell death in chronic lymphocytic leukemia mediated through a reactive oxygen species-dependent pathway. Oncotarget 7, 2809–2822 (2016).
Azad, B. B. et al. A fully human CXCR4 antibody demonstrates diagnostic utility and therapeutic efficacy in solid tumor xenografts. Oncotarget 7, 12344–12358 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02305563 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01120457 (2015).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01359657 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02472977 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02666209 (2016).
Broussas, M. et al. A new anti-CXCR4 antibody that blocks the CXCR4/SDF-1 axis and mobilizes effector cells. Mol. Cancer Ther. 15, 1890–1899 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01139788 (2012).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02954653 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01345201 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02005315 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01973309 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01957007 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01608867 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02050178 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02092363 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02069145 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01469975 (2015).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02482441 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02321709 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02726334 (2017).
Bird Rock Bio. Bird Rock Bio submits clinical trial authorization for the first in human clinical trial for namacizumab. birdrockbio.comhttp://www.birdrockbio.com/wp-content/uploads/2016/10/20161027.pdf (2016).
Schiffgens, S. et al. Sex-specific clinicopathological significance of novel (Frizzled-7) and established (MGMT, IDH1) biomarkers in glioblastoma. Oncotarget 7, 55169–55180 (2016).
Blagodatski, A., Poteryaev, D. & Katanaev, V. L. Targeting the Wnt pathways for therapies. Mol. Cell. Ther. 2, 28 (2014).
Kemper, K. et al. Monoclonal antibodies against LGR5 identify human colorectal cancer stem cells. Stem Cells 30, 2378–2386 (2012).
Gong, X. et al. LGR5- targeted antibody-drug conjugate eradicates gastrointestinal tumors and prevents recurrence. Mol. Cancer Ther. 15, 1580–1590 (2016).
Rajapaksa, K. S. et al. Preclinical safety profile of a depleting antibody against CRTh2 for asthma: well tolerated despite unexpected CRTh2 expression on vascular pericytes in the central nervous system and gastric mucosa. Toxicol. Sci. 152, 72–84 (2016).
Yan, H. et al. Fully human monoclonal antibodies antagonizing the glucagon receptor improve glucose homeostasis in mice and monkeys. J. Pharmacol. Exp. Ther. 329, 102–111 (2009).
Lau, Y. Y. et al. Pharmacokinetic and pharmacodynamic modelling of a monoclonal antibody antagonist of glucagon receptor in male ob/ob mice. AAPS J. 11, 700–709 (2009).
Wang, M. Y. et al. Glucagon receptor antibody completely suppresses type 1 diabetes phenotype without insulin by disrupting a novel diabetogenic pathway. Proc. Natl Acad. Sci. USA 112, 2503–2508 (2015).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02715193 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03117998 (2017).
Viney, J. M. et al. Distinct conformation of the chemokine receptor CCR4 with implications for targeting in allergy. J. Immunol. 192, 3419–3427 (2014).
Mariani, M., Lang, R., Binda, E., Panina-Bordignon, P. & D'Ambrosio, D. Dominance of CCL22 over CCL17 in induction of chemokine receptor CCR4 desensitization and internalization on human Th2 cells. Eur. J. Immunol. 34, 231–240 (2004).
Sun, W. et al. Blockade of MCP-1/CCR4 signaling-induced recruitment of activated regulatory cells evokes an antitumor immune response in head and neck squamous cell carcinoma. Oncotarget 7, 37714–37727 (2016). This study reports CCR4 biology, including MCP1-induced signalling that recruits activated T reg cells.
Kurose, K. et al. Increase in activated Treg in TIL in lung cancer and in vitro depletion of Treg by ADCC using an antihuman CCR4 mAb (KM2760). J. Thorac. Oncol. 10, 74–83 (2015).
Kurose, K. et al. Phase 1a study of FoxP3+ CD4 Treg depletion by infusion of a humanized anti-CCR4 antibody, KW-0761, in cancer patients. Clin. Cancer Res. 21, 4327–4336 (2015).
Andrews, G., Jones, C. & Wreggett, K. A. An intracellular allosteric site for a specific class of antagonists of the CC chemokine G protein-coupled receptors CCR4 and CCR5. Mol. Pharmacol. 73, 855–867 (2008).
Ajram, L. et al. Internalization of the chemokine receptor CCR4 can be evoked by orthosteric and allosteric receptor antagonists. Eur. J. Pharmacol. 729, 75–85 (2014).
Ishii, T. et al. Defucosylated humanized anti-CCR4 monoclonal antibody KW-0761 as a novel immunotherapeutic agent for adult T-cell leukaemia/lymphoma. Clin. Cancer Res. 16, 1520–1531 (2010).
Balkwill, F. R. The chemokine system and cancer. J. Pathol. 226, 148–157 (2012).
Hagemann, U. B. et al. Fully human antagonistic antibodies against CCR4 potently inhibit cell signaling and chemotaxis. PLoS ONE 9, e103776 (2014).
Chang, D. K. et al. Humanization of an anti-CCR antibody that kills cutaneous T-cell lymphoma cells and abrogates suppression by T-regulatory cells. Mol. Cancer Ther. 11, 2451–2461 (2012).
Solari, R. & Pease, J. E. Targeting chemokine receptors in disease — a case study of CCR4. Eur. J. Pharmacol. 763, 169–177 (2015).
Bayry, J., Tartour, E. & Tough, D. F. Targeting CCR4 as an emerging strategy for cancer therapy and vaccines. Trends Pharmacol. Sci. 35, 163–165 (2014).
Sugaya, M. et al. CCR4 is expressed on infiltrating cells in lesional skin of early mycosis fungoides and atopic dermatitis. J. Dermatol. 42, 613–615 (2015).
Yamamoto, K. et al. Phase I study of KW-0761, a defucosylated humanized anti-CCR4 antibody, in relapsed patients with adult T-cell leukemia-lymphoma and peripheral T-cell lymphoma. J. Clin. Oncol. 28, 1591–1598 (2010).
Ishida, T. et al. Defucosylated anti-CCR4 monoclonal antibody (KW-0761) for relapsed adult T-cell leukemia-lymphoma: a multicenter phase II study. J. Clin. Oncol. 30, 837–842 (2012).
Ogura, M. et al. Multicenter phase II study of mogamulizumab (KW-0761), a defucosylated anti-cc chemokine receptor 4 antibody, in patients with relapsed peripheral T-cell lymphoma and cutaneous T-cell lymphoma. J. Clin. Oncol. 32, 1157–1163 (2014).
Vela, M., Aris, M., Llorente, M., Garcia-Sanz, J. A. & Kremer, L. Chemokine receptor-specific antibodies in cancer immunotherapy: achievements and challenges. Front. Immunol. 6, 12 (2015).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02301130 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02444793 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02476123 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02705105 (2017).
Furukawa, M. et al. Persistent complete remission of acute leukemic-phase CCR4-positive gamma-delta peripheral T-cell lymphoma by autologous stem cell transplantation with mogamulizumab. Int. J. Hematol. 102, 498–505 (2015).
Haji, S. et al. Administration of an anti-CC chemokine receptor 4 monoclonal antibody, mogamulizumab, before allogeneic bone marrow transplantation for adult T-cell leukemia/lymphoma. Bone Marrow Transplant. 51, 432–434 (2016).
Tanaka, H. et al. Development of engineered T cells expressing a chimeric CD16-CD3ζ receptor to improve the clinical efficacy of mogamulizumab therapy against adult T-cell leukemia. Clin. Cancer Res. 22, 4405–4416 (2016).
Kato, K. et al. Diffuse panbronchiolitis after humanized anti-CCR4 monoclonal antibody therapy for relapsed adult T-cell leukemia/lymphoma. Int. J. Hematol. 97, 430–432 (2013).
Ohyama, Y., Kumode, T., Eguchi, G., Yamaguchi, T. & Maeda, Y. Induction of molecular remission by using anti-CC-chemokine receptor 4 (anti-CCR4) antibodies for adult T cell leukemia: a risk of opportunistic infection after treatment with anti-CCR4 antibodies. Ann. Hematol. 93, 169–171 (2014).
Ishii, Y. et al. Cytomegalovirus pneumonia after anti-CC-chemokine receptor 4 monoclonal antibody (mogamulizumab) therapy in an angioimmunoblastic T-cell lymphoma patient. Intern. Med. 55, 673–675 (2016).
Morichika, K. et al. Recurrence of psoriasis vulgaris accompanied by treatment with C-C chemokine receptor type 4 (CCR4) antibody (mogamulizumab) therapies in a patient with adult T cell leukemia/ lymphoma: insight into autoinflammatory diseases. Intern. Med. 55, 1345–1349 (2016).
Ishitsuka, K. et al. Colitis mimicking graft-versus-host disease during treatment with the anti-CCR4 monoclonal antibody, mogamulizumab. Int. J. Hematol. 102, 493–497 (2015).
Ifuku, H. et al. Fatal reactivation of hepatitis B virus infection in a patient with adult T-cell leukemia-lymphoma receiving the anti-CC chemokine receptor 4 antibody mogamulizumab. Hepatol. Res. 45, 1363–1367 (2015).
Ishiida, T. et al. Stevens-Johnson syndrome associated with mogamulizumab treatment of adult T-cell leukemia / lymphoma. Cancer Sci. 104, 647–650 (2013).
Fuji, S. Pretransplantation anti-CCR4 antibody mogamulizumab against adult T-cell leukemia/lymphoma is associated with significantly increased risks of severe and corticosteroid-refractory graft-versus-host disease, nonrelapse mortality, and overall mortality. J. Clin. Oncol. 34, 3426–3433 (2016).
Ureshino, H. et al. Effector regulatory T cells reflect the equilibrium between antitumor immunity and autoimmunity in adult T-cell leukemia. Cancer Immunol. Res. 4, 644–649 (2016).
Wilcox, R. A. Mogamulizumab: 2 birds, 1 stone. Blood 125, 1847–1848 (2015).
Ueda, R. Clinical application of anti-CCR4 monoclonal antibody. Oncology 89 (Suppl. 1), 16–21 (2015).
Zinzani, P. L. et al. Panoptic clinical review of the current and future treatment of relapsed/refractory T-cell lymphomas: peripheral T-cell lymphomas. Crit. Rev. Oncol. Hematol. 99, 214–227 (2016).
Bachelerie, F. et al. International Union of Basic and Clinical Pharmacology. LXXXIX. Update on the extended family of chemokine receptors and introducing a new nomenclature for atypical chemokine receptors. Pharmacol. Rev. 66, 1–79 (2014).
Lee, B., Sharron, M., Montaner, L. J., Weissman, D. & Doms, R. W. Quantification of CD4, CCR5, and CXCR4 levels on lymphocyte subsets, dendritic cells, and differentially conditioned monocyte-derived macrophages. Proc. Natl Acad. Sci. USA 96, 5215–5220 (1999).
Martinson, J. J., Chapman, N. H., Rees, D. C., Liu, Y. T. & Clegg, J. B. Global distribution of the CCR5 gene 32-basepair deletion. Nat. Genet. 16, 100–103 (1997).
Glass, W. G. et al. CCR5 deficiency increases risk of symptomatic West Nile virus infection. J. Exp. Med. 203, 35–40 (2006).
Glass, W. G. et al. Chemokine receptor CCR5 promotes leukocyte trafficking to the brain and survival in West Nile virus infection. J. Exp. Med. 202, 1087–1098 (2005).
Kindberg, E. et al. A deletion in the chemokine receptor 5 (CCR5) gene is associated with tickborne encephalitis. J. Infect. Dis. 197, 266–269 (2008).
Smyth, D. J. et al. Shared and distinct genetic variants in type 1 diabetes and celiac disease. N. Engl. J. Med. 359, 2767–2777 (2008).
Maddon, P. J. et al. The T4 gene encodes the AIDS virus receptor and is expressed in the immune system and the brain. Cell 47, 333–348 (1986).
Trkola, A. et al. CD4-dependent, antibody-sensitive interactions between HIV-1 and its co-receptor CCR-5. Nature 384, 184–187 (1996).
Feng, Y., Broder, C. C., Kennedy, P. E. & Berger, E. A. HIV-1 entry cofactor: functional cDNA cloning of a seven-transmembrane, G protein-coupled receptor. Science 272, 872–877 (1996).
Dean, M. et al. Genetic restriction of HIV-1 infection and progression to AIDS by a deletion allele of the CKR5 structural gene. Hemophilia Growth and Development Study, Multicenter AIDS Cohort Study, Multicenter Hemophilia Cohort Study, San Francisco City Cohort, ALIVE Study. Science 273, 1856–1862 (1996).
Paquet, A. C. et al. A decade of HIV-1 drug resistance in the United States: trends and characteristics in a large protease/reverse transcriptase and co-receptor tropism database from 2003 to 2012. Antivir. Ther. 19, 435–441 (2014).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02483078 (2016).
Brown, T. R. I am the Berlin patient: a personal reflection. AIDS Res. Hum. Retroviruses 31, 2–3 (2015). This article is the account of the Berlin patient and discusses HIV remission and subsequent research to find a cure.
Trkola, A. et al. Potent, broad-spectrum inhibition of human immunodeficiency virus type 1 by the CCR5 monoclonal antibody PRO 140. J. Virol. 75, 579–588 (2001).
Olson, W. C. et al. Differential inhibition of human immunodeficiency virus type 1 fusion, gp120 binding, and CC-chemokine activity by monoclonal antibodies to CCR5. J. Virol. 73, 4145–4155 (1999). This study is the first description of the discovery of PRO 140.
Ji, C. et al. Novel CCR5 monoclonal antibodies with potent and broad-spectrum anti-HIV activities. Antiviral Res. 74, 125–137 (2007).
Rusert, P. et al. Virus isolates during acute and chronic human immunodeficiency virus type 1 infection show distinct patterns of sensitivity to entry inhibitors. J. Virol. 79, 8454–8469 (2005).
Cilliers, T. et al. The CCR5 and CXCR4 coreceptors are both used by human immunodeficiency virus type 1 primary isolates from subtype C. J. Virol. 77, 4449–4456 (2003).
Ketas, T. J. et al. Human immunodeficiency virus type 1 attachment, coreceptor, and fusion inhibitors are active against both direct and trans infection of primary cells. J. Virol. 77, 2762–2767 (2003).
Trkola, A. et al. HIV-1 escape from a small molecule, CCR5-specific entry inhibitor does not involve CXCR4 use. Proc. Natl Acad. Sci. USA 99, 395–400 (2002).
Pugach, P. et al. HIV-1 clones resistant to a small molecule CCR5 inhibitor use the inhibitor-bound form of CCR5 for entry. Virology 361, 212–228 (2007).
Murga, J. D., Franti, M., Pevear, D. C., Maddon, P. J. & Olson, W. C. Potent antiviral synergy between monoclonal antibody and small-molecule CCR5 inhibitors of human immunodeficiency virus type 1. Antimicrob. Agents Chemother. 50, 3289–3296 (2006).
Shearer, W. T. et al. Susceptibility of pediatric HIV-1 isolates to recombinant CD4-IgG2 (PRO 542) and humanized mAb to the chemokine receptor CCR5 (PRO 140). J. Allergy Clin. Immunol. 118, 518–521 (2006).
Binley, J. M. et al. Redox-triggered infection by disulfide-shackled human immunodeficiency virus type 1 pseudovirions. J. Virol. 77, 5678–5684 (2003).
Tan, Q. et al. Structure of the CCR5 chemokine receptor-HIV entry inhibitor maraviroc complex. Science 341, 1387–1390 (2013).
Garcia-Perez, J. et al. Allosteric model of maraviroc binding to CC chemokine receptor 5 (CCR5). J. Biol. Chem. 286, 33409–33421 (2011).
Maeda, K. et al. Involvement of the second extracellular loop and transmembrane residues of CCR5 in inhibitor binding and HIV-1 fusion: insights into the mechanism of allosteric inhibition. J. Mol. Biol. 381, 956–974 (2008).
Farzan, M. et al. Tyrosine sulfation of the amino terminus of CCR5 facilitates HIV-1 entry. Cell 96, 667–676 (1999).
Cormier, E. G. et al. Specific interaction of CCR5 amino-terminal domain peptides containing sulfotyrosines with HIV-1 envelope glycoprotein gp120. Proc. Natl Acad. Sci. USA 97, 5762–5767 (2000).
Qin, L. et al. Crystal structure of the chemokine receptor CXCR4 in complex with a viral chemokine. Science 347, 1117–1122 (2015).
Burg, J. S. et al. Structural biology. Structural basis for chemokine recognition and activation of a viral G protein-coupled receptor. Science 347, 1113–1117 (2015).
Tamamis, P. & Floudas, C. A. Elucidating a key anti-HIV-1 and cancer-associated axis: the structure of CCL5 (Rantes) in complex with CCR5. Sci. Rep. 4, 5447 (2014).
Jacobson, J. M. et al. Phase 2a study of the CCR5 monoclonal antibody PRO 140 administered intravenously to HIV-infected adults. Antimicrob. Agents Chemother. 54, 4137–4142 (2010).
Jacobson, J. M. et al. Anti-HIV-1 activity of weekly or biweekly treatment with subcutaneous PRO 140, a CCR5 monoclonal antibody. J. Infect. Dis. 201, 1481–1487 (2010).
Jacobson, J. M. et al. Antiviral activity of single-dose PRO 140, a CCR5 monoclonal antibody, in HIV-infected adults. J. Infect. Dis. 198, 1345–1352 (2008).
Mani, N. et al. Novel clinical trial designs for the development of new antiretroviral agents. AIDS 26, 899–907 (2012).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01272258 (2017).
Gunthard, H. F. et al. Antiretroviral treatment of adult HIV infection: 2014 recommendations of the International Antiviral Society-USA Panel. JAMA 312, 410–425 (2014).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02175680 (2016).
Maddon, P. J. et al. PRO140 SC monotherapy provides long-term, full virologic suppression in HIV patients. ASM Microbe (Boston, 2016).
Li, J. Z. et al. The size of the expressed HIV reservoir predicts timing of viral rebound after treatment interruption. AIDS 30, 343–353 (2016).
Walker, C. S. et al. A second trigeminal CGRP receptor: function and expression of the AMY1 receptor. Ann. Clin. Transl Neurol. 2, 595–608 (2015).
Ho, T. W. et al. Randomized controlled trial of the CGRP receptor antagonist telcagepant for migraine prevention. Neurology 83, 958–966 (2014).
Giamberardino, M. A. et al. Anti-CGRP monoclonal antibodies in migraine: current perspectives. Intern. Emerg. Med. 11, 1045–1057 (2016). This is a review of current CGRP-targeting mAbs in migraine.
Sun, H. et al. Safety and efficacy of AMG 334 for prevention of episodic migraine: a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Neurol. 15, 382–390 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02456740 (2017).
Reichert, J. M. Antibodies to watch in 2016: mid-year update. The Antibody Society www.antibodysociety.org (updated 18 Aug 2016).
Beck, A. et al. Strategies and challenges for the next generation of antibody–drug conjugates. Nat. Rev. Drug Discov. 16, 315–337 (2017).
Junttila, M. R. et al. Targeting LGR5+ cells with an antibody-drug conjugate for the treatment of colon cancer. Sci. Transl Med. 7, 314ra186 (2015).
Kularatne, S. A. et al. A CXCR4-targeted site-specific antibody-drug conjugate. Angew. Chem. Int. Ed. 53, 11863–11867 (2014).
Wang, E. et al. An immunosuppressive antibody-drug conjugate. J. Am. Chem. Soc. 137, 3229–3232 (2015).
Asundi, J. et al. An antibody-drug conjugate targeting the endothelin B receptor for the treatment of melanoma. Clin. Cancer Res. 17, 965–975 (2011).
Asundi, J. et al. MAPK pathway inhibition enhances the efficacy of an anti-endothelin B receptor drug conjugate by inducing target expression in melanoma. Mol. Cancer Ther. 13, 1599–1610 (2014).
Spiess, C., Zhai, Q. & Carter, P. J. Alternative molecular formats and therapeutic applications for bispecific antibodies. Mol. Immunol. 67, 95–106 (2015).
Panjideh, H. et al. Immunotherapy of B-cell non-Hodgkin lymphoma by targeting the chemokine receptor CXCR5 in a preclinical mouse model. Int. J. Cancer 135, 2623–2632 (2014).
Pardridge, W. M. Targeted delivery of proteins and gene medicines through the blood-brain barrier. Clin. Pharmacol. Ther. 97, 347–361 (2015).
Abulrob, A., Sprong, H., Van Bergen en Henegouwen, P. & Stanimirovic, D. The blood-brain barrier transmigrating single domain antibody: mechanisms of transport and antigenic epitopes in human brain endothelial cells. J. Neurochem. 95, 1201–1214 (2005).
Webster, C. I. et al. Brain penetration, target engagement, and disposition of the blood-brain barrier-crossing bispecific antibody antagonist of metabotropic glutamate receptor type 1. FASEB J. 30, 1927–1940 (2016).
Manglik, A., Kobilka, B. K. & Steyaert, J. Nanobodies to study G protein-coupled receptor structure and function. Annu. Rev. Pharmacol. Toxicol. 57, 19–37 (2017). This study discusses the application of nanobodies used to study GPCR structure and function.
Bradley, M. E. et al. Potent and efficacious inhibition of CXCR2 signaling by biparatopic nanobodies combining two distinct modes of action. Mol. Pharmacol. 87, 251–262 (2015). This study describes the discovery and characterization of a biparatopic nanobody targeting CXCR2 that is now in phase I trials.
Griffiths, K. et al. i-Bodies, human single domain antibodies that antagonize chemokine receptor CXCR4. J. Biol. Chem. 291, 12641–12657 (2016).
Wang, F. et al. Reshaping antibody diversity. Cell 153, 1379–1393 (2013).
Chames, P., Van Regenmortel, M., Weiss, E. & Baty, D. Therapeutic antibodies: successes, limitations and hopes for the future. Br. J. Pharmacol. 157, 220–233 (2009).
Bell, A. et al. Differential tumor-targeting abilities of three single-domain antibody formats. Cancer Lett. 289, 81–90 (2010).
Kearns, J. D. et al. Enhanced targeting of the EGFR network with MM-151, and oligoclonal anti-EGFR antibody therapeutic. Mol. Cancer Ther. 14, 1625–1636 (2015).
Yang, X. et al. Developability studies before initiation of process development: improving manufacturability of monoclonal antibodies. mAbs 5, 787–794 (2013).
Jarasch, A. et al. Developability assessment during the selection of novel therapeutic antibodies. J. Pharm. Sci. 104, 1885–1898 (2015).
Tohidkia, M. R., Asadi, F., Barar, J. & Omidi, Y. Selection of potential therapeutic human single-chain Fv antibodies against cholecystokinin-B/gastrin receptor by phage display technology. BioDrugs 27, 55–67 (2013).
Hermann, T. & Bueltmann, A. Antibodies targeting specifically human CXCR2. US Patent application 20160060347 (2016)
Rossant, C. J. et al. Phage display and hybridoma generation of antibodies to human CXCR2 yields antibodies with distinct mechanisms and epitopes. mAbs 6, 1425–1438 (2014).
Douthwaite, J. A. et al. Affinity maturation of a novel antagonistic human monoclonal antibody with a long VH CDR3 targeting the Class A GPCR formyl-peptide receptor 1. mAbs 7, 152–166 (2015).
Koth, C. M. et al. Molecular basis for negative regulation of the glucagon receptor. Proc. Natl Acad. Sci. USA 109, 14393–14398 (2012).
Mumaw, M. M., de la Fuente, M., Arachiche, A., Wahl, J. K. & Nieman, M. T. Development and characterization of monoclonal antibodies against Protease Activated Receptor 4 (PAR4). Thromb. Res. 135, 1165–1171 (2015).
Harris, G. L., Creason, M. B., Brulte, G. B. & Herr, D. R. In vitro and in vivo antagonism of a G protein-coupled receptor (S1P3) with a novel blocking monoclonal antibody. PLoS ONE 7, e35129 (2012).
Acknowledgements
The authors thank B. Tehan (Heptares Therapeutics) for his assistance in compiling figure 1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
C.J.H. is a shareholder and owns stock options of Heptares Therapeutics and Sosei Group, and has provided consultancy services to Heptares Therapeutics, TetraGenetics, Crescendo Biologics, Abcam, Tusk Therapeutics and xCella Biosciences. M.K. and F.H.M. are employees, shareholders and hold stock options of Heptares Therapeutics and Sosei Group. W.C.O. is an employee, a shareholder and holds stock options of Regeneron Pharmaceuticals.
Related links
DATABASES
FURTHER INFORMATION
Glossary
- Antibody-dependent cellular cytotoxicity
-
(ADCC). A mechanism of cell death that forms part of the humoral immune response, is mediated by the crystallizable fragment (Fc) region of an antibody and is isotype-dependent. In ADCC, an effector cell of the immune system (such as a natural killer cell, macrophage, neutrophil or eosinophil) actively lyses a target cell whose membrane surface antigens have been bound by specific antibodies. The Fc receptor on the immune cell binds to the Fc portion of the antibody.
- Complement-dependent cytotoxicity
-
(CDC). A mechanism of cell death that is mediated by the crystallizable fragment (Fc) region of an antibody. This is a function of the complement system and is initiated by C1q binding. CDC kills pathogens by damaging their membranes and is one of the mechanisms by which antibodies can have an antitumour effect.
- Antibody-dependent cellular phagocytosis
-
(ADCP). This is a key effector function that is mediated by the crystallizable fragment (Fc) region. It is increasingly becoming important in antibody development and relies on macrophages to attack and assimilate tumour cells following antibody binding.
- Antibody–drug conjugates
-
(ADCs). These are composed of an antibody that is linked to a toxic payload or drug and form an important modality for the treatment of cancer.
- Amphipols
-
These are compounds that are formed by the joining of smaller, usually repeating, amphiphilic polymers linked by covalent bonds. They are used to make membrane proteins water-soluble by trapping them with polymers rather than detergents.
- Syngenic
-
Genetically identical, or sufficiently identical, and immunologically compatible as to allow for transplantation. In this context, it refers to the use of syngenic cells of the animal to overexpress the target and then transfer the transfected cells back into the animal as a broader species-related approach rather than an individual approach.
- Hybridoma technology
-
This technology is a means of immortalizing an antibody-producing B cell by fusing it with a cell that is derived from a myeloma cell line to generate large amounts of antibody. However, hybridoma cell lines can be notoriously unstable.
- Single-domain antibody
-
A single-domain antibody is the smallest antibody-derived binding structure based on the separate variable domains of an IgG molecule and is monomeric in nature.
- Biparatopic
-
A biparatopic antibody is a type of bispecific antibody that is bivalent and directed against two different non-overlapping epitopes on the same target antigen molecule.
Rights and permissions
About this article
Cite this article
Hutchings, C., Koglin, M., Olson, W. et al. Opportunities for therapeutic antibodies directed at G-protein-coupled receptors. Nat Rev Drug Discov 16, 787–810 (2017). https://doi.org/10.1038/nrd.2017.91
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd.2017.91
This article is cited by
-
Structural basis for the allosteric modulation of rhodopsin by nanobody binding to its extracellular domain
Nature Communications (2023)
-
Isoform- and ligand-specific modulation of the adhesion GPCR ADGRL3/Latrophilin3 by a synthetic binder
Nature Communications (2023)
-
Structural basis of α1A-adrenergic receptor activation and recognition by an extracellular nanobody
Nature Communications (2023)
-
ERR-activated GPR35 promotes immune infiltration level of macrophages in gastric cancer tissues
Cell Death Discovery (2022)
-
Heterocomplexes between the atypical chemokine MIF and the CXC-motif chemokine CXCL4L1 regulate inflammation and thrombus formation
Cellular and Molecular Life Sciences (2022)