Key Points
-
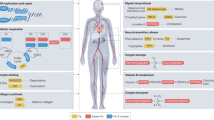
Iron deficiency has detrimental effects in patients with coronary artery disease, heart failure (HF), pulmonary hypertension, and possibly in patients undergoing cardiac surgery
-
One-third of all patients with HF and more than one-half of patients with pulmonary hypertension present with iron deficiency
-
Improved exercise capacity, quality of life, or both have been shown after iron administration in patients with iron deficiency and HF or pulmonary hypertension
-
Recipients of cardiac transplantations should probably not be treated with intravenous iron preparations
Abstract
Iron deficiency affects up to one-third of the world's population, and is particularly common in elderly individuals and those with certain chronic diseases. Iron excess can be detrimental in cardiovascular illness, and research has now also brought anaemia and iron deficiency into the focus of cardiovascular medicine. Data indicate that iron deficiency has detrimental effects in patients with coronary artery disease, heart failure (HF), and pulmonary hypertension, and possibly in patients undergoing cardiac surgery. Around one-third of all patients with HF, and more than one-half of patients with pulmonary hypertension, are affected by iron deficiency. Patients with HF and iron deficiency have shown symptomatic improvements from intravenous iron administration, and some evidence suggests that these improvements occur irrespective of the presence of anaemia. Improved exercise capacity has been demonstrated after iron administration in patients with pulmonary hypertension. However, to avoid iron overload and T-cell activation, it seems that recipients of cardiac transplantations should not be treated with intravenous iron preparations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
DeMaeyer, E. & Adiels-Tegman, M. The prevalence of anaemia in the world. World Health Stat. Q. 38, 302–316 (1985).
Stoltzfus, R. Defining iron-deficiency anemia in public health terms: a time for reflection. J. Nutr. 131, 565S–567S (2001).
Lundqvist, H. & Sjöberg, F. Food interaction of oral uptake of iron / a clinical trial using 59Fe. Arzneimittelforschung 57, 401–416 (2007).
Andrews, N. C. & Schmidt, P. J. Iron homeostasis. Annu. Rev. Physiol. 69, 69–85 (2007).
Andrews, N. C. Disorders of iron metabolism. N. Engl. J. Med. 341, 1986–1995 (1999).
Zimmermann, M. B. The influence of iron status on iodine utilization and thyroid function. Annu. Rev. Nutr. 26, 367–389 (2006).
Moos, T. Brain iron homeostasis. Dan. Med. Bull. 49, 279–301 (2002).
Christian, P. et al. Prenatal micronutrient supplementation and intellectual and motor function in early school-aged children in Nepal. JAMA 304, 2716–2723 (2010).
Fairweather-Tait, S. J., Wawer, A. A., Gillings, R., Jennings, A. & Myint, P. K. Iron status in the elderly. Mech. Ageing Dev. 136–137, 22–28 (2014).
Finch, C. A. et al. Iron deficiency in the rat. Physiological and biochemical studies of muscle dysfunction. J. Clin. Invest. 58, 447–453 (1976).
Beard, J. L. Iron biology in immune function, muscle metabolism and neuronal functioning. J. Nutr. 131, 568S–579S (2001).
Silverberg, D. S. et al. The use of subcutaneous erythropoietin and intravenous iron for the treatment of the anemia of severe, resistant congestive heart failure improves cardiac and renal function and functional cardiac class, and markedly reduces hospitalizations. J. Am. Coll. Cardiol. 35, 1737–1744 (2000).
van Veldhuisen, D. J., Anker, S. D., Ponikowski, P. & Macdougall, I. C. Anemia and iron deficiency in heart failure: mechanisms and therapeutic approaches. Nat. Rev. Cardiol. 8, 485–493 (2011).
Sullivan, J. L. Iron and the sex difference in heart disease risk. Lancet 317, 1293–1294 (1981).
Ganz, T. & Nemeth, E. Iron imports. IV. Hepcidin and regulation of body iron metabolism. Am. J. Physiol. Gastrointest. Liver Physiol. 290, G199–G203 (2006).
Jankowska, E. A, von Haehling, S., Anker, S. D., Macdougall, I. C. & Ponikowski, P. P. Iron deficiency and heart failure: diagnostic dilemmas and therapeutic perspectives. Eur. Heart J. 34, 816–829 (2013).
Monsen, E. R. Iron and absorption: dietary factors which impact iron bioavailability. J. Am. Diet. Assoc. 88, 786–790 (1988).
Nemeth, E. & Ganz, T. The role of hepcidin in iron metabolism. Acta Haematol. 122, 78–86 (2009).
Schaefer, B., Effenberger, M. & Zoller, H. Iron metabolism in transplantation. Transpl. Int. 27, 1109–1117 (2014).
Cook, J. D. Diagnosis and management of iron-deficiency anaemia. Best Pract. Res. Clin. Haematol. 18, 319–332 (2005).
Geisser, P. Safety and efficacy of iron(III)-hydroxide polymaltose complex / a review of over 25 years experience. Arzneimittelforschung 57, 439–452 (2007).
von Haehling, S & Anker, S. D. in Iron Deficiency and Anaemia in Heart Failure 2nd edn (Anker, S. D. & von Haehling, S.) 68–83 (UNI-MED, 2012).
Auerbach, M. & Ballard, H. Clinical use of intravenous iron: administration, efficacy, and safety. Hematology Am. Soc. Hematol. Educ. Program 2010, 338–347 (2010).
Lauffer, R. B. Iron depletion and coronary disease. Am. Heart J. 119, 1448–1449 (1990).
Salonen, J. T. et al. High stored iron levels are associated with excess risk of myocardial infarction in eastern Finnish men. Circulation 86, 803–811 (1992).
de Valk, B. & Marx, J. J. Iron, atherosclerosis, and ischemic heart disease. Arch. Intern. Med. 159, 1542–1548 (1999).
Ma, J. & Stampfer, M. J. Body iron stores and coronary heart disease. Clin. Chem. 48, 601–603 (2002).
Danesh, J. & Appleby, P. Coronary heart disease and iron status: meta-analyses of prospective studies. Circulation 99, 852–854 (1999).
Wood, R. J. The iron-heart disease connection: is it dead or just hiding? Ageing Res. Rev. 3, 355–367 (2004).
Das De, S., Krishna, S. & Jethwa, A. Iron status and its association with coronary heart disease: systematic review and meta-analysis of prospective studies. Atherosclerosis 238, 296–303 (2015).
Kang, P., Liu, T., Tian, C., Zhou, Y. & Jia, C. Association of total iron binding capacity with coronary artery disease. Clin. Chim. Acta 413, 1424–1429 (2012).
Ascherio, A., Rimm, E. B., Giovannucci, E., Willett, W. C. & Stampfer, M. J. Blood donations and risk of coronary heart disease in men. Circulation 103, 52–57 (2001).
Grammer, T. B. et al. Hemoglobin, iron metabolism and angiographic coronary artery disease (The Ludwigshafen Risk and Cardiovascular Health Study). Atherosclerosis 236, 292–300 (2014).
Ponikowska, B. et al. Iron status and survival in diabetic patients with coronary artery disease. Diabetes Care 36, 4147–4156 (2013).
Jankowska, E. A. et al. Bone marrow iron depletion is common in patients with coronary artery disease. Int. J. Cardiol. 182, 517–522 (2015).
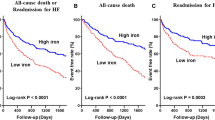
Anand, I. S. et al. Anemia and change in hemoglobin over time related to mortality and morbidity in patients with chronic heart failure: results from Val-HeFT. Circulation 112, 1121–1127 (2005).
Felker, G. M., Shaw, L. K., Stough, W. G. & O'Connor, C. M. Anemia in patients with heart failure and preserved systolic function. Am. Heart J. 151, 457–462 (2006).
Go, A. S. et al. Hemoglobin level, chronic kidney disease, and the risks of death and hospitalization in adults with chronic heart failure: the Anemia in Chronic Heart Failure: Outcomes and Resource Utilization (ANCHOR) Study. Circulation 113, 2713–2723 (2006).
Komajda, M. et al. The impact of new onset anaemia on morbidity and mortality in chronic heart failure: results from COMET. Eur. Heart J. 27, 1440–1446 (2006).
O'Meara, E. et al. Clinical correlates and consequences of anemia in a broad spectrum of patients with heart failure: results of the Candesartan in Heart Failure: assessment of Reduction in Mortality and Morbidity (CHARM) Program. Circulation 113, 986–994 (2006).
von Haehling, S. et al. Anaemia among patients with heart failure and preserved or reduced ejection fraction: results from the SENIORS study. Eur. J. Heart Fail. 13, 656–663 (2011).
Anker, S. D. et al. Prevalence, incidence, and prognostic value of anaemia in patients after an acute myocardial infarction: data from the OPTIMAAL trial. Eur. Heart J. 30, 1331–1339 (2009).
von Haehling, S., Anker, M. S., Jankowska, E. A., Ponikowski, P. & Anker, S. D. Anemia in chronic heart failure: can we treat? What to treat? Heart Fail. Rev. 17, 203–210 (2012).
Anand, I. S. Anemia and chronic heart failure implications and treatment options. J. Am. Coll. Cardiol. 52, 501–511 (2008).
Westenbrink, B. D. et al. Anaemia in chronic heart failure is not only related to impaired renal perfusion and blunted erythropoietin production, but to fluid retention as well. Eur. Heart J. 28, 166–171 (2007).
Weiss, G. & Goodnough, L. T. Anemia of chronic disease. N. Engl. J. Med. 352, 1011–1023 (2005).
Ebner, N. & von Haehling, S. Iron deficiency in heart failure: a practical guide. Nutrients 5, 3730–3739 (2013).
Ezekowitz, J. A., McAlister, F. A. & Armstrong, P. W. Anemia is common in heart failure and is associated with poor outcomes: insights from a cohort of 12 065 patients with new-onset heart failure. Circulation 107, 223–225 (2003).
Nanas, J. N. et al. Etiology of anemia in patients with advanced heart failure. J. Am. Coll. Cardiol. 48, 2485–2489 (2006).
Jankowska, E. A. et al. Iron deficiency: an ominous sign in patients with systolic chronic heart failure. Eur. Heart J. 31, 1872–1880 (2010).
Parikh, A., Natarajan, S., Lipsitz, S. R. & Katz, S. D. Iron deficiency in community-dwelling US adults with self-reported heart failure in the National Health and Nutrition Examination Survey III: prevalence and associations with anemia and inflammation. Circ. Heart Fail. 4, 599–606 (2011).
Silverberg, D. S. et al. The effect of correction of mild anemia in severe, resistant congestive heart failure using subcutaneous erythropoietin and intravenous iron: a randomized controlled study. J. Am. Coll. Cardiol. 37, 1775–1780 (2001).
Silverberg, D. S. et al. The effect of correction of anaemia in diabetics and non-diabetics with severe resistant congestive heart failure and chronic renal failure by subcutaneous erythropoietin and intravenous iron. Nephrol. Dial. Transplant. 18, 141–146 (2003).
Mancini, D. M. et al. Effect of erythropoietin on exercise capacity in patients with moderate to severe chronic heart failure. Circulation 107, 294–299 (2003).
Palazzuoli, A. et al. Erythropoietin improves anemia exercise tolerance and renal function and reduces B-type natriuretic peptide and hospitalization in patients with heart failure and anemia. Am. Heart J. 152, 1096.e9–1096.e15 (2006).
Ponikowski, P. et al. Effect of darbepoetin alfa on exercise tolerance in anemic patients with symptomatic chronic heart failure: a randomized, double-blind, placebo-controlled trial. J. Am. Coll. Cardiol. 49, 753–762 (2007).
van Veldhuisen, D. J. et al. Randomized, double-blind, placebo-controlled study to evaluate the effect of two dosing regimens of darbepoetin alfa in patients with heart failure and anaemia. Eur. Heart J. 28, 2208–2216 (2007).
Ghali, J. K. et al. Randomized double-blind trial of darbepoetin alfa in patients with symptomatic heart failure and anemia. Circulation 117, 526–535 (2008).
Swedberg, K. et al. Treatment of anemia with darbepoetin alfa in systolic heart failure. N. Engl. J. Med. 368, 1210–1219 (2013).
Bolger, A. P. et al. Intravenous iron alone for the treatment of anemia in patients with chronic heart failure. J. Am. Coll. Cardiol. 48, 1225–1227 (2006).
Toblli, J. E., Lombraña, A., Duarte, P. & Di Gennaro, F. Intravenous iron reduces NT-pro-brain natriuretic peptide in anemic patients with chronic heart failure and renal insufficiency. J. Am. Coll. Cardiol. 50, 1657–1665 (2007).
Okonko, D. O. et al. Effect of intravenous iron sucrose on exercise tolerance in anemic and nonanemic patients with symptomatic chronic heart failure and iron deficiency FERRIC-HF: a randomized, controlled, observer-blinded trial. J. Am. Coll. Cardiol. 51, 103–112 (2008).
Anker, S. D. et al. Rationale and design of Ferinject assessment in patients with IRon deficiency and chronic Heart Failure (FAIR-HF) study: a randomized, placebo-controlled study of intravenous iron supplementation in patients with and without anaemia. Eur. J. Heart Fail. 11, 1084–1091 (2009).
Anker, S. D. et al. Ferric carboxymaltose in patients with heart failure and iron deficiency. N. Engl. J. Med. 361, 2436–2448 (2009).
McMurray, J. J. et al. ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur. J. Heart Fail. 14, 803–869 (2012).
Krum, H. et al. 2011 update to National Heart Foundation of Australia and Cardiac Society of Australia and New Zealand Guidelines for the prevention, detection and management of chronic heart failure in Australia, 2006. Med. J. Aust. 194, 405–409 (2011).
Yancy, C. W. et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 62, e147–e239 (2013).
Ponikowski, P. et al. Rationale and design of the CONFIRM-HF study: a double-blind, randomized, placebo-controlled study to assess the effects of intravenous ferric carboxymaltose on functional capacity in patients with chronic heart failure and iron deficiency. ESC Heart Failure 1, 52–58 (2014).
Clark, A. L. & Pellicori, P. Clinical trials update from the European Society of Cardiology meeting 2014: PARADIGM-HF, CONFIRM-HF, SIGNIFY, atrial fibrillation, beta-blockers and heart failure, and vagal stimulation in heart failure. ESC Heart Failure 1, 82–86 (2014).
Ponikowski, P. et al. Beneficial effects of long-term intravenous iron therapy with ferric carboxymaltose in patients with symptomatic heart failure and iron deficiency. Eur. Heart J. 36, 657–668 (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
von Haehling, S. Inflammation in right ventricular dysfunction due to thromboembolic pulmonary hypertension. Int. J. Cardiol. 144, 206–211 (2010).
Soon, E. et al. Unexplained iron deficiency in idiopathic and heritable pulmonary arterial hypertension. Thorax 66, 326–332 (2011).
Ruiter, G. et al. Iron deficiency is common in idiopathic pulmonary arterial hypertension. Eur. Respir. J. 37, 1386–1391 (2011).
van Empel, V. P., Lee, J., Williams, T. J. & Kaye, D. M. Iron deficiency in patients with idiopathic pulmonary arterial hypertension. Heart Lung Circ. 23, 287–292 (2014).
Rhodes, C. J. et al. Iron deficiency and raised hepcidin in idiopathic pulmonary arterial hypertension: clinical prevalence, outcomes, and mechanistic insights. J. Am. Coll. Cardiol. 58, 300–309 (2011).
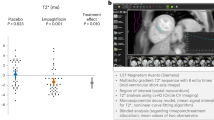
Viethen, T. et al. Ferric carboxymaltose improves exercise capacity and quality of life in patients with pulmonary arterial hypertension and iron deficiency: a pilot study. Int. J. Cardiol. 175, 233–239 (2014).
Gleissner, C. A. et al. Reduced hemoglobin after heart transplantation is no independent risk factor for survival but is associated closely with impaired renal function. Transplantation 77, 710–717 (2004).
Przybylowski, P., Malyszko, J. & Malyszko, J. Anemia is a predictor of outcome in heart transplant recipients. Transplant. Proc. 41, 3228–3231 (2009).
Taegtmeyer, A. B. et al. The effects of pre- and post-transplant anemia on 1-year survival after cardiac transplantation. J. Heart Lung Transplant. 27, 394–399 (2008).
Przybylowski, P. et al. Prevalence of iron deficiency in heart and kidney allograft recipients. Transplant. Proc. 43, 3885–3887 (2011).
Schaefer, B., Effenberger, M. & Zoller, H. Iron metabolism in transplantation. Transpl. Int. 27, 1109–1117 (2014).
Menasché, P., Grousset, C., Mouas, C. & Piwnica, A. A promising approach for improving the recovery of heart transplants. Prevention of free radical injury through iron chelation by deferoxamine. J. Thorac. Cardiovasc. Surg. 100, 13–21 (1990).
Macedo, M. F. et al. Transferrin is required for early T-cell differentiation. Immunology 112, 543–549 (2004).
Zheng, Y. et al. A role for mammalian target of rapamycin in regulating T cell activation versus anergy. J. Immunol. 178, 2163–2170 (2007).
Brekelmans, P., Van Soest, P., Leenen, P. J. & Van Ewijk, W. Inhibition of proliferation and differentiation during early T cell development by anti-transferrin receptor antibody. Eur. J. Immunol. 24, 2896–2902 (1994).
David, O., Sinha, R., Robinson, K. & Cardone, D. The prevalence of anaemia, hypochromia and microcytosis in preoperative cardiac surgical patients. Anaesth. Intensive Care 41, 316–321 (2013).
Hung, M. et al. A prospective observational cohort study to identify the causes of anaemia and association with outcome in cardiac surgical patients. Heart 101, 107–112 (2015).
Piednoir, P. et al. Preoperative iron deficiency increases transfusion requirements and fatigue in cardiac surgery patients: a prospective observational study. Eur. J. Anaesthesiol. 28, 796–801 (2011).
Madi-Jebara, S. N. et al. Postoperative intravenous iron used alone or in combination with low-dose erythropoietin is not effective for correction of anemia after cardiac surgery. J. Cardiothorac. Vasc. Anesth. 18, 59–63 (2004).
Garrido-Martín, P. et al. The effect of intravenous and oral iron administration on perioperative anaemia and transfusion requirements in patients undergoing elective cardiac surgery: a randomized clinical trial. Interact. Cardiovasc. Thorac. Surg. 15, 1013–1018 (2012).
Wish, J. B. Assessing iron status: beyond serum ferritin and transferrin saturation. Clin. J. Am. Soc. Nephrol. 1 (Suppl. 1), S4–S8 (2006).
Usmanov, R. I., Zueva, E. B., Silverberg, D. S. & Shaked, M. Intravenous iron without erythropoietin for the treatment of iron deficiency anemia in patients with moderate to severe congestive heart failure and chronic kidney insufficiency. J. Nephrol. 21, 236–242 (2008).
Author information
Authors and Affiliations
Contributions
S.v.H. determined the content of the article, and wrote the first draft of the article. E.A.J., D.J.v.V., P.P., and S.D.A. contributed to the review and editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
S.v.H. has received consulting honoraria from Novartis, Pfizer, Respicardia, Thermo Fisher Scientific, and Vifor Pharma, as well as lecture fees from Amgen and Sorin. E.A.J. has received honoraria for lectures and participation in advisory boards from Vifor Pharma. D.J.v.V. has received board membership fees from Amgen and Vifor Pharma. P.P. is a consultant for, and has received honoraria for speaking from, Amgen and Vifor Pharma, as well as research grants from Vifor Pharma. S.D.A. has received consulting fees and honoraria from Bayer, BRAHMS, Novartis, and Vifor Pharma, consulting fees from Keryx, and research support from Abbott Vascular and Vifor Pharma.
Rights and permissions
About this article
Cite this article
von Haehling, S., Jankowska, E., van Veldhuisen, D. et al. Iron deficiency and cardiovascular disease. Nat Rev Cardiol 12, 659–669 (2015). https://doi.org/10.1038/nrcardio.2015.109
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2015.109
This article is cited by
-
Novel Bis-pyrazoline Fluorescent Probe for Cu2+ and Fe3+ Detection and Application in Cell Imaging
Journal of Fluorescence (2024)
-
Association between latent profile of dietary intake and cardiovascular diseases (CVDs): Results from Fasa Adults Cohort Study (FACS)
Scientific Reports (2023)
-
Efficacy and safety of intravenous iron repletion in patients with heart failure: a systematic review and meta-analysis
Clinical Research in Cardiology (2023)
-
Interactive Effects of Obesity and Hypertension on Patterns of Hair Essential Trace Element and Mineral Content in Adult Women
Biological Trace Element Research (2023)
-
Non-anemic iron deficiency predicts prolonged hospitalisation following surgical aortic valve replacement: a single-centre retrospective study
Journal of Cardiothoracic Surgery (2022)