Key Points
-
Many new anticancer treatments are molecular targeted agents and only benefit a subgroup of patients within a histologically defined cancer
-
Heterogeneity of treatment effects requires development of biomarkers to identify patients who could benefit from new treatments
-
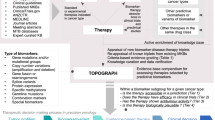
Phase III trial designs for evaluation of targeted treatments use biomarker-driven enrichment strategies that range from limiting evaluation to the biomarker-positive subgroup to sequential testing of biomarker-positive, biomarker-negative and overall populations
-
To provide compelling evidence for informing clinical practice, the choice of an appropriate phase III trial design should be guided by the strength of the biomarker's credentials
Abstract
The use of biomarkers to identify patients who can benefit from treatment with a specific anticancer agent has the potential to both improve patient care and accelerate drug development. The development of targeted agents and their accompanying biomarkers frequently occurs contemporaneously, and confidence in a putative biomarker's performance might, therefore, be insufficient to restrict the definitive testing of a new agent to the subgroup of biomarker-positive patients. This Review considers which clinical trial designs and analysis strategies are appropriate for use in phase III, biomarker-driven, randomized clinical trials, on the basis of pre-existing evidence that the biomarker can successfully identify patients who will respond to the treatment in question. The types of interim monitoring that are appropriate for these trials are also discussed. In addition, enrichment strategies based on the use of prognostic biomarkers to separate a population into subgroups with better and worse outcomes, regardless of treatment, are described. Finally, the possibility of formally using a biomarker during phase II drug development, to select what type of biomarker-driven strategy should be used in the phase III trial, is discussed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
U.S. Department of Health and Human Services. Enrichment strategies for clinical trials to support approval of human drugs and biological products [online], (2012).
Temple, R. Enrichment of clinical study populations. Clin. Pharmacol. Ther. 88, 774–778 (2010).
Van Cutsem, E. et al. Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer. N. Engl. J. Med. 360, 1408–1417 (2009).
Jimeno, A., Messersmith, W. A., Hirsch, F. R., Franklin, W. A. & Eckhardt, S. G. KRAS mutations and sensitivity to epidermal growth factor receptor inhibitors in colorectal cancer: practical application of patient selection. J. Clin. Oncol. 27, 1130–1136 (2009).
Van Custem, E. et al. Cetuximab plus irinotecan, fluorouracil, and leucovorin as first-line treatment for metastatic colorectal cancer: updated analysis of overall survival according to tumour KRAS and BRAF mutation status. J. Clin. Oncol. 29, 2011–2019 (2011).
Leaf, C. Do clinical trials work? New York Times [online], (13 July 2013).
Williams, P. M., Lively, T. G., Jessup, J. M. & Conley, B. A. Bridging the gap: moving predictive and prognostic assays from research to clinical use. Clin. Cancer Res. 18, 1531–1539 (2012).
Simon, R. M., Paik, S. & Hayes, D. F. Use of archived specimens in evaluation of prognostic and predictive biomarkers. J. Natl Cancer. Inst. 101, 1446–1452 (2009).
Polley, M. Y. et al. Statistical and practical considerations for clinical evaluation of predictive biomarkers. J. Natl Cancer Inst. http://dx.doi.org/10.1093/jnci/djt282.
Simon, R. & Maitournam, A. Evaluating the efficiency of targeted designs for randomized clinical trials. Clin. Cancer Res. 10, 6759–6763 (2004).
Chapman, P. B. et al. Improved survival with vemurafenib in melanoma with BRAFV600E mutation. N. Engl. J. Med. 364, 2507–16 (2011).
Romond, E. H. et al. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N. Engl. J. Med. 353, 1673–1684 (2005).
Mandrekar, S. J. & Sargent, D. J. Clinical trial designs for predictive biomarker validation: theoretical considerations and practical challenges. J. Clin. Oncol. 27, 4027–4034 (2009).
Paik, S., Kim, C. & Wolmark, N. HER2 status and benefit from adjuvant trastuzumab in breast cancer. N. Engl. J. Med. 358, 1409–1411 (2008).
Perez, E. A. et al. HER2 and chromosome 17 effect on patient outcome in the N9831 adjuvant trastuzumab trial. J. Clin. Oncol. 28, 4307–4315 (2010).
Freidlin, B., McShane, L. M. & Korn, E. L. Randomized clinical trials with biomarkers: design issues. J. Natl Cancer Inst. 102, 152–160 (2010).
Douillard, J. Y. et al. Randomized, phase III trial of panitumumab with infusional fluorouracil, leucovorin, and oxaliplatin (FOLFOX4) versus FOLFOX4 alone as first-line treatment in patients with previously untreated metastatic colorectal cancer: the PRIME study. J. Clin. Oncol. 28, 4697–4705 (2010).
Johnston, S. et al. Lapatinib combined with letrozole versus letrozole and placebo as first-line therapy for postmenopausal hormone receptor-positive metastatic breast cancer. J. Clin. Oncol. 27, 5538–5546 (2009).
Cappuzzo, F. et al. Erlotinib as maintenance treatment in advanced non-small-cell lung cancer: a multicentre, randomised, placebo-controlled phase 3 study. Lancet Oncol. 11, 521–529 (2010).
Rothmann, M. D., Zhang, J. J., Lu, L. & Fleming, T. R. Testing in a prespecified subgroup and the intent-to-treat population. Drug Inf. J. 46, 175–179 (2012).
Karuri, S. W. & Simon, R. A two-stage Bayesian design for co-development of new drugs and companion diagnostics. Stat. Med. 31, 901–914 (2012).
Freidlin, B., Korn, E. L. & Gray, R. Marker sequential test (MaST) design. Clin. Trials. http://dx.doi.org/10.1177/1740774513503739.
Freidlin, B., Sun, Z., Gray, R. & Korn, E. L. Phase III clinical trials that integrate treatment and biomarker evaluation. J. Clin. Oncol. 31, 3158–3161 (2013).
Simon, R. The use of genomics in clinical trial design. Clin. Cancer Res. 14, 5984–5993 (2008).
Jiang, W., Freidlin, B. & Simon, R. Biomarker-adaptive threshold design: a procedure for evaluating treatment with possible biomarker-defined subset effect. J. Natl Cancer Inst. 99, 1036–1043 (2007).
Freidlin, B. & Simon, R. Adaptive signature design: an adaptive clinical trial design for generating and prospectively testing a gene expression signature for sensitive patients. Clin. Cancer Res. 11, 7872–7878 (2005).
McShane, L. M. et al. Criteria for the use of omics-based predictors in clinical trials: explanation and elaborations. BMC Med. 11, 220 (2013).
Green, S., Benedetti, J., Smith, A. & Crowley, J. Clinical Trials in Oncology 2nd edn 97–122 (Chapman & Hall/CRC Press, 2002).
Korn, E. L. & Freidlin, B. in Oncology Clinical Trials Ch. 19 (eds Kelly, W. K. & Halabi, S.) 163–177 (Demos Medical Publishing, 2010).
Liu, A., Liu, C., Li, Q., Yu, K. F. & Yuan, V. W. A threshold sample-enrichment approach in a clinical trial with heterogeneous subpopulations. Clin. Trials 7, 537–545 (2010).
Rubinstein, L. V. et al. Design issues of randomized phase II trials and a proposal for phase II screening trials. J. Clin. Oncol. 23, 7199–7206 (2005).
Amatu, A. et al. Promoter CpG island hypermethylation of the DNA repair enzyme MGMT predicts clinical response to dacarbazine in a phase II study for metastatic colorectal cancer. Clin. Cancer Res. 19, 2265–2272 (2013).
Edelman, M. J. et al. Randomized phase II study of ixabepilone or paclitaxel plus carboplatin in patients with non-small-cell lung cancer prospectively stratified by β-3 tubulin status. J. Clin. Oncol. 31, 1990–1996 (2013).
Flaherty, K. T. et al. Inhibition of mutated, activated BRAF in metastatic melanoma. N. Engl. J. Med. 363, 809–819 (2010).
Spigel, D. R. et al. Treatment rationale study design for the MetLung trial: a randomized, double-blind phase III study of onartuzumab (MetMAb) in combination with erlotinib versus erlotinib alone in patients who have received standard chemotherapy for stage IIIB or IV Met-positive non-small-cell lung cancer. Clin. Lung Cancer 13, 500–504 (2012).
Jones, C. L. & Holmgren, E. An adaptive Simon two-stage design for phase 2 studies of targeted therapies. Contemp. Clin. Trials 28, 654–661 (2007).
Freidlin, B., McShane, L. M., Polley, M. Y. & Korn, E. L. Randomized phase II trial designs with biomarkers. J. Clin. Oncol. 30, 3304–3309 (2012).
Hayes, D. F. Targeting adjuvant chemotherapy: a good idea that needs to be proven! J. Clin. Oncol. 30, 1264–1267 (2012).
Paik, S. et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N. Engl. J. Med. 351, 2817–2826 (2004).
Paik, S. et al. Gene expression and benefit of chemotherapy in women with node-negative, oestrogen receptor-positive breast cancer. J. Clin. Oncol. 24, 3726–3734 (2006).
Sparano, J. A. TAILORx: trial assigning individualized options for treatment (Rx). Clin. Breast Cancer 7, 347–350 (2006).
Ramsey, S. D. et al. Integrating comparative effectiveness design elements and endpoints into a phase III, randomized clinical trial (SWOG S1007) evaluating Oncotype DX-guided management for women with breast cancer involving lymph nodes. Contemp. Clin. Trials 34, 1–9 (2013).
Rosell, R. et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 13, 239–246 (2012).
Baselga, J. et al. Pertuzumab plus trastuzumab plus docetaxel for metastatic breast cancer. N. Engl. J. Med. 366, 109–119 (2012).
Shaw, A. T. et al. Crizotinib versus chemotherapy in advanced ALK-positive lung cancer. N. Engl. J. Med. 368, 2385–2394 (2013).
Sequist, L. V. et al. Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations. J. Clin. Oncol. 31, 3327–3334 (2013).
Peeters, M. et al. Randomized phase III study of panitumumab with fluorouracil, leucovorin, and irinotecan (FOLFIRI) compared with FOLFIRI alone as second-line treatment in patients with metastatic colorectal cancer. J. Clin. Oncol. 28, 4706–4713 (2010).
Sargent, D. J. & Mandrekar, S. J. Statistical issues in the validation of prognostic, predictive, and surrogate biomarkers. Clin. Trials 10, 647–652 (2013).
Herbst, R. S. et al. Vandetanib plus docetaxel versus docetaxel as second-line treatment for patients with advanced non-small-cell lung cancer (ZODIAC): a double-blind, randomized, phase 3 trial. Lancet Oncol. 11, 619–626 (2010).
Scagliotti, G. V. et al. International, randomized, placebo-controlled, double-blind phase III study of motesanib plus carboplatin/paclitaxel in patients with advanced nonsquamous non-small-cell lung cancer: MONET1. J. Clin. Oncol. 30, 2829–2836 (2012).
Redman, M. W. et al. Design of a phase III clinical trial with prospective biomarker validation: SWOG S0819. Clin. Cancer Res. 18, 4004–4012 (2012).
Boyer, M. et al. Rationale and study design for ARCHER: a randomized, double-blind, phase III study of dacomitinib versus erlotinib for advanced non-small-cell lung cancer. Clin. Invest. 3, 29–35 (2013).
Miles, D. et al. MERiDiAN: A phase III, randomized, double-blind study of efficacy, safety, and associated biomarker of bevacizumab plus paclitaxel compared with paclitaxel plus placebo, as first-line therapy of patients with HER2-negative breast cancer [abstract]. J. Clin. Oncol. 31 (Suppl.), TPS1142 (2013).
Author information
Authors and Affiliations
Contributions
Both authors contributed to all aspects of this article, including researching the data for the article, discussions of its content, writing the article, and review and editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Freidlin, B., Korn, E. Biomarker enrichment strategies: matching trial design to biomarker credentials. Nat Rev Clin Oncol 11, 81–90 (2014). https://doi.org/10.1038/nrclinonc.2013.218
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2013.218
This article is cited by
-
New insights into adaptive enrichment designs
Statistical Papers (2023)
-
The Future Glioblastoma Clinical Trials Landscape: Early Phase 0, Window of Opportunity, and Adaptive Phase I–III Studies
Current Oncology Reports (2023)
-
Cardiac-sparing radiotherapy for locally advanced non-small cell lung cancer
Radiation Oncology (2021)
-
IL-8 and cancer prognosis on immunotherapy
Nature Medicine (2020)