Key Points
-
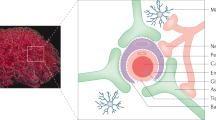
The blood–brain barriers (BBBs) are dynamic, adaptable, interactive monolayers of cells, including endothelial, ependymal and tanycytic cells, that participate in central nervous system (CNS) protection, are responsible for CNS nutrition and homeostasis, and facilitate serum-based brain–body communications.
-
The cells forming the BBB are in communication with other cells of the CNS, thus forming the neurovascular unit. This communication informs the BBB of the needs of the CNS, allowing it to adapt to the needs of the CNS.
-
The BBB also communicates with circulating immune cells and via blood-borne signals with the peripheral tissues. Through transport, secretion and other mechanisms, the BBB relays information between the periphery and the CNS.
-
The complexity of the BBB complicates CNS drug delivery, but also provides many unique opportunities for drug delivery. Manipulation of transporters, secretory functions, the extracellular pathways, and adsorptive transcytosis are examples of promising approaches to drug development.
-
The complexity of the BBB predisposes it to dysfunctions that can result in or promote disease. Such dysfunctions include BBB disruption as well as dysfunctions related to BBB transporters, neurovascular unit communication and secretion. Thus, the BBB itself can be a therapeutic target.
Abstract
One of the biggest challenges in the development of therapeutics for central nervous system (CNS) disorders is achieving sufficient blood–brain barrier (BBB) penetration. Research in the past few decades has revealed that the BBB is not only a substantial barrier for drug delivery to the CNS but also a complex, dynamic interface that adapts to the needs of the CNS, responds to physiological changes, and is affected by and can even promote disease. This complexity confounds simple strategies for drug delivery to the CNS, but provides a wealth of opportunities and approaches for drug development. Here, I review some of the most important areas that have recently redefined the BBB and discuss how they can be applied to the development of CNS therapeutics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Neuwelt, E. et al. Strategies to advance translational research into brain barriers. Lancet Neurol. 7, 84–96 (2008).
Hawkins, B. T. & Davis, T. P. The blood–brain barrier/neurovascular unit in health and disease. Pharmacol. Rev. 57, 173–185 (2005).
Daneman, R., Zhou, L., Kebede, A. A. & Barres, B. A. Pericytes are required for blood–brain barrier integrity during embryogenesis. Nature 468, 562–566 (2010). Demonstrates the role of pericytes in establishing the BBB and shows that barrier function is present even during the fetal period.
Abbott, N. J., Ronnback, L. & Hansson, E. Astrocyte–endothelial interactions at the blood–brain barrier. Nat. Rev. Neurosci. 7, 41–53 (2006).
Banks, W. A. The blood–brain barrier in neuroimmunology: tales of separation and assimilation. Brain Behav. Immun. 44, 1–8 (2015). Review of mechanisms by which the BBB defines the neuroimmune system.
Bell, R. D. et al. Apolipoprotein E controls cerebrovascular integrity via cyclophilin A. Nature 485, 512–516 (2012).
Oldendorf, W. H. Brain uptake of radio-labelled amino acids, amines and hexoses after arterial injection. Am. J. Physiol. 221, 1629–1639 (1971).
Kastin, A. J. & Pan, W. Blood–brain barrier and feeding: regulatory roles of saturable transport systems for ingestive peptides. Curr. Pharm. Design 14, 1615–1619 (2008).
Pan, W. & Kastin, A. J. Interactions of cytokines with blood–brain barrier: implications for feeding. Curr. Pharm. Design 9, 827–831 (2003).
Banks, W. A. et al. Delivery across the blood–brain barrier of antisense directed againt amyloid β: reversal of learning and memory deficits in mice overexpressing amyloid precursor protein. J. Pharmacol. Exp. Ther. 297, 1113–1121 (2001).
Soilu-Hanninen, M. et al. Treatment of experimental autoimmune encephalomyelitis with antisense oligonucleotides against the low affinity neurotrophin receptor. J. Neurosci. Res. 59, 712–721 (2000).
Erickson, M. A. et al. Peripheral administration of antisense oligonucleotides targeting the amyloid-β protein precursor reverses AβPP and LRP-1 overexpression in aged SAMP8 mouse brain. J. Alzheimers Dis. 28, 951–960 (2012).
Farr, S. A. et al. Antisense oligonucelotide against GSK-3β in brain of SAMP8 mice improves learning and memory and decreases oxidative stress: involvement of transcription factor Nrf2 and implications for Alzheimer's disease. Free Radic. Biol. Med. 67, 387–395 (2013).
Dogrukol-Ak, D. et al. Isolation of peptide transport system-6 from brain endothelial cells: therapeutic effects with antisense inhibition in Alzheimer's and stroke models. J. Cereb. Blood Flow Metab. 29, 411–422 (2009).
Poon, H. F. et al. Proteomic identification of less oxidized brain proteins in aged senescence-accelerated mice following administration of antisense oligonucleotide directed at the Aβ region of amyloid precursor protein. Brain Res. Mol. Brain Res. 138, 8–13 (2005).
Farr, S. A., Erickson, M. A., Niehoff, M. L., Banks, W. A. & Morley, J. E. Central and peripheral administration of antisense oligonucleotide targeting amyloid precursor protein improves learning and memory and reduces neuroinflammatory cytokines in Tg2576 (APPswe) mice. J. Alzheimers Dis. 40, 1005–1016 (2014).
Poduslo, J. F. & Curran, G. L. Permeability at the blood–brain barrier and blood–nerve barriers of the neurotrophic factors: NGF, CNTF, NT-3, BDNF. Mol. Brain Res. 36, 280–286 (1996).
Pan, W., Banks, W. A., Fasold, M. B., Bluth, J. & Kastin, A. J. Transport of brain-derived neurotrophic factor across the blood–brain barrier. Neuropharmacology 37, 1553–1561 (1998).
Fernstrom, J. D. Branched-chain amino acids and brain function. J. Nutr. 135, 1439S–1546S (2005).
Elinav, E. et al. Pegylated leptin antagonist is a potent orexigenic agent: preparation and mechanism of activity. Endocrinology 150, 3083–3091 (2009).
Georgieva, J. V., Hoekstra, D. & Zuhorn, I. S. Smuggling drugs into the brain: an overview of ligands targeting transcytosis for drug delivery across the blood–brain barrier. Pharmaceutics 6, 557–583 (2014).
Pardridge, W. M. Blood–brain barrier drug delivery of IgG fusion proteins with a transferrin receptor monoclonal antibody. Expert Opin. Drug Deliv. 20, 1–16 (2014).
Wang, T. et al. Toll-like receptor 3 mediates West Nile virus entry into the brain causing lethal encephalitis. Nat. Med. 10, 1366–1373 (2004).
Dohgu, S., Ryerse, J. S., Robinson, S. M. & Banks, W. A. Human immunodeficiency virus-1 uses the mannose-6-phosphate receptor to cross the blood–brain barrier. PLoS ONE 7, e41623 (2012).
Hambleton, S. Chickenpox. Curr. Opin. Infect. Dis. 18, 235–240 (2005).
McCall, R. L. et al. Pathogen-inspired drug delivery to the central nervous system. Tissue Barriers 8, 3944449 (2014).
Atwal, J. K. et al. A therapeutic antibody targeting BACE1 inhibits amyloid-β production in vivo. Sci. Transl Med. 3, 84ra43 (2011).
Yu, Y. J. et al. Boosting brain uptake of the therapeutic antibody by reducing its affinity for a transcytosis target. Sci. Transl Med. 3, 84ra44 (2011).
Niewoehner, J. et al. Increased brain penetration and potency of a therapeutic antibody using a monovalent molecular shuttle. Neuron 81, 49–60 (2014).
Moinuddin, A., Morley, J. E. & Banks, W. A. Regional variations in the transport of interleukin-1α across the blood–brain barrier in ICR and aging SAMP8 mice. Neuroimmunomodulation 8, 165–170 (2000).
Maness, L. M., Banks, W. A., Zadina, J. E. & Kastin, A. J. Selective transport of blood-borne interleukin-1α into the posterior division of the septum of the mouse brain. Brain Res. 700, 83–88 (1995).
Begley, D. J. ABC transporters and the blood–brain barrier. Curr. Pharm. Design 10, 1295–1312 (2004). Classic and detailed description of P-gp and other ABC transporter CNS-to-blood efflux systems.
Nicita, F. et al. Efficacy of verapamil as an adjunctive treatment in children with drug-resistant epilepsy: a pilot study. Seizure 23, 36–40 (2014).
Faraci, F. M. & Heistad, D. D. Regulation of the cerebral circulation: role of endothelium and potassium channels. Physiol. Rev. 78, 53–97 (1998).
Reyes, T. M., Fabry, Z. & Coe, C. L. Brain endothelial cell production of a neuroprotective cytokine, interleukin-6, in response to noxious stimuli. Brain Res. 851, 215–220 (1999).
Kis, B. et al. Cerebral endothelial cells are a major source of adrenomedullin. J. Neuroendocrinol. 14, 283–293 (2002).
Dohgu, S., Fleegal-DeMotta, M. A. & Banks, W. A. Lipopolysaccharide-enhanced transcellular transport of HIV-1 across the blood–brain barrier is mediated by luminal microvessel IL-6 and GM-CSF. J. Neuroinflamm. 8, 167 (2011).
Cao, C., Matsumura, K., Yamagata, K. & Watanabe, Y. Involvement of cyclooxygenase-2 in LPS-induced fever and regulation of its mRNA by LPS in the rat brain. Am. J. Physiol. 272, R1712–R1725 (1997).
Dohgu, S. & Banks, W. A. Brain pericytes increase the lipopolysaccharide-enhanced transcytosis of HIV-1 free virus across the in vitro blood–brain barrier: evidence for cytokine-mediated pericyte-endothelial cell cross talk. Fluids Barriers CNS 10, 23 (2013).
Deli, M. A., Abraham, C. R., Kataoka, Y. & Niwa, M. Permeability studies on in vitro blood–brain barrier models: physiology, pathology, and pharmacology. Cell. Mol. Neurobiol. 25, 59–127 (2005). An authoritative and thorough review on the utility of, and approaches to, the gold standard in vitro monolayer model of the vascular BBB.
Johanson, C. E. in Neuromethods; The Neuronal Microenvironment (eds Boulton, A. et al.) 33–104 (The Humana Press, 1988).
Verma, S., Nakaoke, R., Dohgu, S. & Banks, W. A. Release of cytokines by brain endothelial cells: a polarized response to lipopolysaccharide. Brain Behav. Immun. 20, 449–455 (2006).
Engstrom, L. et al. Lipopolysaccharide-induced fever depends on prostaglandin E2 production specifically in brain endothelial cells. Endocrinology 153, 4849–4861 (2012). An elegant example of the BBB relay arm of the neuroimmune axis: blood-borne LPS binds to the luminal surface of the BEC, stimulating release of PGE2 from the abluminal surface into the CNS, thus inducing fever.
Banks, W. A. & Robinson, S. M. Minimal penetration of lipopolysaccharide across the murine blood–brain barrier. Brain Behav. Immun. 24, 102–109 (2010).
Parepally, J. M., Mandula, H. & Smith, Q. R. Brain uptake of nonsteroidal anti-inflammatory drugs: ibuprofen, flurbiprofen, and indomethacin. Pharm. Res. 23, 873–881 (2006).
Morimoto, A., Murakami, N., Nakamori, T. & Watanabe, T. Evidence for separate mechanisms of induction of biphasic fever inside and outside the blood–brain barrier. J. Physiol. 383, 629–637 (1987).
Saunders, N. R., Daneman, R., Dziegielewska, K. M. & Liddelow, S. A. Transporters of the blood–brain and blood–CSF interfaces in development and in the adult. Mol. Aspects Med. 34, 742–752 (2013).
Kastin, A. J. & Akerstrom, V. Fasting, but not adrenalectomy, reduces transport of leptin into the brain. Peptides 21, 679–682 (2000).
Banks, W. A., Burney, B. O. & Robinson, S. M. Effects of triglycerides, obesity, and starvation on ghrelin transport across the blood–brain barrier. Peptides 29, 2061–2065 (2008).
Pan, W., Cain, C., Yu, Y. & Kastin, A. J. Receptor-mediated transport of LIF across blood–spinal cord barrier is upregulated after spinal cord injury. J. Neuroimmunol. 174, 119–125 (2006).
Pan, W. et al. Stroke upregulates TNFα transport across the blood–brain barrier. Exp. Neurol. 198, 222–233 (2006).
Somogyvari-Vigh, A., Pan, W., Reglodi, D., Kastin, A. J. & Arimura, A. Effect of middle cerebral artery occulsion on the passage of pituitary adenylate cyclase activating polypeptide across the blood–brain barrier in the rat. Regul. Pept. 91, 89–95 (2000).
Yu, C. et al. Neuroinflammation activates Mdr1b efflux transport through NFκB: promoter analysis in BBB endothelia. Cell Physiol. Biochem. 22, 745–756 (2008).
Yu, C., Pan, W., Tu, H., Waters, S. & Kastin, A. J. TNF activates MDR1 (P-glycoprotein) in cerebral microvascular endothelial cells. Cell Physiol. Biochem. 20, 853–858 (2007).
Bauer, B., Hartz, A. M. S. & Miller, D. S. Tumor necrosis factor α and endothelin-1 increase P-glycoprotein expression and transport activity at the blood–brain barrier. Mol. Pharmacol. 71, 667–675 (2007).
Chikale, E. G., Burton, P. S. & Borchardt, R. T. The effect of verapamil on the transport of peptides across the blood–brain barrier in rats: kinetic evidence for an apically polarized efflux mechanism. J. Pharmacol. Exp. Ther. 273, 298–303 (1995).
Drion, N., Lemaire, M., Lefauconnier, J. M. & Scherrmann, J. M. Role of P-glycoprotein in the blood–brain transport of colchicine and vinblastine. J. Neurochem. 67, 1688–1693 (1996).
Banks, W. A. Enhanced leptin transport across the blood–brain barrier by α1-adrenergic agents. Brain Res. 899, 209–217 (2001).
Vogler, C. et al. Enzyme replacement in murine mucopolysaccharidosis type VII: neuronal and glial response to β-glucuronidase requires early initiation of enzyme replacement therapy. Pediatr. Res. 45, 838–844 (1999).
Urayama, A., Grubb, J. H., Banks, W. A. & Sly, W. S. Epinephrine enhances lysosomal enzyme delivery across the blood–brain barrier by up-regulation of the mannose 6-phosphate receptor. Proc. Natl Acad. Sci. USA 31, 12873–12878 (2007).
van Vliet, E. A. et al. COX-2 inhibition controls P-glycoprotein expression and promotes brain delivery of phenytoin in chronic epilipetic rats. Neuropharmacology 58, 404–412 (2010).
Loscher, W. & Potschka, H. Role of multidrug transporters in pharmacoresistance to antiepileptic drugs. J. Pharmacol. Exp. Ther. 30, 7–14 (2002).
Robins, S. J., Collins, D., McNamara, J. R. & Bloomfield, H. E. Body weight, plasma insulin, and coronary events with gemfibrozil in the Veterans Affairs High-Density Lipoprotein Intervenation Trial (VA-HIT). Atherosclerosis 196, 849–855 (2007).
Mandi, Y. et al. Nitric oxide production and MDR expression by human brain endothelial cells. Anticancer Res. 18, 3049–3052 (1998).
Banks, W. A. Is obesity a disease of the blood–brain barrier? Physiological, pathological, and evolutionary considerations. Curr. Pharm. Design 9, 801–809 (2003).
Liu, J. Y. et al. Neuropathology of the blood–brain barrier and pharmaco-resistance in human epilepsy. Brain 135, 3115–3133 (2012).
Kumar, A., Tripathi, D., Paliwal, V. K., Neyaz, Z. & Agarwal, V. Role of P-glycoprotein in refractoriness of seizures to antiepileptic drugs in Lennox–Gastaut syndrome. J. Child Neurol. 30, 223–227 (2014).
Greig, N. H., Brossi, A., Pei, X.-F., Ingram, D. K. & Soncrant, T. T. in New Concepts of a Blood–Brain Barrier (eds Greenwood, J. et al.) 251–264 (Plenum Press, 1995). A clear, concise review of the major factors that control drug entry into the CNS.
Cornford, E. M., Braun, L. D., Oldendorf, W. H. & Hill, M. A. Comparison of lipid-mediated blood–brain-barrier penetrability in neonates and adults. Am. J. Physiol. 243, C161–C168 (1982).
Oldendorf, W. H. Lipid solubility and drug penetration of the blood–brain barrier. Proc. Soc. Exp. Biol. Med. 147, 813–816 (1974).
Rall, D. P., Stabenau, J. R. & Zubrod, C. G. Distribution of drugs between blood and cerebrospinal fluid: general methodology and effect of pH gradients. J. Pharmacol. Exp. Ther. 125, 185–193 (1959).
Banks, W. A. & Kastin, A. J. Peptides and the blood–brain barrier: lipophilicity as a predictor of permeability. Brain Res. Bull. 15, 287–292 (1985).
Chikhale, E. G., Ng, K. Y., Burton, P. S. & Borchardt, R. T. Hydrogen bonding potential as a determinant of the in vitro and in situ blood–brain barrier permeability of peptides. Pharm. Res. 11, 412–419 (1994).
Gray, R. A. et al. Delta-sleep-inducing peptide: solution conformational studies of a membrane-permeable peptide. Biochemistry 33, 1323–1331 (1994).
Begley, D. J. Strategies for delivery of peptide drugs to the central nervous system: exploiting molecular structure. J. Control. Release 29, 293–306 (1994).
Brownson, E. A., Abbruscato, T. J., Gillespie, T. J., Hruby, V. J. & Davis, T. P. Effect of peptidases at the blood brain barrier on the permeability of enkephalin. J. Pharmacol. Exp. Ther. 270, 675–680 (1994).
Hardebo, J. E. & Owman, C. in Pathophysiology of the Blood–Brain Barrier (eds Johansson, B. B., Owman, C. & Widner, H.) 41–55 (Elsevier, 1990).
Kalaria, R. N., Mitchell, M. J. & Harik, S. I. Correlation of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine neurotoxicity with blood–brain barrier monoamine oxidase activity. Proc. Natl Acad. Sci. USA 84, 3521–3525 (1987). A classic example of the enzymatic barrier and an early example of the NVU.
Svendgaard, N. A., Bjorklund, A., Hardebo, J. & Stenevi, U. Axonal degeneration associated with a defective blood–brain barrier in cerebral implants. Nature 225, 334–336 (1975).
van Gelder, N. M. in Brain Barrier Systems (eds Lajtha, A. & Ford, D. H.) 259–271 (Elsevier, 1968).
Novakovic, Z. M., Anderson, B. M. & Grasso, P. Myristic acid conjugation of [D-Leu-4]-OB3, a biologically active leptin-related synthetic peptide amide, significantly improves its pharmacokinetic profile and efficacy. Peptides 62, 176–182 (2014).
Grubb, J. H. et al. Chemically modified β-glucuronidase crosses blood–brain barrier and clears neuronal storage in murine mucopolysaccharidosis VII. Proc. Natl Acad. Sci. USA 105, 2616–2621 (2008).
Drewes, L. R., Conway, W. P. & Gilboe, D. D. Net amino acid transport between plasma and erythrocytes and perfused dog brain. Am. J. Physiol. 2, E320–E325 (1977).
Jacquez, J. A. Red blood cell as glucose carrier: significance for placental and cerebral glucose transfer. Am. J. Physiol. 246, R289–R298 (1984).
Patel, A. et al. Soluble interleukin-6 receptor induces motor sterotypies and co-localizes with Gp130 in regions linked to cortico–striato–thalamo-cortical circuits. PLoS ONE 7, e1623 (2012).
Banks, W. A. Are the extracellular pathways a conduit for the delivery of therapeutics to the brain? Curr. Pharm. Design 10, 1365–1370 (2004).
Broadwell, R. D. & Sofroniew, M. V. Serum proteins bypass the blood–brain barrier for extracellular entry to the central nervous system. Exp. Neurol. 120, 245–263 (1993). The first elucidation of the extracellular pathways to the CNS. Later studies showed that therapeutic antibodies and other drugs with similar pharmacokinetic properties can use this route to access the CNS.
Garg, A. & Balthasar, J. P. Investigation of the influence of FcRn on the distribution of IgG to the brain. AAPS J. 11, 553–557 (2009).
Banks, W. A. et al. Anti-amyloid beta protein antibody passage across the blood–brain barrier in the SAMP8 mouse model of Alzheimer's disease: an age related selective uptake with reversal of learning impairment. Exp. Neurol. 206, 248–256 (2007).
Mellman, I., Fuchs, R. & Helenius, A. Acidification of the endocytic and exocytic pathways. Annu. Rev. Biochem. 55, 663–700 (1986).
Hardebo, J. E. & Kahrstrom, J. Endothelial negative surface charge areas and blood–brain barrier function. Acta Physiol. Scand. 125, 495–499 (1985).
Villegas, J. C. & Broadwell, R. D. Transcytosis of protein through the mammalian cerebral epithelium and endothelium: II. Adsorptive transcytosis of WGA-HRP and the blood–brain and brain–blood barriers. J. Neurocytol. 22, 67–80 (1993).
Banks, W. A., Kastin, A. J. & Akerstrom, V. HIV-1 protein gp120 crosses the blood–brain barrier: role of adsorptive endocytosis. Life Sci. 61, L119–L125 (1997).
Vorbrodt, A. W., Dobrogowska, D. H., Ueno, M. & Lossinsky, A. S. Immunocytochemical studies of protamine-induced blood–brain barrier opening to endogenous albumin. Acta Neuropathol. 89, 491–499 (1995).
Herve, F., Ghinea, N. & Scherrmann, J. M. CNS delivery via adsorptive transcytosis. AAPS J. 10, 455–472 (2008). A clear, lucid review of potential of adsorptive transcytosis for CNS drug delivery.
Chekhonin, V. P., Kabanov, A. V., Zhirkov, Y. A. & Morozov, G. V. Fatty acid acylated Fab-fragments of antibodies to neurospecific proteins as carriers for neuroleptic targeted delivery in brain. FEBS Lett. 287, 149–152 (1991).
Peter, J. C. et al. A pharmacologically active monoclonal antibody against the human melanocortin-4 receptor: effectiveness after peripheral and central administration. J. Pharmacol. Exp. Ther. 333, 478–490 (2010).
Kroll, R. A. & Neuwelt, E. A. Outwitting the blood–brain barrier for therapeutic purposes: osmotic opening and other means. Neurosurgery 42, 1083–1099 (1998).
Saaber, D., Wollenhaupt, S., Baumann, K. & Reichl, S. Recent progress in tight junction modulation for improving bioavailability. Expert Opin. Drug Deliv. 9, 347–381 (2014).
Cheng, Z. et al. Central nervous system penetration for small molecule therapeutic agents does not increase in multiple sclerosis- and Alzheimer's disease-related animal models despite reported blood–brain barrier disruption. Drug Metab. Dispos. 38, 135–161 (2010).
Somjen, G. G., Segal, M. B. & Herreras, O. Osmotic-hypertensive opening of the blood–brain barrier in rats does not necessarily provide access for potassium to cerebral interstitial fluid. Exp. Physiol. 76, 507–514 (1991).
Sengillo, J. D. et al. Deficiency in mural vascular cells coincides with blood–brain barrier disruption in Alzheimer's disease. Brain Pathol. 23, 303–310 (2012).
Price, T. O., Eranki, V., Banks, W. A., Ercal, N. & Shah, G. N. Topiramate treatment protects blood–brain barrier pericytes from hyperglycemia-induced oxidative damage in diabetic mice. Endocrinology 153, 362–372 (2012).
Hammes, H. P. et al. Pericytes and the pathogenesis of diabetic retinopathy. Diabetes 51, 3107–3112 (2002).
Avison, M. J. et al. Inflammatory changes and breakdown of microvascular integrity in early human immunodeficiency virus dementia. J. Neurovirol. 10, 223–232 (2004).
Boven, L. A., Middel, J., Verhoef, J., De Groot, C. J. & Nottet, H. S. Monocytes infiltration is highly associated with loss of tight junction protein zonula occludens in HIV-1-associated dementia. Neuropathol. Appl. Neurobiol. 26, 356–362 (2000).
Lossinsky, A. S., Vorbrodt, A. W. & Wisniewski, H. M. Ultracytochemical studies of vesicular and canalicular transport structures in the injured mammalian blood–brain barrier. Acta Neuropathol. 61, 239–245 (1983).
Wahl, M., Unterberg, A., Baethmann, A. & Schilling, L. Mediators of blood–brain barrier dysfunction and formation of vasogenic brain edema. J. Cereb. Blood Flow Metab. 8, 621–634 (1988).
Beauchesne, E., Desjardins, P., Hazell, A. S. & Butterworth, R. F. eNOS gene deletion restores blood–brain barrier integrity and attenuates neurodegeneration in the thiamine-deficient mouse brain. J. Neurochem. 111, 425–459 (2009).
Halliday, M. R. et al. Relationship between cyclophilin A levels and matrix metalloproteinase 9 activity in cerebrospinal fluid of cognitively normal apolipoprotein E4 carriers and blood–brain barrier breakdown. JAMA 70, 1198–1200 (2013).
Shah, G. N. et al. Pharmacological inhibition of mitochondrial carbonic anhydrases protects mouse cerebral pericytes from high glucose-induced oxidative stress and apoptosis. J. Pharmacol. Exp. Ther. 344, 637–645 (2013).
Candelario-Jalil, E. et al. Cyclooxygenase inhibition limits blood–brain barrier disruption following intracerebral injection of tumor necrosis factor-α in the rat. J. Pharmacol. Exp. Ther. 323, 488–498 (2007).
Frank, T. et al. Pegylated granulocyte colony-stimultating factor conveys long-term neuroprotection and improves functional outcome in a model of Parkinson's disease. Brain 135, 1914–1925 (2012).
Polt, R., Dhanasekaran, M. & Keyari, C. M. Glycosylated neuropeptides: a new vista for neuropsychopharmacology. Med. Res. Rev. 25, 557–585 (2005).
Batrakova, E. V., Gendelman, H. E. & Kabanov, A. V. Cell-mediated drug delivery. Expert Opin. Drug Deliv. 8, 415–433 (2011).
Meng, Y. et al. Effectve intravenous therapy for neurodegenerative disease with a therapeutic enzyme and a peptide that mediates delivery to the brain. Mol. Ther. 22, 547–543 (2014).
Yi, X. & Kabanov, A. V. Brain delivery of proteins via their fatty acid and block copolymer modifications. J. Drug Target. 21, 940–955 (2013).
De Vivo, D. C. et al. Defective glucose transport across the blood–brain barrier as a cause of persistent hypoglycorrhachia, seizures, and developmental delay. N. Engl. J. Med. 325, 703–709 (1991).
Erickson, M. A. & Banks, W. A. Blood–brain barrier dysfunction as a cause and consequence of Alzheimer's disease. J. Cereb. Blood Flow Metab. 33, 1500–1513 (2013).
Boulton, M. et al. Contribution of extracranial lymphatics and arachnoid villi to the clearance of a CSF tracer in the rat. Am. J. Physiol. 276, R818–R823 (1999).
Alafuzoff, I., Adolfsson, R., Grundke-Iqbal, I. & Winblad, B. Blood–brain barrier in Alzheimer dementia and in non-demented elderly. Acta Neuropathol. 73, 160–166 (1987).
Iliff, J. J. et al. A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid β. Sci. Transl Med. 4, 147ra111 (2012). Key study demonstrating that the glymphatic pathway is important to CSF and brain interstitial fluid circulations, and clearance of toxins from the CNS.
Erickson, M. A. et al. Lipopolysaccharide impairs amyloid β efflux from brain: altered vascular sequestration, cerebrospinal fluid reabsorption, peripheral clearance and transporter function at the blood–brain barrier. J. Neuroinflamm. 9, 150 (2012).
Grammas, P. Neurovascular dysfunction, inflammation and endothelial activation: implications for the pathogenesis of Alzheimer's disease. J. Neuroinflammation 8, 26 (2011).
Zlokovic, B. V. Neurovascular mechanisms of Alzheimer's neurodegeneration. Trends Neurosci. 28, 202–208 (2005). Introduction of the neurovascular hypothesis, which states that impaired BBB clearance of Aβ peptide from the CNS is a fundamental contributor to Alzheimer disease.
Zlokovic, B. V., Deane, R., Sagare, A. P., Bell, R. D. & Winkler, E. A. Low density lipoprotein receptor related protein-1: a serial clearance homeostatic mechanism controlling Alzheimer's amyloid β-peptide elimination fromt the brain. J. Neurochem. 115, 1077–1089 (2010).
Hartz, A. M. S., Miller, D. S. & Bauer, B. Restoring blood–brain barrier P-glycoprotein reduces brain amyloid-β in a mouse model of Alzheimer's disease. Mol. Pharmacol. 77, 715–723 (2010).
Donahue, J. E. et al. RAGE, LRP-1, and amyloid-beta protein in Alzheimer's disease. Acta Neuropathol. 112, 405–415 (2006).
Wijesuriya, J. C., Bullock, J. Y., Faull, R. L. M., Hladky, S. B. & Barrand, M. A. ABC efflux transporters in brain vasculature of Alzheimer's subjects. Brain Res. 1358, 228–238 (2010).
Owen, J. B. et al. Oxidative modification to LDL receptor-related protein 1 in hippocampus from subjects with Alzheimer's disease: implications for Aβ accumulation in AD brain. Free Radic. Biol. Med. 49, 1798–1803 (2010).
van Assema, D. M. et al. Blood–brain barrier P-glycoprotein function in Alzheimer's disease. Brain 135, 181–189 (2012).
Jaeger, L. B. et al. Testing the neurovascular hypothesis of Alzheimer's disease: LRP-1 antisense reduces blood–brain barrier clearance, increases brain levels of amyloid-β protein, and impairs cognition. J. Alzheimers Dis. 17, 553–570 (2009). Provides experimental support for the neurovascular hypothesis first proposed in reference 125.
Butterfield, D. A. & Boyd-Kimball, D. The critical role of methionine 35 in Alzheimer's amyloid β peptide (1–42)-induced oxidative stress and neurotoxicity. Biochim. Biophys. Acta 1703, 149–156 (2005).
Banks, W. A. et al. Impairments in brain-to-blood transport of amyloid-β and reabsorption of cerebrospinal fluid in an animal model of Alzheimer's disease are reversed by antisense directed against amyloid-β protein precursor. J. Alzheimers Dis. 23, 599–605 (2011).
Sagare, A. P. et al. Pericyte loss influences Alzheimer-like neurodegeneration in mice. Nature Commun. 4, 2932 (2013).
Craft, S. et al. Cerebrosinal fluid and plasma insulin levels in Alzheimer's disease: relationship to severity of dementia and apolipoprotein E genotype. Neurology 50, 164–168 (1998).
Talbot, K. et al. Demonstrated brain insulin resistance in Alzheimer's disease patients is associated with IGF-1 resistance, IRS-1 dyregulation, and cognitive decline. J. Clin. Invest. 122, 1316–1338 (2012).
Reger, M. A. et al. Intranasal insulin administration dose-dependently modulates verbal memory and plasma amyloid-β in memory-impaired older adults. J. Alzheimers Dis. 13, 323–331 (2008).
Urayama, A. & Banks, W. A. Starvation and triglycerides reverse the obesity-induced impairment of insulin transport at the blood–brain barrier. Endocrinology 149, 3592–3597 (2008).
Berthoud, H. R. Interactions between 'cognitive' and 'metabolic' brain in the control of food intake. Physiol. Behav. 91, 486–498 (2007).
Butter, C., Baker, D., O'Neill, J. K. & Turk, J. L. Mononuclear cell trafficking and plasma protein extravasation into the CNS during chronic relapsing experimental allergic encephalomyelitis in Biozzi AB/H mice. J. Neurol. Sci. 104, 9–12 (1991).
Hsuchou, H., Pan, W., Wu, X. & Kastin, A. J. Cessation of blood-to-brain influx of interleukin-15 during development of EAE. J. Cereb. Blood Flow Metab. 29, 1568–1578 (2009).
Juhler, M. et al. Blood–brain and blood–spinal cord barrier permeability during the course of experimental allergic encephalomyelitis in the rat. Brain Res. 302, 347–355 (1984).
Mishra, P. K. et al. Loss of astrocytic leptin signaling worsens experimental autoimmune encephalomyelitis. Brain Behav. Immun. 34, 98–107 (2013).
Hudson, L. C., Bragg, D. C., Tompkins, M. B. & Meeker, R. B. Astrocytes and microglia differentially regulate trafficking of lymphocyte subsets across brain endothelial cells. Brain Res. 1058, 148–160 (2005).
Stuve, O. The effects of natalizumab on the innate and adaptive immune system in the central nervous system. J. Neurol. Sci. 274, 39–41 (2008).
Correale, J. & Villa, A. The blood–brain barrier in multiple sclerosis: functional roles and therapeutic targeting. Autoimmunity 40, 148–160 (2007).
Sandoval, D. A., Obici, S. & Seeley, R. J. Targeting the CNS to treat type 2 diabetes. Nat. Rev. Drug Discov. 8, 386–398 (2009). Shows the fundamental role of the BBB in controlling blood glucose levels via its transport of insulin into the CNS.
Scherer, T. et al. Brain insulin controls adipose tissue lipolysis and lipogenesis. Cell Metab. 13, 183–194 (2011).
Banks, W. A., DiPalma, C. R. & Farrell, C. L. Impaired transport of leptin across the blood–brain barrier in obesity. Peptides 20, 1341–1345 (1999).
Romeo, G., Liu, W. H., Asnaghi, V., Kern, T. S. & Lorenzi, M. Activation of nuclear factor-κB induced by diabetes and high glucose regulates a proapoptotic program in retinal pericytes. Diabetes 51, 2241–2248 (2002).
Huber, J. D., VanGilder, R. L. & Houser, K. A. Streptozotocin-induced diabetes progressively increases blood–brain barrier permeability in specific brain regions in rats. Am. J. Physiol. 291, H2660–H2668 (2006).
Starr, J. M. et al. Increased blood–brain barrier permeability in type II diabetes demonstrated by gadolinium magnetic resonance imaging. J. Neurol. Neurosurg. Psychiatry 74, 70–76 (2003). References 152 and 153 establish that BBB disruption occurs in diabetes.
Shah, G. N., Morofuji, Y., Banks, W. A. & Price, T. O. High glucose-induced mitochondrial resistance and reactive oxygen species in mouse cerebral pericytes is reversed by pharmacological inhibition of mitochondrial carbonic anhydrase: implications for cerbral microvascular disease in diabetes. Biochem. Biophys. Res. Commun. 440, 354–358 (2013). Demonstrates that BBB disruption occurs because of oxidative stress arising from excess mitochondrial respiration.
Kowluru, R. A. Diabetic retinopathy: mitochondrial dysfunction and retinal capillary cell death. Antioxid. Redox Signal. 7, 1581–1587 (2005).
Weiwei, Z. & Hu, R. Targeting carbonic anhydrase to treat diabetic retinopathy: emerging evidences and encouraging results. Biochem. Biophys. Res. Commun. 390, 368–371 (2009).
Banks, W. A. et al. Triglycerides induce leptin resistance at the blood–brain barrier. Diabetes 53, 1253–1260 (2004).
Kastin, A. J. & Akerstrom, V. Glucose and insulin increase the transport of leptin through the blood–brain barrier in normal mice but not in streptozotocin-diabetic mice. Neuroendocrinology 73, 237–242 (2001).
Ito, S. et al. 1α,25-dihydroxyvitam D3 enhances cerebral clearance of human amyloid-β peptide(1-40) from mouse brain across the blood–brain barrier. Fluids Barriers CNS 8, 20 (2011).
Moon, J. H. et al. The effect of rosiglitazone on LRP1 expression and amyloid β uptake in human brain microvascular endothelial cells: a possible role of a low-dose thiazolidinedione for dementia treatment. Int. J. Neuropsychopharmacol. 1, 1–8 (2011).
O'Donnell, M. E., Lam, T. I., Tran, L. Q., Foroutan, S. & Anderson, S. E. Estradiol reduces activity of the blood–brain barrier Na-K-Cl cotransporter and decreases edema formation in permenent middle cerebral artery occlusion. J. Cereb. Blood Flow Metab. 26, 1234–1249 (2006).
Lyden, P. et al. Phase 1 safety, tolerability and pharmacokinetics of 3K3A-APC in healthy adult vounteers. Curr. Pharm. Design 19, 7479–7485 (2013).
McGuire, T. R. et al. Release of prostaglandin E-2 in bovine brain endothelial cells after exposure to three unique forms of the antifungal drug amphotericin-B: role of COX-2 in amphotericin-B induced fever. Life Sci. 72, 2581–2590 (2003).
Sury, M. D. et al. Evidence that N-acetylcysteine inhibits TNF-α-induced cerebrovascular endothelin-1 upregulation via inhibition of mitogen- and stress-activated protein kinase. Free Radic. Biol. Med. 41, 1372–1383 (2006).
Didier, N., Banks, W. A., Creminon, C., Dereuddre-Bosquet, N. & Mabondzo, A. HIV-1-induced production of endothelin-1 in an in vitro model of the human blood–brain barrier. Neuroreport 13, 1179–1183 (2002).
Rolinski, B. et al. Endothelin-1 elevated in the cerebrospinal fluid of HIV-infected patients with encephalopathy. Infection 27, 244–247 (1999).
Vangilder, R. L., Rosen, C. L., Barr, T. L. & Huber, J. D. Targeting the neurovascular unit for treatment of neurological disorders. Pharmacol. Ther. 130, 239–247 (2011).
Li, J. et al. Immune activation of human brain microvascular endothelial cells inhibits HIV replication in macrophages. Blood 121, 2934–2942 (2013).
Kubie, L. S. & Shults, G. M. Studies on the relationship of the chemical constituents of blood and cerebrospinal fluid. J. Exp. Med. 42, 565–591 (1925).
Pincus, J. B. & Kramer, B. Comparative study of the concentration of various anions and cations in cerebrospinal fluid and serum. J. Biol. Chem. 57, 463–470 (1923).
Davson, H. & Smith, H. V. Physiological aspects of the penetration of drugs into the cerebrospinal fluid. Proc. R. Soc. Med. 50, 963–966 (1957).
Roth, L. J. & Barlow, C. F. Drugs in the brain. Science 134, 22–31 (1961).
Reese, T. S. & Karnovsky, M. J. Fine structural localization of a blood–brain barrier to endogenous peroxidase. J. Cell Biol. 34, 207–217 (1967). A classic paper demonstrating the ultrastructural basis for the BBB: the presence of tight junctions and decreased transcytotic vesicles.
Coisne, C., Mao, W. & Engelhardt, B. Cutting edge: natalizumab blocks adhesion but not initial contact of human T cells to the blood–brain barrier in vivo in an animal model of multiple sclerosis. J. Immunol. 182, 5909–5913 (2009).
Acknowledgements
The author is supported by the US Department of Veterans Affairs and a grant from the US National Institute on Aging (grant R01 AG046619).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Glossary
- Blood–brain barrier
-
(BBB). The modified capillary bed of the brain; can be conceptualized as those processes that, taken together, control the exchange of substances between the blood and the fluids (cerebrospinal fluid and brain interstitial fluid) of the central nervous system (CNS).
- Neurovascular unit
-
(NVU). For the purposes of this Review, this refers to the concept that the cells forming the BBB are in communication with other cells of the central nervous system (CNS) and, by extension, with the circulating immune cells, and with the peripheral tissues via blood-borne secretions.
- Transporters
-
Proteins that provide a mechanism by which substances can be carried from one side of the blood–brain barrier (BBB) to the other, thus greatly increasing (for blood-to-brain transporters) or greatly decreasing (for brain-to-blood transporters) the central nervous system (CNS) uptake of a substance in comparison with that predicted based on its physicochemical characteristics.
- Polarization
-
A characteristic of the blood–brain barrier (BBB), arising from different characteristics of its abluminal and luminal surfaces, including differing levels of enzymes, glycoproteins, lipid composition and transporters.
- Active transport
-
Transport by an energy-requiring transporter that can move its ligand against a concentration gradient.
- Facilitated diffusion
-
Transport by a non-energy-requiring transporter that moves it ligand down a concentration gradient.
- Adsorptive transcytosis
-
A mechanism by which glycoproteins or highly chargedmolecules bind to brain endothelial cell (BEC) glycoproteins, inducing vesicles that are routed to the opposite membrane.
- Passive diffusion
-
The mechanism by which a substance crosses the blood–brain barrier (BBB) by non-saturable means, with the degree of passage depending on the physicochemical characteristics of the substance.
- Extracellular pathways
-
Areas such as the pial surface and subarachnoid space that are deficient in a blood–brain barrier (BBB) and thus allow small amounts of blood-borne substances, including albumin and immunoglobulins, to access the brain primarily through the Virchow–Robin spaces.
- Neurovascular hypothesis
-
The hypothesis that the impaired ability of the blood–brain barrier (BBB) to remove amyloid-β peptide from the central nervous system (CNS) contributes to amyloid-β accumulation and the progression of Alzheimer disease.
Rights and permissions
About this article
Cite this article
Banks, W. From blood–brain barrier to blood–brain interface: new opportunities for CNS drug delivery. Nat Rev Drug Discov 15, 275–292 (2016). https://doi.org/10.1038/nrd.2015.21
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd.2015.21
This article is cited by
-
Cocaine regulates antiretroviral therapy CNS access through pregnane-x receptor-mediated drug transporter and metabolizing enzyme modulation at the blood brain barrier
Fluids and Barriers of the CNS (2024)
-
Bi-directional regulation of AIMP2 and its splice variant on PARP-1-dependent neuronal cell death; Therapeutic implication for Parkinson's disease
Acta Neuropathologica Communications (2024)
-
Blood–brain borders: a proposal to address limitations of historical blood–brain barrier terminology
Fluids and Barriers of the CNS (2024)
-
Long and Short-Term Effect of mTOR Regulation on Cerebral Organoid Growth and Differentiations
Tissue Engineering and Regenerative Medicine (2024)
-
Open source board based acoustofluidic transwells for reversible disruption of the blood–brain barrier for therapeutic delivery
Biomaterials Research (2023)