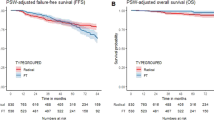
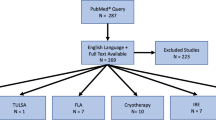
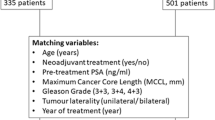
Abstract
Tissue-preserving focal therapies, such as brachytherapy, cryotherapy, high-intensity focused ultrasound and photodynamic therapy, aim to target individual cancer lesions rather than the whole prostate. These treatments have emerged as potential interventions for localized prostate cancer to reduce treatment-related adverse-effects associated with whole-gland treatments, such as radical prostatectomy and radiotherapy. In this article, the Prostate Cancer RCT Consensus Group propose that a novel cohort-embedded randomized controlled trial (RCT) would provide a means to study men with clinically significant localized disease, which we defined on the basis of PSA level (≤15 ng/ml or ≤20 ng/ml), Gleason grade (Gleason pattern ≤4 + 4 or ≤4 + 3) and stage (≤cT2cN0M0). This RCT should recruit men who stand to benefit from treatment, with the control arm being whole-gland surgery or radiotherapy. Composite outcomes measuring rates of local and systemic salvage therapies at 3–5 years might best constitute the basis of the primary outcome on which to change practice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Change history
12 September 2017
Nature Reviews Clinical Oncology 11, 482–491 (2014) In the published version of this article, the affiliation and position held by Dr Viktor Berge was incorrectly stated as Professor of Urology and Director of Research at the University Hospital Oslo. Dr Berge is a Consultant Urologist at the Department of Urology, at the Oslo University Hospital.
References
Wilt, T. J. & Ahmed, H. U. Prostate cancer screening and the management of clinically localized disease. BMJ 346, f325 (2009).
Welch, H. G. & Albertsen, P. C. Prostate cancer diagnosis and treatment after the introduction of prostate-specific antigen screening: 1986–2005. J. Natl Cancer Inst. 101, 1325–1329 (2009).
Chou, R. et al. Screening for prostate cancer: a review of the evidence for the U. S. preventive services task force. Ann. Intern. Med. 155, 762–771 (2011).
McVey, G. P. et al. Initial management of low-risk localized prostate cancer in the UK: analysis of the British Association of Urological Surgeons Cancer Registry. BJU Int. 106, 1161–1164 (2010).
Cooperberg, M. R. et al. The changing face of low-risk prostate cancer: trends in clinical presentation and primary management. J. Clin. Oncol. 22, 2141–2149 (2004).
Bill-Axelson, A. et al. Radical prostatectomy versus watchful waiting in early prostate cancer. N. Engl. J. Med. 364, 1708–1717 (2011).
Wilt, T. J. et al. Radical prostatectomy versus observation for localized prostate cancer. N. Engl. J. Med. 367, 203–213 (2012).
Resnick, M. J. et al. Long-term functional outcomes after treatment for localized prostate cancer. N. Engl. J. Med. 368, 436–445 (2013).
Ahmed, H. U. et al. Will focal therapy become a standard of care for men with localized prostate cancer? Nat. Clin. Pract. Oncol. 4, 632–642 (2007).
Ahmed, H. U. The index lesion and the origin of prostate cancer. N. Engl. J. Med. 361, 1704–1706 (2009).
Valerio, M. et al. The role of focal therapy in the management of localised prostate cancer: a systematic review. Eur. Urol. http://dx.doi.org/10.1016/j.eururo.2013.05.048.
Ahmed, H. U. et al. Transatlantic consensus group on active surveillance and focal therapy for prostate cancer. BJU Int. 109, 1636–1647 (2012).
Arumainayagam, N. et al. Multiparametric MR imaging for detection of clinically significant prostate cancer: a validation cohort study with transperineal template prostate mapping as the reference standard. Radiology 268, 761–769 (2013).
Kasivisvanathan, V. et al. Transperineal magnetic resonance image targeted prostate biopsy versus transperineal template prostate biopsy in the detection of clinically significant prostate cancer. J. Urol. 189, 860–866 (2013).
Puech, P. et al. Prostate cancer diagnosis: multiparametric MR-targeted biopsy with cognitive and transrectal US-MR fusion guidance versus systematic biopsy—prospective multicenter study. Radiology 268, 461–469 (2013).
Puech, P. et al. Dynamic contrast-enhanced-magnetic resonance imaging evaluation of intraprostatic prostate cancer: correlation with radical prostatectomy specimens. Urology 74, 1094–1099 (2009).
Turkbey, B. et al. Correlation of magnetic resonance imaging tumor volume with histopathology. J. Urol. 188, 1157–1163 (2012).
Losa, A. et al. Complications and quality of life after template-assisted transperineal prostate biopsy in patients eligible for focal therapy. Urology 81, 1291–1296 (2013).
Crawford, E. D. et al. Clinical-pathologic correlation between transperineal mapping biopsies of the prostate and three-dimensional reconstruction of prostatectomy specimens. Prostate 73, 778–787 (2013).
Huo, A. S. et al. Accuracy of primary systematic template guided transperineal biopsy of the prostate for locating prostate cancer: a comparison with radical prostatectomy specimens. J. Urol. 187, 2044–2049 (2012).
Ahmed, H. U. et al. Characterizing clinically significant prostate cancer using template prostate mapping biopsy. J. Urol. 186, 458–464 (2011).
Taira, A. V. et al. Performance of transperineal template-guided mapping biopsy in detecting prostate cancer in the initial and repeat biopsy setting. Prostate Cancer Prostatic Dis. 13, 71–77 (2010).
US National Library of Medicine. ClinicalTrials.gov[online], (2011).
Simmons, L. A. et al. The PICTURE study—Prostate Imaging (multi-parametric MRI and Prostate HistoScanning™) Compared to Transperineal Ultrasound guided biopsy for significant prostate cancer Risk Evaluation. Contemp. Clin. Trials 37, 69–83 (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Bahn, D. et al. Focal cryotherapy for clinically unilateral, low-intermediate risk prostate cancer in 73 men with a median follow-up of 3.7 years. Eur. Urol. 62, 55–63 (2012).
Ward, J. F. & Jones J. S. Focal cryotherapy for localized prostate cancer: a report from the national Cryo On-Line Database (COLD) Registry. BJU Int. 109, 1648–1654 (2012).
Ahmed, H. U. et al. Focal therapy for localised unifocal and multifocal prostate cancer: a prospective development study. Lancet Oncol. 13, 622–632 (2012).
Ahmed, H. U. et al. Focal therapy for localized prostate cancer: a phase I/II trial. J. Urol. 185, 1246–1254 (2011).
Ahmed, H. U. et al. Focal salvage therapy for localized prostate cancer recurrence after external beam radiotherapy: A pilot study. Cancer 118, 4148–4155 (2012).
Lindner, U. et al. Image guided photothermal focal therapy for localized prostate cancer: phase I trial. J. Urol. 182, 1371–1377 (2013).
Oto, A. et al. MR Imaging-guided focal laser ablation for prostate cancer: phase I trial. Radiology 267, 932–940 (2013).
Preston, M. A., Harisinghani, M. G., Mucci, L., Witiuk, K. & Breau, R. H. Diagnostic tests in urology: magnetic resonance imaging (MRI) for the staging of prostate cancer. BJU Int. 111, 514–517 (2013).
Ahmed, H. U., Arya, M., Freeman, A. & Emberton, M. Do low-grade and low-volume prostate cancers bear the hallmarks of malignancy? Lancet Oncol. 13, e509–e517 (2012).
Nevoux, P. et al. Quantitative tissue analyses of prostate cancer foci in an unselected cystoprostatectomy series. BJU Int. 110, 517–523 (2012).
Karavitakis, M., Ahmed, H. U., Abel, P. D., Hazell, S. & Winkler, M. H. Tumor focality in prostate cancer: implications for focal therapy. Nat. Rev. Clin. Oncol. 8, 48–55 (2011).
Mazzucchelli, R. et al. Pathology of prostate cancer and focal therapy ('male lumpectomy'). Anticancer Res. 29, 5155–5161 (2009).
Ahmed, H. U., Emberton, M., Kepner, G. & Kepner, J. A biomedical engineering approach to mitigate the errors of prostate biopsy. Nat. Rev. Urol. 9, 227–231 (2012).
Han, M. et al. Geometric evaluation of systematic transrectal ultrasound guided prostate biopsy. J. Urol. 188, 2404–2409 (2012).
Wong, L. M. et al. A negative confirmatory biopsy among men on active surveillance for prostate cancer does not protect them from histologic grade progression. Eur. Urol. http://dx.doi.org/10.1016/j.eururo.2013.04.038.
Tsivian, M., Hruza, M., Mouraviev, V., Rassweiler, J. & Polascik, T. J. Prostate biopsy in selecting candidates for hemiablative focal therapy. J. Endourol. 24, 849–853 (2010).
MRC. Developing and evaluating complex interventions: new guidance [online], (2008).
Arumainayagam, N. et al. Multiparametric MR imaging for detection of clinically significant prostate cancer: a validation cohort study with transperineal template prostate mapping as the reference standard. Radiology 268, 761–769 (2013).
Muller, B. G. et al. The role of magnetic resonance imaging in focal therapy for prostate cancer: recommendations from a Consensus Panel. BJU Int. http://dx.doi.org/10.1111/bju.12243.
Yerram, N. K. et al. Low suspicion lesions on multiparametric magnetic resonance imaging predict for the absence of high-risk prostate cancer. BJU Int. 110, E783–E788 (2012).
Robertson, N. L., Emberton, M. & Moore, C. M. MRI-targeted prostate biopsy: a review of technique and results. Nat. Rev. Urol. 10, 589–597 (2013).
Puech, P. et al. Prostate cancer diagnosis: multiparametric MR-targeted biopsy with cognitive and transrectal US-MR fusion guidance versus systematic biopsy—prospective multicenter study. Radiology 268, 461–469 (2013).
Sonn, G. A. et al. Targeted biopsy in the detection of prostate cancer using an office based magnetic resonance ultrasound fusion device. J. Urol. 189, 86–91 (2013).
Kasivisvanathan, V. et al. Transperineal magnetic resonance image targeted prostate biopsy versus transperineal template prostate biopsy in the detection of clinically significant prostate cancer. J. Urol. 189, 860–866 (2013).
Moore, C. M. et al. Image-guided prostate biopsy using magnetic resonance imaging-derived targets: a systematic review. Eur. Urol. 63, 125–140 (2013).
Wysock, J. S. et al. A prospective, blinded comparison of magnetic resonance (MR) imaging-ultrasound fusion and visual estimation in the performance of MR-targeted prostate biopsy: the PROFUS trial. Eur. Urol. http://dx.doi.org/10.1016/j.eururo.2013.10.048.
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Jarow, J. P. et al. Drug and device development for localized prostate cancer: report of a Food and Drug Administration/American Urological Association Public Workshop. Urology http://dx.doi.org/10.1016/j.urology.2013.10.087.
Williamson, P. R. et al. Developing core outcome sets for clinical trials: issues to consider. Trials 13, 132 (2012).
Williamson, P., Altman, D., Blazeby, J., Clarke, M. & Gargon, E. Driving up the quality and relevance of research through the use of agreed core outcomes. J. Health Serv. Res. Policy 17, 1–2 (2012).
Freemantle, N. & Calvert, M. J. Interpreting composite outcomes in trials. BMJ 341, c3529 (2010).
Hagberg, K. W., Taylor, A., Hernandez, R. K. & Jick, S. Incidence of bone metastases in breast cancer patients in the United Kingdom: results of a multi-database linkage study using the general practice research database. Cancer Epidemiol. 37, 240–246 (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2011).
Mayer, E. et al. Randomised controlled trial (RCT) of Laparoscopic, OPEn and Robot-Assisted prostatectomy as treatment for organ-confined prostate cancer. LopeRA Feasibility Study (CRUK/09/008) [abstract]. NCRI UK Annual Conference, a49 (2011).
Eccles, B. K. et al. SABRE 1 (Surgery Against Brachytherapy—a Randomised Evaluation): feasibility randomised controlled trial (RCT) of brachytherapy vs radical prostatectomy in low-intermediate risk clinically localised prostate cancer. BJU Int. 112, 330–337 (2013).
Crook, J. M. et al. Comparison of health-related quality of life 5 years after SPIRIT: Surgical Prostatectomy Versus Interstitial Radiation Intervention Trial. J. Clin. Oncol. 29, 362–368 (2011).
Wallace, K., Fleshner, N., Jewett, M., Basiuk, J. & Crook, J. Impact of a multi-disciplinary patient education session on accrual to a difficult clinical trial: the Toronto experience with the surgical prostatectomy versus interstitial radiation intervention trial. J. Clin. Oncol. 24, 4158–4162 (2006).
Donnelly, B. J. et al. A randomized trial of external beam radiotherapy versus cryoablation in patients with localized prostate cancer. Cancer 116, 323–330 (2010).
Robinson, J. W. et al. A randomized trial of external beam radiotherapy versus cryoablation in patients with localized prostate cancer: quality of life outcomes. Cancer 115, 4695–4704 (2009).
Chin, J. L. et al. Randomized trial comparing cryoablation and external beam radiotherapy for T2C-T3B prostate cancer. Prostate Cancer Prostatic Dis. 11, 40–45 (2008).
Crawford, E. D. et al. Southwest Oncology Group strategies in prostatic carcinoma. Semin. Surg. Oncol. 11, 60–64 (1995).
O'Reilly, P., Martin, L. & Collins, G. Few patients with prostate cancer are willing to be randomised to treatment. BMJ 318, 1556 (1999).
PR06 Collaborators. Early closure of a randomized controlled trial of three treatment approaches to early localised prostate cancer: the MRC PR06 trial. BJU Int. 94, 1400–1401 (2004).
Scandinavian Prostate Cancer Group–SPCG. http://www.spcginfo.com/ (2014).
Donovan, J. et al. Quality improvement report: Improving design and conduct of randomised trials by embedding them in qualitative research: ProtecT (prostate testing for cancer and treatment) study. Commentary: presenting unbiased information to patients can be difficult. BMJ 325, 766–770 (2002).
The ProtecT trial—evaluating the effectiveness of treatments for clinically localised prostate cancer. http://www.nets.nihr.ac.uk/projects/hta/962099 (2012).
Thompson, I. M. Jr & Tangen, C. M. Prostate cancer—uncertainty and a way forward. N. Engl. J. Med. 367, 270–271 (2012).
Wilt, T. J. et al. Systematic review: comparative effectiveness and harms of treatments for clinically localized prostate cancer. Ann. Intern. Med. 148, 435–448 (2008).
Al-Shahi Salman, R. et al. Increasing value and reducing waste in biomedical research regulation and management. Lancet 383, 176–185 (2014).
National Research Council. Initial National Priorities for Comparative Effectiveness Research [online], (2009).
Relton, C., Torgerson, D., O'Cathain, A. & Nicholl, J. Rethinking pragmatic randomised controlled trials: introducing the “cohort multiple randomised controlled trial” design. BMJ 340, c1066 (2010).
Collaborative care in screen-positive elders (CASPER) trial. http://www.controlled-trials.com/ISRCTN02202951 (2012).
Relton, C. et al. South Yorkshire Cohort: a 'cohort trials facility' study of health and weigh—protocol for the recruitment phase. BMC Public Health 11, 640 (2011).
Multifaceted podiatry intervention for fall prevention in patients over 65 years of age. http://www.controlled-trials.com/ISRCTN68240461 (2013).
Comparison of infliximab and ciclosporin in steroid resistant ulcerative colitis: a trial. http://www.controlled-trials.com/ISRCTN22663589 (2012).
Thombs, B. D. et al. New directions for patient-centred care in scleroderma: the Scleroderma Patient-centred Intervention Network (SPIN). Clin. Exp. Rheumatol. 30 (Suppl. 71), S23–S29 (2012).
Verkooijen, H. M., Roes, K. & van Gils, C. H. Cohort multiple randomized controlled trial: a solution for the evaluation of multiple interventions. Ned. Tijdschr. Geneeskd. 157, A5762 (2013).
Altman, D. G. et al. Randomised consent designs in cancer clinical trials. Eur. J. Cancer 31A, 1934–1944 (1995).
Ergina, P. L. et al. Challenges in evaluating surgical innovation. Lancet 374, 1097–1104 (2009).
Vickers, A. J. et al. Feasibility study of a clinically-integrated randomized trial of modifications to radical prostatectomy. Trials 13, 23 (2012).
Current Controlled Trials. www.controlled-trials.com[online], (2011).
Acknowledgements
The Medical Research Council (UK) provided support for travel and accommodation costs to all attendees (ref G1002509). Pelican Cancer Foundation charity provided logistical support in organizing the event. No commercial bodies were involved.
Author information
Authors and Affiliations
Consortia
Contributions
H.U.A. wrote the first draft. All named co-authors provided editorial input to revisions and authorized the final draft. All attendees named in Box 2 were sent the draft manuscript and had input into the drafting and editing process. All attendees gave permission to be named.
Corresponding author
Ethics declarations
Competing interests
H.U.A. has received research funding from the following companies: Advanced Medical Diagnostics, GlaxoSmithKline and Sonacare. M.E. has received research funding from the following companies: Advanced Medical Diagnostics, GlaxoSmithKline, Steba Biotech and Sonacare. M.E. is a paid consultant for GlaxoSmithKline, Steba Biotech and Sonacare. M.E. is a director on the board of and has share options in Nuada Medical. The other authors declare no competing interests.
Supplementary information
Supplementary Table 1
List of Protocol Development Group attendees (DOC 79 kb)
Rights and permissions
About this article
Cite this article
Ahmed, H., Berge, V., Bottomley, D. et al. Can we deliver randomized trials of focal therapy in prostate cancer?. Nat Rev Clin Oncol 11, 482–491 (2014). https://doi.org/10.1038/nrclinonc.2014.44
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2014.44
This article is cited by
-
The Trial within Cohorts (TwiCs) study design in oncology: experience and methodological reflections
BMC Medical Research Methodology (2023)
-
Feasibility of Comparative Health Research Outcome of Novel Surgery in prostate cancer (IP4-CHRONOS): statistical analysis plan for the randomised feasibility phase of the CHRONOS study
Trials (2021)
-
Making a case “against” focal therapy for intermediate-risk prostate cancer
World Journal of Urology (2021)
-
Cryoablation, high-intensity focused ultrasound, irreversible electroporation, and vascular-targeted photodynamic therapy for prostate cancer: a systemic review and meta-analysis
International Journal of Clinical Oncology (2021)
-
Focal therapy compared to radical prostatectomy for non-metastatic prostate cancer: a propensity score-matched study
Prostate Cancer and Prostatic Diseases (2021)