Key Points
-
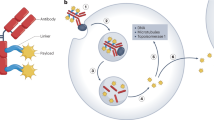
Immunoconjugates represent an efficient strategy to exploit the tumor-targeting properties of monoclonal antibodies and the tumor-killing properties of various types of toxins
-
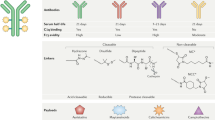
Immunoconjugates are composed of an antibody, a linker and a therapeutic entity
-
Pharmacologics, radioisotopes, and toxins can all be used as therapeutic entity in an immunoconjugate; all three types of therapeutic entities are actively being investigated
-
Future immunoconjugate development will entail the repurposing of existing tumor-targeting antibodies as immunoconjugate delivery vehicles, as well as the design of antibodies with novel tumor-targeting specificities
Abstract
Immunoconjugates are specific, highly effective, minimally toxic anticancer therapies that are beginning to show promise in the clinic. Immunoconjugates consist of three separate components: an antibody that binds to a cancer cell antigen with high specificity, an effector molecule that has a high capacity to kill the cancer cell, and a linker that will ensure the effector does not separate from the antibody during transit and will reliably release the effector to the cancer cell or tumour stroma. The high affinity antibody–antigen interaction allows specific and selective delivery of a range of effectors, including pharmacologic agents, radioisotopes, and toxins, to cancer cells. Some anticancer molecules are not well tolerated when administered systemically owing to unacceptable toxicity to the host. However, this limitation can be overcome through the linking of such cytotoxins to specific antibodies, which mask the toxic effects of the drug until it reaches its target. Conversely, many unconjugated antibodies are highly specific for a cancer target, but have low therapeutic potential and can be repurposed as delivery vehicles for highly potent effectors. In this Review, we summarize the successes and shortcomings of immunoconjugates, and discuss the future potential for the development of these therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Paul Ehrlich—Biographical. Nobelprize.org[online], (2014).
Anderson, C. S., Quinones, R. & Olson, M. R. Determining efficacy and side effects from the concurrent use of chemotherapy and radiation therapy for the management of adult solid tumors [abstract]. J. Clin. Oncol. 32 (Suppl.), e17640 (2014).
Van Cutsem, E. et al. Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer. N. Engl. J. Med. 360, 1408–1417 (2009).
Fuchs, C. S. et al. Randomized, controlled trial of irinotecan plus infusional, bolus, or oral fluoropyrimidines in first-line treatment of metastatic colorectal cancer: results from the BICC-C study. J. Clin. Oncol. 25, 4779–4786 (2007).
Hochster, H. S. et al. Safety and efficacy of oxaliplatin-fluoropyrimidine regimens with or without bevacizumab as first-line treatment of metastatic colorectal cancer (mCRC): results of the TREE-Study. J. Clin. Oncol. 26, 3523–3529 (2008).
Reichert, J. M. et al. Monoclonal antibody successes in the clinic. Nat. Biotechnol. 23, 1073–1078 (2005).
Pro, B. et al. Brentuximab vedotin (SGN-35) in patients with relapsed or refractory systemic anaplastic large-cell lymphoma: results of a phase II study. J. Clin. Oncol. 30, 2190–2196 (2012).
Younes, A. et al. Results of a pivotal phase II study of brentuximab vedotin for patients with relapsed or refractory Hodgkin's lymphoma. J. Clin. Oncol. 30, 2183–2189 (2012).
Verma, S. et al. Trastuzumab emtansine for HER2-positive advanced breast cancer. N. Engl. J. Med. 367, 1783–1791 (2012).
Sievers, E. L. & Senter, P. D. Antibody-drug conjugates in cancer therapy. Annu. Rev. Med. 64, 15–29 (2013).
Teicher, B. A. & Chari, R. V. J. Antibody conjugate therapeutics: challenges and potential. Clin. Cancer Res. 17, 6389–6397 (2011).
Thurber, G. M., Schmidt, M. M. & Wittrup, K. D. Antibody tumor penetration: transport opposed by systemic and antigen-mediated clearance. Adv. Drug Deliv. Rev. 60, 1421–1434 (2008).
Chari, R. V. J. Targeted cancer therapy: conferring specificity to cytotoxic drugs. Acc. Chem. Res. 41, 98–107 (2008).
Kapoor, P. et al. Anti-CD20 monoclonal antibody therapy in multiple myeloma. Br. J. Haematol. 141, 135–148 (2008).
Yarden, Y. The EGFR family and its ligands in human cancer: signaling mechanisms and therapeutic opportunities. Eur. J. Cancer 37, S3–S8 (2001).
Colcher, D. et al. Pharmacokinetics and biodistribution of genetically-engineered antibodies. Q. J. Nucl. Med. 42, 225–241 (1998).
Wells, A. EGF receptor. Int. J. Biochem. Cell Biol. 31, 637–643 (1999).
Colby, D. W. et al. Development of a human light chain variable domain (V(L)) intracellular antibody specific for the amino terminus of huntingtin via yeast surface display. J. Mol. Biol. 342, 901–912 (2004).
Binz, H. K. Amstutz, P. & Pluckthun, A. Engineering novel binding proteins from nonimmunoglobulin domains. Nat. Biotechnol. 23, 1257–1268 (2005).
Cao, Y. et al. Single-chain antibody-based immunotoxins targeting Her2/neu: design optimization and impact of affinity on antitumor efficacy and off-target toxicity. Mol. Cancer Ther. 11, 143–153 (2012).
Ducancel, F. & Muller, B. H. Molecular engineering of antibodies for therapeutic and diagnostic purposes. mAbs 4, 445–457 (2012).
Weiner, L. M. et al. I. A human tumor xenograft model of therapy with a bispecific monoclonal antibody targeting c-erbB-2 and CD16. Cancer Res. 53, 94–110 (1993).
Yokota, T. et al. Microautoradiographic analysis of the normal organ distribution of radioiodinated single-chain Fv and other immunoglobulin forms. Cancer Res. 53, 3776–3783 (1993).
Adams, G. P. et al. Optimization of in vivo tumor targeting in SCID mice with divalent forms of 741F8 anti-c-erbB-2 single-chain Fv: effects of dose escalation and repeated i.v. administration. Cancer Immunol. Immunother. 40, 299–306 (1995).
Adams, G. P. et al. Enhanced tumor specificity of 741F8–1 (sFv')2, an anti-c-erbB-2 single-chain Fv dimer, mediated by stable radioiodine conjugation. J. Nucl. Med. 36, 2276–2281 (1995).
Saga, T. et al. Targeting cancer micrometastases with monoclonal antibodies: a binding-site barrier. Proc. Natl Acad. Sci. USA 92, 8999–9003 (1995).
Adams, G. P. et al. Increased affinity leads to improved selective tumor delivery of single-chain Fv antibodies. Cancer Res. 58, 485–490 (1998).
Zhang, Y. & Pastan, I. High shed antigen levels within tumours: an additional barrier to immunoconjugate therapy. Clin. Cancer Res. 14, 7981–7986 (2008).
Adams, G. P. et al. High affinity restricts the localization and tumor penetration of single-chain fv antibody molecules. Cancer Res. 61, 4750–4755 (2001).
Ackerman, M. E., Pawlowski, D. & Wittrup, K. D. Effect of antigen turnover rate and expression level on antibody penetration into tumor spheroids. Mol. Cancer Ther. 7, 2233–2240 (2008).
Mayer, A. et al. Radioimmunoguided surgery in colorectal cancer using a genetically engineered anti-CEA single-chain Fv antibody. Clin. Cancer Res. 5, 1711–1719 (2000).
Singh, R., Zhang, Y., Pastan, I. & Kreitman, R. J. Synergistic antitumor activity of anti-CD25 recombinant immunotoxin LMB-2 with chemotherapy. Clin. Cancer Res. 18, 152–160 (2012).
Pasquetto, M. V., Vecchia, L., Covini, D., Digilio, R. & Scotti, C. Targeted drug delivery using immunoconjugates: principles and applications. J. Immunother. 34, 611–628 (2011).
Marks, J. D. et al. Human antibody fragments specific for human blood group antigens from a phage display library. Biotechnology 11, 1145–1149 (1993).
Winter, G., Griffiths, A. D., Hawkins, R. E. & Hoogenboom, H. R. Making antibodies by phage display technology. Annu. Rev. Immunol. 12, 433–455 (1994).
Griffiths, A. D. & Duncan, A. R. Strategies for selection of antibodies by phage display. Curr. Opin. Biotechnol. 1, 102–108 (1998).
Feldhaus, M. J. & Siegel, R. W. Yeast display of antibody fragments: a discovery and characterization platform. J. Immunol. Methods 290, 69–80 (2004).
Graff, C. P., Chester, K., Begent, R. & Wittrop, K. D. Directed evolution of an anti-carcinoembrynoic antigen sc Fv with a 4-day monovalent dissociation half-time at 37°C. Protein Eng. Des. Sel. 17, 293–304 (2004).
Dubreuil, O. et al. Fine tuning of specificity of an anti-progesterone antibody by first and second sphere residue engineering. J. Biol. Chem. 280, 24880–24887 (2005).
Clark, L. A. et al. Affinity enhancement of an in vivo matured therapeutic antibody using structure-based computational design. Protein Sci. 15, 949–960 (2006).
Lippow, S. M., Wittrup, K. D. & Tidor, B. Computational design of antibody-affinity improvement beyond in vivo maturation. Nat. Biotechnol. 25, 1171–1176 (2007).
Ward, E. S., Zhou, J., Ghetie, V. & Ober, R. J. Evidence to support the cellular mechanism involved in serum IgG homeostasis in humans. Int. Immunol. 15, 187–195 (2003).
Covell, D. G. et al. Pharmacokinetics of monoclonal immunoglobulin G1, F(ab')2, and Fab' in mice. Cancer Res. 46, 3969–3978 (1986).
Adams, G. P. et al. Prolonged in vivo tumour retention of a human diabody targeting the extracellular domain of human HER2/neu. Br. J. Cancer 77, 1405–1412 (1998).
Wu, A. M. Engineered antibodies for molecular imaging of cancer. Methods 65, 139–147 (2014).
Olafsen, T., Sirk, S. J., Olma, S., Shen, C. K. & Wu, A. M. ImmunoPET using engineered antibody fragments: fluorine-18 labeled diabodies for same-day imaging. Tumour Biol. 33, 669–677 (2012).
Sanderson, R. J. In vivo drug-linker stability of an anti-CD30 dipeptide-liked auristatin immunoconjugate. Clin. Cancer Res. 11, 843–852 (2005).
Ducry, L. & Stump, B. Antibody-drug congjugates: linking cytotoxic payloads to monoclonal antibodies Bioconjug. Chem. 21, 5–13 (2010).
Junutula, J. R. et al. Site-specific conjugation of a cytotoxic drug to an antibody improves the therapeutic index. Nat. Biotechnol. 26, 925–932 (2008).
Koblinski, J. E., Ahran, M. & Sloane, B. F. Unraveling the role of proteases in cancer. Clin. Chim. Acta 291, 113–135 (2000).
Sjogren, H. O. et al. Human carcinomas in athymic mice and rats and syngeneic rat carcinomas in immunocompetant rates. Cancer Res. 57, 4530–4536 (1997).
Saleh, M. N. et al. Phase I trial of the anti-lewis Y drug immunoconjugate BR96-doxorubicin in patients with lewis Y-expressing epithelial tumors. J. Clin. Oncol. 18, 2282–2292 (2000).
Wong, J. Y. C. et al. Pilot trial evaluating an 123I-labeled 80-kilodalton engineered anticarcinoembryonic antigen antibody fragment in patients with colorectal cancer. Clin. Cancer Res. 10, 5014–5021 (2004).
Kreitman, R. J. et al. Phase I trial of anti-CD22 recombinant immunotoxin moxetumomab pasudotox in patients with hairy cell leukemia. J. Clin. Oncol. 30, 1822–1828 (2012).
Hassan, R. et al. Major cancer regressions in mesothelioma after treatment with an anti-mesothelin immunotoxin and immune suppression. Sci. Transl. Med. 5, 208ra147 (2013).
Dosio, F., Brusa, P. & Cattel, L. Immunotoxins and anticancer drug conjugate assemblies: the role of the linkage between components. Toxins 3, 848–883 (2011).
Sutherland, M. S. et al. Lysosomal trafficking and cysteine protease metabolism confer target-specific cytotoxicity by peptide-linked anti-CD30-auristatin conjugates. J. Biol. Chem. 281, 10540–10547 (2006).
US Department of Health and Human Services. Brentuximab vedotin information. US Food and Drug Administration [online], (2012).
Ujjani, C. & Cheson B. D. The current status and future impact of targeted therapies in non-Hodgkin lymphoma. Expert Rev. Hematol. 6, 191–203 (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Ansell, S. M. et al. Phase I/II study of an anti-CD30 monoclonal antibody in Hodgkin's lymphoma and anaplastic large-cell lymphoma. J. Clin. Oncol. 25, 2764–2769 (2007).
LoRusso, P. M., Weiss, D., Guardino, E., Grish, S. & Sliwkowski, M. X. Trastuzumab emtansine: a unique antibody-drug conjugate in development for human epidermal growth factor receptor 2-positive cancer. Clin. Cancer Res. 17, 6437–6447 (2011).
Lewis Phillips, G. D. et al. Targeting HER2-positive breast cancer with trastuzumab-DM1, an antibody-cytotoxic drug conjugate. Cancer Res. 68, 9280–9290 (2008).
Barok, M., Tanner, M., Koninki, K. & Isola, J. Trasuzumab-DM1 causes tumour growth inhibition by mitotic catastrophe in trastuzumab-resistant breast cancer cells in vivo. Breast Cancer Res. 13, R46 (2011).
Burris, H. A. et al. Phase II study of the antibody drug conjugate trastuzumab-DM1 for the treatment of human epidermal growth factor receptor 2 (HER2)-positive breast cancer after prior HER2-directed therapy. J. Clin. Oncol. 29, 398–405 (2011).
Krop, I. E. et al. A phase II study of trastuzumab emtansine in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer who were previously treated with trasuzumab, lapatinib, an anthracycline, a taxane, and capecitabine. J. Clin. Oncol. 30, 3234–3241 (2012).
US Department of Health and Human Services. Ado-trastuzumab emtansine. US Food and Drug Administration [online], (2013).
Bross, P. F. et al. Approval summary: gemtuzumab ozogamicin in relapsed acute myeloid leukemia. Clin. Cancer Res. 7, 1490–1496 (2001).
Ricart, A. D. Antibody-drug conjugates of calicheamicin derivative: gemtuzumab ozogamicin and inotuzumab ozogamicin. Clin. Cancer Res. 17, 6417–6427 (2011).
Petersdorf, S. et al. Preliminary results of Southwest Oncology Group Study S0106: an international intergroup phase 3 randomized trial comparing the addition of gemtuzumab ozogamicin to standard induction therapy versus standard induction therapy followed by a second randomization to post-consolidation gemtuzumab ozogamicin versus no additional therapy for previously untreated acute myeloid leukemia [abstract]. Blood 114, a790 (2009).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Ribrag, V. et al. A dose-escalation study of SAR3419, an anti-CD19 antibody maytansinoid conjugate, administered by intravenous infusion once weekly in patients with relapsed/refractory B-cell non-Hodgkin lymphoma. Clin. Cancer Res. 20, 213–220 (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Maric, G., Rose, A. A., Annis, M. G. & Siegel, P. M. Glycoprotein non-metastatic b (GPNMB): A metastatic mediator and emerging therapeutic target in cancer. Onco. Targets Ther. 6, 839–852 (2013).
Celdex Therapeutics. Final Data from Celldex Therapeutic's CDX-011 phase 2 study in metastatic breast cancer supports overall survival benefit in patients with high GPNMB expression [online], (2012).
Naumovski, L. & Junutula, J. R. Glembatumumab vedotin, a conjugate of an anti-glycoprotein non-metastatic melanoma protein B mAb and monomethyl auristatin E for the treatment of melanoma and breast cancer. Curr. Opin. Mol. Ther. 12, 248–257 (2010).
Hamid, O. et al. Frequent dosing and GPNMB expression with CDX-011, an antibody-drug conjugate, in patients with advanced melanoma. J. Clin. Oncol. 28, 15s (2010).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Woll, P. Abstract B237: Clinical experience of IMGN901 (BB-10901, huN901-DM1) in patients with Merkel cell carcinoma (MCC). Mol. Cancer Ther. 8, B237 (2009).
Gan, H. K. et al. A phase I study evaluating ABT-414 in combination with temozolamide for subjects with recurrent or unresectable glioblastoma. J. Clin. Oncol. 32, 5s (2014).
Burris, H. A. et al. A phase I study of DNIB0600A, an antibody-drug conjugate targeting NaPi2b, in patients with non-small cell lung cancer or platinum-resistant ovarian cancer. J. Clin. Oncol. 32, 5s (2014).
Marx, J. L. in A Revolulation in Biotechnology (ed. Marx, J. L.) 145–158 (The Press Syndicate of the University of Cambridge, 1989).
Witzig, T. E. et al. Treatment with ibritumomab tiuxetan radioimmunotherapy in patients with rituximab-refractory follicular non-Hodgkin's lymphoma. J. Clin. Oncol. 20, 3262–3269 (2002).
US Department of Health and Human Services. Ibritumomab tiuxetan product approval information. US Food and Drug Administration [online], (2009).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Kaminski, M. S. et al. Pivotal study of iodine I 131 tositumomab for chemotherapy-refractory low-grade or transformed low-grade B-cell non-Hodgkin's lymphomas. J. Clin. Oncol. 19, 3918–3928 (2001).
Timmerman, L. Why good drugs sometimes fail: the Bexxar story. Xconomy [online], (2013).
Steiner, M. & Neri, D. Antibody-radionucleotide conjugates for cancer therapy: historical considerations and new trends. Clin. Cancer Res. 17, 6406–6416 (2011).
Song, H. & Sgouros, G. Radioimmunotherapy of solid tumors: searching for the right target. Current Drug Deliv. 8, 26–44 (2011).
Sharkey, R. M., Karacay, H., Govindan, S. V. & Goldenberg, D. M. Combination radioimmunotherapy and chemoimmunotherapy involving different or the same targets improves therapy of human pancreatic carcinoma xenograft models. Mol. Cancer Ther. 10, 1072–1081 (2011).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Press, O. W. et al. A comparative evaluation of conventional and pretargeted radioimmunotherapy of CD-20 expressing lymphoma xenografts. Blood 98, 2535–2543 (2001).
Knox, S. J. et al. Phase II trial of Yttrium-90-DOTA-biotin pretargeted by NR-LU-10 antibody/streptavidin in patients with metastatic colorectal cancer. Clin. Cancer Res. 6, 406–414 (2000).
Park, S. I. et al. Conventional and pretargeted radioimmunotherapy using bismuth-213 to target and treat non-Hodgkin lymphomas expressing CD20: a preclinical model toward optimal consolidation therapy to eradicate minimal residual disease. Blood 116, 4231–4239 (2010).
Baidoo, K. E., Yong, K. & Brechbiel, M. W. Molecular pathoways: targeted α-particle radiation therapy. Clin. Cancer Res. 19, 530–537 (2013).
Kim, Y. S. & Brechbiel, M. W. An overview of targeted alpha therapy. Tumour Biol. 33, 573–590 (2012).
Gould, B. J. et al. Phase I study of an anti-breast cancer immunotoxin by continuous infusion: report of a targeted toxic effect not predicted by animal studies. J. Natl Cancer Inst. 81, 775–781 (1989).
Weiner, L. M. et al. Phase I evaluation of an anti-breast cancer monoclonal antibody 260F9-recombinant ricin A chain immunoconjugate. Cancer Res. 49, 4062–4067, (1989).
Fitzgerald, D. J., Wayne, A. S., Kreitman, R. J. & Pastan, I. Treatment of hematologic malignancies with immunotoxins and antibody-drug conjugates. Cancer Res. 71, 6300–6309 (2011).
Chaudhary, V. K. et al. A recombinant immunotoxin consisting of two antibody variable domains fused to Pseudomonas exotoxin. Nature 339, 394–397 (1989).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Bogner, C. et al. Immunotoxin BL22 induces apoptosis in mantle cell lymphoma (MCL) cells dependent on Bcl-2 expression. Br. J. Haematol. 148, 99–109 (2009).
Traini, R. et al. ABT-737 overcomes resistance to immunotoxin-mediated apoptosis and enhances the delivery of Pseudomonas exotoxin-based proteins to the cell cytosol. Mol. Cancer Ther. 9, 2007–2015 (2010).
Amundson, S. A. et al. An informatics approach identifying markers of chemosensitivity in human cancer cell lines. Cancer Res. 60, 6101–6110 (2000).
Mattoo, A. R., Pastan, I. & Fitzgerald, D. Combination treatmens with the PKC inhibitor, enzastaurin, enhance the cytotoxicity of the anti-mesothelin immunotoxin, SS1P. PLoS ONE 8, e75576 (2013).
Liu, W. et al. Recombinant immunotoxin engineered for low immunogenicity and antigenicity by identifying and silencing human B-cell epitopes. Proc. Natl Acad. Sci. USA 109, 11782–11787 (2012).
Onda, M. et al. An immunotoxin with greatly reduced immunogenicity by identification and removal of B cell epitopes. Proc. Natl Acad. Sci. USA 105, 11311–11316 (2008).
Pai, L. H. et al. Inhibition of antibody response to Pseudomonas exotoxin by 15-deoxyspergualin in mice. Cancer Res. 50, 7750–7753 (1990).
Gelber, E. E. et al. Effect of immunosuppressive agents on the immunogenicity and efficacy of an immunotoxin in mice. Clin. Cancer Res. 4, 1297–1304 (1998).
Hassan, R., Williams-Gould, J., Watson, T., Pai-Scherf, L. & Pastan, I. Pretreatment with rituximab does not inhibit the human immune response against the immunogenic protein LMB-1. Clin. Cancer Res. 10, 16–18 (2004).
Mazor, R. et al. Identification and elimination of an immunodominant T-cell epitope in recombinant immunotoxins based on Pseudomonas exotoxin A. Proc. Natl Acad. Sci. USA 109, E3597–E3603 (2012).
Basu, A. et al. Structure-function engineering of interferon-beta-1b for improving stability, solubility, potency, immunogenicity, and pharmacokinetic properties by site-selective mono-PEGylation. Bioconjugate Chem. 17, 618–630 (2006).
Kreitman, R. J. Immunotoxins for targeted cancer therapy. AAPS J. 8, E532–E551 (2006)
Becerril, B., Poul, M. A. & Marks, J. D. Towards selection of internalizing antibodies from phage libraries. Biochem. Biophys. Res. Commun. 255, 386–393 (1999).
Nielsen, U. B. et al. Therapeutic efficacy of anti-ErbB2 immunoliposomes targeted by phage antibody selected for cellular endocytosis. Biochim. Biophys. Acta 1591, 109–118 (2002).
Noble, C. O. et al. Characterization of highly stable liposomal and immunoliposomal formulations of vincristine and vinblastine. Cancer Chemother. Pharmacol. 64, 741–751 (2009).
El-Bayoumi, T. A. & Tochilin, V. P. Tumor-targeted nanomedicines: enhanced antitumor efficacy in vivo of doxorubicin-loaded, long-circulating liposomes modified with cancer-specific monoclonal antibody. Clin. Cancer Res. 15, 1973–1980 (2009).
Xu, L. et al. Systemic tumor-targeted gene delivery by anti-transferrin receptor scFv-immunoliposomes. Mol. Cancer Ther. 5, 337–346 (2002).
Mamot, C. Tolerability, safety, pharmacokinetics, and efficacy of doxorubicin-loaded anti-EGFR immunoliposomes in advanced solid tumours: a phase 1 dose-escalation study. Lancet Oncol. 13, 1234–1241 (2012).
Senzer, N. et al. Phase I study of a systemically delivered p53 nanoparticle in advanced solid tumors. Mol. Ther. 21, 1096–1103 (2013).
Kelly, E. J., Hadac, E. M., Greiner, S. & Russell, S. J. Engineering microRNA responsiveness to decrease virus pathogenicity. Nat. Med. 14, 1278–1283 (2008).
Galanis, E. et al. Phase I trial of intraperitoneal administration of an oncolytic measles virus strain engineered to express carcinoembryonic antigen for recurrent ovarian cancer. Cancer Res. 70, 875–882 (2010).
Liu, X. et al. Enhanced pancreatic cancer gene therapy by combination of adenoviral vector expressing c-erb-B2-targeted immunotoxin with a replication-competent adenovirus or etoposide. Hum. Gene Ther. 21, 157–170 (2010).
Sharkey, R. M. & Goldenberg, D. M. Targeted therapy of cancer: new prospects for antibodies and immunoconjugates. CA Cancer J. Clin. 56, 226–243 (2006).
Fournier, P. & Schirrmacher, V. Bispecific antibodies and trispecific immunocytokines for targeting the immune system against cancer. BioDrugs, 27, 35–53 (2013).
List, T. & Neri, D. Immunocytokines: a review of molecules in clinical development for cancer therapy. Clin. Pharmacol. 5, 29–45 (2013).
Pasche, N. & Neri, D. Immunocytokines: a novel class of potent armed antibodies. Drug Discov. Today 17, 583–590 (2012).
Carnemolla, B. et al. Enhancement of the antitumor properties of interleukin-2 by its targeted delivery to the tumor blood vessel extracellular matrix. Blood 99, 1659–1665 (2002).
Borsi, L. et al. Selective targeted delivery of TNF-alpha to tumor blood vessels. Blood 102, 4384–4392 (2003).
Halin, C. et al. Enhancement of the antitumor activity of interleukin-12 by targeted delivery to neovasculature. Nat. Biotechnol. 20, 264–269 (2002).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Vogelstein, B., Papadopoulos, N., Velculescu, V. E., Zhou, S., Diaz, L. A. & Kinzler, K. W. Cancer genome landscapes. Science 339, 1546–1558 (2013).
Robinson, M. K. et al. Quantitative immune-positron emission tomography imaging of HER2-positive tumor xenografts with an iodine-124 labeled anti-HER2 diabody. Cancer Res. 65, 1471–1478 (2005).
Rudnick, S. I. et al. Influsence of affinity and antigen internalization on the uptake and penetration of anti-HER2 antibodies in solid tumors. Cancer Res. 71, 2250–2259 (2011).
Schlom, J. Therapeutic cancer vaccines: current status and moving forward. J. Natl Cancer Inst. 104, 599–613 (2012).
Disis, M. L. et al. Concurrent trastuzumab and HER2/neu-specific vaccination in patients with metastatic breast cancer. J. Clin. Oncol. 27, 4685–4692 (2009).
Acknowledgements
L.M.W. is supported by National Institute of Health Awards CA51880 and CA50633.
Author information
Authors and Affiliations
Contributions
B.G.S., D.A. and L.M.W. researched data for this article, reviewed and edited the manuscript before submission, provided substantial contribution to discussion of content and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
L.M.W. is an advisor and a consultant for Abbvie, Celldex Pharmaceuticals, Jounce Pharmaceuticals, and Merrimack Pharmaceuticals; he has stock options in Celldex Pharmaceuticals, Jounce Pharmaceuticals and Merrimack Pharmaceuticals and his research has been funded by Symphogen AG. The other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Smaglo, B., Aldeghaither, D. & Weiner, L. The development of immunoconjugates for targeted cancer therapy. Nat Rev Clin Oncol 11, 637–648 (2014). https://doi.org/10.1038/nrclinonc.2014.159
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2014.159