Key Points
-
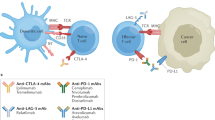
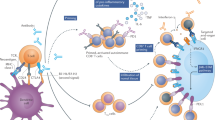
CTLA-4 regulates T-cell activation upon initiation of an immune response, in the lymphoid organs, where naive T cells are primed, and potentially in the periphery via regulatory T-cell (TREG) depletion
-
This diverse role of CTLA-4 in initiating and mounting immune responses might explain the plethora of immune-related adverse events (irAEs) experienced by patients receiving treatment with anti-CTLA-4 antibodies
-
PD-1 suppresses T-cell activity, mostly within the peripheral tissues and in the tumour microenvironment, which might explain the distinct spectrum and reduced incidence of adverse effects of anti-PD-1 antibodies
-
Thyroid disorders are more frequent adverse effects of treatment with anti-PD-1 antibodies (pembrolizumab and nivolumab) whereas colitis and hypophysitis are more frequent with anti-CTLA-4 antibodies (ipilimumab)
-
General guidelines on the management of irAEs recommend treatment of symptoms; corticosteroids are generally indicated together with dose skipping or discontinuation in patients with persistent grade ≥2 adverse events
Abstract
Inhibition of immune checkpoints using anti-programmed cell death-1 (PD-1) or anti cytotoxic-T-lymphocyte-associated antigen 4 (CTLA-4) monoclonal antibodies has revolutionized the management of patients with advanced-stage melanoma and is among the most promising treatment approaches for many other cancers. Use of CTLA-4 and PD-1 inhibitors, either as single agents, or in combination, has been approved by the US FDA for the treatment of metastatic melanoma. Treatment with these novel immunotherapies results in a unique and distinct spectrum of adverse events, which are mostly related to activation of the immune system and are, therefore, an unwanted consequence of their mechanisms of action. Adverse effects of CTLA-4 and/or PD-1 inhibition are most commonly observed in the skin, gastrointestinal tract, liver and endocrine systems and include pruritus, rash, nausea, diarrhoea and thyroid disorders. In this Review, the authors describe the adverse event profile of checkpoint inhibitors targeting CTLA-4 and PD-1, used both as monotherapies and in combination and aim to provide some general guidelines, based upon the mechanisms of action of these therapies and on the management of these immune-related adverse events.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Greenwald, R. J. et al. The B7 family revisited. Annu. Rev. Immunol. 23, 515–548 (2005).
Zou, W. et al. Inhibitory B7-family molecules in the tumour microenvironment. Nat. Rev. Immunol. 8, 467–477 (2008).
Sjöblom, T. et al. The consensus coding sequences of human breast and colorectal cancers. Science 314, 268–274 (2006).
Segal, N. H. et al. Epitope landscape in breast and colorectal cancer. Cancer Res. 68, 889–892 (2008).
Dunn, G. P. et al. Cancer immunoediting: from immunosurveillance to tumor escape. Nat. Immunol. 3, 991–998 (2002).
Drake, C. G. et al. Mechanisms of immune evasion by tumors. Adv. Immunol. 90, 51–81 (2006).
Hanahan, D. et al. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Topalian, S. L. et al. Cancer immunotherapy comes of age. J. Clin. Oncol. 29, 4828–4836 (2011).
Mellman, I. et al. Cancer immunotherapy comes of age. Nature 480, 480–489 (2011).
Brunner, M. C. et al. CTLA-4-mediated inhibition of early events of T cell proliferation. J. Immunol. 162, 5813–5820 (1999).
Leach, D. R. et al. Enhancement of antitumor immunity by CTLA-4 blockade. Science 271, 1734–1736 (1996).
O'Day, S. J. et al. Targeting cytotoxic T-lymphocyte antigen-4 (CTLA-4): a novel strategy for the treatment of melanoma and other malignancies. Cancer 110, 2614–2627 (2007).
Camacho, L. H. et al. Phase I/II trial of tremelimumab in patients with metastatic melanoma. J. Clin. Oncol. 27, 1075–1081 (2009).
Ribas, A. et al. Phase III randomized clinical trial comparing tremelimumab with standard-of-care chemotherapy in patients with advanced melanoma. J. Clin. Oncol. 31, 616–622 (2013).
Wolchock, J. D. et al. Ipilimumab monotherapy in patients with pretreated advanced melanoma: a randomised, double-blind, multicentre, phase 2, dose-ranging study. Lancet Oncol. 11, 155–164 (2010).
Hodi, F. S. et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 363, 711–723 (2010).
The Department of Health and Human Services. FDA approves new treatment for a type of late-stage skin cancer. FDA [online] http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm1193237.htm (2011).
The European Medicines Agency. Assessment report for Yervoy (ipilimumab). [online] http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/002213/WC500109302.pdf (2011).
Lynch, T. J. et al. Ipilimumab in combination with paclitaxel and carboplatin as first-line treatment in stage IIIB/IV non-small-cell lung cancer: results from a randomized, double-blind, multicenter phase II study. J. Clin. Oncol. 30, 2046–2054 (2012).
Slovin, S. F. et al. Ipilimumab alone or in combination with radiotherapy in metastatic castration-resistant prostate cancer: results from an open-label, multicenter phase I/II study. Ann. Oncol. 24, 1813–1821 (2013).
Kwon, E. D. et al. Ipilimumab versus placebo after radiotherapy in patients with metastatic castration-resistant prostate cancer that had progressed after docetaxel chemotherapy (CA184-043): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol. 15, 700–712 (2014).
Calabrò, L. et al. Tremelimumab for patients with chemotherapy-resistant advanced malignant mesothelioma: an open-label, single-arm, phase 2 trial. Lancet Oncol. 14, 1104–1111 (2013).
Topalian, S. L. et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N. Engl. J. Med. 366, 2443–2454 (2012).
Patnaik, A. et al. Phase I study of MK-3475 (anti-PD-1 monoclonal antibody) in patients with advanced solid tumors [abstract]. J. Clin. Oncol. 33 (Suppl.), a2512 (2012).
Westin, J. R. et al. Safety and activity of PD1 blockade by pidilizumab in combination with rituximab in patients with relapsed follicular lymphoma: a single group, open-label, phase 2 trial. Lancet Oncol. 15, 69–77 (2014).
Hamanishi, J. et al. Safety and antitumor activity of anti-PD-1 antibody, nivolumab, in patients with platinum-resistant ovarian cancer. J. Clin. Oncol. 33, 4015–4022 (2015).
Brahmer, J. R. et al. Phase I study of single-agent anti-programmed death-1 (MDX-1106) in refractory solid tumors: safety, clinical activity, pharmacodynamics, and immunologic correlates. J. Clin. Oncol. 28, 3167–3175 (2010).
Topalian, S. L. et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J. Clin. Oncol. 32, 1020–1030 (2014).
Robert, C. et al. Nivolumab in previously untreated melanoma without BRAF mutation. N. Engl. J. Med. 372, 320–330 (2015).
Hamid, O. et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N. Engl. J. Med. 369, 134–144 (2013).
Robert, C. et al. Anti-programmed-death-receptor-1 treatment with pembrolizumab in ipilimumab-refractory advanced melanoma: a randomised dose-comparison cohort of a phase 1 trial. Lancet 384, 1109–1117 (2014).
Ribas, A. et al. Pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractory melanoma (KEYNOTE-002): a randomised, controlled, phase 2 trial. Lancet Oncol. 16, 908–918 (2015).
Robert, C. et al. Pembrolizumab versus ipilimumab in advanced melanoma. N. Engl. J. Med. 372, 2521–2532 (2015).
Daud, A. et al. Long-term efficacy of pembrolizumab (pembro; MK-3475) in a pooled analysis of 655 patients (pts) with advanced melanoma (MEL) enrolled in KEYNOTE-001 [abstract]. J. Clin. Oncol. 33 (Suppl.), a9005 (2015).
The Department of Health and Human Services. FDA approves Keytruda for advanced melanoma. FDA [online] http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm412802.htm (2014).
American Association for Cancer Research. PD-1 inhibitor approved for melanoma. Cancer Discov. 4, 1249 (2014).
Poole, R. M. Pembrolizumab: first global approval. Drugs 74, 1973–1981 (2014).
The Department of Health and Human Services. Pembrolizumab label updated with new clinical trial information FDA [online] http://www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm478493.htm (2015).
The European Medicines Agency. Assessment report: Keytruda. International non-proprietary name: pembrolizumab. [online] http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/003820/WC500190992.pdf (2015).
The Department of Health and Human Services. FDA approves Opdivo for advanced melanoma. FDA [online] http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm427716.htm (2014).
The European Medicines Agency. Keytruda. [online] http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/003820/human_med_001886., (2015).
Wolchok, J. D. et al. Nivolumab plus ipilimumab in advanced melanoma. N. Engl. J. Med. 369, 122–133 (2013).
Sznol, M. et al. Survival, response duration, and activity by BRAF mutation (MT) status of nivolumab (NIVO, anti-PD-1, BMS-936558, ONO-4538) and ipilimumab (IPI) concurrent therapy in advanced melanoma (MEL) [abstract]. J. Clin. Oncol. 32 (Suppl. 5), a9003 (2014).
Postow, M. A. et al. Nivolumab and ipilimumab versus ipilimumab in untreated melanoma. N. Engl. J. Med. 372, 2006–2017 (2015).
Larkin, J. et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N. Engl. J. Med. 373, 23–34 (2015).
Pardoll, D. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 12, 252–264 (2012).
Fecher, L. A. et al. Ipilimumab and its toxicities: a multidisciplinary approach. Oncologist 18, 733–743 (2013).
Schwartz, R. H. Costimulation of T lymphocytes: the role of CD28, CTLA-4, and B7/BB1 in interleukin-2 production and immunotherapy. Cell 71, 1065–1068 (1992).
Rudd, C. E. et al. CD28 and CTLA-4 coreceptor expression and signal transduction. Immunol. Rev. 229, 12–26 (2009).
Hathcock, K. S. et al. Identification of an alternative CTLA-4 ligand costimulatory for T cell activation. Science 262, 905–907 (1993).
Cutler, C. W. et al. Dendritic cells: immune saviors or Achilles' heel? Infect. Immun. 69, 4703–4708 (2001).
Sharpe, A. H. et al. T-cell costimulation — biology, therapeutic potential, and challenges. N. Engl. J. Med. 355, 973–975 (2006).
Konig, R. Interactions between MHC molecules and co-receptors of the TCR. Curr. Opin. Immunol. 14, 75–83 (2002).
Freeman, G. J. et al. Cloning of B7-2: a CTLA-4 counter-receptor that costimulates human T cell proliferation. Science 262, 909–911 (1993).
Azuma, M. et al. B70 antigen is a second ligand for CTLA-4 and CD28. Nature 366, 76–79 (1993).
Peggs, K. S. et al. Blockade of CTLA-4 on both effector and regulatory T cell compartments contributes to the antitumor activity of anti-CTLA-4 antibodies. J. Exp. Med. 206, 1717–1725 (2009).
Bulliard, Y. et al. Activating Fc γ receptors contribute to the antitumor activities of immunoregulatory receptor-targeting antibodies. J. Exp. Med. 210, 1685–1693 (2013).
Wang, C. J. et al. Cutting edge: cell-extrinsic immune regulation by CTLA-4 expressed on conventional T cells. J. Immunol. 189, 1118–1122 (2012).
Romano, E. et al. Ipilimumab-dependent cell-mediated cytotoxicity of regulatory T cells ex vivo by nonclassical monocytes in melanoma patients. Proc. Natl Acad. Sci. USA 112, 6140–6145 (2015).
Selby, M. J. et al. Anti-CTLA-4 antibodies of IgG2a isotype enhance antitumor activity through reduction of intratumoral regulatory T cells. Cancer Immunol. Res. 1, 32–42 (2013).
Postow, M. A. et al. Immune checkpoint blockade in cancer therapy. J. Clin. Oncol. 33, 1974–1982 (2015).
Smigiel, K. S. et al. Regulatory T-cell homeostasis: steady-state maintenance and modulation during inflammation. Immunol. Rev. 259, 40–59 (2014).
Khattri, R. et al. Lymphoproliferative disorder in CTLA-4 knockout mice is characterized by CD28-regulated activation of Th2 responses. J. Immunol. 162, 5784–5791 (1999).
Waterhouse, P. et al. Lymphoproliferative disorders with early lethality in mice deficient in Ctla-4. Science 270, 985–988 (1995).
Wang, H. B. et al. Anti-CTLA-4 antibody treatment triggers determinant spreading and enhances murine myasthenia gravis. J. Immunol. 166, 6430–6436 (2001).
Ueda, H. et al. Association of the T-cell regulatory gene CTLA4 with susceptibility to autoimmune disease. Nature 423, 506–511 (2003).
Sanderson, K. et al. Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III and IV melanoma. J. Clin. Oncol. 23, 741–750 (2005).
Mora, J. R. et al. T-cell homing specificity and plasticity: new concepts and future challenges. Trends Immunol. 27, 235–243 (2006).
Ott, P. A. et al. CTLA-4 and PD-1/PD-L1 blockade: new immunotherapeutic modalities with durable clinical benefit in melanoma patients. Clin. Cancer Res. 19, 5300–5309 (2013).
Keir, M. E. et al. PD-1 and its ligands in tolerance and immunity. Annu. Rev. Immunol. 26, 677–704 (2008).
Francisco, L. M. et al. The PD-1 pathway in tolerance and autoimmunity. Immunol. Rev. 236, 219–242 (2010).
Okazaki, T. et al. A rheostat for immune responses: the unique properties of PD-1 and their advantages for clinical application. Nat. Immunol. 14, 1212–1218 (2013).
Fife, B. T. et al. Interactions between PD-1 and PD-L1 promote tolerance by blocking the TCR-induced stop signal. Nat. Immunol. 10, 1185–1192 (2009).
Freeman, G. J. et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J. Exp. Med. 192, 1027–1034 (2000).
Latchman, Y. et al. PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat. Immunol. 2, 261–268 (2001).
Topalian, S. L. et al. Targeting the PD-1/B7-H1(PD-L1) pathway to activate anti-tumor immunity. Curr. Opin. Immunol. 24, 207–212 (2012).
Blank, C. et al. PD-L1/B7H-1 inhibits the effector phase of tumor rejection by T cell receptor (TCR) transgenic CD8+ T cells. Cancer Res. 64, 1140–1145 (2004).
Nishimura, H. et al. Development of lupus-like autoimmune diseases by disruption of the PD-1 gene encoding an ITIM motif-carrying immunoreceptor. Immunity 11, 141–151 (1999).
Nishimura, H. et al. Autoimmune dilated cardiomyopathy in PD-1 receptor-deficient mice. Science 291, 319–322 (2001).
Liang, S. C. et al. Regulation of PD-1, PD-L1, and PD-L2 expression during normal and autoimmune responses. Eur. J. Immunol. 33, 2706–2716 (2003).
Weber, J. S. et al. Phase I/II study of ipilimumab for patients with metastatic melanoma. J. Clin. Oncol. 26, 5950–5956 (2008).
Margolin, K. et al. Ipilimumab in patients with melanoma and brain metastases: an open-label, phase 2 trial. Lancet Oncol. 13, 459–465 (2012).
Di Giacomo, A. M. et al. Ipilimumab and fotemustine in patients with advanced melanoma (NIBIT-M1): an open-label, single-arm phase 2 trial. Lancet Oncol. 13, 879–886 (2012).
Robert, C. et al. Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N. Engl. J. Med. 364, 2517–2526 (2011).
Weber, J. S. et al. Patterns of onset and resolution of immune-related adverse events of special interest with ipilimumab: detailed safety analysis from a phase 3 trial in patients with advanced melanoma. Cancer 119, 1675–1682 (2013).
Eggermont, A. M. et al. Adjuvant ipilimumab versus placebo after complete resection of high-risk stage III melanoma (EORTC 18071): a randomised, double-blind, phase 3 trial. Lancet Oncol. 16, 522–530 (2015).
Gupta, A. et al. Systematic review: colitis associated with anti-CTLA-4 therapy. Aliment. Pharmacol. Ther. 42, 406–417 (2015).
Weber, J. S. et al. Management of immune-related adverse events and kinetics of response with ipilimumab. J. Clin. Oncol. 30, 2691–2697 (2012).
Marthey, L. et al. Ipilimumab colitis: a GETAID multicentric study [poster P202]. J. Chrohns Colitits 8, S146 (2014).
Dignass, A. et al. Second European evidence-based consensus on the diagnosis and management of ulcerative colitis part 2: current management. J. Crohns Colitis 6, 991–1030 (2012).
Beck, K. E. et al. Enterocolitis in patients with cancer after antibody blockade of cytotoxic T-lymphocyte-associated antigen 4. J. Clin. Oncol. 24, 2283–2289 (2006).
Berman, D. et al. Blockade of cytotoxic T-lymphocyte antigen-4 by ipilimumab results in dysregulation of gastrointestinal immunity in patients with advanced melanoma. Cancer Immun. 10, 11 (2010).
Lacouture, M. E. et al. Ipilimumab in patients with cancer and the management of dermatologic adverse events. J. Am. Acad. Dermatol. 71, 161–169 (2014).
Voskens, C. J. et al. The price of tumor control: an analysis of rare side effects of anti-CTLA-4 therapy in metastatic melanoma from the ipilimumab network. PLoS ONE 8, e53745 (2013).
Delyon, J. et al. Hemophilia A induced by ipilimumab. N. Engl. J. Med. 365, 1747–1748 (2011).
Robinson, M. R. et al. Cytotoxic T lymphocyte-associated antigen 4 blockade in patients with metastatic melanoma: a new cause of uveitis. J. Immunother. 27, 478–479 (2004).
Ribas, A. et al. A randomized controlled comparison of pembrolizumab and chemotherapy in patients with ipilimumab-refractory melanoma [late breaker oral presentation]. Soc. Melanoma Res. Congress SMR (2014).
Orlov, S. et al. Induction of painless thyroiditis in patients receiving programmed death 1 receptor immunotherapy for metastatic malignancies. J. Clin. Endocrinol. Metab. 100, 1738–1741 (2015).
Garon, E. B. et al. Pembrolizumab for the treatment of non-small-cell lung cancer. N. Engl. J. Med. 372, 2018–2028 (2015).
Weber, J. S. et al. Nivolumab versus chemotherapy in patients with advanced melanoma who progressed after anti-CTLA-4 treatment (CheckMate 037): a randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 16, 375–384 (2015).
Weber, J. S. et al. Safety profile of nivolumab (NIVO) in patients (pts) with advanced melanoma (MEL): a pooled analysis [abstract]. J. Clin. Oncol. 33 (Suppl.), a9018 (2015).
Bristol-Myers Squibb Company. Risk evaluation and mitigation strategy (REMS). FDA [online] http://www.fda.gov/downloads/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/UCM249435.pdf, (2012).
Lichtiger, S. et al. Cyclosporine in severe ulcerative colitis refractory to steroid therapy. N. Engl. J. Med. 330, 1841–1845 (1994).
Pages, C. et al. Ipilimumab-induced acute severe colitis treated by infliximab. Melanoma Res. 23, 227–230 (2013).
Minor, D. R. et al. Infliximab in the treatment of anti-CTLA4 antibody (ipilimumab) induced immune-related colitis. Cancer Biother. Radiopharm. 24, 321–325 (2009).
Merrill, S. P. et al. Early administration of infliximab for severe ipilimumab-related diarrhea in a critically ill patient. Ann. Pharmacother. 48, 806–810 (2014).
Douglas, B. J. et al. Ipilimumab in metastatic melanoma patients with pre-existing autoimmune disorders [abstract]. J. Clin. Oncol. 33 (Suppl.), a9019 (2015).
Prashanth, P. et al. Efficacy and toxicity of treatment with the anti-CTLA-4 antibody ipilimumab in patients with metastatic melanoma who have progressed on anti-PD-1 therapy [abstract]. J. Clin. Oncol. 33 (Suppl.), a9059 (2015).
Lutzky, J. et al. Association between immune-related adverse events (irAEs) and disease control or overall survival in patients (pts) with advanced melanoma treated with 10 mg/kg ipilimumab in three phase II clinical trials [abstract]. J. Clin. Oncol. 33 (Suppl. 15), a9034 (2009).
Weber, J. S. et al. Phase II trial of extended dose anti-CTLA-4 antibody ipilimumab (formerly MDX-010) with a multipeptide vaccine for resected stages IIIC and IV melanoma [abstract]. J. Clin. Oncol. 33 (Suppl. 15), a9023 (2009).
Hua, C. et al. Association of vitiligo with tumor response in patients with metastatic melanoma treated with pembrolizumab. JAMA Dermatol. 152, 45–51 (2016).
Freeman-Keller, M. et al. Nivolumab in resected and unresectable metastatic melanoma: characteristics of immune-related adverse events and association with outcomes. Clin. Cancer Res. http://dx.doi.org/10.1158/1078-0432.CCR-15-1136 (2015).
Author information
Authors and Affiliations
Contributions
C.B., E.L. and C.R. researched data for this article, C.B., A.T. and C.R. made a substantial contribution to discussions of content. C.B., A.T. and C.R. wrote the manuscript, and all authors edited and/or reviewed the manuscript prior to submission. C.B. and A.T. contributed equally to this Review.
Corresponding author
Ethics declarations
Competing interests
A.T. has acted as consultant of BMS and Merck. He has also conducted contract research for Amgen, BMS, Merck and Novartis. E.R. has acted as a consultant of and BMS and Roche. O.L. has acted as a consultant of Genzyme and MSD. F.C. has acted as consultant of Abbvie, Enterome, Ferring, Genentech, Hospira, Jansen, Mayoly, MSD, Otsuka, Splindler, and Takeda and Vifor. A.M. has acted as an advisor and has received honoraria from Amgen, BMS, Pfizer, MSD, Novartis and Roche. A.E. is a member of the scientific advisory board of BMS, Incyte, Medimmune and Merck. J-C.S. has acted as a consultant of Astra-Zeneca, Merus, MSD, Pfizer, Roche, Servier and Symphogen. C.M. has acted as consultant of BMS and Merck. C.R. has acted as a consultant of Amgen, BMS, GSK, Merck, Novartis and Roche. C.B, F.L.L, H.I., S.C., A.B., E.L., M.T. and C.L. declare no competing interests.
Supplementary information
Supplementary information S1 (table)
Incidence per 1000 person-months of all grade and grade 3 to 5 adverse events under immunotherapy using the SAS Sytstem. The results include data from the following studies: CA-184-002, KEYNOTE-001, KEYNOTE-001 (randomized cohorts), KEYNOTE-002, KEYNOTE-006, CheckMate-037, CheckMate-066, CheckMate-067, and CheckMate-069. (PDF 2014 kb)
Rights and permissions
About this article
Cite this article
Boutros, C., Tarhini, A., Routier, E. et al. Safety profiles of anti-CTLA-4 and anti-PD-1 antibodies alone and in combination. Nat Rev Clin Oncol 13, 473–486 (2016). https://doi.org/10.1038/nrclinonc.2016.58
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2016.58
This article is cited by
-
Turning up the heat: CTLA4 blockade in urothelial cancer
Nature Reviews Urology (2024)
-
Evaluation of an established oncology triage hotline in a model of emergency department avoidance: assessing the UKONS triage tool and call outcomes
Supportive Care in Cancer (2024)
-
Targeting monoamine oxidase A: a strategy for inhibiting tumor growth with both immune checkpoint inhibitors and immune modulators
Cancer Immunology, Immunotherapy (2024)
-
The OX40-TRAF6 axis promotes CTLA-4 degradation to augment antitumor CD8+ T-cell immunity
Cellular & Molecular Immunology (2023)
-
The current status and prospect of immunotherapy in colorectal cancer
Clinical and Translational Oncology (2023)