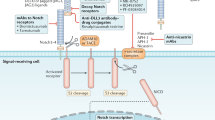
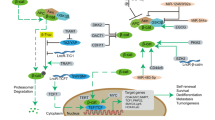
Key Points
-
Preclinical models provide evidence of cancer stem cells (CSCs) contributing to cancer proliferation, relapse and metastasis; this theory is being examined and validated in the clinical setting, currently in advanced malignancies
-
Over the past few years, new investigational agents have been developed to block the Notch, Hedgehog (HH) or Wnt signalling pathways for targeting CSCs
-
To date, robust antitumour activity has not been observed by targeting CSCs using Notch, HH or Wnt inhibitors, either as single agents or in combination with standard chemotherapy, in clinical trials
-
Combination approaches to overcome the crosstalk among Notch, HH and Wnt pathways, as well as other signalling pathways, has been examined mostly in preclinical models, with promising results
-
The success of the combination therapy in clinical trials might depend on CSC–tumour microenvironment interactions, perhaps in the context of the genotypes and phenotypes of the bulk tumour, CSCs, and the tumour microenvironment
-
A number of clinical trials have incorporated surrogate CSC assays to measure the effects of an investigational agent on CSCs, but further technological improvements in assays are needed
Abstract
During the past decade, cancer stem cells (CSCs) have been increasingly identified in many malignancies. Although the origin and plasticity of these cells remain controversial, tumour heterogeneity and the presence of small populations of cells with stem-like characteristics is established in most malignancies. CSCs display many features of embryonic or tissue stem cells, and typically demonstrate persistent activation of one or more highly conserved signal transduction pathways involved in development and tissue homeostasis, including the Notch, Hedgehog (HH), and Wnt pathways. CSCs generally have slow growth rates and are resistant to chemotherapy and/or radiotherapy. Thus, new treatment strategies targeting these pathways to control stem-cell replication, survival and differentiation are under development. Herein, we provide an update on the latest advances in the clinical development of such approaches, and discuss strategies for overcoming CSC-associated primary or acquired resistance to cancer treatment. Given the crosstalk between the different embryonic developmental signalling pathways, as well as other pathways, designing clinical trials that target CSCs with rational combinations of agents to inhibit possible compensatory escape mechanisms could be of particular importance. We also share our views on the future directions for targeting CSCs to advance the clinical development of these classes of agents.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Vermeulen, L., Sprick, M. R., Kemper, K., Stassi, G. & Medema, J. P. Cancer stem cells—old concepts, new insights. Cell Death Differ. 15, 947–958 (2008).
Espinoza, I. & Miele, L. Deadly crosstalk: notch signaling at the intersection of EMT and cancer stem cells. Cancer Lett. 341, 41–45 (2013).
Krivtsov, A. V. et al. Transformation from committed progenitor to leukaemia stem cell initiated by MLL–AF9. Nature 442, 818–822 (2006).
Reya, T., Morrison, S. J., Clarke, M. F. & Weissman, I. L. Stem cells, cancer, and cancer stem cells. Nature 414, 105–111 (2001).
Pattabiraman, D. R. & Weinberg, R. A. Tackling the cancer stem cells—what challenges do they pose? Nat. Rev. Drug Discov. 13, 497–512 (2014).
Tirino, V. et al. Cancer stem cells in solid tumors: an overview and new approaches for their isolation and characterization. FASEB J. 27, 13–24 (2013).
Lehmann, C., Jobs, G., Thomas, M., Burtscher, H. & Kubbies, M. Established breast cancer stem cell markers do not correlate with in vivo tumorigenicity of tumor-initiating cells. Int. J. Oncol. 41, 1932–1942 (2012).
Karsten, U. & Goletz, S. What makes cancer stem cell markers different? Springerplus 2, 301 (2013).
Sachs, N. & Clevers, H. Organoid cultures for the analysis of cancer phenotypes. Curr. Opin. Genet. Dev. 24, 68–73 (2014).
Chen, J. et al. A restricted cell population propagates glioblastoma growth after chemotherapy. Nature 488, 522–526 (2012).
Driessens, G., Beck, B., Caauwe, A., Simons, B. D. & Blanpain, C. Defining the mode of tumour growth by clonal analysis. Nature 488, 527–530 (2012).
Schepers, A. G. et al. Lineage tracing reveals Lgr5+ stem cell activity in mouse intestinal adenomas. Science 337, 730–735 (2012).
Hoey, T. et al. DLL4 blockade inhibits tumor growth and reduces tumor-initiating cell frequency. Cell Stem Cell 5, 168–177 (2009).
Varnat, F., Siegl-Cachedenier, I., Malerba, M., Gervaz, P. & Ruiz i Altaba, A. Loss of WNT–TCF addiction and enhancement of HH–GLI1 signalling define the metastatic transition of human colon carcinomas. EMBO Mol. Med. 2, 440–457 (2010).
Vermeulen, L. et al. Wnt activity defines colon cancer stem cells and is regulated by the microenvironment. Nat. Cell Biol. 12, 468–476 (2010).
LaBarge, M. A. The difficulty of targeting cancer stem cell niches. Clin. Cancer Res. 16, 3121–3129 (2010).
Merchant, A. A. & Matsui, W. Targeting Hedgehog—a cancer stem cell pathway. Clin. Cancer Res. 16, 3130–3140 (2010).
Takebe, N., Harris, P. J., Warren, R. Q. & Ivy, S. P. Targeting cancer stem cells by inhibiting Wnt, Notch, and Hedgehog pathways. Nat. Rev. Clin. Oncol. 8, 97–106 (2011).
Espinoza, I. & Miele, L. Notch inhibitors for cancer treatment. Pharmacol. Ther. 139, 95–110 (2013).
Gomez-del Arco, P. et al. Alternative promoter usage at the Notch1 locus supports ligand-independent signaling in T cell development and leukemogenesis. Immunity 33, 685–698 (2010).
Murthy, A. et al. Notch activation by the metalloproteinase ADAM17 regulates myeloproliferation and atopic barrier immunity by suppressing epithelial cytokine synthesis. Immunity 36, 105–119 (2012).
Gu, J. W. et al. Notch signals in the endothelium and cancer “stem-like” cells: opportunities for cancer therapy. Vasc. Cell 4, 7 (2012).
Andersson, E. R. & Lendahl, U. Therapeutic modulation of Notch signalling—are we there yet? Nat. Rev. Drug Discov. 13, 357–378 (2014).
Perumalsamy, L. R., Marcel, N., Kulkarni, S., Radtke, F. & Sarin, A. Distinct spatial and molecular features of notch pathway assembly in regulatory T cells. Sci. Signal. 5, ra53 (2012).
Perumalsamy, L. R., Nagala, M., Banerjee, P. & Sarin, A. A hierarchical cascade activated by non-canonical Notch signaling and the mTOR–Rictor complex regulates neglect-induced death in mammalian cells. Cell Death Differ. 16, 879–889 (2009).
Perumalsamy, L. R., Nagala, M. & Sarin, A. Notch-activated signaling cascade interacts with mitochondrial remodeling proteins to regulate cell survival. Proc. Natl Acad. Sci. USA 107, 6882–6887 (2010).
Raafat, A. et al. Rbpj conditional knockout reveals distinct functions of Notch4/Int3 in mammary gland development and tumorigenesis. Oncogene 28, 219–230 (2009).
Sade, H., Krishna, S. & Sarin, A. The anti-apoptotic effect of Notch-1 requires p56lck-dependent, Akt/PKB-mediated signaling in T cells. J. Biol. Chem. 279, 2937–2944 (2004).
Lee, K. S. et al. Roles of PINK1, mTORC2, and mitochondria in preserving brain tumor-forming stem cells in a noncanonical Notch signaling pathway. Genes Dev. 27, 2642–2647 (2013).
Takebe, N., Warren, R. Q. & Ivy, S. P. Breast cancer growth and metastasis: interplay between cancer stem cells, embryonic signaling pathways and epithelial-to-mesenchymal transition. Breast Cancer Res. 13, 211 (2011).
Deangelo, D. J. et al. A phase I clinical trial of the notch inhibitor MK-0752 in patients with T-cell acute lymphoblastic leukemia/lymphoma (T-ALL) and other leukemias [abstract]. J. Clin. Oncol. 24 (Suppl.), a6585 (2006).
Pandya, K. et al. Targeting both Notch and ErbB-2 signalling pathways is required for prevention of ErbB-2-positive breast tumour recurrence. Br. J. Cancer 105, 796–806 (2011).
Abel, E. V. et al. The Notch pathway is important in maintaining the cancer stem cell population in pancreatic cancer. PLoS ONE 9, e91983 (2014).
Grudzien, P. et al. Inhibition of Notch signaling reduces the stem-like population of breast cancer cells and prevents mammosphere formation. Anticancer Res. 30, 3853–3867 (2010).
Hassan, K. A. et al. Notch pathway activity identifies cells with cancer stem cell-like properties and correlates with worse survival in lung adenocarcinoma. Clin. Cancer Res. 19, 1972–1980 (2013).
Saito, N. et al. A high Notch pathway activation predicts response to γ-secretase inhibitors in proneural subtype of glioma tumor-initiating cells. Stem Cells 32, 301–312 (2014).
Messersmith, W. A. et al. A phase I, dose-finding study in patients with advanced solid malignancies of the oral γ-secretase inhibitor PF-03084014. Clin. Cancer Res. 21, 60–67 (2015).
Milano, J. et al. Modulation of Notch processing by γ-secretase inhibitors causes intestinal goblet cell metaplasia and induction of genes known to specify gut secretory lineage differentiation. Toxicol. Sci. 82, 341–358 (2004).
Searfoss, G. H. et al. Adipsin, a biomarker of gastrointestinal toxicity mediated by a functional γ-secretase inhibitor. J. Biol. Chem. 278, 46107–46116 (2003).
Wong, G. T. et al. Chronic treatment with the γ-secretase inhibitor LY-411575 inhibits β-amyloid peptide production and alters lymphopoiesis and intestinal cell differentiation. J. Biol. Chem. 279, 12876–12882 (2004).
van Es, J. H. et al. Notch/γ-secretase inhibition turns proliferative cells in intestinal crypts and adenomas into goblet cells. Nature 435, 959–963 (2005).
Krop, I. et al. Phase I pharmacologic and pharmacodynamic study of the gamma secretase (Notch) inhibitor MK-0752 in adult patients with advanced solid tumors. J. Clin. Oncol. 30, 2307–2313 (2012).
Dumortier, A. et al. Atopic dermatitis-like disease and associated lethal myeloproliferative disorder arise from loss of Notch signaling in the murine skin. PLoS ONE 5, e9258 (2010).
Roderick, J. E. et al. Therapeutic targeting of NOTCH signaling ameliorates immune-mediated bone marrow failure of aplastic anemia. J. Exp. Med. 210, 1311–1329 (2013).
Real, P. J. & Ferrando, A. A. NOTCH inhibition and glucocorticoid therapy in T-cell acute lymphoblastic leukemia. Leukemia 23, 1374–1377 (2009).
Yun, J. et al. Crosstalk between PKCα and Notch-4 in endocrine-resistant breast cancer cells. Oncogenesis 2, e60 (2013).
Means-Powell, J. et al. A phase Ib dose escalation trial of RO4929097 (a γ-secretase inhibitor) in combination with exemestane in patients with ER+ metastatic breast cancer [abstract P2-14-04]. Cancer Res. 72 (Suppl.), 280s (2012).
Albain, K. Combination of Notch inhibitor MK-0752 and endocrine therapy for early stage ERα breast cancer in a pre-surgical window study. Cancer Res. 70 (Suppl.), 113s–114s (2010).
Tolcher, A. W. et al. Phase I study of RO4929097, a gamma secretase inhibitor of Notch signaling, in patients with refractory metastatic or locally advanced solid tumors. J. Clin. Oncol. 30, 2348–2353 (2012).
Harrison, H. et al. Regulation of breast cancer stem cell activity by signaling through the Notch4 receptor. Cancer Res. 70, 709–718 (2010).
Hellstrom, M. et al. DLL4 signalling through Notch1 regulates formation of tip cells during angiogenesis. Nature 445, 776–780 (2007).
Jimeno, A. et al. Phase I study of REGN421/SAR153192, a fully-human delta-like ligand 4 (DLL4) monoclonal antibody (mAb), in patients with advanced solid tumors [abstract]. J. Clin. Oncol. 31 (Suppl.), a2502 (2013).
Yan, M. Therapeutic promise and challenges of targeting DLL4/NOTCH1. Vasc. Cell 3, 17 (2011).
Xie, M., He, C. S., He, Z. & Lin, Q. Clinical and prognostic implications of delta-like ligand 4 and hypoxia-inducible factors in metastatic renal cell carcinoma (mRCC) patients treated with sunitinib as first-line therapy [abstract]. J. Clin. Oncol. 31 (Suppl.), e15567 (2013).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
Sahebjam, S. et al. A phase I study of the combination of RO4929097 and cediranib in patients with advanced solid tumours (PJC-004/NCI 8503). Br. J. Cancer 109, 943–949 (2013).
Sagert, J. et al. Tumor-specific inhibition of Jagged-dependent Notch signaling using a Probody™ therapeutic [abstract]. Mol. Cancer Ther. 12 (Suppl. 11), C158 (2013).
Mohammed, T. A. et al. A pilot phase II study of valproic acid for treatment of low-grade neuroendocrine carcinoma. Oncologist 16, 835–843 (2011).
Rajguru, S. et al. A phase II study of the histone deacetylase inhibitor panobinostat (LBH589) in low-grade neuroendocrine tumors [abstract]. J. Clin. Oncol. 30 (Suppl.), e14554 (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
Paryan, M. et al. Investigation of deregulated genes of Notch signaling pathway in human T cell acute lymphoblastic leukemia cell lines and clinical samples. Mol. Biol. Rep. 40, 5531–5540 (2013).
Fogelstrand, L. et al. Prognostic implications of mutations in NOTCH1 and FBXW7 in childhood T-ALL treated according to the NOPHO ALL-1992 and ALL-2000 protocols. Pediatr. Blood Cancer 61, 424–430 (2014).
Wang, H. et al. NOTCH1–RBPJ complexes drive target gene expression through dynamic interactions with superenhancers. Proc. Natl Acad. Sci. USA 111, 705–710 (2014).
Takebe, N., Nguyen, D. & Yang, S. X. Targeting Notch signaling pathway in cancer: clinical development advances and challenges. Pharmacol. Ther. 141, 140–149 (2014).
Andersen, P., Uosaki, H., Shenje, L. T. & Kwon, C. Non-canonical Notch signaling: emerging role and mechanism. Trends Cell Biol. 22, 257–265 (2012).
Stoeck, A. et al. Discovery of biomarkers predictive of GSI response in triple-negative breast cancer and adenoid cystic carcinoma. Cancer Discov. 4, 1154–1167 (2014).
Nguyen, D. et al. Notch1 phenotype and clinical stage progression in non-small cell lung cancer. J. Hematol. Oncol. 8, 9 (2015).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
Rota, L. M., Lazzarino, D. A., Ziegler, A. N., LeRoith, D. & Wood, T. L. Determining mammosphere-forming potential: application of the limiting dilution analysis. J. Mammary Gland Biol. Neoplasia 17, 119–123 (2012).
Espinoza, I., Pochampally, R., Xing, F., Watabe, K. & Miele, L. Notch signaling: targeting cancer stem cells and epithelial-to-mesenchymal transition. Onco Targets Ther. 6, 1249–1259 (2013).
Fernandez-Majada, V. et al. Nuclear IKK activity leads to dysregulated notch-dependent gene expression in colorectal cancer. Proc. Natl Acad. Sci. USA 104, 276–281 (2007).
Fre, S. et al. Notch and Wnt signals cooperatively control cell proliferation and tumorigenesis in the intestine. Proc. Natl Acad. Sci. USA 106, 6309–6314 (2009).
Kode, A. et al. Leukaemogenesis induced by an activating β-catenin mutation in osteoblasts. Nature 506, 240–244 (2014).
Sjolund, J. et al. The Notch and TGF-β signaling pathways contribute to the aggressiveness of clear cell renal cell carcinoma. PLoS ONE 6, e23057 (2011).
Izrailit, J., Berman, H. K., Datti, A., Wrana, J. L. & Reedijk, M. High throughput kinase inhibitor screens reveal TRB3 and MAPK–ERK/TGFβ pathways as fundamental Notch regulators in breast cancer. Proc. Natl Acad. Sci. USA 110, 1714–1719 (2013).
Albain, K. et al. Modulation of cancer stem cell biomarkers by the Notch inhibitor MK0752 added to endocrine therapy for early stage ER+ breast cancer [abstract S1-5]. Cancer Res. 71 (Suppl.), (2011).
Osipo, C. et al. ErbB-2 inhibition activates Notch-1 and sensitizes breast cancer cells to a γ-secretase inhibitor. Oncogene 27, 5019–5032 (2008).
Zhang, C. C. et al. Synergistic effect of the γ-secretase inhibitor PF-03084014 and docetaxel in breast cancer models. Stem Cells Transl. Med. 2, 233–242 (2013).
Zhang, S., Chung, W. C., Miele, L. & Xu, K. Targeting Met and Notch in the Lfng-deficient, Met-amplified triple-negative breast cancer. Cancer Biol. Ther. 15, 633–642 (2014).
Strosberg, J. R. et al. A phase II study of RO4929097 in metastatic colorectal cancer. Eur. J. Cancer 48, 997–1003 (2012).
Beachy, P. A., Hymowitz, S. G., Lazarus, R. A., Leahy, D. J. & Siebold, C. Interactions between Hedgehog proteins and their binding partners come into view. Genes Dev. 24, 2001–2012 (2010).
Odoux, C. et al. A stochastic model for cancer stem cell origin in metastatic colon cancer. Cancer Res. 68, 6932–6941 (2008).
Amakye, D., Jagani, Z. & Dorsch, M. Unraveling the therapeutic potential of the Hedgehog pathway in cancer. Nat. Med. 19, 1410–1422 (2013).
Ng, J. M. & Curran, T. The Hedgehog's tale: developing strategies for targeting cancer. Nat. Rev. Cancer 11, 493–501 (2011).
Kool, M. et al. Genome sequencing of SHH medulloblastoma predicts genotype-related response to Smoothened inhibition. Cancer Cell 25, 393–405 (2014).
Johnson, R. L. et al. Human homolog of Patched, a candidate gene for the basal cell nevus syndrome. Science 272, 1668–1671 (1996).
Roma, J., Almazan-Moga, A., Sanchez de Toledo, J. & Gallego, S. Notch, Wnt, and Hedgehog pathways in rhabdomyosarcoma: from single pathways to an integrated network. Sarcoma 2012, 695603 (2012).
Dennler, S. et al. Induction of Sonic hedgehog mediators by transforming growth factor-β: Smad3-dependent activation of Gli2 and Gli1 expression in vitro and in vivo. Cancer Res. 67, 6981–6986 (2007).
Jagani, Z. et al. Loss of the tumor suppressor Snf5 leads to aberrant activation of the Hedgehog–Gli pathway. Nat. Med. 16, 1429–1433 (2010).
Ji, Z., Mei, F. C., Xie, J. & Cheng, X. Oncogenic KRAS activates Hedgehog signaling pathway in pancreatic cancer cells. J. Biol. Chem. 282, 14048–14055 (2007).
Riobo, N. A., Lu, K., Ai, X., Haines, G. M. & Emerson, C. P. Jr. Phosphoinositide 3-kinase and Akt are essential for Sonic Hedgehog signaling. Proc. Natl Acad. Sci. USA 103, 4505–4510 (2006).
Wang, Y. et al. The crosstalk of mTOR/S6K1 and Hedgehog pathways. Cancer Cell 21, 374–87 (2012).
Dirix, L. Discovery and exploitation of novel targets by approved drugs. J. Clin. Oncol. 32, 720–721 (2014).
Sekulic, A. et al. Efficacy and safety of vismodegib in advanced basal-cell carcinoma. N. Engl. J. Med. 366, 2171–2179 (2012).
Lewis, K. D. et al. Vismodegib in the treatment of patients with metastatic basal cell carcinoma (mBCC) and distant metastases: survival in the pivotal phase II and phase I studies [abstract]. J. Clin. Oncol. 32 (5s Suppl.), a9012 (2014).
Mortier, L. et al. A phase II study to assess vismodegib in the neoadjuvant treatment of locally advanced basal cell carcinoma (laBCC): the Vismodegib Neoadjuvant (VISMONEO) study [abstract]. J. Clin. Oncol. 32 (5s Suppl.), TPS9014 (2014).
Sloan, A. E. et al. Targeting glioma-initiating cells in GBM: ABTC-0904, a randomized phase 0/II study targeting the Sonic Hedgehog-signaling pathway [abstract]. J. Clin. Oncol. 32 (5s Suppl.), a2026 (2014).
Belani, C. P. et al. Three-arm randomized phase II study of cisplatin and etoposide (CE) versus CE with either vismodegib (V) or cixutumumab (Cx) for patients with extensive stage-small cell lung cancer (ES-SCLC) (ECOG 1508) [abstract]. J. Clin. Oncol. 31 (Suppl.), a7508 (2013).
Catenacci, D. V. T. et al. Final analysis of a phase IB/randomized phase II study of gemcitabine (G.) plus placebo (P) or vismodegib (V), a hedgehog (Hh) pathway inhibitor, in patients (pts) with metastatic pancreatic cancer (PC): a University of Chicago phase II consortium study [abstract]. J. Clin. Oncol. 31 (Suppl.), a4012 (2013).
Cohen, D. J. et al. Vismodegib (V), a Hedgehog (HH) pathway inhibitor, combined with FOLFOX for first-line therapy of patients (pts) with advanced gastric and gastroesophageal junction (GEJ) carcinoma: a New York Cancer Consortium led phase II randomized study [abstract]. J. Clin. Oncol. 31 (Suppl.), a4011 (2013).
De Jesus-Acosta, A. et al. A phase II study of vismodegib, a Hedgehog (Hh) pathway inhibitor, combined with gemcitabine and nab-paclitaxel (nab-P) in patients (pts) with untreated metastatic pancreatic ductal adenocarcinoma (PDA) [abstract]. J. Clin. Oncol. 32 (Suppl. 3), a257 (2014).
Von Hoff, D. E. et al. Randomized phase III study of weekly nab-paclitaxel plus gemcitabine versus gemcitabine alone in patients with metastatic adenocarcinoma of the pancreas (MPACT) [abstract]. J. Clin. Oncol. 30 (Suppl. 34), LBA148 (2012).
Berlin, J. et al. A randomized phase II trial of vismodegib versus placebo with FOLFOX or FOLFIRI and bevacizumab in patients with previously untreated metastatic colorectal cancer. Clin. Cancer Res. 19, 258–267 (2013).
Kaye, S. B. et al. A phase II, randomized, placebo-controlled study of vismodegib as maintenance therapy in patients with ovarian cancer in second or third complete remission. Clin. Cancer Res. 18, 6509–6518 (2012).
Madden, J. I. Infinity Reports Update from Phase 2 Study of Saridegib Plus Gemcitabine in Patients with Metastatic Pancreatic Cancer. Infinity Pharmaceuticals [online], (2012).
Madden, J. I. Infinity Stops Phase 2 Trials of Saridegib in Chondrosarcoma and Myelofibrosis. FierceBiotech [online], (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2015).
Migden, M. R. et al. Randomized, double-blind study of sonidegib (LDE225) in patients (pts) with locally advanced (La) or metastatic (m) basal-cell carcinoma (BCC) [abstract]. J. Clin. Oncol. 32 (5s Suppl.), a9009a (2014).
Pietanza, M. C. et al. Phase I trial of the Hedgehog (Hh) inhibitor, LDE225, in combination with etoposide and cisplatin (EP) for initial treatment of extensive stage small cell lung cancer (ES-SCLC) [abstract]. J. Clin. Oncol. 32 (5s Suppl.), a7602 (2014).
Zahreddine, H. A. et al. The Sonic hedgehog factor GLI1 imparts drug resistance through inducible glucuronidation. Nature 511, 90–93 (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2015).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2015).
Yauch, R. L. et al. A paracrine requirement for Hedgehog signalling in cancer. Nature 455, 406–410 (2008).
Ellison, D. W. et al. Medulloblastoma: clinicopathological correlates of, SHH, WNT, and non-SHH/WNT molecular subgroups. Acta Neuropathol. 121, 381–396 (2011).
Shou, Y. et al. A five-gene Hedgehog signature developed as a patient preselection tool for hedgehog inhibitor therapy in medulloblastoma. Clin. Cancer Res. 21, 585–593 (2015).
Von Hoff, D. D. et al. Inhibition of the Hedgehog pathway in advanced basal-cell carcinoma. N. Engl. J. Med. 361, 1164–1172 (2009).
Graham, R. A. et al. Pharmacokinetics of Hedgehog pathway inhibitor vismodegib (GDC-0449) in patients with locally advanced or metastatic solid tumors: the role of alpha-1-acid glycoprotein binding. Clin. Cancer Res. 17, 2512–2520 (2011).
Ramaswamy, B. et al. Hedgehog signaling is a novel therapeutic target in tamoxifen-resistant breast cancer aberrantly activated by PI3K/AKT pathway. Cancer Res. 72, 5048–5059 (2012).
Yauch, R. L. et al. Smoothened mutation confers resistance to a Hedgehog pathway inhibitor in medulloblastoma. Science 326, 572–574 (2009).
Buonamici, S. et al. Interfering with resistance to Smoothened antagonists by inhibition of the PI3K pathway in medulloblastoma. Sci. Transl. Med. 2, 51ra70 (2010).
Yoon, J. W. et al. Noncanonical regulation of the Hedgehog mediator GLI1 by c-MYC in Burkitt lymphoma. Mol. Cancer Res. 11, 604–615 (2013).
Hollingshead, M. G. Antitumor efficacy testing in rodents. J. Natl Cancer Inst. 100, 1500–1510 (2008).
Brennan, D., Chen, X., Cheng, L., Mahoney, M. & Riobo, N. A. Noncanonical Hedgehog signaling. Vitam. Horm. 88, 55–72 (2012).
Gruber Filbin, M. et al. Coordinate activation of SHH and PI3K signaling in PTEN-deficient glioblastoma: new therapeutic opportunities. Nat. Med. 19, 1518–1523 (2013).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
Kim, D. J. et al. Open-label, exploratory phase II trial of oral itraconazole for the treatment of basal cell carcinoma. J. Clin. Oncol. 32, 745–751 (2014).
Kim, J. et al. Itraconazole, a commonly used antifungal that inhibits Hedgehog pathway activity and cancer growth. Cancer Cell 17, 388–399 (2010).
Kim, J., Lee, J. J., Kim, J., Gardner, D. & Beachy, P. A. Arsenic antagonizes the Hedgehog pathway by preventing ciliary accumulation and reducing stability of the Gli2 transcriptional effector. Proc. Natl Acad. Sci. USA 107, 13432–13437 (2010).
Kim, J. et al. Itraconazole and arsenic trioxide inhibit Hedgehog pathway activation and tumor growth associated with acquired resistance to Smoothened antagonists. Cancer Cell 23, 23–34 (2013).
Chien, A. J., Conrad, W. H. & Moon, R. T. A Wnt survival guide: from flies to human disease. J. Invest. Dermatol. 129, 1614–1627 (2009).
Willert, K. et al. Wnt proteins are lipid-modified and can act as stem cell growth factors. Nature 423, 448–452 (2003).
Takada, R. et al. Monounsaturated fatty acid modification of Wnt protein: its role in Wnt secretion. Dev. Cell 11, 791–801 (2006).
Nusse, R. Wnt signaling in disease and in development. Cell Res. 15, 28–32 (2005).
He, X., Semenov, M., Tamai, K. & Zeng, X. LDL receptor-related proteins 5 and 6 in Wnt/β-catenin signaling: arrows point the way. Development 131, 1663–1677 (2004).
Logan, C. Y. & Nusse, R. The Wnt signaling pathway in development and disease. Annu. Rev. Cell Dev. Biol. 20, 781–810 (2004).
Itasaki, N. et al. Wise, a context-dependent activator and inhibitor of Wnt signalling. Development 130, 4295–4305 (2003).
Gao, C. & Chen, Y. G. Dishevelled: the hub of Wnt signaling. Cell Signal. 22, 717–727 (2010).
Habas, R. & Dawid, I. B. Dishevelled and Wnt signaling: is the nucleus the final frontier? J. Biol. 4, 2 (2005).
Behrens, J. Control of β-catenin signaling in tumor development. Ann. N. Y. Acad. Sci. 910, 21–33 (2000).
Arce, L., Yokoyama, N. N. & Waterman, M. L. Diversity of LEF/TCF action in development and disease. Oncogene 25, 7492–7504 (2006).
Nguyen, L. V., Vanner, R., Dirks, P. & Eaves, C. J. Cancer stem cells: an evolving concept. Nat. Rev. Cancer 12, 133–143 (2012).
Malanchi, I. et al. Cutaneous cancer stem cell maintenance is dependent on β-catenin signalling. Nature 452, 650–653 (2008).
Vincan, E. & Barker, N. The upstream components of the Wnt signalling pathway in the dynamic EMT and MET associated with colorectal cancer progression. Clin. Exp. Metastasis 25, 657–663 (2008).
Lee, H. J., Wang, N. X., Shi, D. L. & Zheng, J. J. Sulindac inhibits canonical Wnt signaling by blocking the PDZ domain of the protein Dishevelled. Angew. Chem. Int. Ed. Engl. 48, 6448–6452 (2009).
Barker, N. & Clevers, H. Mining the Wnt pathway for cancer therapeutics. Nat. Rev. Drug Discov. 5, 997–1014 (2006).
Smith, M. L., Hawcroft, G. & Hull, M. A. The effect of non-steroidal anti-inflammatory drugs on human colorectal cancer cells: evidence of different mechanisms of action. Eur. J. Cancer 36, 664–674 (2000).
Yoshizumi, T. et al. Thiazolidinedione, a peroxisome proliferator-activated receptor-γ ligand, inhibits growth and metastasis of HT-29 human colon cancer cells through differentiation-promoting effects. Int. J. Oncol. 25, 631–639 (2004).
Huang, S. M. et al. Tankyrase inhibition stabilizes axin and antagonizes Wnt signalling. Nature 461, 614–620 (2009).
Waaler, J. et al. A novel tankyrase inhibitor decreases canonical Wnt signaling in colon carcinoma cells and reduces tumor growth in conditional APC mutant mice. Cancer Res. 72, 2822–2832 (2012).
Waaler, J. et al. Novel synthetic antagonists of canonical Wnt signaling inhibit colorectal cancer cell growth. Cancer Res. 71, 197–205 (2011).
Fujii, N. et al. An antagonist of dishevelled protein–protein interaction suppresses β-catenin-dependent tumor cell growth. Cancer Res. 67, 573–579 (2007).
Grandy, D. et al. Discovery and characterization of a small molecule inhibitor of the PDZ domain of dishevelled. J. Biol. Chem. 284, 16256–16263 (2009).
Su, Y., Ishikawa, S., Kojima, M. & Liu, B. Eradication of pathogenic β-catenin by Skp1/Cullin/F box ubiquitination machinery. Proc. Natl Acad. Sci. USA 100, 12729–12734 (2003).
Yao, H. et al. AV-65, a novel Wnt/β-catenin signal inhibitor, successfully suppresses progression of multiple myeloma in a mouse model. Blood Cancer J. 1, e43 (2011).
El-Khoueiry, A. B. et al. A phase I first-in-human study of PRI-724 in patients (pts) with advanced solid tumors [abstract]. J. Clin. Oncol. 31 (Suppl.), a2501 (2013).
Ma, H., Nguyen, C., Lee, K. S. & Kahn, M. Differential roles for the coactivators CBP and p300 on TCF/β-catenin-mediated survivin gene expression. Oncogene 24, 3619–3631 (2005).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2015).
Li, X. et al. Prostate tumor progression is mediated by a paracrine TGF- β/Wnt3a signaling axis. Oncogene 27, 7118–7130 (2008).
Kahn, M. Can we safely target the WNT pathway? Nat. Rev. Drug Discov. 13, 513–532 (2014).
Smith, D. C. et al. First-in-human evaluation of the human monoclonal antibody vantictumab (OMP-18R5; anti-Frizzled) targeting the Wnt pathway in a phase I study for patients with advanced solid tumors [abstract]. J. Clin. Oncol. 31 (Suppl.), a2540 (2013).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
US National Library of Medicine. ClinicalTrials.gov [online], (2015).
Smith, D. C. et al. A first-in-human phase I study of anti-cancer stem cell (CSC) agent OMP-54F28 (FZD8-Fc) targeting the Wnt pathway in patients with advanced solid tumors [abstract]. Mol. Cancer Ther. 12 (11 Suppl.), B79 (2013).
Jimeno, A. et al. A first-in-human phase I study of anticancer stem cell agent OMP-54F28 (FZD8-Fc), decoy receptor for Wnt ligands, in patients with advanced solid tumors [abstract]. J. Clin. Oncol. 32 (5s Suppl.), a2505 (2014).
Katoh, M. Networking of Wnt, FGF, Notch, BMP, and Hedgehog signaling pathways during carcinogenesis. Stem Cell Rev. 3, 30–38 (2007).
Kwon, C. et al. Notch post-translationally regulates β-catenin protein in stem and progenitor cells. Nat. Cell Biol. 13, 1244–1251 (2011).
Rodilla, V. et al. Jagged1 is the pathological link between Wnt and Notch pathways in colorectal cancer. Proc. Natl Acad. Sci. USA 106, 6315–6320 (2009).
Arcaroli, J. J. et al. Tumours with elevated levels of the Notch and Wnt pathways exhibit efficacy to PF-03084014, a γ-secretase inhibitor, in a preclinical colorectal explant model. Br. J. Cancer 109, 667–675 (2013).
Escobar, C., Munker, R., Thomas, J. O., Li, B. D. & Burton, G. V. Update on desmoid tumors. Ann. Oncol. 23, 562–569 (2012).
Messersmith, W. A. et al. First-in-patient phase I study of the novel gamma secretase inhibitor PF-03084014 in patients with advanced solid tumor malignancies [abstract 588]. Eur. J. Cancer 48 (Suppl. 6), 180 (2012).
Carothers, A. M. et al. Mesenchymal stromal cell mutations and wound healing contribute to the etiology of desmoid tumors. Cancer Res. 72, 346–355 (2012).
Doody, R. S. et al. A phase 3 trial of semagacestat for treatment of Alzheimer's disease. N. Engl. J. Med. 369, 341–350 (2013).
He, J. et al. Suppressing Wnt signaling by the Hedgehog pathway through sFRP-1. J. Biol. Chem. 281, 35598–35602 (2006).
Schneider, F. T. et al. Sonic hedgehog acts as a negative regulator of β-catenin signaling in the adult tongue epithelium. Am. J. Pathol. 177, 404–414 (2010).
Rabadan, M. A. et al. Jagged2 controls the generation of motor neuron and oligodendrocyte progenitors in the ventral spinal cord. Cell Death Differ. 19, 209–219 (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2015).
Dakubo, G. D., Mazerolle, C. J. & Wallace, V. A. Expression of Notch and Wnt pathway components and activation of Notch signaling in medulloblastomas from heterozygous patched mice. J. Neurooncol. 79, 221–227 (2006).
Ingram, W. J., McCue, K. I., Tran, T. H., Hallahan, A. R. & Wainwright, B. J. Sonic hedgehog regulates Hes1 through a novel mechanism that is independent of canonical Notch pathway signalling. Oncogene 27, 1489–1500 (2008).
Di Marcotullio, L. et al. Numb is a suppressor of Hedgehog signalling and targets Gli1 for Itch-dependent ubiquitination. Nat. Cell Biol. 8, 1415–1423 (2006).
Domingo-Domenech, J. et al. Suppression of acquired docetaxel resistance in prostate cancer through depletion of Notch- and Hedgehog-dependent tumor-initiating cells. Cancer Cell 22, 373–388 (2012).
Cilloni, D. & Saglio, G. Molecular pathways: BCR–ABL. Clin. Cancer Res. 18, 930–937 (2012).
Chau, W. K., Ip, C. K., Mak, A. S., Lai, H. C. & Wong, A. S. c-Kit mediates chemoresistance and tumor-initiating capacity of ovarian cancer cells through activation of Wnt/β-catenin-ATP-binding cassette G2 signaling. Oncogene 32, 2767–2781 (2013).
Espinosa, L., Ingles-Esteve, J., Aguilera, C. & Bigas, A. Phosphorylation by glycogen synthase kinase-3β down-regulates Notch activity, a link for Notch and Wnt pathways. J. Biol. Chem. 278, 32227–32235 (2003).
Saint Just Ribeiro, M., Hansson, M. L., Lindberg, M. J., Popko-Scibor, A. E. & Wallberg, A. E. GSK3β is a negative regulator of the transcriptional coactivator MAML1. Nucleic Acids Res. 37, 6691–6700 (2009).
Foltz, D. R., Santiago, M. C., Berechid, B. E. & Nye, J. S. Glycogen synthase kinase-3β modulates Notch signaling and stability. Curr. Biol. 12, 1006–1011 (2002).
Rizzo, P. et al. Cross-talk between notch and the estrogen receptor in breast cancer suggests novel therapeutic approaches. Cancer Res. 68, 5226–5235 (2008).
Hao, L. et al. Notch-1 activates estrogen receptor-α-dependent transcription via IKKα in breast cancer cells. Oncogene 29, 201–213 (2010).
Acknowledgements
We gratefully acknowledge Dr Richard Swerdlow of PSI INTERNATIONAL, Inc., Rockville, MD, USA, for his assistance in organizing the references and formatting the manuscript, and Melissa Maher at Technical Resources International, Inc., Bethesda, MD, USA, for drafting the Figures.
Author information
Authors and Affiliations
Contributions
All authors contributed substantially to researching the data for the article, discussions of content, writing the article, and review/editing of the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
L.M. has participated in collaborative research on Notch inhibitors with CytoMX and Merck Oncology. M.K. is an equity holder in Prism Pharma. The other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Takebe, N., Miele, L., Harris, P. et al. Targeting Notch, Hedgehog, and Wnt pathways in cancer stem cells: clinical update. Nat Rev Clin Oncol 12, 445–464 (2015). https://doi.org/10.1038/nrclinonc.2015.61
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2015.61
This article is cited by
-
GPR68-ATF4 signaling is a novel prosurvival pathway in glioblastoma activated by acidic extracellular microenvironment
Experimental Hematology & Oncology (2024)
-
Crosstalk between colorectal CSCs and immune cells in tumorigenesis, and strategies for targeting colorectal CSCs
Experimental Hematology & Oncology (2024)
-
Non-canonical non-genomic morphogen signaling in anucleate platelets: a critical determinant of prothrombotic function in circulation
Cell Communication and Signaling (2024)
-
SLC26A9 promotes colorectal tumorigenesis by modulating Wnt/β-catenin signaling
Cell Death Discovery (2024)
-
Targeted activation of ferroptosis in colorectal cancer via LGR4 targeting overcomes acquired drug resistance
Nature Cancer (2024)