Abstract
KRAS mutations may be predictive of resistance to anti-EGFR monoclonal-based therapy in patients with colorectal cancer (CRC). Screening for KRAS mutations in patients with CRC and non-small-cell lung cancer (NSCLC) may provide additional information on optimizing treatment options with targeted therapies. Only limited studies, however, have assessed the predictive value of KRAS mutations in response to conventional chemotherapy. We reviewed all relevant papers investigating the association of KRAS mutations and conventional chemotherapy-related outcome in NSCLC, CRC, and other solid tumors, both in the adjuvant and advanced settings. Our Review strongly suggests that KRAS mutations have no value in response prediction to conventional chemotherapy in NSCLC, CRC and other solid tumors. Therefore, KRAS mutations should not be used as molecular predictors of response to conventional chemotherapy.
Key Points
-
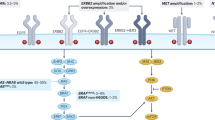
Activating RAS mutations occur in 30% of human cancers and are the most common gain-of-function mutations
-
KRAS mutations are molecular predictors for the lack of benefit of anti-EGFR monoclonal-based therapy in colorectal cancer
-
In lung cancer, colorectal cancer, and other solid tumors, KRAS mutations have no value for predicting response to conventional chemotherapy
-
KRAS mutations should not be used within the decision algorithm of chemotherapy
-
All these findings need to be confirmed in prospective trials with appropriate multivariate analysis taking into account other clinical and biological predictors
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Schubbert, S., Shannon, K. & Bollag, G. Hyperactive Ras in developmental disorders and cancer. Nat. Rev. Cancer 7, 295–308 (2007).
Rodenhuis, S. et al. Mutational activation of the K-ras oncogene and the effect of chemotherapy in advanced adenocarcinoma of the lung: a prospective study. J. Clin. Oncol. 15, 285–291 (1997).
Marks, J. L. et al. Prognostic and therapeutic implications of EGFR and KRAS mutations in resected lung adenocarcinoma. J. Thorac. Oncol. 3, 111–116 (2008).
Mascaux, C. et al. The role of RAS oncogene in survival of patients with lung cancer: a systematic review of the literature with meta-analysis. Br. J. Cancer 92, 131–139 (2005).
Eberhard, D. A. et al. Mutations in the epidermal growth factor receptor and in KRAS are predictive and prognostic indicators in patients with non-small-cell lung cancer treated with chemotherapy alone and in combination with erlotinib. J. Clin. Oncol. 23, 5900–5909 (2005).
Lievre, A. et al. KRAS mutation status is predictive of response to cetuximab therapy in colorectal cancer. Cancer Res. 66, 3992–3995 (2006).
Di Fiore, F. et al. Clinical relevance of KRAS mutation detection in metastatic colorectal cancer treated by cetuximab plus chemotherapy. Br. J. Cancer 96, 1166–1169 (2007).
Karapetis, C. S. et al. K-ras mutations and benefit from cetuximab in advanced colorectal cancer. N. Engl. J. Med. 359, 1757–1765 (2008).
Amado, R. G. et al. Wild-type KRAS is required for panitumumab efficacy in patients with metastatic colorectal cancer. J. Clin. Oncol. 26, 1626–1634 (2008).
Tsai, C. M. et al. Correlation of intrinsic chemoresistance of non-small-cell lung cancer cell lines with HER-2/neu gene expression but not with ras gene mutations. J. Natl Cancer Inst. 85, 897–901 (1993).
Kelland, L. The resurgence of platinum-based cancer chemotherapy. Nat. Rev. Cancer 7, 573–584 (2007).
Chin, K. V., Ueda, K., Pastan, I. & Gottesman, M. M. Modulation of activity of the promoter of the human MDR1 gene by Ras and p53. Science 255, 459–462 (1992).
Wishart, G. C., Plumb, J. A., Spandidos, D. A. & Kerr, D. J. H-ras infection in mink lung epithelial cells may induce 'atypical' multidrug resistance. Eur. J. Cancer 27, 673 (1991).
Yagüe, E. et al. Ability to acquire drug resistance arises early during the tumorigenesis process. Cancer Res. 67, 1130–1137 (2007).
Schmidt, C. J. & Hamer, D. H. Cell specificity and an effect of ras on human metallothionein gene expression. Proc. Natl Acad. Sci. USA 83, 3346–3350 (1986).
Isonishi, S. et al. Expression of the c-Ha-RAS oncogene in mouse NIH 3T3 cells induces resistance to cisplatin. Cancer Res. 51, 5903–5909 (1991).
Niimi, S. et al. Resistance to anticancer drugs in NIH3T3 cells transfected with c-myc and/or c-H-ras genes. Br. J. Cancer 63, 237–241 (1991).
Merkel, P., Khoury, N., Bertolotto, C. & Perfetti, R. Insulin and glucose regulate the expression of the DNA repair enzyme XPD. Mol. Cell. Endocrinol. 201, 75–85 (2003).
Lee-Kwon, W., Park, D. & Bernier, M. Involvement of the Ras/extracellular signal-regulated kinase signalling pathway in the regulation of ERCC-1 mRNA levels by insulin. Biochem. J. 331, 591–597 (1998).
Mabry, M. et al. v-Ha-ras oncogene insertion: a model for tumor progression of human small cell lung cancer. Proc. Natl Acad. Sci. USA 85, 6523–6527 (1988).
Kaufmann, S. H., Kalemkerian, G. P., Jasti, R. & Mabry, M. Effect of v-rasH on sensitivity of NCI-H82 human small cell lung cancer cells to cisplatin, etoposide, and camptothecin. Biochem. Pharmacol. 50, 1987–1993 (1995).
Rosell, R. et al. Mutated K-ras gene analysis in a randomized trial of preoperative chemotherapy plus surgery versus surgery in stage IIIA non-small cell lung cancer. Lung Cancer 12 (Suppl. 1), S59–S70 (1995).
Schiller, J. H. et al. Lack of prognostic significance of p53 and K-ras mutations in primary resected non-small-cell lung cancer on E4592: a Laboratory Ancillary Study on an Eastern Cooperative Oncology Group Prospective Randomized Trial of Postoperative Adjuvant Therapy. J. Clin. Oncol. 19, 448–457 (2001).
Tsao, M. S. et al. Prognostic and predictive importance of p53 and RAS for adjuvant chemotherapy in non small-cell lung cancer. J. Clin. Oncol. 25, 5240–5247 (2007).
Zalcman, G. et al. Use of RAS effector RASSF1A promoter gene methylation and chromosome 9p loss of heterozygosity (LOH) to predict progression-free survival (PFS) in perioperative chemotherapy (CT) phase III trial IFCT-0002 in resectable non-small cell lung cancer [abstract]. ASCO Meeting Abstracts 26, 7500 (2008).
Longley, D. B., Harkin, D. P. & Johnston, P. G. 5-fluorouracil: mechanisms of action and clinical strategies. Nat. Rev. Cancer 3, 330–338 (2003).
Klampfer, L. et al. Oncogenic Ras increases sensitivity of colon cancer cells to 5-FU-induced apoptosis. Oncogene 24, 3932–3941 (2005).
Agarwal, B. et al. Lovastatin augments apoptosis induced by chemotherapeutic agents in colon cancer cells. Clin. Cancer Res. 5, 2223–2239 (1999).
Tseng, Y. S. et al. Ha-ras overexpression mediated cell apoptosis in the presence of 5-fluorouracil. Exp. Cell Res. 288, 403–414 (2003).
Huang, Y., Horvath, C. M. & Waxman, S. Regrowth of 5-fluorouracil-treated human colon cancer cells is prevented by the combination of interferon gamma, indomethacin, and phenylbutyrate. Cancer Res. 60, 3200–3206 (2000).
Houghton, J. A., Ebanks, R., Harwood, F. G. & Tillman, D. M. Inhibition of apoptosis after thymineless stress is conferred by oncogenic K-Ras in colon carcinoma cells. Clin. Cancer Res. 4, 2841–2848 (1998).
Scanlon, K. I. et al. Ribozyme-mediated cleavage of c-fos mRNA reduces gene expression of DNA synthesis enzymes and metallothionein. Proc. Natl Acad. Sci. USA 88, 10591–10595 (1991).
Giovanella, B. C. et al. DNA topoisomerase I—targeted chemotherapy of human colon cancer in xenografts. Science 246, 1046–1048 (1989).
Markowitz, S. et al. Mutant K-ras oncogenes in colon cancers do not predict patient's chemotherapy response or survival. Clin. Cancer Res. 1, 441–445 (1995).
Bleeker, W. A. et al. Prognostic significance of K-ras and TP53 mutations in the role of adjuvant chemotherapy on survival in patients with Dukes C colon cancer. Dis. Colon Rectum 44, 358–363 (2001).
Ahnen, D. J. et al. Ki-ras mutation and p53 overexpression predict the clinical behavior of colorectal cancer: a Southwest Oncology Group study. Cancer Res. 58, 1149–1158 (1998).
Westra, J. L. et al. Determination of TP53 mutation is more relevant than microsatellite instability status for the prediction of disease-free survival in adjuvant-treated stage III colon cancer patients. J. Clin. Oncol. 23, 5635–5643 (2005).
Etienne-Grimaldi, M. C. et al. K-Ras mutations and treatment outcome in colorectal cancer patients receiving exclusive fluoropyrimidine therapy. Clin. Cancer Res. 14, 4830–4835 (2008).
Nemunaitis, J., Cox, J., Meyer, W., Courtney, A. & Mues, G. Irinotecan hydrochloride (CPT-11) resistance identified by K-ras mutation in patients with progressive colon cancer after treatment with 5-fluorouracil (5-FU). Am. J. Clin. Oncol. 20, 527–529 (1997).
Gnanasampanthan, G., Elsaleh, H., McCaul, K. & Iacopetta, B. Ki-RAS mutation type and the survival benefit from adjuvant chemotherapy in Dukes' C colorectal cancer. J. Pathol. 195, 543–548 (2001).
Van Cutsem, E. et al. Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer. N. Engl. J. Med. 360, 1408–1417 (2009).
Bokemeyer, C. et al. KRAS status and efficacy of first-line treatment of patients with metastatic colorectal cancer (mCRC) with FOLFOX with or without cetuximab: The OPUS experience [abstract]. ASCO Meeting Abstracts 26, 4000 (2008).
Gallagher, S. J. et al. p16INK4a expression and absence of activated B-RAF are independent predictors of chemosensitivity in melanoma tumors. Neoplasia 10, 1231–1239 (2008).
Dergham, S. T., Dugan, M. C., Sarkar, F. H. & Vaitkevicius, V. K. Molecular alterations associated with improved survival in pancreatic cancer patients treated with radiation or chemotherapy. J. Hepatobiliary Pancreat. Surg. 5, 269–272 (1998).
Bissada, E. et al. Prevalence of KRAS codon 12 mutations in locally advanced head and neck squamous cell carcinoma and influence with regards to response to chemoradiation therapy [abstract]. ASCO Meeting Abstracts 26, 17005 (2008).
Hachisuga, T. et al. K-ras mutation in the endometrium of tamoxifen-treated breast cancer patients, with a comparison of tamoxifen and toremifene. Br. J. Cancer. 92, 1098–1103 (2005).
Wallén, M., Tomás, E., Visakorpi, T., Holli, K. & Mäenpää, J. Endometrial K-ras mutations in postmenopausal breast cancer patients treated with adjuvant tamoxifen or toremifene. Cancer Chemother. Pharmacol. 55, 343–346 (2005).
Wistuba, I. I. & Gazdar, A. F. Gallbladder cancer: lessons from a rare tumour. Nat. Rev. Cancer 4, 695–706 (2004).
Verslype, C., Prenen, H. & Van Cutsem, E. The role of chemotherapy in biliary tract carcinoma. HPB (Oxford) 10, 164–167 (2008).
Ince, W. L. et al. Association of k-ras, b-raf, and p53 status with the treatment effect of bevacizumab. J. Natl Cancer Inst. 97, 981–989 (2005).
Rosty, C. et al. Determination of microsatellite instability, p53 and K-RAS mutations in hepatic metastases from patients with colorectal cancer: relationship with response to 5-fluorouracil and survival. Int. J. Cancer 95, 162–167 (2001).
Biomarkers Definitions Working Group. Biomarkers and surrogate endpoints: preferred definitions and conceptual framework. Clin. Pharmacol. Ther. 69, 89–95 (2001).
Michiels, S., Koscielny, S. & Hill, C. Interpretation of microarray data in cancer. Br. J. Cancer 96, 1155–1158 (2007).
Acknowledgements
The authors thank H. Lord for her help in editing the manuscript. Y. Loriot and P. Mordant contributed equally to this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Loriot, Y., Mordant, P., Deutsch, E. et al. Are RAS mutations predictive markers of resistance to standard chemotherapy?. Nat Rev Clin Oncol 6, 528–534 (2009). https://doi.org/10.1038/nrclinonc.2009.106
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2009.106
This article is cited by
-
Mechanisms of chemotherapeutic resistance and the application of targeted nanoparticles for enhanced chemotherapy in colorectal cancer
Journal of Nanobiotechnology (2022)
-
RAS oncogenic activity predicts response to chemotherapy and outcome in lung adenocarcinoma
Nature Communications (2022)
-
Actionable Mutation Profiles of Non-Small Cell Lung Cancer patients from Vietnamese population
Scientific Reports (2020)
-
(−)-Epigallocatechin-3-gallate derivatives combined with cisplatin exhibit synergistic inhibitory effects on non-small-cell lung cancer cells
Cancer Cell International (2019)
-
New oncogenes drivers in lung cancer—new therapeutic targets
Current Pulmonology Reports (2016)