Abstract
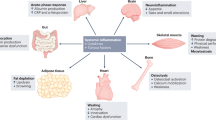
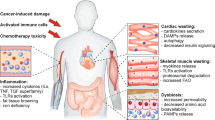
Cancer cachexia is a metabolic syndrome that can be present even in the absence of weight loss ('precachexia'). Cachexia is often compounded by pre-existing muscle loss, and is exacerbated by cancer therapy. Furthermore, cachexia is frequently obscured by obesity, leading to under-diagnosis and excess mortality. Muscle wasting (the signal event in cachexia) is associated not only with reduced quality of life, but also markedly increased toxicity from chemotherapy. Many of the primary events driving cachexia are likely mediated via the central nervous system and include inflammation-related anorexia and hypoanabolism or hypercatabolism. Treatment of cachexia should be initiated early. In addition to active management of secondary causes of anorexia (such as pain and nausea), therapy should target reduced food intake (nutritional support), inflammation-related metabolic change (anti-inflammatory drugs or nutrients) and reduced physical activity (resistance exercise). Advances in the understanding of the molecular biology of the brain, immune system and skeletal muscle have provided novel targets for the treatment of cachexia. The combination of therapies into a standard multimodal package coupled with the development of novel therapeutics promises a new era in supportive oncology whereby quality of life and tolerance to cancer therapy could be improved considerably.
Key Points
-
Cancer cachexia remains an important unmet medical need that affects patients' quality of life and treatment outcomes
-
Cachexia can be missed in an ever-increasingly obese population
-
Therapy should start early and can run in parallel with antineoplastic therapy
-
There is an urgent need to establish best supportive multimodal care for cachexia: beyond good clinical or oncological care, the treatable defects in dietary intake, physical activity and systemic inflammation should be addressed
-
Patients with cachexia should be actively considered for entry into clinical trials
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Fearon, K. et al. Definition and classification of cancer cachexia: an international consensus. Lancet Oncol. 12, 489–495 (2011).
Arends, J. et al. ESPEN Guidelines on Enteral Nutrition: non-surgical oncology. Clin. Nutr. 25, 245–259 (2006).
Kubrak, C. et al. Nutrition impact symptoms: key determinants of reduced dietary intake, weight loss, and reduced functional capacity of patients with head and neck cancer before treatment. Head Neck 32, 290–300 (2010).
Simons, J. P. et al. Effects of medroxyprogesterone acetate on food intake, body composition, and resting energy expenditure in patients with advanced, nonhormone-sensitive cancer: a randomized, placebo-controlled trial. Cancer 82, 553–560 (1998).
Nixon, D. W. et al. Hyperalimentation of the cancer patient with protein-calorie undernutrition. Cancer Res. 41, 2038–2045 (1981).
Dodson, S. et al. Muscle wasting in cancer cachexia: clinical implications, diagnosis, and emerging treatment strategies. Annu. Rev. Med. 62, 265–279 (2011).
van Wetering, C. R. et al. Efficacy and costs of nutritional rehabilitation in muscle-wasted patients with chronic obstructive pulmonary disease in a community-based setting: a prespecified subgroup analysis of the INTERCOM trial. J. Am. Med. Dir. Assoc. 11, 179–187 (2010).
Barratt, S. M., Smith, R. C., Kee, A. J., Mather, L. E. & Cousins, M. J. Multimodal analgesia and intravenous nutrition preserves total body protein following major upper gastrointestinal surgery. Reg. Anesth. Pain Med. 27, 15–22 (2002).
Awad, S. et al. Marked changes in body composition following neoadjuvant chemotherapy for oesophagogastric cancer. Clin. Nutr. 31, 74–77 (2012).
Smith, M. R. et al. Changes in body composition during androgen deprivation therapy for prostate cancer. J. Clin. Endocrinol. Metab. 87, 599–603 (2002).
Antoun, S. et al. Association of skeletal muscle wasting with treatment with sorafenib in patients with advanced renal cell carcinoma: results from a placebo-controlled study. J. Clin. Oncol. 28, 1054–1060 (2010).
Helbekkmo, N. et al. Chemotherapy and quality of life in NSCLC PS 2 patients. Acta Oncol. 48, 1019–1025 (2009).
Kalantar-Zadeh, K. et al. Risk factor paradox in wasting diseases. Curr. Opin. Clin. Nutr. Metab. Care 10, 433–442 (2007).
McAuley, P. A. & Blair, S. N. Obesity paradoxes. J. Sports Sci. 29, 773–782 (2011).
Tan, B. H., Birdsell, L. A., Martin, L., Baracos, V. E. & Fearon, K. C. Sarcopenia in an overweight or obese patient is an adverse prognostic factor in pancreatic cancer. Clin. Cancer Res. 15, 6973–6979 (2009).
Prado, C. M. et al. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: a population-based study. Lancet Oncol. 9, 629–635 (2008).
Martin, L. et al. Prognostic factors in patients with advanced cancer: use of the patient-generated subjective global assessment in survival prediction. J. Clin. Oncol. 28, 4376–4383 (2010).
Pichard, C., Baracos, V. & Attaix, D. Would you buy a new tool to improve your practice? Curr. Opin. Clin. Nutr. Metab. Care 14, 221–222 (2011).
Lieffers, J. R. et al. A viscerally-driven cachexia syndrome in patients with advanced colorectal cancer: contributions of organ and tumor mass to whole body energy demands. Am. J. Clin. Nutr. 89, 1173–1179 (2009).
Stephens, N. A. et al. Sexual dimorphism modulates the impact of cancer cachexia on lower limb muscle mass and function. Clin. Nutr. 31, 499–505 (2012).
Costa, G. & Donaldson, S. S. Current concepts in cancer: effects of cancer and cancer treatment on the nutrition of the host. N. Engl. J. Med. 300, 1471–1474 (1979).
Andreyev, H. J., Norman, A. R., Oates, J. & Cunningham, D. Why do patients with weight loss have a worse outcome when undergoing chemotherapy for gastrointestinal malignancies? Eur. J. Cancer 34, 503–509 (1998).
Nitenberg, G. & Raynard, B. Nutritional support of the cancer patient: issues and dilemmas. Crit. Rev. Oncol. Hematol. 34, 137–168 (2000).
Stewart, G. D., Skipworth, R. J. & Fearon, K. C. Cancer cachexia and fatigue. Clin. Med. 6, 140–143 (2006).
Watanabe, S. & Bruera, E. Anorexia and cachexia, asthenia, and lethargy. Hematol. Oncol. Clin. North Am. 10, 189–206 (1996).
Reid, J., McKenna, H., Fitzsimons, D. & McCance, T. Fighting over food: patient and family understanding of cancer cachexia. Oncol. Nurs. Forum 36, 439–445 (2009).
Dewys, W. D. et al. Prognostic effect of weight loss prior to chemotherapy in cancer patients. Eastern Cooperative Oncology Group. Am. J. Med. 69, 491–497 (1980).
Ross, P. J. et al. Do patients with weight loss have a worse outcome when undergoing chemotherapy for lung cancer? Br. J. Cancer 90, 1905–1911 (2004).
Prado, C. M., Antoun, S., Sawyer, M. B. & Baracos, V. E. Two faces of drug therapy in cancer: drug-related lean tissue loss and its adverse consequences to survival and toxicity. Curr. Opin. Nutr. Metab. Care 14, 250–254 (2011).
Mir, O. et al. Sarcopenia predicts early dose-limiting toxicities and pharmacokinetics of sorafenib in patients with hepatocellular carcinoma. PLoS ONE 7, e37563 (2012).
Ardizzoni, A. et al. Cisplatin- versus carboplatin-based chemotherapy in first-line treatment of advanced non-small-cell lung cancer: an individual patient data meta-analysis. J. Natl Cancer Inst. 99, 847–857 (2007).
Straub, R. H., Cutolo, M., Buttgereit, F. & Pongratz, G. Energy regulation and neuroendocrine-immune control in chronic inflammatory diseases. J. Intern. Med. 267, 543–560 (2010).
Gibb, J., Audet, M. C., Hayley, S. & Anisman, H. Neurochemical and behavioral responses to inflammatory immune stressors. Front. Biosci. (Schol. Ed.) 1, 275–295 (2009).
Myers, J. S. Proinflammatory cytokines and sickness behavior: implications for depression and cancer-related symptoms. Oncol. Nurs. Forum 35, 802–807 (2008).
Fearon, K. C., Glass, D. J. & Guttridge, D. C. Cancer cachexia: mediators, signaling, and metabolic pathways. Cell Metab. 16, 153–166 (2012).
Braun, T. P. et al. Central nervous system inflammation induces muscle atrophy via activation of the hypothalamic-pituitary-adrenal axis. J. Exp. Med. 208, 2449–2463 (2011).
Tan, B. H. et al. P-selectin genotype is associated with the development of cancer cachexia. EMBO Mol. Med. 4, 462–471 (2012).
Das, S. K. et al. Adipose triglyceride lipase contributes to cancer-associated cachexia. Science 333, 233–238 (2011).
Kortebein, P., Ferrando, A., Lombeida, J., Wolfe, R. & Evans, W. J. Effect of 10 days of bed rest on skeletal muscle in healthy older adults. JAMA 297, 1772–1774 (2007).
Ferrando, A. A., Stuart, C. A., Sheffield-Moore, M. & Wolfe, R. R. Inactivity amplifies the catabolic response of skeletal muscle to cortisol. J. Clin. Endocrinol. Metab. 84, 3515–3521 (1999).
Murphy, R. A. et al. Aberrations in plasma phospholipid fatty acids in lung cancer patients. Lipids 47, 363–369 (2012).
Murphy, R. A. et al. Nutritional intervention with fish oil provides a benefit over standard of care for weight and skeletal muscle mass in patients with nonsmall cell lung cancer receiving chemotherapy. Cancer 117, 1775–1782 (2011).
Murphy, R. A. et al. Supplementation with fish oil increases first-line chemotherapy efficacy in patients with advanced nonsmall cell lung cancer. Cancer 117, 3774–3780 (2011).
Fakih, M. G. et al. Chemotherapy is linked to severe vitamin D deficiency in patients with colorectal cancer. Int. J. Colorectal Dis. 24, 219–224 (2009).
Zeisel, S. H. & da Costa, K. A. Choline: an essential nutrient for public health. Nutr. Rev. 67, 615–623 (2009).
Hasselgren, P. O. Glucocorticoids and muscle catabolism. Curr. Opin. Clin. Nutr. Metab. Care 2, 201–205 (1999).
Sheffield-Moore, M. et al. Androgen therapy induces muscle protein anabolism in older women. J. Clin. Endocrinol. Metab. 91, 3844–3849 (2006).
Gullett, N. P., Hebbar, G. & Ziegler, T. R. Update on clinical trials of growth factors and anabolic steroids in cachexia and wasting. Am. J. Clin. Nutr. 91, 1143S–1147S (2010).
Bodine, S. C. et al. Akt/mTOR pathway is a crucial regulator of skeletal muscle hypertrophy and can prevent muscle atrophy in vivo. Nat. Cell Biol. 3, 1014–1019 (2001).
Glass, D. J. PI3 kinase regulation of skeletal muscle hypertrophy and atrophy. Curr. Top. Microbiol. Immunol. 346, 267–278 (2010).
Trendelenburg, A. U. et al. Myostatin reduces Akt/TORC1/p70S6K signaling, inhibiting myoblast differentiation and myotube size. Am. J. Physiol. Cell Physiol. 296, C1258–1270 (2009).
Spiro, A., Baldwin, C., Patterson, A., Thomas, J. & Andreyev, H. J. The views and practice of oncologists towards nutritional support in patients receiving chemotherapy. Br. J. Cancer 95, 431–434 (2006).
Heyland, D. K. et al. Total parenteral nutrition in the surgical patient: a meta-analysis. Can. J. Surg. 44, 102–111 (2001).
Jatoi, A., Kumar, S., Sloan, J. A. & Nguyen, P. L. On appetite and its loss. J. Clin. Oncol. 18, 2930–2932 (2000).
Ries, A. L. Pulmonary rehabilitation: summary of an evidence-based guideline. Respir. Care 53, 1203–1207 (2008).
de Gramont, A. et al. The evolution of adjuvant therapy in the treatment of early-stage colon cancer. Clin. Colorectal Cancer 10, 218–226 (2011).
Vigano, A., Del Fabbro, E., Bruera, E. & Borod, E. The cachexia clinic: from staging to managing nutritional and functional problems in advanced cancer patients. Crit. Rev. Oncog. 17, 293–304 (2012).
Manini, T. M. Energy expenditure and aging. Ageing Res. Rev. 9, 1–11 (2010).
Moses, A. W., Slater, C., Preston, T., Barber, M. D. & Fearon, K. C. Reduced total energy expenditure and physical activity in cachectic patients with pancreatic cancer can be modulated by an energy and protein dense oral supplement enriched with n-3 fatty acids. Br. J. Cancer 90, 996–1002 (2004).
Fearon, K. C. et al. Effect of a protein and energy dense N-3 fatty acid enriched oral supplement on loss of weight and lean tissue in cancer cachexia: a randomised double blind trial. Gut 52, 1479–1486 (2003).
Lundholm, K., Daneryd, P., Bosaeus, I., Korner, U. & Lindholm, E. Palliative nutritional intervention in addition to cyclooxygenase and erythropoietin treatment for patients with malignant disease: effects on survival, metabolism, and function. Cancer 100, 967–977 (2004).
Baldwin, C., Spiro, A., Ahern, R. & Emery, P. W. Oral nutritional interventions in malnourished patients with cancer: a systematic review and meta-analysis. J. Natl Cancer Inst. 7, 371–385 (2012).
Ferriolli, E. et al. Physical activity monitoring: a responsive and meaningful patient-centered outcome for surgery, chemotherapy, or radiotherapy? J. Pain Symptom Manage. 43, 1025–1035 (2012).
Lenk, K., Schuler, G. & Adams, V. Skeletal muscle wasting in cachexia and sarcopenia: molecular pathophysiology and impact of exercise training. J. Cachexia Sarcopenia Muscle 1, 9–21 (2010).
Oldervoll, L. M. et al. Physical exercise for cancer patients with advanced disease: a randomized controlled trial. Oncologist 16, 1649–1657 (2011).
Argilés, J. M., Busquets, S., López-Soriano, F. J., Costelli, P. & Penna, F. Are there any benefits of exercise training in cancer cachexia? J. Cachexia Sarcopenia Muscle 3, 73–76 (2012).
Loprinzi, C. L. et al. Randomized comparison of megestrol acetate versus dexamethasone versus fluoxymesterone for the treatment of cancer anorexia/cachexia. J. Clin. Oncol. 17, 3299–3306 (1999).
Strasser, F. et al. Comparison of orally administered cannabis extract and δ-9-tetrahydrocannabinol in treating patients with cancer-related anorexia-cachexia syndrome: a multicenter, phase III, randomized, double-blind, placebo-controlled clinical trial from the Cannabis-In-Cachexia-Study-Group. J. Clin. Oncol. 24, 3394–3400 (2006).
Lundholm, K. et al. Anti-inflammatory treatment may prolong survival in undernourished patients with metastatic solid tumors. Cancer Res. 54, 5602–5606 (1994).
Lundholm, K., Daneryd, P., Körner, U., Hyltander, A. & Bosaeus, I. Evidence that long-term COX-treatment improves energy homeostasis and body composition in cancer patients with progressive cachexia. Int. J. Oncol. 24, 505–512 (2004).
Schmitz, G. & Ecker, J. The opposing effects of n-3 and n-6 fatty acids. Prog. Lipid Res. 47, 147–155 (2008).
Dewey, A., Baughan, C., Dean, T., Higgins, B. & Johnson, I. Eicosapentaenoic acid (EPA, an ω-3 fatty acid from fish oils) for the treatment of cancer cachexia. Cochrane Database of Systematic Reviews, Issue 1, Art. No.: CD004597. http://dx.doi.org/10.1002/14651858.CD004597.pub2.
van der Meij, B. S. et al. Oral nutritional supplements containing (n-3) polyunsaturated fatty acids affect the nutritional status of patients with stage III non-small cell lung cancer during multimodality treatment. J. Nutr. 140, 1774–1780 (2010).
Noman, A. S. et al. Thalidomide inhibits lipopolysaccharide-induced tumor necrosis factor-α production via down-regulation of MyD88 expression. Innate Immun. 15, 33–41 (2009).
Bruera, E. et al. Thalidomide in patients with cachexia due to terminal cancer: preliminary report. Ann. Oncol. 10, 857–859 (1999).
Khan, Z. H. et al. Oesophageal cancer and cachexia: the effect of short-term treatment with thalidomide on weight loss and lean body mass. Aliment. Pharmacol. Ther. 17, 677–682 (2003).
Gordon, J. N. et al. Thalidomide in the treatment of cancer cachexia: a randomised placebo controlled trial. Gut 54, 540–545 (2005).
Moehler, T. Clinical experience with thalidomide and lenalidomide in multiple myeloma. Curr. Cancer Drug Targets 12, 372–390 (2012).
Heslin, M. J., Newman, E., Wolf, R. F., Pisters, P. W. & Brennan, M. F. Effect of systemic hyperinsulinemia in cancer patients. Cancer Res. 52, 3845–3850 (1992).
Newman, E., Heslin, M. J., Wolf, R. F., Pisters, P. W. & Brennan, M. F. The effect of insulin on glucose and protein metabolism in the forearm of cancer patients. Surg. Oncol. 1, 257–267 (1992).
Lundholm, K. et al. Insulin treatment in cancer cachexia: effects on survival, metabolism, and physical functioning. Clin. Cancer Res. 13, 2699–2706 (2007).
Neary, N. M. et al. Ghrelin increases energy intake in cancer patients with impaired appetite: acute, randomized, placebo-controlled trial. J. Clin. Endocrinol. Metab. 89, 2832–2836 (2004).
Garcia, J. M., Friend, J. & Allen, S. Therapeutic potential of anamorelin, a novel, oral ghrelin mimetic, in patients with cancer-related cachexia: a multicenter, randomized, double-blind, crossover, pilot study. Support. Care Cancer http://dx.doi.org/10.1007/s00520-012-1500-1.
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
Dalton, J. T. et al. The selective androgen receptor modulator GTx-024 (enobosarm) improves lean body mass and physical function in healthy elderly men and postmenopausal women: results of a double-blind, placebo-controlled phase II trial. J. Cachexia Sarcopenia Muscle 2, 153–161 (2011).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
Zaki, M. H., Nemeth, J. A. & Trikha, M. CNTO 328, a monoclonal antibody to IL-6, inhibits human tumor-induced cachexia in nude mice. Int. J. Cancer 111, 592–595 (2004).
Bayliss, T. J., Smith, J. T., Schuster, M., Dragnev, K. H. & Rigas, J. R. A humanized anti-IL-6 antibody (ALD518) in non-small cell lung cancer. Expert Opin. Biol. Ther. 11, 1663–1668 (2011).
Prado, C. M. et al. Skeletal muscle anabolism is a side effect of therapy with the MEK inhibitor: selumetinib in patients with cholangiocarcinoma. Br. J. Cancer 106, 1583–1586 (2012).
Han, H. Q. & Mitch, W. E. Targeting the myostatin signaling pathway to treat muscle wasting diseases. Curr. Opin. Support Palliat. Care 5, 334–341 (2011).
Zhou, X. et al. Reversal of cancer cachexia and muscle wasting by ActRIIB antagonism leads to prolonged survival. Cell 142, 531–543 (2010).
Benny Klimek, M. E. et al. Acute inhibition of myostatin-family proteins preserves skeletal muscle in mouse models of cancer cachexia. Biochem. Biophys. Res. Commun. 391, 1548–1554 (2010).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
American Dietetic Association. Oncology evidence-based nutrition practice guideline [online], (2007).
August, D. A., Huhmann, M. B. & American Society for Parenteral and Enteral Nutrition (A. S. P. E. N.) Board of Directors. A. S. P. E. N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J. Parenter. Enteral Nutr. 33, 472–500 (2009).
Bauer, J. D. et al. Evidence based practice guidelines for the nutritional management of cancer cachexia. Nutr. Dietetics 63 (Suppl. s2), S3–S32 (2006).
Bozzetti, F. et al. ESPEN Guidelines on Parenteral Nutrition: non-surgical oncology. Clin. Nutr. 28, 445–454 (2009).
Moore, F. A., Ziegler, T. R., Heyland, D. K., Marik, P. E. & Bistrian, B. R. Developing research programs in clinical and translational nutrition. JPEN J. Parenter. Enteral. Nutr. 34 (Suppl. 6), 97S–105S (2010).
Commission on Cancer. Cancer Program Standards 2012: Ensuring Patient-Centered Care [online], (2012).
OnkoZert [online], (2012).
German Society of Hematology and Oncology [German, online], (2012).
Boomsma, F. et al. Organisation of European Cancer Institutes Accreditation and Designation User Manual (eds Pierotti, M. A., van Harten W., Hummel, H. & Otter, R.) [online], (2011).
Council of Europe Committee of Ministers. Resolution ResAP (2003)3 on food and nutritional care in hospitals [online], (2003).
Author information
Authors and Affiliations
Contributions
All authors researched data for the article, made a substantial contribution to the discussion of the content, wrote the manuscript, and reviewed and edited it prior to submission.
Corresponding author
Ethics declarations
Competing interests
K. Fearon acts either a consultant for or receives honoraria or research support from Abbott, Alder Biopharmaceuticals, Nutricia and Novartis. J. Arends receives honoraria or research support from Abbott, Baxter, B. Braun, Fresenius-Kabi and Nutricia. V. Baracos declares no competing interests.
Rights and permissions
About this article
Cite this article
Fearon, K., Arends, J. & Baracos, V. Understanding the mechanisms and treatment options in cancer cachexia. Nat Rev Clin Oncol 10, 90–99 (2013). https://doi.org/10.1038/nrclinonc.2012.209
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2012.209
This article is cited by
-
Platelet-to-albumin ratio and radiation-induced lymphopenia—prognostic biomarker for carcinoma esophagus
Journal of the Egyptian National Cancer Institute (2024)
-
Effect of cancer cachexia on first-line chemotherapy in patients with advanced pancreatic cancer: a claims database study in Japan
International Journal of Clinical Oncology (2024)
-
A novel nutrition-related nomogram for the survival prediction of colorectal cancer-results from a multicenter study
Nutrition & Metabolism (2023)
-
Cancer cachexia: molecular mechanisms and treatment strategies
Journal of Hematology & Oncology (2023)
-
The underestimated impact of excess body weight on colorectal cancer risk: Evidence from the UK Biobank cohort
British Journal of Cancer (2023)