Key Points
-
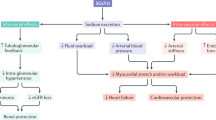
Heart failure is now the most common cardiovascular complication of type 2 diabetes mellitus (T2DM), with its incidence exceeding that of myocardial infarction or stroke
-
Since 2008, the FDA and the European Medicines Agency require proof of cardiovascular safety for all glucose-lowering agents, resulting in an increase in trials focusing on the cardiovascular effects of T2DM drugs
-
No large randomized, controlled trials have been conducted to examine the effects of insulin, metformin, or sulfonylureas in patients with established heart failure
-
Use of thiazolidinediones is cautioned in all patients with signs and symptoms of heart failure, and they are contraindicated in patients with established heart failure
-
Different types of dipeptidyl peptidase 4 inhibitors are associated with varying levels of heart failure risk, possibly owing to different selectivity for glucagon-like peptide 1 receptor agonists
-
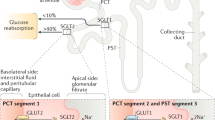
Sodium/glucose cotransporter 2 inhibitors (specifically empagliflozin) are associated with significant reductions in heart failure hospitalization and prevention of heart failure-related and arrhythmia-related deaths
Abstract
Heart failure is one of the most common comorbidities of diabetes mellitus. Glucose-lowering therapies that can prevent heart failure or improve outcomes in patients with established heart failure are of critical importance among those with type 2 diabetes. Several types of glucose-lowering drugs have been assessed in this setting. Metformin has been shown to modestly improve the outcomes of patients with heart failure, whereas the effect of insulin in those with established heart failure is less clear. The effect of sulfonylureas on improving heart failure is controversial; observational reports have suggested that they are harmful in these patients, but these data have not been confirmed in randomized, controlled trials. Thiazolidinediones are contraindicated in patients with established heart failure and have also been known to cause heart failure. Furthermore, certain dipeptidyl peptidase 4 inhibitors seem to increase heart failure hospitalization. The effects of glucagon-like peptide 1 receptor agonists might differ in patients with or without established heart failure, particularly those with decompensated heart failure with a reduced ejection fraction. However, perhaps the most important finding has been that sodium/glucose cotransporter 2 (SGLT2; also known as SLC5A2) inhibitors reduce heart failure hospitalizations and, in the case of empagliflozin, markedly reduce the rate of cardiovascular death. Given the known neutral (or even harmful) effects of other glucose-lowering drugs on heart failure outcomes, SGLT2 inhibitors might well be considered the drug class of choice in patients with diabetes and heart failure, or in those at high risk of developing heart failure.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Cohen-Solal, A., Beauvais, F. & Logeart, D. Heart failure and diabetes mellitus: epidemiology and management of an alarming association. J. Card. Fail. 14, 615–625 (2008).
McMurray, J. J. et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N. Engl. J. Med. 371, 993–1004 (2014).
Kannel, W. B., Hjortland, M. & Castelli, W. P. Role of diabetes in congestive heart failure: the Framingham study. Am. J. Cardiol. 34, 29–34 (1974).
Adams, K. F. Jr. et al. Characteristics and outcomes of patients hospitalized for heart failure in the United States: rationale, design, and preliminary observations from the first 100,000 cases in the Acute Decompensated Heart Failure National Registry (ADHERE). Am. Heart J. 149, 209–216 (2005).
Kristensen, S. L. et al. Risk related to pre–diabetes mellitus and diabetes mellitus in heart failure with reduced ejection fraction. Insights from prospective comparison of ARNI with ACEI to determine impact on global mortality and morbidity in heart failure trial. Heart Fail. 9, e002560 (2016).
Parving, H.-H. et al. Cardiorenal end points in a trial of aliskiren for type 2 diabetes. N. Engl. J. Med. 367, 2204–2213 (2012).
Kannel, W. B. & McGee, D. L. Diabetes and cardiovascular disease. The Framingham study. JAMA 241, 2035–2038 (1979).
Mak, K. H. & Topol, E. J. Emerging concepts in the management of acute myocardial infarction in patients with diabetes mellitus. J. Am. Coll. Cardiol. 35, 563–568 (2000).
Devereux, R. B. et al. Impact of diabetes on cardiac structure and function: the strong heart study. Circulation 101, 2271–2276 (2000).
Abbott, R. D., Donahue, R. P., Kannel, W. B. & Wilson, P. W. The impact of diabetes on survival following myocardial infarction in men versus women. The Framingham Study. JAMA 260, 3456–3460 (1988).
Bella, J. N. et al. Separate and joint effects of systemic hypertension and diabetes mellitus on left ventricular structure and function in American Indians (the Strong Heart Study). Am. J. Cardiol. 87, 1260–1265 (2001).
Palmieri, V. et al. Effect of type 2 diabetes mellitus on left ventricular geometry and systolic function in hypertensive subjects: Hypertension Genetic Epidemiology Network (HyperGEN) study. Circulation 103, 102–107 (2001).
Struthers, A. D. & Morris, A. D. Screening for and treating left-ventricular abnormalities in diabetes mellitus: a new way of reducing cardiac deaths. Lancet 359, 1430–1432 (2002).
Di Bonito, P. et al. Early detection of diabetic cardiomyopathy: usefulness of tissue Doppler imaging. Diabet Med. 22, 1720–1725 (2005).
Liu, J. E. et al. The impact of diabetes on left ventricular filling pattern in normotensive and hypertensive adults: the Strong Heart Study. J. Am. Coll. Cardiol. 37, 1943–1949 (2001).
Kasznicki, J. & Drzewoski, J. Heart failure in the diabetic population — pathophysiology, diagnosis and management. Arch. Med. Sci. 10, 546–556 (2014).
Fang, Z. Y., Prins, J. B. & Marwick, T. H. Diabetic cardiomyopathy: evidence, mechanisms, and therapeutic implications. Endocr. Rev. 25, 543–567 (2004).
Selvin, E. et al. Diabetes mellitus, prediabetes, and incidence of subclinical myocardial damage. Circulation 130, 1374–1382 (2014).
Bertoni, A. G. et al. Heart failure prevalence, incidence, and mortality in the elderly with diabetes. Diabetes Care 27, 699–703 (2004).
Demant, M. N. et al. Association of heart failure severity with risk of diabetes: a Danish nationwide cohort study. Diabetologia 57, 1595–1600 (2014).
Aguilar, D., Bozkurt, B., Ramasubbu, K. & Deswal, A. Relationship of hemoglobin A1C and mortality in heart failure patients with diabetes. J. Am. Coll. Cardiol. 54, 422–428 (2009).
Turnbull, F. M. et al. Intensive glucose control and macrovascular outcomes in type 2 diabetes. Diabetologia 52, 2288–2298 (2009).
Turner, R. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 352, 837–853 (1998).
Group, T. A. C. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N. Engl. J. Med. 358, 2560–2572 (2008).
Duckworth, W. et al. Glucose control and vascular complications in veterans with type 2 diabetes. N. Engl. J. Med. 360, 129–139 (2009).
Group, T. A. S. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N. Engl. J. Med. 362, 1575–1585 (2010).
Eshaghian, S., Horwich, T. B. & Fonarow, G. C. An unexpected inverse relationship between HbA1c levels and mortality in patients with diabetes and advanced systolic heart failure. Am. Heart J. 151, 91.e1–91.e6 (2006).
Action to Control Cardiovascular Risk in Diabetes Study Group. Effects of intensive glucose lowering in type 2 diabetes. N. Engl. J. Med. 358, 2545–2559 (2008).
Stades, A. M., Heikens, J. T., Erkelens, D. W., Holleman, F. & Hoekstra, J. B. Metformin and lactic acidosis: cause or coincidence? A review of case reports. J. Intern. Med. 255, 179–187 (2004).
Eurich, D. T. et al. Metformin treatment in diabetes and heart failure: when academic equipoise meets clinical reality. Trials 10, 12 (2009).
Eurich, D. T., Majumdar, S. R., McAlister, F. A., Tsuyuki, R. T. & Johnson, J. A. Improved clinical outcomes associated with metformin in patients with diabetes and heart failure. Diabetes Care 28, 2345–2351 (2005).
Eurich, D. T. et al. Comparative safety and effectiveness of metformin in patients with diabetes mellitus and heart failure: systematic review of observational studies involving 34,000 patients. Circ. Heart Fail. 6, 395–402 (2013).
Bailey, C. J. & Turner, R. C. Metformin. N. Engl. J. Med. 334, 574–579 (1996).
Giamouzis, G., Triposkiadis, F. & Butler, J. Metformin use in patients with diabetes mellitus and heart failure: friend or foe? J. Card. Fail. 16, 207–210 (2010).
Leifer, A. A case of insulin edema. J. Am. Med. Assoc. 90, 610–611 (1928).
Skøtt, P. et al. Effects of insulin on kidney function and sodium excretion in healthy subjects. Diabetologia 32, 694–699 (1989).
DeFronzo, R. A., Cooke, C. R., Andres, R., Faloona, G. R. & Davis, P. J. The effect of insulin on renal handling of sodium, potassium, calcium, and phosphate in man. J. Clin. Invest. 55, 845–855 (1975).
Investigators, T. O. T. Basal insulin and cardiovascular and other outcomes in dysglycemia. N. Engl. J. Med. 367, 319–328 (2012).
Group, T. B. D. S. A. Randomized trial of therapies for type 2 diabetes and coronary artery disease. N. Engl. J. Med. 360, 2503–2515 (2009).
Pocock, S. J. et al. Predictors of mortality and morbidity in patients with chronic heart failure. Eur. Heart J. 27, 65–75 (2006).
Smooke, S., Horwich, T. B. & Fonarow, G. C. Insulin-treated diabetes is associated with a marked increase in mortality in patients with advanced heart failure. Am. Heart J. 149, 168–174 (2005).
Masoudi, F. A. et al. Thiazolidinediones, metformin, and outcomes in older patients with diabetes and heart failure: an observational study. Circulation 111, 583–590 (2005).
Bell, D. S. H. Do sulfonylurea drugs increase the risk of cardiac events? CMAJ 174, 185–186 (2006).
Simpson, S. H., Majumdar, S. R., Tsuyuki, R. T., Eurich, D. T. & Johnson, J. A. Dose-response relation between sulfonylurea drugs and mortality in type 2 diabetes mellitus: a population-based cohort study. CMAJ 174, 169–174 (2006).
Scognamiglio, R. et al. Effects of treatment with sulfonylurea drugs or insulin on ischemia-induced myocardial dysfunction in type 2 diabetes. Diabetes 51, 808–812 (2002).
Bijlstra, P. J., Lutterman, J. A., Russel, F. G., Thien, T. & Smits, P. Interaction of sulphonylurea derivatives with vascular ATP-sensitive potassium channels in humans. Diabetologia 39, 1083–1090 (1996).
Bain, S. et al. Cardiovascular events and all-cause mortality associated with sulphonylureas compared with other antihyperglycaemic drugs: a Bayesian meta-analysis of survival data. Diabetes Obes. Metab. 19, 329–335 (2017).
Tzoulaki, I. et al. Risk of cardiovascular disease and all cause mortality among patients with type 2 diabetes prescribed oral antidiabetes drugs: retrospective cohort study using UK general practice research database. BMJ 339, b4731 (2009).
Pantalone, K. M. et al. The risk of developing coronary artery disease or congestive heart failure, and overall mortality, in type 2 diabetic patients receiving rosiglitazone, pioglitazone, metformin, or sulfonylureas: a retrospective analysis. Acta Diabetol. 46, 145–154 (2009).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01243424 (2017).
Yki-Jarvinen, H. Thiazolidinediones. N. Engl. J. Med. 351, 1106–1118 (2004).
Nolan, J. J., Ludvik, B., Beerdsen, P., Joyce, M. & Olefsky, J. Improvement in glucose tolerance and insulin resistance in obese subjects treated with troglitazone. N. Engl. J. Med. 331, 1188–1193 (1994).
Seki, G. et al. Role of renal proximal tubule transport in thiazolidinedione-induced volume expansion. World J. Nephrol. 1, 146–150 (2012).
Yang, T. & Soodvilai, S. Renal and vascular mechanisms of thiazolidinedione-induced fluid retention. PPAR Res. 2008, 943614 (2008).
Raskin, P. et al. A randomized trial of rosiglitazone therapy in patients with inadequately controlled insulin-treated type 2 diabetes. Diabetes Care 24, 1226–1232 (2001).
Investigators, D. T. et al. Effects of ramipril and rosiglitazone on cardiovascular and renal outcomes in people with impaired glucose tolerance or impaired fasting glucose: results of the Diabetes REduction Assessment with ramipril and rosiglitazone Medication (DREAM) trial. Diabetes Care 31, 1007–1014 (2008).
Dormandy, J. A. et al. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone Clinical Trial in macroVascular Events): A randomised controlled trial. Lancet 366, 1279–1289 (2005).
Dargie, H. J. et al. A randomized, placebo-controlled trial assessing the effects of rosiglitazone on echocardiographic function and cardiac status in type 2 diabetic patients with new york heart association functional class I or II heart failure. J. Am. Coll. Cardiol. 49, 1696–1704 (2007).
Lago, R. M., Singh, P. P. & Nesto, R. W. Congestive heart failure and cardiovascular death in patients with prediabetes and type 2 diabetes given thiazolidinediones: a meta-analysis of randomised clinical trials. Lancet 370, 1129–1136 (2007).
Lincoff, A. et al. Effect of aleglitazar on cardiovascular outcomes after acute coronary syndrome in patients with type 2 diabetes mellitus: the alecardio randomized clinical trial. JAMA 311, 1515–1525 (2014).
Nissen, S. E., Wolski, K. & Topol, E. J. Effect of muraglitazar on death and major adverse cardiovascular events in patients with type 2 diabetes mellitus. JAMA 294, 2581–2586 (2005).
Kernan, W. N. et al. Pioglitazone after ischemic stroke or transient ischemic attack. N. Engl. J. Med. 374, 1321–1331 (2016).
Nesto, R. W. et al. Thiazolidinedione use, fluid retention, and congestive heart failure: a consensus statement from the American Heart Association and American Diabetes Association. Circulation 108, 2941–2948 (2003).
Thrainsdottir, I., Malmberg, K., Olsson, A., Gutniak, M. & Rydén, L. Initial experience with GLP-1 treatment on metabolic control and myocardial function in patients with type 2 diabetes mellitus and heart failure. Diabetes Vasc. Dis. Res. 1, 40–43 (2004).
Liu, F. P. et al. Glucagon-like peptide 1 receptor agonist therapy is more efficacious than insulin glargine for poorly controlled type 2 diabetes: a systematic review and meta-analysis. J. Diabetes 7, 322–328 (2015).
Monami, M., Dicembrini, I., Nardini, C., Fiordelli, I. & Mannucci, E. Effects of glucagon-like peptide-1 receptor agonists on cardiovascular risk: a meta-analysis of randomized clinical trials. Diabetes, Obes. Metabolism 16, 38–47 (2014).
Vilsbøll, T., Christensen, M., Junker, A. E., Knop, F. K. & Gluud, L. L. Effects of glucagon-like peptide-1 receptor agonists on weight loss: systematic review and meta-analyses of randomised controlled trials. BMJ 344, d7771 (2012).
Marso, S. P. et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N. Engl. J. Med. 375, 1834–1844 (2016).
Marso, S. P. et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N. Engl. J. Med. 375, 311–322 (2016).
Bhashyam, S. et al. Glucagon-like peptide-1 increases myocardial glucose uptake via p38alpha MAP kinase-mediated, nitric oxide-dependent mechanisms in conscious dogs with dilated cardiomyopathy. Circ. Heart Fail. 3, 512–521 (2010).
Nikolaidis, L. A. et al. Recombinant glucagon-like peptide-1 increases myocardial glucose uptake and improves left ventricular performance in conscious dogs with pacing-induced dilated cardiomyopathy. Circulation 110, 955–961 (2004).
Margulies, K. B. et al. Effects of liraglutide on clinical stability among patients with advanced heart failure and reduced ejection fraction: a randomized clinical trial. JAMA 316, 500–508 (2016).
Jorsal, A. et al. Effect of liraglutide, a glucagon-like peptide-1 analogue, on left ventricular function in stable chronic heart failure patients with and without diabetes (LIVE) — a multicentre, double-blind, randomised, placebo-controlled trial. Eur. J. Heart Fail. 19, 69–77 (2017).
Lorenz, M. et al. Differential effects of glucagon-like peptide-1 receptor agonists on heart rate. Cardiovasc. Diabetol. 16, 6 (2017).
Meier, J. J. et al. Contrasting effects of lixisenatide and liraglutide on postprandial glycemic control, gastric emptying, and safety parameters in patients with type 2 diabetes on optimized insulin glargine with or without metformin: a randomized, open-label trial. Diabetes Care 38, 1263–1273 (2015).
Bohm, M. et al. Heart rate as a risk factor in chronic heart failure (SHIFT): the association between heart rate and outcomes in a randomised placebo-controlled trial. Lancet 376, 886–894 (2010).
Deacon, C. F. et al. Both subcutaneously and intravenously administered glucagon-like peptide I are rapidly degraded from the NH2-terminus in type II diabetic patients and in healthy subjects. Diabetes 44, 1126–1131 (1995).
Inzucchi, S. E. et al. Management of hyperglycaemia in type 2 diabetes: a patient-centered approach. Position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 55, 1577–1596 (2012).
Scirica, B. M. et al. Heart failure, saxagliptin, and diabetes mellitus: Observations from the SAVOR-TIMI 53 randomized trial. Circulation 130, 1579–1588 (2014).
dos Santos, L. et al. Circulating dipeptidyl peptidase IV activity correlates with cardiac dysfunction in human and experimental heart failure. Circ. Heart Fail. 6, 1029–1038 (2013).
Shigeta, T. et al. Dipeptidyl peptidase-4 modulates left ventricular dysfunction in chronic heart failure via angiogenesis-dependent and -independent actions. Circulation 126, 1838–1851 (2012).
Zannad, F., Cannon, C. P. & Cushman, W. C. Heart failure and mortality outcomes in patients with type 2 diabetes taking alogliptin versus placebo in EXAMINE: a multicentre randomised double-blind trial. Lancet 385, 2067–2076 (2015).
McGuire, D. K. et al. Association between sitagliptin use and heart failure hospitalization and related outcomes in type 2 diabetes mellitus: secondary analysis of a randomized clinical trial. JAMA Cardiol. 1, 126–135 (2016).
Green, J. B. et al. Effect of sitagliptin on cardiovascular outcomes in type 2 diabetes. N. Engl. J. Med. 373, 232–242 (2015).
Kankanala, S. R. et al. Cardiovascular safety of dipeptidyl peptidase-4 inhibitors: recent evidence on heart failure. Am. J. Transl Res. 8, 2450–2458 (2016).
McMurray, J. J. V. et al. Effects of vildagliptin on ventricular function in patients with type 2 diabetes mellitus and heart failure: a randomized placebo-controlled trial. JACC Heart Fail. http://dx.doi.org/10.1016/j.jchf.2017.08.004 (2017).
McMurray, J. The Vildagliptin in Ventricular Dysfunction Diabetes trial (VIVIDD) in European Heart Failure Congress (Lisbon, 2013).
Scirica, B. M. The safety of dipeptidyl peptidase 4 inhibitors and the risk for heart failure. JAMA Cardiol. 1, 123–125 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01897532 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02917031 (2017).
Inzucchi, S. E. et al. SGLT-2 inhibitors and cardiovascular risk: proposed pathways and review of ongoing outcome trials. Diab. Vasc. Dis. Res. 12, 90–100 (2015).
Neal, B. et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N. Engl. J. Med. 377, 644–657 (2017).
Zinman, B. et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N. Engl. J. Med. 373, 2117–2128 (2015).
Fitchett, D. et al. Heart failure outcomes with empagliflozin in patients with type 2 diabetes at high cardiovascular risk: results of the EMPA-REG OUTCOME® trial. Eur. Heart J. 37, 1526–1534 (2016).
Wanner, C. et al. Empagliflozin and progression of kidney disease in type 2 diabetes. N. Engl. J. Med. 375, 323–334 (2016).
Neal, B. et al. Rationale, design, and baseline characteristics of the Canagliflozin Cardiovascular Assessment Study (CANVAS) — a randomized placebo-controlled trial. Am. Heart J. 166, 217–223.e11 (2013).
Kosiborod, M. et al. Lower risk of heart failure and death in patients initiated on sglt-2 inhibitors versus other glucose-lowering drugs: the CVD-REAL study. Circulation 136, 249–259 (2017).
Scheerer, M. et al. Gesamtereignisraten für die hospitalisierung bei herzinsuffizienz (HHI) bei neueinstellung auf SGLT-2-hemmer im vergleich zu anderen antidiabetika–daten aus dem klinischen alltag aus Deutschland, Schweden und Norwegen mit mehr als 45.000 typ 2 diabetes patienten (CVD-Real). Diabetologie und Stoffwechsel 12, 222–228 (2017).
[No authors listed.] FDA approves Jardiance to reduce cardiovascular death in adults with type 2 diabetes. U.S. Food and Drug Administration http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm531517.htm (2016).
Husten, L. CardioBrief: Specialty rift brewing over empagliflozin. MedPage Today http://www.medpagetoday.com/cardiology/cardiobrief/58907 (2016).
Oelze, M. et al. The sodium-glucose co-transporter 2 inhibitor empagliflozin improves diabetes-induced vascular dysfunction in the streptozotocin diabetes rat model by interfering with oxidative stress and glucotoxicity. PLoS ONE 9, e112394 (2014).
Lambers Heerspink, H. J., de Zeeuw, D., Wie, L., Leslie, B. & List, J. Dapagliflozin a glucose-regulating drug with diuretic properties in subjects with type 2 diabetes. Diabetes Obes. Metab. 15, 853–862 (2013).
Mudaliar, S., Alloju, S. & Henry, R. R. Can a shift in fuel energetics explain the beneficial cardiorenal outcomes in the EMPA-REG OUTCOME study? A unifying hypothesis. Diabetes Care 39, 1115–1122 (2016).
Sattar, N., McLaren, J., Kristensen, S. L., Preiss, D. & McMurray, J. J. SGLT2 Inhibition and cardiovascular events: why did EMPA-REG Outcomes surprise and what were the likely mechanisms? Diabetologia 59, 1333–1339 (2016).
Januzzi, J. L. et al. Effects of Canagliflozin on cardiovascular biomarkers in older adults with type 2 diabetes. J. Am. Coll. Cardiol. 70, 704–712 (2017).
Ponikowski, P. et al. 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. J. Heart Fail. 18, 891–975 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03057951 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03057977 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03036124 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02653482 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03030222 (2017).
Nissen, S. E. & Wolski, K. Effect of rosiglitazone on the risk of myocardial infarction and death from cardiovascular causes. N. Engl. J. Med. 356, 2457–2471 (2007).
[No authors listed.] Guideline on clinical investigation of medicinal products in the treatment or prevention of diabetes mellitus. U.S. Food and Drug Administration http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/ucm071627.pdf (2008).
[No authors listed.] Guideline on clinical investigation of medicinal products in the treatment or prevention of diabetes mellitus. European Medicines Agency http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2012/06/WC500129256.pdf (2012).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01730534 (2017).
Pfeffer, M. A. et al. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N. Engl. J. Med. 373, 2247–2257 (2015).
Author information
Authors and Affiliations
Contributions
Both authors researched data for the article, discussed the content, wrote the manuscript, and reviewed and edited it before submission.
Corresponding author
Ethics declarations
Competing interests
M.K. declares that he is on the advisory boards for Amgen, AstraZeneca, Boehringer Ingelheim, Eisai, Glytec, GSK, Merck, Novo Nordisk, Sanofi, and ZS Pharma; is a consultant for AstraZeneca, Sanofi, and ZS Pharma; and has received research grants from AstraZeneca and Boehringer Ingelheim. M.N. declares no competing interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Nassif, M., Kosiborod, M. Effect of glucose-lowering therapies on heart failure. Nat Rev Cardiol 15, 282–291 (2018). https://doi.org/10.1038/nrcardio.2017.211
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2017.211
This article is cited by
-
Association between serum chloride and in-hospital mortality in congestive heart failure with diabetes: Data from the MIMIC-IV database
Journal of Diabetes & Metabolic Disorders (2023)
-
Cardioprotective mechanism of SGLT2 inhibitor against myocardial infarction is through reduction of autosis
Protein & Cell (2022)
-
The effects of antidiabetic agents on heart failure
Netherlands Heart Journal (2022)
-
Herzinsuffizienzprotektion bei Patienten mit Diabetes mellitus Typ 2 durch SGLT2-Inhibitoren – Evidenzlage und mögliche Mechanismen
Herz (2021)
-
GLP-1 mediated diuresis and natriuresis are blunted in heart failure and restored by selective afferent renal denervation
Cardiovascular Diabetology (2020)