Abstract
There are two hypotheses to explain melanoma dissemination: first, simultaneous lymphatic and hematogeneous spread, with regional lymph nodes as indicators of metastatic disease; and second, orderly progression, with regional lymph nodes as governors of metastatic disease. The sentinel node (SN) has been defined as the first draining lymph node from a tumor and is harvested with the use of the triple technique and is processed by an extensive pathology protocol. The SN status is a strong prognostic factor for survival (83–94% for SN negative, 56–75% SN-positive patients). False-negative rates are considerable (9–21%). Preliminary results of the MSLT-1 trial did not demonstrate a survival benefit for the SN procedure, although a subgroup analysis indicates a possible benefit. A mathematical model has demonstrated 24% prognostic false positivity. SN tumor burden represents a heterogenous patient population and is classified most frequently with the Starz, Dewar or Rotterdam Criteria. A completion lymph-node dissection might not be indicated in all SN-positive patients. Patients classified with metastases <0.1 mm by the Rotterdam Criteria have excellent survival rates. Ultrasound-guided fine-needle aspiration cytology is emerging as a staging tool for high-risk patients, but more research is necessary before this can change clinical practice.
Key Points
-
The sentinel lymph-node biopsy (SLNB) is a very accurate staging procedure for stage I–II melanoma patients, which provides the most important prognostic information for survival
-
The SLNB procedure followed by immediate completion lymph-node dissection (CLND) does not improve survival, although further studies are needed to determine if it might be beneficial for some patients
-
Sentinel node tumor burden determines survival and the need for CLND
-
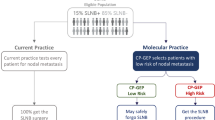
Patients with sentinel node micrometastases <0.1 mm have a clinical course that is indistinguishable from sentinel-node-negative patients and routine CLND may not be indicated in these patients
-
The EORTC Melanoma Group recommends the use of the Rotterdam Criteria for the measurement of sentinel node tumor burden
-
Sentinel node staging is the most accurate staging in melanoma patients, but future staging approaches might include a role for ultrasound-guided fine-needle aspiration cytology
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Surveillance, Epidemiology and End Results (SEER) Database (2010).
MacKie, R. M., Hauschild, A. & Eggermont, A. M. Epidemiology of invasive cutaneous melanoma. Ann. Oncol. 20 (Suppl. 6), vi1–7 (2009).
de Vries, E. & Coebergh, J. W. Melanoma incidence has risen in Europe. BMJ 331, 698 (2005).
Morton, D. L. et al. Technical details of intraoperative lymphatic mapping for early stage melanoma. Arch. Surg. 127, 392–399 (1992).
Meier, F. et al. Metastatic pathways and time courses in the orderly progression of cutaneous melanoma. Br. J. Dermatol. 147, 62–70 (2002).
Slingluff, C. L. Jr, Stidham, K. R., Ricci, W. M., Stanley, W. E. & Seigler, H. F. Surgical management of regional lymph nodes in patients with melanoma. Experience with 4682 patients. Ann. Surg. 219, 120–130 (1994).
Veronesi, U. et al. Inefficacy of immediate node dissection in stage 1 melanoma of the limbs. N. Engl. J. Med. 297, 627–630 (1977).
Sim, F. H., Taylor, W. F., Ivins, J. C., Pritchard, D. J. & Soule, E. H. A prospective randomized study of the efficacy of routine elective lymphadenectomy in management of malignant melanoma. Preliminary results. Cancer 41, 948–956 (1978).
Cascinelli, N., Morabito, A., Santinami, M., MacKie, R. M. & Belli, F. Immediate or delayed dissection of regional nodes in patients with melanoma of the trunk: a randomised trial. WHO Melanoma Programme. Lancet 351, 793–796 (1998).
Balch, C. M. et al. Long-term results of a multi-institutional randomized trial comparing prognostic factors and surgical results for intermediate thickness melanomas (1.0 to 4.0 mm). Intergroup Melanoma Surgical Trial. Ann. Surg. Oncol. 7, 87–97 (2000).
Reintgen, D. et al. The orderly progression of melanoma nodal metastases. Ann. Surg. 220, 759–767 (1994).
Morton, D. L. et al. Sentinel-node biopsy or nodal observation in melanoma. N. Engl. J. Med. 355, 1307–1317 (2006).
Balch, C. M. et al. Prognostic factors analysis of 17,600 melanoma patients: validation of the American Joint Committee on Cancer melanoma staging system. J. Clin. Oncol. 19, 3622–3634 (2001).
Guggenheim, M. et al. The influence of sentinel lymph node tumour burden on additional lymph node involvement and disease-free survival in cutaneous melanoma—a retrospective analysis of 392 cases. Br. J. Cancer 98, 1922–1928 (2008).
van Akkooi, A. C. et al. High positive sentinel node identification rate by EORTC melanoma group protocol. Prognostic indicators of metastatic patterns after sentinel node biopsy in melanoma. Eur. J. Cancer 42, 372–380 (2006).
Doubrovsky, A., De Wilt, J. H., Scolyer, R. A., McCarthy, W. H. & Thompson, J. F. Sentinel node biopsy provides more accurate staging than elective lymph node dissection in patients with cutaneous melanoma. Ann. Surg. Oncol. 11, 829–836 (2004).
Wong, S. L., Brady, M. S., Busam, K. J. & Coit, D. G. Results of sentinel lymph node biopsy in patients with thin melanoma. Ann. Surg. Oncol. 13, 302–309 (2006).
Wright, B. E. et al. Importance of sentinel lymph node biopsy in patients with thin melanoma. Arch. Surg. 143, 892–899; discussion 899–900 (2008).
Cochran, A. J. et al. Update on lymphatic mapping and sentinel node biopsy in the management of patients with melanocytic tumours. Pathology 36, 478–484 (2004).
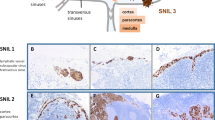
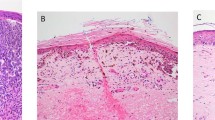
Scolyer, R. A., Murali, R., McCarthy, S. W. & Thompson, J. F. Pathologic examination of sentinel lymph nodes from melanoma patients. Semin. Diagn. Pathol. 25, 100–111 (2008).
Cook, M. G. et al. The development of optimal pathological assessment of sentinel lymph nodes for melanoma. J. Pathol. 200, 314–319 (2003).
Cochran, A. J., Wen, D. R. & Morton, D. L. Occult tumor cells in the lymph nodes of patients with pathological stage I malignant melanoma. An immunohistological study. Am. J. Surg. Pathol. 12, 612–618 (1988).
Starz, H., Balda, B. R., Kramer, K. U., Buchels, H. & Wang, H. A micromorphometry-based concept for routine classification of sentinel lymph node metastases and its clinical relevance for patients with melanoma. Cancer 91, 2110–2121 (2001).
Abrahamsen, H. N., Hamilton-Dutoit, S. J., Larsen, J. & Steiniche, T. Sentinel lymph nodes in malignant melanoma: extended histopathologic evaluation improves diagnostic precision. Cancer 100, 1683–1691 (2004).
Gietema, H. A. et al. Sentinel lymph node investigation in melanoma: detailed analysis of the yield from step sectioning and immunohistochemistry. J. Clin. Pathol. 57, 618–620 (2004).
Spanknebel, K. et al. Characterization of micrometastatic disease in melanoma sentinel lymph nodes by enhanced pathology: recommendations for standardizing pathologic analysis. Am. J. Surg. Pathol. 29, 305–317 (2005).
Chao, C. et al. Patterns of early recurrence after sentinel lymph node biopsy for melanoma. Am. J. Surg. 184, 520–524; discussion 525 (2002).
Gershenwald, J. E. et al. Patterns of recurrence following a negative sentinel lymph node biopsy in 243 patients with stage I or II melanoma. J. Clin. Oncol. 16, 2253–2260 (1998).
Gogel, B. M. et al. Sentinel lymph node biopsy for melanoma. Am. J. Surg. 176, 544–547 (1998).
Zogakis, T. G. et al. Melanoma recurrence patterns after negative sentinel lymphadenectomy. Arch. Surg. 140, 865–871; discussion 871–862 (2005).
Carlson, G. W. et al. Regional recurrence after negative sentinel lymph node biopsy for melanoma. Ann. Surg. 248, 378–386 (2008).
Rossi, C. R., Pasquali, S. & Mocellin, S. Actual false-negative rate prompts the routine use of ultrasound scan before and after sentinel node biopsy in melanoma. Ann. Surg. Oncol. 15, 2976–2977 (2008).
Jansen, L. et al. Reliability of sentinel lymph node biopsy for staging melanoma. Br. J. Surg. 87, 484–489 (2000).
Nieweg, O. E. & Estourgie, S. H. What is a sentinel node and what is a false-negative sentinel node? Ann. Surg. Oncol. 11, 169S–173S (2004).
Wasserberg, N., Tulchinsky, H., Schachter, J., Feinmesser, M. & Gutman, H. Sentinel-lymph-node biopsy (SLNB) for melanoma is not complication-free. Eur. J. Surg. Oncol. 30, 851–856 (2004).
Scolyer, R. A., Murali, R., Satzger, I. & Thompson, J. F. The detection and significance of melanoma micrometastases in sentinel nodes. Surg. Oncol. 17, 165–174 (2008).
de Wilt, J. H., van Akkooi, A. C., Verhoef, C. & Eggermont, A. M. Detection of melanoma micrometastases in sentinel nodes—the cons. Surg. Oncol. 7, 175–181 (2008).
Karim, R. Z. et al. False negative sentinel lymph node biopsies in melanoma may result from deficiencies in nuclear medicine, surgery, or pathology. Ann. Surg. 247, 1003–1010 (2008).
Nowecki, Z. I., Rutkowski, P., Nasierowska-Guttmejer, A. & Ruka, W. Survival analysis and clinicopathological factors associated with false-negative sentinel lymph node biopsy findings in patients with cutaneous melanoma. Ann. Surg. Oncol. 13, 1655–1663 (2006).
Cochran, A. J. & Thompson, J. F. Lymphatic mapping and sentinel node biopsy: the data unclouded by speculation. Arch. Dermatol. 144, 687–688; author reply 688–689 (2008).
Morton, D. L., Cochran, A. J. & Thompson, J. F. The rationale for sentinel-node biopsy in primary melanoma. Nat. Clin. Pract. Oncol. 5, 510–511 (2008).
Morton, D. L. & Elashoff, R. Sentinel node biopsy: facts to clear the alleged clouds. Arch. Dermatol. 144, 685–686; author reply 687 (2008).
Ross, M. I. & Gershenwald, J. E. How should we view the results of the Multicenter Selective Lymphadenectomy Trial-1 (MSLT-1)? Ann. Surg. Oncol. 15, 670–673 (2008).
Gershenwald, J. E. & Ross, M. I. Is sentinel-node biopsy superior to nodal observation in melanoma? Nat. Clin. Pract. Oncol. 4, 278–279 (2007).
Koskivuo, I. et al. Sentinel lymph node biopsy in cutaneous melanoma: a case-control study. Ann. Surg. Oncol. 14, 3566–3574 (2007).
Kretschmer, L. et al. Patients with lymphatic metastasis of cutaneous malignant melanoma benefit from sentinel lymphonodectomy and early excision of their nodal disease. Eur. J. Cancer 40, 212–218 (2004).
De Giorgi, V. et al. Sentinel lymph nodes in melanoma patients: evaluating the evidence. Oncology 71, 460–462 (2006).
Gonzalez, U. Cloud over sentinel node biopsy: unlikely survival benefit in melanoma. Arch. Dermatol. 143, 775–776 (2007).
Kanzler, M. H. The current status of evaluation and treatment of high-risk cutaneous melanoma: therapeutic breakthroughs remain elusive. Arch. Dermatol. 143, 785–787 (2007).
Thomas, J. M. Prognostic false-positivity of the sentinel node in melanoma. Nat. Clin. Pract. Oncol. 5, 18–23 (2008).
van Akkooi, A. C. et al. Multivariable analysis comparing outcome after sentinel node biopsy or therapeutic lymph node dissection in patients with melanoma. Br. J. Surg. 94, 1293–1299 (2007).
van Akkooi, A. C. et al. Clinical relevance of melanoma micrometastases (<0.1 mm) in sentinel nodes: are these nodes to be considered negative? Ann. Oncol. 17, 1578–1585 (2006).
van Akkooi, A. C. et al. Sentinel node tumor burden according to the Rotterdam criteria is the most important prognostic factor for survival in melanoma patients: a multicenter study in 388 patients with positive sentinel nodes. Ann. Surg. 248, 949–955 (2008).
Gershenwald, J. E. et al. Microscopic tumor burden in sentinel lymph nodes predicts synchronous nonsentinel lymph node involvement in patients with melanoma. J. Clin. Oncol. 26, 4296–4303 (2008).
Govindarajan, A., Ghazarian, D. M., McCready, D. R. & Leong, W. L. Histological features of melanoma sentinel lymph node metastases associated with status of the completion lymphadenectomy and rate of subsequent relapse. Ann. Surg. Oncol. 14, 906–912 (2007).
van Akkooi, A. C. J. et al. Minimal sentinel node (SN) tumor burden according to the Rotterdam criteria is the most important prognostic factor for survival in melanoma patients. A multicenter study in 388 SN positive patients. Ann. Surg. 248, 949–955 (2008).
Dewar, D. J. et al. The microanatomic location of metastatic melanoma in sentinel lymph nodes predicts nonsentinel lymph node involvement. J. Clin. Oncol. 22, 3345–3349 (2004).
Starz, H., Siedlecki, K. & Balda, B. R. Sentinel lymphonodectomy and s-classification: a successful strategy for better prediction and improvement of outcome of melanoma. Ann. Surg. Oncol. 11, 162S–168S (2004).
Vuylsteke, R. J. et al. Sentinel lymph node tumor load: an independent predictor of additional lymph node involvement and survival in melanoma. Ann. Surg. Oncol. 12, 440–448 (2005).
Cochran, A. J. et al. Prediction of metastatic melanoma in nonsentinel nodes and clinical outcome based on the primary melanoma and the sentinel node. Mod. Pathol. 17, 747–755 (2004).
Reeves, M. E., Delgado, R., Busam, K. J., Brady, M. S. & Coit, D. G. Prediction of nonsentinel lymph node status in melanoma. Ann. Surg. Oncol. 10, 27–31 (2003).
Sabel, M. S. et al. Predictors of nonsentinel lymph node positivity in patients with a positive sentinel node for melanoma. J. Am. Coll. Surg. 201, 37–47 (2005).
Satzger, I. et al. Prognostic significance of histopathological parameters in sentinel nodes of melanoma patients. Histopathology 50, 764–772 (2007).
Roka, F. et al. Prediction of non-sentinel node status and outcome in sentinel node-positive melanoma patients. Eur. J. Surg. Oncol. 34, 82–88 (2008).
Frankel, T. L. et al. Do micromorphometric features of metastatic deposits within sentinel nodes predict nonsentinel lymph node involvement in melanoma? Ann. Surg. Oncol. 15, 2403–2411 (2008).
Lindemann, F., Schlimok, G., Dirschedl, P., Witte, J. & Riethmuller, G. Prognostic significance of micrometastatic tumour cells in bone marrow of colorectal cancer patients. Lancet 340, 685–689 (1992).
Scheri, R. P., Essner, R., Turner, R. R., Ye, X. & Morton, D. L. Isolated tumor cells in the sentinel node affect long-term prognosis of patients with melanoma. Ann. Surg. Oncol. 14, 2861–2866 (2007).
van Akkooi, A. C. J. et al. Long-term follow-up of patients with minimal sentinel node tumor burden (< 0.1mm) according to Rotterdam criteria: A study of the EORTC Melanoma Group [abstract]. J. Clin. Oncol. 15S, a9005 (2009).
van Akkooi, A. C. J. et al. Excellent long-term survival of patients with minimal sentinel node tumor burden (<0.1 mm) according to Rotterdam Criteria: a study of the EORTC melanoma group. Eur. J. Cancer 7 (Suppl.), 576 (2009).
Murali, R. et al. Interobserver reproducibility of histologic parameters of melanoma deposits in sentinel lymph nodes: implications for management of patients with melanoma. Cancer 115, 5026–5037 (2009).
van Akkooi, A. C., Spatz, A., Eggermont, A. M., Mihm, M. & Cook, M. G. Expert opinion in melanoma: the sentinel node; EORTC Melanoma Group recommendations on practical methodology of the measurement of the microanatomic location of metastases and metastatic tumour burden. Eur. J. Cancer 45, 2736–2742 (2009).
de Boer, M. et al. Micrometastases or isolated tumor cells and the outcome of breast cancer. N. Engl. J. Med. 361, 653–663 (2009).
Rossi, C. R. et al. The role of preoperative ultrasound scan in detecting lymph node metastasis before sentinel node biopsy in melanoma patients. J. Surg. Oncol. 83, 80–84 (2003).
Starritt, E. C., Uren, R. F., Scolyer, R. A., Quinn, M. J. & Thompson, J. F. Ultrasound examination of sentinel nodes in the initial assessment of patients with primary cutaneous melanoma. Ann. Surg. Oncol. 12, 18–23 (2005).
van Rijk, M. C. et al. Ultrasonography and fine-needle aspiration cytology in the preoperative evaluation of melanoma patients eligible for sentinel node biopsy. Ann. Surg. Oncol. 13, 1511–1516 (2006).
Voit, C. et al. Ultrasound morphology criteria predict metastatic disease of the sentinel nodes in patients with melanoma. J. Clin. Oncol. 28, 847–852 (2010).
Voit, C. A. et al. Rotterdam Criteria for sentinel node (SN) tumor burden and the accuracy of ultrasound (US)-guided fine-needle aspiration cytology (FNAC): can US-guided FNAC replace SN staging in patients with melanoma? J. Clin. Oncol. 27, 4994–5000 (2009).
van Akkooi, A. C., Voit, C. A., Verhoef, C. & Eggermont, A. M. Potential cost-effectiveness of US-guided FNAC in melanoma patients as a primary procedure and in follow-up. Ann. Surg. Oncol. 17, 660–662 (2009).
Sassen, S., Shaw, H. M., Colman, M. H., Scolyer, R. A. & Thompson, J. F. The complex relationships between sentinel node positivity, patient age, and primary tumor desmoplasia: analysis of 2303 melanoma patients treated at a single center. Ann. Surg. Oncol. 15, 630–637 (2008).
Cascinelli, N. et al. Sentinel and nonsentinel node status in stage IB and II melanoma patients: two-step prognostic indicators of survival. J. Clin. Oncol. 24, 4464–4471 (2006).
Yee, V. S. et al. Outcome in 846 cutaneous melanoma patients from a single center after a negative sentinel node biopsy. Ann. Surg. Oncol. 12, 429–439 (2005).
Ranieri, J. M. et al. Prognostic importance of lymph node tumor burden in melanoma patients staged by sentinel node biopsy. Ann. Surg. Oncol. 9, 975–981 (2002).
Carlson, G. W. et al. The amount of metastatic melanoma in a sentinel lymph node: does it have prognostic significance? Ann. Surg. Oncol. 10, 575–581 (2003).
Pearlman, N. W. et al. Size of sentinel node metastases predicts other nodal disease and survival in malignant melanoma. Am. J. Surg. 192, 878–881 (2006).
Debarbieux, S. et al. Sentinel lymph node biopsy in melanoma: a micromorphometric study relating to prognosis and completion lymph node dissection. Br. J. Dermatol. 157, 58–67 (2007).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
van Akkooi, A., Verhoef, C. & Eggermont, A. Importance of tumor load in the sentinel node in melanoma: clinical dilemmas. Nat Rev Clin Oncol 7, 446–454 (2010). https://doi.org/10.1038/nrclinonc.2010.100
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2010.100
This article is cited by
-
Induction of potassium channel regulator KCNE4 in a submandibular lymph node metastasis model
Scientific Reports (2022)
-
Current management of melanoma patients with nodal metastases
Clinical & Experimental Metastasis (2022)
-
Melanoma-derived small extracellular vesicles induce lymphangiogenesis and metastasis through an NGFR-dependent mechanism
Nature Cancer (2021)
-
SLN melanoma micrometastasis predictivity of nodal status: a long term retrospective study
Journal of Experimental & Clinical Cancer Research (2013)
-
Sentinel lymph node biopsy for high-risk cutaneous squamous cell carcinoma: clinical experience and review of literature
World Journal of Surgical Oncology (2011)