Abstract
The optimal development of novel molecularly targeted agents for the treatment of cancer requires a re-evaluation of the current drug development paradigm. Selection of patients, optimal biologic dose versus maximum tolerated dose, definition of response and clinical benefit and trial designs that address these considerations are the focus of debate in the field of early cancer therapeutics. We present a review of the opportunities and challenges facing drug development in oncology through the phases of clinical development starting with first-in-human trials.
Key Points
-
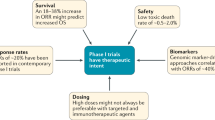
The design of trials to test molecularly targeted agents need to be revised to achieve objectives such as optimal biologic dose, which might require incorporation of analytically validated assays into initial clinical studies
-
The design of early phase trials of molecularly targeted agents might need to have end points such as progression-free survival or time to progression as opposed to objective response rate
-
The design of subsequent phase II studies will need to take into consideration the biological expectations of the molecular target agent (tumor shrinkage versus nonshrinkage)
-
Phase III studies should be pursued only in the presence of sufficient signs of activity, and ideally only initiated after randomized phase II studies of adequate power
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Kola, I. & Landis, J. Can the pharmaceutical industry reduce attrition rates? Nat. Rev. Drug Discov. 3, 711–716 (2004).
Kummar, S. et al. Drug development in oncology: classical cytotoxics and molecularly targeted agents. Br. J. Clin. Pharmacol. 62, 15–26 (2006).
Fox, E. et al. Clinical Trial Design for Target-Based Therapy. Oncologist 7, 401–409 (2002).
[No authors listed] Steps to consider in pharmacodynamic assay development http://dtp.nci.nih.gov/docs/phase0/PharmacoDynamicAssayDeveloment.html (accessed 25 September (2008).
Perez-Soler, R. et al. Determinants of tumor response and survival with erlotinib in patients with non-small-cell lung cancer. J. Clin. Oncol. 22, 3238–3247 (2004).
Ciardiello, F. et al. Antitumor effect and potentiation of cytotoxic drug activity in human cancer cells by ZD-1839 (Iressa), an epidermal growth factor receptor-selective tyrosine kinase inhibitor. Clin. Cancer Res. 6, 2053–2063 (2000).
Herbst, R. S. et al. Phase I/II trial evaluating the anti-vascular endothelial growth factor monoclonal antibody bevacizumab in combination with the HER-1/epidermal growth factor receptor tyrosine kinase inhibitor erlotinib for patients with recurrent non-small-cell lung cancer. J. Clin. Oncol. 23, 2544–2555 (2005).
Therasse, P. et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J. Natl Cancer Inst. 92, 205–216 (2000).
Park, J. O. et al. Measuring response in solid tumors: comparison of RECIST and WHO response criteria. Jpn J. Clin. Oncol. 33, 533–537 (2003).
Schuetze, S. M. et al. Selection of Response Criteria for Clinical Trials of Sarcoma Treatment. Oncologist 13 (Suppl. 2), 32–40 (2008).
Millar, A. W. & Lynch, L. P. Rethinking clinical trials for cytostatic drugs. Nat. Rev. Cancer 3, 540–545 (2003).
Weinstein, I. B. & Joe, A. K. Mechanisms of disease: oncogene addiction—-a rationale for molecular targeting in cancer therapy. Nat. Clin. Pract. Oncol. 3, 448–457 (2006).
Sharma, S. V. et al. A common signaling cascade may underlie “addiction” to the Src, BCR-ABL, and EGF receptor oncogenes. Cancer Cell 10, 425–435 (2006).
Kris, M. G. et al. Efficacy of gefitinib, an inhibitor of the epidermal growth factor receptor tyrosine kinase, in symptomatic patients with non-small cell lung cancer: A randomized trial. JAMA 290, 2149–2158 (2003).
Fukuoka, M. et al. Multi-institutional randomized phase II trial of gefitinib for previously treated patients with advanced non-small-cell lung cancer. J. Clin. Oncol. 21, 2237–2246 (2003).
Yang, J. C. et al. A randomized trial of bevacizumab, an anti-vascular endothelial growth factor antibody, for metastatic renal cancer. N. Engl. J. Med. 349, 427–434 (2003).
Rugo, H. S. Bevacizumab in the treatment of breast cancer: rationale and current data. Oncologist 9 (Suppl. 1), 43–49 (2004).
Cannistra, S. A. et al. Bevacizumab in patients with advanced platinum-resistant ovarian cancer [Abstract]. ASCO Meeting Abstracts 24, 5006 (2006).
Gambacorti-Passerini, C. et al. Inhibition of the ABL kinase activity blocks the proliferation of BCR/ABL+ leukemic cells and induces apoptosis. Blood Cells Mol. Dis. 23, 380–394 (1997).
Blanke, C. D. et al. Evaluation of the safety and efficacy of an oral molecularly-targeted therapy, STI571, in patients (Pts) with unresectable or metastatic gastrointestinal stromal tumors (GISTS) expressing C-KIT (CD117) [Abstract]. Proc. Am. Soc. Clin. Oncol. 20, 1 (2001).
Deininger, M. et al. The development of imatinib as a therapeutic agent for chronic myeloid leukemia. Blood 105, 2640–2653 (2005).
Deininger, M. W. & Druker, B. J. Specific targeted therapy of chronic myelogenous leukemia with imatinib. Pharmacol. Rev. 55, 401–423 (2003).
Sequist, L. V. et al. First-line gefitinib in patients with advanced non-small-cell lung cancer harboring somatic EGFR mutations. J. Clin. Oncol. 26, 2442–2449 (2008).
Thatcher, N. et al. Gefitinib plus best supportive care in previously treated patients with refractory advanced non-small-cell lung cancer: results from a randomised, placebo-controlled, multicentre study (Iressa Survival Evaluation in Lung Cancer). Lancet 366, 1527–1537 (2005).
Chang, A. et al. Gefitinib (IRESSA) in patients of Asian origin with refractory advanced non-small cell lung cancer: subset analysis from the ISEL study. J. Thorac. Oncol. 1, 847–855 (2006).
Clark, G. M. et al. Smoking history and epidermal growth factor receptor expression as predictors of survival benefit from erlotinib for patients with non-small-cell lung cancer in the National Cancer Institute of Canada Clinical Trials Group study BR.21. Clin. Lung Cancer 7, 389–394 (2006).
Tamura, K. et al. Multicentre prospective phase II trial of gefitinib for advanced non-small cell lung cancer with epidermal growth factor receptor mutations: results of the West Japan Thoracic Oncology Group trial (WJTOG0403). Br. J. Cancer 98, 907–914 (2008).
Amado, R. G. et al. Wild-type KRAS is required for panitumumab efficacy in patients with metastatic colorectal cancer. J. Clin. Oncol. 26, 1626–1634 (2008).
Morgillo, F. et al. Implication of the insulin-like growth factor-IR pathway in the resistance of non-small cell lung cancer cells to treatment with gefitinib. Clin. Cancer Res. 13, 2795–2803 (2007).
Dancey, J. E. & Chen, H. X. Strategies for optimizing combinations of molecularly targeted anticancer agents. Nat. Rev. Drug Discov. 5, 649–659 (2006).
Heinrich, M. C. et al. Sunitinib (SU) response in imatinib-resistant (IM-R) GIST correlates with KIT and PDGFRA mutation status [Abstract]. ASCO Meeting Abstracts 24, 9502 (2006).
Xu, J. et al. Human multidrug transporter ABCG2, a target for sensitizing drug resistance in cancer chemotherapy. Curr. Med. Chem. 14, 689–701 (2007).
Amery, W. & Dony, J. A clinical trial design avoiding undue placebo treatment. J. Clin. Pharmacol. 15, 674–679 (1975).
Kopec, J. A. et al. Randomized discontinuation trials: utility and efficiency. J. Clin. Epidemiol. 46, 959–971 (1993).
Ratain, M. J. et al. Phase II placebo-controlled randomized discontinuation trial of sorafenib in patients with metastatic renal cell carcinoma. J. Clin. Oncol. 24, 2505–2512 (2006).
Escudier, B. et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N. Engl. J. Med. 356, 125–134 (2007).
Stadler, W. M. et al. Successful implementation of the randomized discontinuation trial design: an application to the study of the putative antiangiogenic agent carboxyaminoimidazole in renal cell carcinoma—CALGB 69901. J. Clin. Oncol. 23, 3726–3732 (2005).
Freidlin, B. & Simon, R. Evaluation of randomized discontinuation design. J. Clin. Oncol. 23, 5094–5098 (2005).
Freidlin, B. & Simon, R. Adaptive signature design: an adaptive clinical trial design for generating and prospectively testing a gene expression signature for sensitive patients. Clin. Cancer Res. 11, 7872–7878 (2005).
Kabbinavar, F. et al. Phase II, randomized trial comparing bevacizumab plus fluorouracil (FU)/leucovorin (LV) with FU/LV alone in patients with metastatic colorectal cancer. J. Clin. Oncol. 21, 60–65 (2003).
Hurwitz, H. et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N. Engl. J. Med. 350, 2335–2342 (2004).
Kirkwood, J. M. et al. Interferon alfa-2b adjuvant therapy of high-risk resected cutaneous melanoma: the Eastern Cooperative Oncology Group Trial EST 1684. J. Clin. Oncol. 14, 7–17 (1996).
Cascinelli, N. et al. Effect of long-term adjuvant therapy with interferon alpha-2a in patients with regional node metastases from cutaneous melanoma: a randomised trial. Lancet 358, 866–869 (2001).
Eggermont, A. M. et al. EORTC 18961: Post-operative adjuvant ganglioside GM2-KLH21 vaccination treatment vs observation in stage II (T3-T4N0M0) melanoma: 2nd interim analysis led to an early disclosure of the results [Abstract]. ASCO Meeting Abstracts 26, 9004 (2008).
Kummar, S. et al. Compressing drug development timelines in oncology using phase '0' trials. Nat. Rev. Cancer 7, 131–139 (2007).
Murgo, A. J. et al. Designing phase 0 cancer clinical trials. Clin. Cancer Res. 14, 3675–3682 (2008).
FDA Center for Drug Evaluation and Research (CDER). Guidance for Industry, Investigators, and Reviewers Exploratory IND Studies [2006] http://www.fda.gov/cder/guidance/7086fnl.htm (accessed 25 September 2008).
Collins, J. M. Imaging and other biomarkers in early clinical studies: one step at a time or re-engineering drug development? J. Clin. Oncol. 23, 5417–5419 (2005).
Pegram, M. D. et al. Targeted therapy: wave of the future. J. Clin. Oncol. 23, 1776–1781 (2005).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Gutierrez, M., Kummar, S. & Giaccone, G. Next generation oncology drug development: opportunities and challenges. Nat Rev Clin Oncol 6, 259–265 (2009). https://doi.org/10.1038/nrclinonc.2009.38
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2009.38
This article is cited by
-
First-line single-agent regorafenib in frail patients with metastatic colorectal cancer: a pilot phase II study of the Spanish Cooperative Group for the Treatment of Digestive Tumours (TTD)
BMC Cancer (2019)
-
A review of new agents evaluated against pediatric acute lymphoblastic leukemia by the Pediatric Preclinical Testing Program
Leukemia (2016)
-
In vivo RAF signal transduction as a potential biomarker for sorafenib efficacy in patients with neuroendocrine tumours
British Journal of Cancer (2013)
-
Uncovering early, lineage-dependent effects of TPMT genotype in adult acute lymphoblastic leukemia by minimal residual disease
Leukemia (2013)
-
Möglichkeiten und Grenzen der stratifizierenden Medizin am Beispiel der Onkologie
Forum (2012)