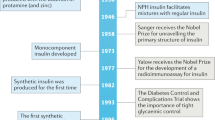
Key Points
-
Insulin therapy has been the cornerstone of diabetes treatment for nearly a century, and numerous commercial alternatives are available to control blood glucose. Nonetheless, insulin therapy remains sub-optimal, as manifested by infrequent normalization of blood glucose and the risk of experiencing dangerously low blood glucose levels.
-
Numerous novel insulin analogues and formulations are in development with the goal of further optimizing the time–action profile. Several once-weekly candidates that might minimize the requirement for daily injection in certain patients are currently in early clinical trials.
-
Alternative methods to subcutaneous injection of insulin delivery should improve the convenience of treatment and should probably lead to enhanced diabetic care, especially in patients that resist using injectable therapy despite treatment failure on oral medications. Oral and inhalable insulin formulations currently represent the most promising prospects in non-injectable delivery and constitute a priority area in the refinement of insulin therapy.
-
Substantial interest persists in the development of glucose-sensitive therapy that is responsive to real-time changes in blood glucose. Direct and indirect methods to render insulin therapy less prone to life-threatening hypoglycaemia are being aggressively pursued. Most notably, the combination of insulin and incretin therapy has delivered superior clinical results as measured by improvements in mean plasma glucose concentration (HbA1C), with fewer occurrences of hypoglycaemia and less body weight gain. Separately, advances in glucagon therapy move the field closer to achieving closed-loop insulin pump therapy.
-
Advances in insulin synthesis have stimulated a renewed interest in structural analogues that enhance pharmacodynamic properties, through tissue targeting (hepatospecific), supplemental pharmacology (insulin sensitizers) or selective action only in hyperglycaemia. Given the seminal importance of insulin as a drug we anticipate the continued pursuit of perfect insulin. Normalization of glucose control without the risk of hypoglycaemia and delivery in a patient-friendly form remain the central objectives.
Abstract
Insulin remains indispensable in the treatment of diabetes, but its use is hampered by its narrow therapeutic index. Although advances in peptide chemistry and recombinant DNA-based macromolecule synthesis have enabled the synthesis of structurally optimized insulin analogues, the growing epidemics of obesity and diabetes have emphasized the need for diabetes therapies that are more efficacious, safe and convenient. Accordingly, a broad set of drug candidates, targeting hyperglycaemia plus other disease abnormalities, is now progressing through the clinic. The development of an insulin therapy that is responsive to glucose concentration remains an ultimate goal, with initial prototypes now reaching the proof-of-concept stage. Simultaneously, the first alternatives to injectable delivery have progressed to registration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Joslin, E. P. & Kahn, C. R. Joslin's Diabetes Mellitus (Lippincott Williams & Wilkins, 2005).
Goeddel, D. V. et al. Expression in Escherichia coli of chemically synthesized genes for human insulin. Proc. Natl Acad. Sci. USA 76, 106–110 (1979).
Keefer, L. M., Piron, M.-A. & De Meyts, P. Human insulin prepared by recombinant DNA techniques and native human insulin interact identically with insulin receptors. Proc. Natl Acad. Sci. USA 78, 1391–1395 (1981).
Johnson, I. S. Human insulin from recombinant DNA technology. Science 219, 632–637 (1983).
Lipska, K. J. et al. Use and out of pocket costs of insulin for type 2 diabetes mellitus from 2000 through 2010. JAMA 311, 2331–2333 (2014).
Hirsch, I. B. Insulin analogues. N. Engl. J. Med. 352, 174–183 (2005). An overview of insulin analogues used in treatment at the turn of the century.
Home, P. et al. Insulin therapy in people with type 2 diabetes: opportunities and challenges? Diabetes Care 37, 1499–1508 (2014). A panel of specialists provides guidelines to initiating insulin therapy in the context of recent findings and novel treatment options.
Cryer, P. Hypoglycemia in Diabetes: Pathophysiology, Prevalence, and Prevention (American Diabetes Association, 2012).
Caparrotta, T. M. & Evans, M. PEGylated insulin Lispro, (LY2605541) — a new basal insulin analogue. Diabetes Obes. Metab. 16, 388–395 (2014).
Gough, S. C. L., Harris, S., Woo, V. & Davies, M. Insulin degludec: overview of a novel ultra long-acting basal insulin. Diabetes Obes. Metab. 15, 301–309 (2013).
Edgerton, D. S. et al. Changes in glucose and fat metabolism in response to the administration of a hepato-preferential insulin analog. Diabetes 63, 3946–3954 (2014).
Glendorf, T. et al. Engineering of insulin receptor isoform-selective insulin analogues. PloS ONE 6, e20288 (2011).
Gough, S. C. et al. One-year efficacy and safety of a fixed combination of insulin degludec and liraglutide in patients with type 2 diabetes: results of a 26-week extension to a 26-week main trial. Diabetes Obes. Metab. 17, 965–973 (2015). This extended clinical study underscores the benefits of combination therapy of insulin with GLP1 analogues.
Hovorka, R. Closed-loop insulin delivery: from bench to clinical practice. Nat. Rev. Endocrinology 7, 385–395 (2011).
Mo, R., Jiang, T., Di, J., Tai, W. & Gu, Z. Emerging micro- and nanotechnology based synthetic approaches for insulin delivery. Chem. Soc. Rev. 43, 3595–3629 (2014).
Mayer, J. P., Zhang, F. & DiMarchi, R. D. Insulin structure and function. Biopolymers 88, 687–713 (2007). This review focuses on the history of insulin chemical synthesis and the insulin structure–activity relationship.
Romans, R. G., Scott, D. A. & Fisher, A. M. Preparation of crystalline insulin. Ind. Eng. Chem. 32, 908–910 (1940).
Scott, D. A. & Best, C. H. The preparation of insulin. Ind. Eng. Chem. 17, 238–240 (1925).
Hallas-Møller, K. K., Jersild, M. M., Petersen, K. K. & Schlichtkrull, J. J. Zinc insulin preparations for single daily injection: Clinical studies of new preparations with prolonged action. J. Am. Med. Assoc. 150, 1667–1671 (1952).
Hallas-Møller, K. The lente insulins. Diabetes 5, 7–14 (1956).
Patel, H. M. & Ryman, B. E. Oral administration of insulin by encapsulation within liposomes. FEBS Letters 62, 60–63 (1976).
Best, C. The prolongation of insulin action. Ohio J. Science 37, 362–377 (1937).
Fonte, P., Araújo, F., Reis, S. & Sarmento, B. Oral insulin delivery: how far are we? J. Diabetes Sci. Technol. 7, 520–531 (2013). An overview of available delivery systems for oral insulin administration.
Santos Cavaiola, T. & Edelman, S. Inhaled insulin: a breath of fresh air? A review of inhaled insulin. Clin. Ther. 36, 1275–1289 (2014). A recent review discussing the benefits and challenges of inhaled insulin formulations.
Clarke, S. & Foster, J. A history of blood glucose meters and their role in self-monitoring of diabetes mellitus. Br. J. Biomed. Sci. 69, 83–93 (2012).
Alsaleh, F., Smith, F., Keady, S. & Taylor, K. Insulin pumps: from inception to the present and toward the future. J. Clin. Pharm. Ther. 35, 127–138 (2010).
Sanger, F. & Tuppy, H. The amino-acid sequence in the phenylalanyl chain of insulin. 2. The investigation of peptides from enzymic hydrolysates. Biochem. J. 49, 481 (1951).
Sanger, F. & Tuppy, H. The amino-acid sequence in the phenylalanyl chain of insulin. 1. The identification of lower peptides from partial hydrolysates. Biochem. J. 49, 463 (1951).
Bliss, M. Rewriting medical history: Charles Best and the Banting and Best myth. J. Hist. Med. Allied Sci. 48, 253–274 (1993).
Katsoyannis, P. G., Fukuda, K., Tometsko, A., Suzuki, K. & Tilak, M. Insulin peptides. X. The synthesis of the B chain of insulin and its combination with natural or synthetis A chin to generate insulin activity. J. Am. Chem. Soc. 86, 930–932 (1964).
Kung, Y.-T., Du, Y., Huang, W., Chen, C. & Ke, L. Total synthesis of crystalline bovine insulin. Sci. Sin. 14, 1710 (1965).
Marglin, B. & Merrifield, R. The synthesis of bovine insulin by the solid phase method1. J. Am. Chem. Soc. 88, 5051–5052 (1966).
Ruttenberg, M. A. Human insulin: facile synthesis by modification of porcine insulin. Science 177, 623–626 (1972).
Thim, L. et al. Secretion and processing of insulin precursors in yeast. Proc. Natl Acad. Sci. USA 83, 6766–6770 (1986).
The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N. Engl. J. Med. 329, 977–986 (1993).
Howey, D. C., Bowsher, R. R., Brunelle, R. L. & Woodworth, J. R. [Lys (B28), Pro (B29)]-human insulin: a rapidly absorbed analogue of human insulin. Diabetes 43, 396–402 (1994).
Torlone, E. et al. Pharmacokinetics, pharmacodynamics and glucose counterregulation following subcutaneous injection of the monomeric insulin analogue [Lys (B28), Pro (B29)] in IDDM. Diabetologia 37, 713–720 (1994).
Anderson, J. H. et al. Improved mealtime treatment of diabetes mellitus using an insulin analogue. Clin. Ther. 19, 62–72 (1997).
Home, P., Lindholm, A. & Riis, A. Insulin aspart versus human insulin in the management of long-term blood glucose control in Type 1 diabetes mellitus: a randomized controlled trial. Diabet. Med. 17, 762–770 (2000).
Home, P. D., Lindholm, A., Hylleberg, B. & Round, P. Improved glycemic control with insulin aspart: a multicenter randomized double-blind crossover trial in type 1 diabetic patients. UK Insulin Aspart Study Group. Diabetes Care 21, 1904–1909 (1998).
Brange, J. et al. Monomeric insulins obtained by protein engineering and their medical implications. Nature 333, 679–682 (1988).
Becker, R. H. & Frick, A. D. Clinical pharmacokinetics and pharmacodynamics of insulin glulisine. Clin. Pharmacokinet. 47, 7–20 (2008).
Becker, R., Frick, A., Burger, F., Potgieter, J. & Scholtz, H. Insulin glulisine, a new rapid-acting insulin analogue, displays a rapid time-action profile in obese non-diabetic subjects. Exp. Clin. Endocrinol. Diabetes 113, 435–443 (2005).
Dreyer, M. et al. Efficacy and safety of insulin glulisine in patients with type 1 diabetes. Horm. Metab. Res. 37, 702–707 (2005).
Owens, D. R., Matfin, G. & Monnier, L. Basal insulin analogues in the management of diabetes mellitus: what progress have we made? Diabetes Metab. Res. Rev. 30, 104–119 (2014).
Zinman, B. Newer insulin analogs: advances in basal insulin replacement. Diabetes Obes. Metab. 15, 6–10 (2013).
Oakley, W., Hill, D. & Oakley, N. Combined use of regular and crystalline protamine (NPH) insulins in the treatment of severe diabetes. Diabetes 15, 219–222 (1966).
Hilgenfeld, R. et al. Controlling insulin bioavailability by crystal contact engineering. Diabetologia 35 (Suppl.), A193 (1992).
Rosenstock, J. et al. Basal insulin therapy in type 2 diabetes 28 week comparison of insulin glargine (HOE 901) and NPH insulin. Diabetes Care 24, 631–636 (2001).
Guthrie, R. Is there a need for a better basal insulin? Clinical Diabetes 19, 66–70 (2001).
Jørgensen, S., Vaag, A., Langkjær, L., Hougaard, P. & Markussen, J. NovoSol Basal: pharmacokinetics of a novel soluble long acting insulin analogue. BMJ 299, 415–419 (1989).
Myers, S. et al. W99-S32 a soluble, basal insulin analog. Diabetologia 38 (suppl. 1), A4 (1995).
Hoeg-Jensen, T. in Peptide and Protein Design for Biopharmaceutical Applications (ed K. J., Jensen) 249–286 (Wiley, 2009).
Havelund, S. et al. The mechanism of protraction of insulin detemir, a long-acting, acylated analog of human insulin. Pharm. Res. 21, 1498–1504 (2004).
Hermansen, K. et al. 26 week, randomized, parallel, treat to target trial comparing insulin detemir with NPH insulin as add on therapy to oral glucose-lowering drugs in insulin-naive people with type 2 diabetes. Diabetes Care 29, 1269–1274 (2006).
Hu, Y. et al. Short-term intensive therapy in newly diagnosed type 2 diabetes partially restores both insulin sensitivity and β cell function in subjects with long-term remission. Diabetes Care 34, 1848–1853 (2011).
Retnakaran, R. & Zinman, B. Short-term intensified insulin treatment in type 2 diabetes: long-term effects on β-cell function. Diabetes Obes. Metab. 14, 161–166 (2011).
DCCT. Hypoglycemia in the diabetes control and complications trial. Diabetes 46, 271–286 (1997).
Vora, J. & Heise, T. Variability of glucose cowering effect as a limiting factor in optimizing basal insulin therapy: a review. Diabetes Obes. Metab. 15, 701–712 (2013). The identification of inter-and intra-patient variability as the major issue of the current insulin therapies and establishing it as a primary consideration for future treatments.
Barag, S. H. Insulin therapy for management of type 2 diabetes mellitus: strategies for initiation and long-term patient adherence. J. Am. Osteopath. Assoc. 111, S13–S19 (2011). A discussion of the psychological barriers associated with insulin therapy.
AntriaBio. Corporation Presentation, Q1 2015. AntriaBio Inc. [online], (2015).
Ascendis Pharma. TransCon Diabetes Program. Ascendis Pharma Inc [online], (2010).
Sanofi-Aventis. Sanofi-Aventis acquires from Ascendis Pharma worldwide rights on drug-delivery technology in diabetes and related disorders. Sanofi [online], (2010).
Hwang, S. Y. et al. Novel very long-acting insulin analog (HM12470) with potential for once-weekly dosing has a favorable PK, PD and mitogenic profile. American Diabetes Association's 74th Scientific Sessions [online], (2014).
Huh, Y. et al. Use of PKPD model to design and analyze results of a euglycemic clamp study for a very long-acting insulin analogue HM12470. American Diabetes Association's 75th Scientific Sessions [online], (2015).
Arnold, S., Jowett, J. & Ballance, J. Synergistic action of PE0139, a super-long-acting basal insulin & PB1023 a weekly GLP1 receptor agonist. American Diabetes Association's 75th Scientific Sessions [online], (2015).
Jowett, J. & Woods, C. Therapeutic agents comprising insulin amino acid sequences. US Patent 20130150291 (2012).
Marquez, F. et al. PE0139, the first recombinant fully human monomeric super-long-acting basal insulin to display a sustained nearly peakless insulin profile following a single subcutaneous dose in subjects with T2DM supporting weekly dosing. American Diabetes Association's 75th Scientific Sessions [online], (2015).
Novo Nordisk. Novo Nordisk Receives FDA Approval for Tresiba® (insulin degludec injection) for Adults with Type 1 and Type 2 Diabetes. Novo Nordisk [online], (2015).
Hartman, M. L. et al. Liver enzyme results from 7 basal insulin peglispro (BIL) clinical trials in T1D and T2D. American Diabetes Association's 75th Scientific Sessions [online], (2015).
Rosenstock, J. et al. better glycemic control and weight loss with the novel long-acting basal insulin LY2605541 compared with insulin glargine in type 1 diabetes: a randomized, crossover study. Diabetes Care 36, 522–528 (2013).
Cobry, E. et al. Timing of meal insulin boluses to achieve optimal postprandial glycemic control in patients with type 1 diabetes. Diabetes Technol. Ther. 12, 173–177 (2010).
Shah, V. N., Shoskes, A., Tawfik, B. & Garg, S. K. Closed-loop system in the management of diabetes: past, present, and future. Diabetes Technol. Ther. 16, 477–490 (2014).
Zhang, Z., Tang, Y., Yao, S., Zhu, S. & Feng, Y. Protein engineering of insulin: two novel fast-acting insulins [B16Ala] insulin and [B26Ala] insulin. Sci. China C Life Sci. 46, 474–480 (2003).
Weiss, M. Insulin analogues with chlorinated amino acids, US Patent 9079975 (2015).
Weiss, M. Halogen-stabilized insulin. US Patent 8921313 (2014).
Krasner, A. et al. Safety and efficacy of ultra-rapid-acting human insulin formulation BIOD-123 in patients with type 1 diabetes. American Diabetes Association's 74th Scientific Sessions [online], (2014).
Andersen, G. et al. Ultra-rapid BioChaperone insulin lispro (BC LIS): linear dose-response and faster absorption than insulin Lispro (LIS). Diabetologia 58 (Suppl 1), S449–S449 (2015).
Hua, Q.-X. et al. Design of an active ultrastable single-chain insulin analog: synthesis, structure, and therapeutic implications. J. Biol. Chem. 283, 14703–14716 (2008).
Duttaroy, A. et al. Development of a long-acting insulin analog using albumin fusion technology. Diabetes 54, 251–258 (2005).
DiMarchi, R. D. et al. Single-chain insulin agonists exhibiting high activity at the insulin receptor. US Patent 8940860 (2011).
Kaur, Z. P. et al. Discovery of high potency, single-chain insulin analogs with a shortened B-chain and nonpeptide linker. ACS Chem. Biol. 8, 1822–1829 (2013).
Andersen, A. S., et al. Backbone cyclic insulin. J. Pept. Sci. 16, 473–479 (2010).
Stowell, M. H. & Plam, M. Chemically and thermodynamically stable insulin analogues and improved methods for their production. US Patent 9006176 (2015).
Wang, Y., Shao, J., Zaro, J. L. & Shen, W.-C. Proinsulin-transferrin fusion protein as a novel long-acting insulin analog for the inhibition of hepatic glucose production. Diabetes 63, 1779–1788 (2014).
Phillips, N. B., Whittaker, J., Ismail-Beigi, F. & Weiss, M. A. Insulin fibrillation and protein design: topological resistance of single-chain analogs to thermal degradation with application to a pump reservoir. J. Diabetes Sci. Technol. 6, 277–288 (2012).
Polonsky, K. S. & Rubenstein, A. H. C peptide as a measure of the secretion and hepatic extraction of insulin. Pitfalls and limitations. Diabetes 33, 486–494 (1984).
Eaton, R. P., Allen, R. C. & Schade, D. S. Hepatic removal of insulin in normal man: dose response to endogenous insulin secretion. J. Clin. Endocrinol. Metab. 56, 1294–1300 (1983).
Meier, J. J., Veldhuis, J. D. & Butler, P. C. Pulsatile insulin secretion dictates systemic insulin delivery by regulating hepatic insulin extraction in humans. Diabetes 54, 1649–1656 (2005).
Meyer, C., Woerle, H. J., Dostou, J. M., Welle, S. L. & Gerich, J. E. Abnormal renal, hepatic, and muscle glucose metabolism following glucose ingestion in type 2 diabetes. Am. J. Physiol. Endocrinol. Metab. 287, E1049–E1056 (2004).
Canavan, J., Flecknell, P., New, J., Alberti, K. & Home, P. The effect of portal and peripheral insulin delivery on carbohydrate and lipid metabolism in a miniature pig model of human IDDM. Diabetologia 40, 1125–1134 (1997).
Sekigami, T. et al. Comparison between closed-loop portal and peripheral venous insulin delivery systems for an artificial endocrine pancreas. J. Artif. Organs 7, 91–100 (2004).
Herring, R., Jones, R. H. & Russell-Jones, D. L. Hepatoselectivity and the evolution of insulin. Diabetes Obes. Metab. 16, 1–8 (2014). A review discussing an evolutionary reason for, and the physiological importance of, portal delivery of endogenous insulin.
Edgerton, D. S. et al. Insulin's direct effects on the liver dominate the control of hepatic glucose production. J. Clin. Invest. 116, 521–527 (2006).
Tompkins, C. V., Brandenburg, D., Jones, R. H. & Sönksen, P. H. Mechanism of action of insulin and insulin analogues. Diabetologia 20, 94–101 (1981).
Glauber, H. S. et al. In vivo deactivation of proinsulin action on glucose disposal and hepatic glucose production in normal man. Diabetes 35, 311–317 (1986).
Smeeton, F. et al. Differential effects of insulin detemir and neutral protamine hagedorn (NPH) insulin on hepatic glucose production and peripheral glucose uptake during hypoglycaemia in type 1 diabetes. Diabetologia 52, 2317–2323 (2009).
Henry, R. R. et al. Basal insulin peglispro demonstrates preferential hepatic versus peripheral action relative to insulin glargine in healthy subjects. Diabetes Care 37, 2609–2615 (2014).
Shojaee-Moradie, F. et al. Novel hepatoselective insulin analog: studies with a covalently linked thyroxyl-insulin complex in humans. Diabetes Care 23, 1124–1129 (2000).
Seino, S. & Bell, G. I. Alternative splicing of human insulin receptor messenger RNA. Biochem. Biophys. Res. Commun. 159, 312–316 (1989).
Belfiore, A., Frasca, F., Pandini, G., Sciacca, L. & Vigneri, R. Insulin receptor isoforms and insulin receptor/insulin-like growth factor receptor hybrids in physiology and disease. Endocr. Rev. 30, 586–623 (2009). A comprehensive review pertaining to insulin receptor structure, its isoforms, interactions with a ligand and signalling pathways.
Mosthaf, L. et al. Journal 9, 2409 (1990).
Moller, D. E., Yokota, A., Caro, J. F. & Flier, J. S. Tissue-specific expression of two alternatively spliced insulin receptor mRNAs in man. Mol. Endocrinol. 3, 1263–1269 (1989).
Leibiger, B. et al. Selective insulin signaling through A and B insulin receptors regulates transcription of insulin and glucokinase genes in pancreatic β cells. Mol. Cell 7, 559–570 (2001).
Sara, G. V. et al. Receptor-isoform-selective insulin analogues give tissue-preferential effects. Biochem. J. 440, 301–308 (2011).
Siddle, K. Signalling by insulin & IGF receptors: supporting acts and new players. J. Mol. Endocrinol. 47, R1–R10 (2011).
Owens, D. R., Zinman, B. & Bolli, G. Alternative routes of insulin delivery. Diabet. Med. 20, 886–898 (2003).
Khafagy, E.-S., Morishita, M., Onuki, Y. & Takayama, K. Current challenges in non-invasive insulin delivery systems: a comparative review. Adv. Drug Deliv. Rev. 59, 1521–1546 (2007).
Mack, G. S. Pfizer dumps Exubera. Nat. Biotechnol. 25, 1331–1332 (2007).
Iyer, H., Khedkar, A. & Verma, M. Oral insulin — a review of current status. Diabetes Obes. Metab. 12, 179–185 (2010).
Heinemann, L. Insulin pens and new ways of insulin delivery. Diabetes Technol. Ther. 16, S44–S55 (2014).
Kling, J. Sanofi to propel inhalable insulin Afrezza into market. Nat. Biotechnol. 32, 851–852 (2014).
Neumiller, J. & Campbell, R. K. Technosphere® Insulin. BioDrugs 24, 165–172 (2010).
Rosenstock, J. et al. Prandial inhaled insulin plus basal insulin glargine versus twice daily biaspart insulin for type 2 diabetes: a multicentre randomised trial. Lancet 375, 2244–2253 (2010).
MannKind Corporation. MannKind Corporation announces termination of license and collaboration agreement with Sanofi. MannKind Corpoartion [online], (2016).
Eldor, R., Arbit, E., Corcos, A. & Kidron, M. Glucose-reducing effect of the ORMD 0801 oral insulin preparation in patients with uncontrolled type 1 diabetes: a pilot study. PloS ONE 8, e59524 (2013).
Novo Nordisk. OI338GT (NN1953) Phase II trial. Novo Nordisk [online], (2016).
Novo Nordisk. Financial report for the period 1 January 2014 to 31 March 2014. Novo Nordisk Company Announcement [online], (2014).
Wu, Q., Wang, L., Yu, H., Wang, J. & Chen, Z. Organization of glucose-responsive systems and their properties. Chem. Rev. 111, 7855–7875 (2011). A thorough review of glucose-responsive systems and glucose sensors.
Wu, W. & Zhou, S. Responsive materials for self-regulated insulin delivery. Macromol. Biosci 13, 1464–1477 (2013).
Merck & Co., Inc. Merck to Acquire SmartCells, Inc. Merck Press Releases [online], (2010).
Zion, T. C. & Lancaster, T. M. Crystalline insulin-conjugates. US Patent 8906850 (2010).
Chou, D. H.-C. et al. Glucose-responsive insulin activity by covalent modification with aliphatic phenylboronic acid conjugates. Proc. Natl Acad. Sci. USA 112, 2401–2406 (2015).
Hoeg-Jensen, T., Havelund, S., Nielsen, P. K. & Markussen, J. Reversible insulin self-assembly under carbohydrate control. J. Am. Chem. Soc. 127, 6158–6159 (2005).
Hoeg-Jensen, T. et al. Glucose dependent release of insulin from glucose sensing insulin derivatives. US Patent 316999 (2008).
Hoeg-Jensen, T., Jakobsen, P., Sensfuss, U., Fledelius, C. & Ribel-Madsen, U. Insulin derivatives. US Patent WO2011000823 (2010).
Garg, S. K. The role of basal insulin and glucagon-like peptide 1 agonists in the therapeutic management of type 2 diabetes — a comprehensive review. Diabetes Technol. Ther. 12, 11–24 (2010).
Vora, J. Combining incretin-based therapies with insulin realizing the potential in type 2 diabetes. Diabetes Care 36, S226–S232 (2013).
Balena, R., Hensley, I., Miller, S. & Barnett, A. Combination therapy with GLP-1 receptor agonists and basal insulin: a systematic review of the literature. Diabetes Obes. Metab. 15, 485–502 (2013).
Holst, J. & Vilsbøll, T. Combining GLP-1 receptor agonists with insulin: therapeutic rationales and clinical findings. Diabetes Obes. Metab. 15, 3–14 (2013). A review discussing the benefits of insulin and GLP1A combination therapy with early clinical examples.
Baggio, L. L. & Drucker, D. J. Biology of incretins: GLP 1 and GIP. Gastroenterology 132, 2131–2157 (2007).
Mathieu, C. et al. A comparison of adding liraglutide versus a single daily dose of insulin aspart to insulin degludec in subjects with type 2 diabetes (BEGIN: VICTOZA ADD-ON). Diabetes Obes. Metab. 16, 636–644 (2014).
Rosenstock, J. et al. Advancing basal insulin replacement in type 2 diabetes inadequately controlled with insulin glargine plus oral agents: a comparison of adding albiglutide, a weekly GLP-1 receptor agonist, versus thrice-daily prandial insulin lispro. Diabetes Care 37, 2317–2325 (2014).
Gough, S. One-year efficacy and safety of IDegLira in patients with type 2 diabetes. American Diabetes Association's 74th Scientific Sessions [online], (2014).
Vilsbøll, T. et al. The pathophysiology of diabetes involves a defective amplification of the late-phase insulin response to glucose by glucose-dependent insulinotropic polypeptide — regardless of etiology and phenotype. J. Clin. Endocrinol. Metab. 88, 4897–4903 (2003).
Miyawaki, K. et al. Inhibition of gastric inhibitory polypeptide signaling prevents obesity. Nat. Medicine 8, 738–742 (2002).
Kim, S.-J. et al. GIP-overexpressing mice demonstrate reduced diet-induced obesity and steatosis, and improved glucose homeostasis. PLoS ONE 7, e40156 (2012).
Kerr, B. D. et al. Fatty acid derivatised analogues of glucose-dependent insulinotropic polypeptide with improved antihyperglycaemic and insulinotropic properties. Biochem. Pharmacol. 78, 1008–1016 (2009).
Gault, V. A., Porter, D. W., Irwin, N. & Flatt, P. R. Comparison of sub-chronic metabolic effects of stable forms of naturally occurring GIP (1–30) and GIP (1–42) in high-fat fed mice. J. Endocrinol. 208, 265–271 (2011).
Finan, B. et al. Unimolecular dual incretins maximize metabolic benefits in rodents, monkeys, and humans. Sci. Transl Med. 5, 209ra151 (2013). An example of improved pharmacology obtained through combination of two incretin hormones.
Baeshen, N. A. et al. Cell factories for insulin production. Microb. Cell Fact. 13, 141 (2014).
Keasling, J. D., Mendoza, A. & Baran, P. S. Synthesis: a constructive debate. Nature 492, 188–189 (2012).
Liu, F., Luo, E. Y., Flora, D. B. & Mayer, J. P. Concise synthetic routes to human insulin. Org. Lett. 15, 960–963 (2013).
Liu, F., Luo, E. Y., Flora, D. B. & Mezo, A. R. A. Synthetic route to human insulin using isoacyl peptides. Angew. Chem. Int. Ed. Engl. 53, 3983–3987 (2014).
Zaykov, A. N., Mayer, J. P., Gelfanov, V. M. & DiMarchi, R. D. Chemical synthesis of insulin analogs through a novel precursor. ACS Chem. Biol. 9, 683–691 (2013).
Walsh, G. Therapeutic insulins and their large-scale manufacture. Appl. Microbiol. Biotechnol. 67, 151–159 (2005).
Avital-Shmilovici, M. et al. Fully convergent chemical synthesis of ester insulin: determination of the high resolution X-ray structure by racemic protein crystallography. J. Am. Chem. Soc. 135, 3173–3185 (2013).
Sohma, Y., Hua, Q.-X., Whittaker, J., Weiss, M. A. & Kent, S. B. H. Design and folding of [GluA4(OβThrB30)]Insulin (“ester insulin”): a minimal proinsulin surrogate that can be chemically converted into human insulin. Angew. Chem. Int. Ed. Engl. 49, 5489–5493 (2010).
Tofteng, A. P., Jensen, K. J., Schäffer, L. & Hoeg-Jensen, T. Total synthesis of desB30 insulin analogues by biomimetic folding of single-chain precursors. ChemBioChem 9, 2989–2996 (2008).
Qiang, G. et al. Identification of a small molecular insulin receptor agonist with potent antidiabetes activity. Diabetes 63, 1394–1409 (2014).
Bhaskar, V. et al. Fully human, allosteric monoclonal antibody that activates the insulin receptor and improves glycemic control. Diabetes 61, 1263–1271 (2012).
Bedinger, D. H., Goldfine, I. D., Corbin, J. A., Roell, M. K. & Adams, S. H. Differential pathway coupling of the activated insulin receptor drives signaling selectivity by XMetA, an allosteric partial agonist antibody. J. Pharmacol. Exp. Ther. 353, 35–43 (2015).
Schäffer, L. et al. Assembly of high-affinity insulin receptor agonists and antagonists from peptide building blocks. Proc. Natl Acad. Sci. USA 100, 4435–4439 (2003).
Knudsen, L. et al. Agonism and antagonism at the insulin receptor. PloS ONE 7, e51972 (2012).
Jensen, M., Hansen, B., De Meyts, P., Schäffer, L. & Ursø, B. Activation of the insulin receptor by insulin and a synthetic peptide leads to divergent metabolic and mitogenic signaling and responses. J. Biol. Chem. 282, 35179–35186 (2007).
Yunn, N.-O. et al. Agonistic aptamer to the insulin receptor leads to biased signaling and functional selectivity through allosteric modulation. Nucleic Acids Res. 43, 7688–7701 (2015).
Mameli, C. et al. 7 year follow up retrospective, international, multicenter study of insulin pump therapy in children and adolescents with type 1 diabetes. Acta Diabetol. 51, 205–210 (2014).
Pickup, J. C. Diabetes: insulin pump therapy for type 2 diabetes mellitus. Nat. Rev. Endocrinology 10, 647–649 (2014).
Pickup, J. C. Insulin-pump therapy for type 1 diabetes mellitus. N. Engl. J. Med. 366, 1616–1624 (2012).
Bouwens, L., Houbracken, I. & Mfopou, J. K. The use of stem cells for pancreatic regeneration in diabetes mellitus. Nat. Rev. Endocrinology 9, 598–606 (2013). Stem-cell technology offers a prospect of in vitro production of pancreatic β-cells and organ restoration.
Pagliuca, F. W. et al. Generation of functional human pancreatic β cells in vitro. Cell 159, 428–439 (2014).
Goh, S.-K. et al. Perfusion-decellularized pancreas as a natural 3D scaffold for pancreatic tissue and whole organ engineering. Biomaterials 34, 6760–6772 (2013).
Brethauer, S. A. et al. Can diabetes be surgically cured? Long-term metabolic effects of bariatric surgery in obese patients with type 2 diabetes mellitus. Ann. Surg. 258, 628–636 (2013). Discussion of bariatric surgery as a potential cure for T2D.
Sjöström, L. et al. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N. Engl. J. Med. 351, 2683–2693 (2004).
Schauer, P. R. et al. Bariatric surgery versus intensive medical therapy in obese patients with diabetes. N. Engl. J. Med. 366, 1567–1576 (2012).
Rodgers, R. J., Tschöp, M. H. & Wilding, J. P. Anti-obesity drugs: past, present and future. Dis. Model. Mech. 5, 621–626 (2012).
Xu, J. et al. Acute glucose-lowering and insulin-sensitizing action of FGF21 in insulin-resistant mouse models — association with liver and adipose tissue effects. Am. J. Physiol. Endocrinol. Metab. 297, E1105–E1114 (2009).
Holland, W. L. et al. An FGF21 adiponectin-ceramide axis controls energy expenditure and insulin action in mice. Cell Metab. 17, 790–797 (2013).
Denroche, H. C., Huynh, F. K. & Kieffer, T. J. The role of leptin in glucose homeostasis. J. Diabetes Investig. 3, 115–129 (2012).
Fani, L., Bak, S., Delhanty, P., van Rossum, E. & van den Akker, E. The melanocortin 4 receptor as target for obesity treatment: a systematic review of emerging pharmacological therapeutic options. Int. Journal Obes. 38, 163–169 (2014).
Pearson, T. L. Practical aspects of insulin pen devices. J. Diabetes Sci. Technol. 4, 522–531 (2010).
Zijlstra, E., Heinemann, L. & Plum-Mörschel, L. Oral insulin reloaded a structured approach. J. Diabetes Sci. Technol. 8, 458–465 (2014).
Kidron, M., Neutel, J. & Arbit, E. Preprandial oral insulin (ORMD-0801) reduces rapid-acting insulin requirements and fasting glucose levels in T1DM patients (poster). American Diabetes Association's 75th Scientific Sessions [online], (2015).
Geiss, L. S., Herman, W. H. & Smith, P. J. in Diabetes in America (ed R. Aubert) 233–255 (DIANE Publishing, 1995).
Jonassen, I. et al. Design of the novel protraction mechanism of insulin degludec, an ultra-long-acting basal insulin. Pharm. Res. 29, 2104–2114 (2012).
Wang, F., Surh, J. & Kaur, M. Insulin degludec as an ultralong-acting basal insulin once a day: a systematic review. Diabetes Meta. Synd. Obes. 5, 191–204 (2012).
Sorli, C. et al. Elderly patients with diabetes experience a lower rate of nocturnal hypoglycaemia with insulin degludec than with insulin glargine: a meta-analysis of Phase IIIa trials. Drugs Aging 30, 1009–1018 (2013).
Birkeland, K. I. et al. Insulin degludec in a flexible daily dosing regimen provides similar glycaemic control without increasing rates of hypoglycaemia compared to dosing the same time daily in type 2 diabetes. Diabetologia 54 (Suppl. 1), 542 (2011).
Dorey, E. FDA dashes Novo's hopes. Nat. Biotechnol. 31, 266–266 (2013).
Sinha, V. P. et al. Single-dose pharmacokinetics and glucodynamics of the novel, long-acting basal insulin LY2605541 in healthy subjects. J.Clin. Pharmacol. 54, 792–799 (2014).
Bergenstal, R. M. et al. Lower glucose variability and hypoglycemia measured by continuous glucose monitoring with novel long-acting insulin LY2605541 versus insulin glargine. Diabetes Care 37, 659–665 (2013).
Jacober, S. J. et al. Contrasting weight changes with LY2605541, a novel long-acting insulin, and insulin glargine despite similar improved glycaemic control in T1DM and T2DM. Diabetes Obes. Metab. 16, 351–356 (2014).
Wigley, F. M., Londono, J. H., Wood, S. H., Shipp, J. C. & Waldman, R. H. Insulin across respiratory mucosae by aerosol delivery. Diabetes 20, 552–556 (1971).
Eli Lilly and Company. Lilly Ends Basal Insulin Peglispro Development Program. Lilly Investor Press Release [online], (2014).
Buse, J. B. et al. Superior HbA1c reduction with basal insulin peglispro (BIL) versus insulin glargine (GL) alone or with oral antihyperglycemic medications (OAMs) in T2D patients (Pts) previously treated with basal insulin: IMAGINE 5. American Diabetes Association's 75th Scientific Sessions [online], (2015).
Blevins, T. et al. Superior HbA1c reduction with basal insulin peglispro (BIL) versus insulin glargine (GL) and preprandial insulin lispro in a double-blind study in patients (pts) with type 2 diabetes (T2D): IMAGINE 4. American Diabetes Association's 75th Scientific Sessions [online], (2015).
Bergenstal, R. M. et al. Superior reduction of HbA1c in a double-blind, randomized study of basal insulin peglispro (BIL) versus insulin glargine (GL) in patients (pts) with T1D: IMAGINE 3. American Diabetes Association's 75th Scientific Sessions [online], (2015).
Hansen, R. et al. LY2605541: leveraging hydrodynamic size to develop a novel basal insulin. Diabetes 61, A228 (2012).
Zijlstra, E. et al. Dance 501 inhaled human insulin has a dose-linear response and similar within-subject variability as rapid-acting insulin lispro. American Diabetes Association's 75th Scientific Sessions [online], (2015).
Smith, N. B. et al. Ultrasound-mediated transdermal transport of insulin in vitro through human skin using novel transducer designs. Ultrasound Med. Biol. 29, 311–317 (2003).
Transdermal Specialties Inc. The U-Strip — Insulin Patch. Transdermal Specialties [online], (2015).
Gough, S. C. L. et al. Efficacy and safety of a fixed-ratio combination of insulin degludec and liraglutide (IDegLira) compared with its components given alone: results of a Phase 3, open-label, randomised, 26 week, treat to target trial in insulin-naive patients with type 2 diabetes. Lancet Diabetes Endocrinol. 2, 885–893 (2014).
Rosenstock, J. et al. Improved glucose control without increased hypoglycemia risk at any level of HbA1c reduction with insulin glargine/lixisenatide fixed-ratio combination (LixiLan) versus insulin glargine alone both added on to metformin in type 2 diabetes (T2DM). American Diabetes Association's 75th Scientific Sessions [online], (2015).
Sanofi. FDA accepts Sanofi new drug application for once-daily fixed-ratio combination of insulin glargine and lixisenatide. Sanofi [online], (2016).
Onishi, Y., Ono, Y., Rabøl, R., Endahl, L. & Nakamura, S. Superior glycaemic control with once-daily insulin degludec/insulin aspart versus insulin glargine in Japanese adults with type 2 diabetes inadequately controlled with oral drugs: a randomized, controlled phase 3 trial. Diabetes Obes. Metab. 15, 826–832 (2013).
Fulcher, G. R. et al. Comparison of insulin degludec/insulin aspart and biphasic insulin aspart 30 in uncontrolled, insulin-treated type 2 diabetes: a phase 3a, randomized, treat to target trial. Diabetes Care 37, 2084–2090 (2014).
Hirsch, I. B. et al. Insulin degludec/insulin aspart administered once daily at any meal, with insulin aspart at other meals versus a standard basal-bolus regimen in patients with type 1 diabetes. A 26 week, phase 3, randomized, open-label, treat to target trial. Diabetes Care 35, 2174–2181 (2012).
Acknowledgements
The authors wish to dedicate this review to the memory of Ronald Chance. They are also thankful for all the guidance provided to them over the years by multiple international authorities in the biology and chemistry of insulin, specifically J. Amatruda, J. Caro, P. Cryer, B. Frank, J. Galloway, V. Gelfanov, S. Kent, R. Kahn, D. Kelly, P. Li, F. Liu, D. Perez-Tilve, S. Taylor, M. Tschoep, L. Vignati, M. Weiss and R. Whitcomb.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
R.D.D. is a founder and Chief Scientific Officer of Calibrium LLC. J.P.M. is employee and shareholder of Calibrium LLC. A.N.Z. is a consultant and shareholder of Calibrium LLC.
Related links
Glossary
- Diabetes mellitus
-
A metabolic disease associated with elevated levels of glucose that results from pancreatic insufficiency in insulin production and/or reduced target-tissue insulin sensitivity. Type 1 or juvenile diabetes is caused by immunological destruction of insulin-producing pancreatic β-cells. Type 2 or adult-onset diabetes is a progressive condition characterized by insulin resistance and is often associated with obesity.
- Insulin analogues
-
Compounds derived from insulin that has been altered in its structure for the primary purpose of enhanced pharmaceutics or pharmacology. The two main types of insulin analogues are basal or sustained-action analogues, which are used for daytime and night time glucose control, and bolus or rapid-acting analogues, which are used for mealtime glucose control and pump administration.
- Subcutaneous delivery
-
The drug is delivered through injection to the subcutis, the layer of tissue located immediately beneath the skin layer. It remains the most common method of insulin administration.
- Extended time–action profile
-
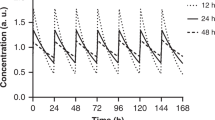
Characteristic of long-acting insulin analogues, which produce relatively stable insulin levels for 12–24 hours after injection. These analogues are sometimes referred to as 'peakless' insulins by virtue of their flat pharmacokinetic profiles.
- Therapeutic index
-
The ratio between the dose of the drug that causes an adverse effect relative to the therapeutic dose. Insulin has an inherently low therapeutic index. This represents a persistent risk for overdosing that can result in life-threatening hypoglycaemia.
- Incretin hormone
-
A gut-derived peptide hormone that stimulates insulin secretion after food consumption. Additional functions of incretin hormones include inhibition of glucagon secretion, restriction of gastric motility and appetite suppression. The two most prominent physiological hormones within this class are glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide 1 (GLP1).
- Single-chain insulin
-
(SCI). An insulin analogue or its precursor in which the two individual peptide chains (A and B) are covalently connected. The two chains can be linked by a connecting sequence (such as the proinsulin C-peptide), synthetic linker or fused directly through an amide bond.
- β-cells
-
Functional endocrine cells that are located in pancreatic islets and are responsible for biosynthesis, storage and secretion of insulin under glucose control.
- Gastric bypass surgery
-
A bariatric procedure that surgically reduces the size of the stomach. This restricts the quantity of absorbed nutrients and alters local hormone production and action to collectively achieve body weight reduction.
Rights and permissions
About this article
Cite this article
Zaykov, A., Mayer, J. & DiMarchi, R. Pursuit of a perfect insulin. Nat Rev Drug Discov 15, 425–439 (2016). https://doi.org/10.1038/nrd.2015.36
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd.2015.36
This article is cited by
-
Simultaneous attenuation of hyperglycemic memory-induced retinal, pulmonary, and glomerular dysfunctions by proinsulin C-peptide in diabetes
BMC Medicine (2023)
-
Enhanced hexamerization of insulin via assembly pathway rerouting revealed by single particle studies
Communications Biology (2023)
-
A stepwise activation model for the insulin receptor
Experimental & Molecular Medicine (2023)
-
Genetically encoded discovery of perfluoroaryl macrocycles that bind to albumin and exhibit extended circulation in vivo
Nature Communications (2023)
-
Genetic-code-expanded cell-based therapy for treating diabetes in mice
Nature Chemical Biology (2022)