Key Points
-
Ionizing radiation (IR) has proved to be a powerful tool in the treatment of cancer. However, it also has serious side effects for normal tissues. The overall sensitivity of a mammalian organism to IR is determined by the pathological alterations that occur in a few sensitive tissues.
-
Organisms that survive acute toxicity of radiation can suffer from long-term remote consequences, including radiation-induced carcinogenesis and fibrosis, which develop in exposed organs (such as the kidneys, liver or lungs) months and years after irradiation.
-
Cellular DNA is the main target of IR; it causes DNA damage (genotoxic stress) by direct and indirect (free-radical-based) mechanisms. All organisms maintain a DNA-repair system that is capable of effective recovery of radiation-damaged DNA; errors in the DNA-repair process might lead to mutations and an increased risk of cancer development.
-
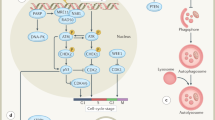
At the molecular level, radiation-induced damage results in activation of DNA repair, coupled with arrest at cell-cycle checkpoints, which allows the cell to repair the damage before proceeding through mitosis. This mechanism is conserved in all eukaryotes.
-
Multicellular organisms have acquired additional response mechanisms to genotoxic stress, which involve activation of the transcription factor p53. p53 can induce growth arrest or apoptosis, and these responses maintain genomic stability. Failure of this system results in cancer development and genomic instability.
-
The ways in which cells respond to IR are tissue specific and vary greatly during embryogenesis. Cells with a high proliferative capacity tend to apoptose, whereas fibroblasts — the structural component of tissues — tend to growth arrest.
-
Tumours are generally highly sensitive to gamma-radiation — because of loss of negative growth regulation and genomic stability — and are treated with many local doses to reduce damage to normal tissues.
-
Apoptosis has a relatively modest role in the tumour response to radiation; most tumours lose the ability to apoptose. The antitumour effect of radiation is realized through mitotic catastrophe or in senescence-like irreversible growth arrest.
-
Radiation sensitivity involves both intrinsic mechanisms and bystander effects, in which the failure of a certain cell type within the complex mix of tumour and normal cells leads to a chain of reactions that results in tissue failure.
-
Facilitation of radiation therapy outcome can be achieved by modulating the molecular mechanisms of the DNA-damage response in normal tissues and in tumours. So, p53 inhibition can reduce normal tissue damage and sensitize tumour cells for treatment.
Abstract
Ionizing radiation (IR) has proven to be a powerful medical treatment in the fight against cancer. Rational and effective use of its killing power depends on understanding IR-mediated responses at the molecular, cellular and tissue levels. Tumour cells frequently acquire defects in the molecular regulatory mechanisms of the response to IR, which sensitizes them to radiation therapy. One of the key molecules involved in a cell's response to IR is p53. Understanding these mechanisms indicates new rational approaches to improving cancer treatment by IR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Santana, P. et al. Acid sphingomyelinase-deficient human lymphoblasts and mice are defective in radiation-induced apoptosis. Cell 86, 189–199 (1996).
Fowler, J. F. Biological factors influencing optimum fractionation in radiation therapy. Acta Oncol. 40, 712–717 (2001).
Wahl, G. M. & Carr, A. M. The evolution of diverse biological responses to DNA damage: insights from yeast and p53. Nature Cell Biol. 3, E277–E286 (2001).
Lowndes, N. F. & Murguia, J. R. Sensing and responding to DNA damage. Curr. Opin. Genet. Dev. 10, 17–25 (2000).
Neta, R., Oppenheim, J. J. & Douches, S. D. Interdependence of the radioprotective effects of human recombinant interleukin 1 alpha, tumor necrosis factor alpha, granulocyte colony-stimulating factor, and murine recombinant granulocyte-macrophage colony-stimulating factor. J. Immunol. 140, 108–111 (1988).
Kemp, C. J., Sun, S. & Gurley, K. E. p53 induction and apoptosis in response to radio- and chemotherapy in vivo is tumor-type-dependent. Cancer Res. 61, 327–332 (2001). Shows that the experimental mouse tumours that respond to cancer treatment by apoptosis originate from apoptosis-prone tissues.
Smith, M. L. p53 regulation of DNA excision repair pathways. Mutagenesis 17, 149–156 (2002).
Dasika, G. K. et al. DNA damage-induced cell cycle checkpoints and DNA strand break repair in development and tumorigenesis. Oncogene 18, 7883–7899 (1999).
Lowe, S. W. & Lin, A. W. Apoptosis in cancer. Carcinogenesis 21, 485–495 (2000).
Ko, L. J. & Prives, C. p53: puzzle and paradigm. Genes Dev. 10, 1054–1072 (1996).
el-Deiry, W. S. Regulation of p53 downstream genes. Semin. Cancer Biol. 8, 345–357 (1998).
Offer, H. et al. The onset of p53-dependent DNA repair or apoptosis is determined by the level of accumulated damaged DNA. Carcinogenesis 23, 1025–1032 (2002).
Vousden, K. H. p53: death star. Cell 103, 691–694 (2000).
Schmitt, C. A., Rosenthal, C. T. & Lowe, S. W. Genetic analysis of chemoresistance in primary murine lymphomas. Nature Med. 6, 1029–1035 (2000).
Samuels-Lev, Y. et al. ASPP proteins specifically stimulate the apoptotic function of p53. Mol. Cell 8, 781–794 (2001).
Oda, K. et al. p53AIP1, a potential mediator of p53-dependent apoptosis, and its regulation by Ser-46-phosphorylated p53. Cell 102, 849–862 (2000).
Khanna, K. K. et al. Cellular Responses to DNA Damage and the Human Chromosome Instability Syndromes (Humana Press, San Diego, 1998).
Shiloh, Y. Ataxia-telangiectasia and the Nijmegen breakage syndrome: related disorders but genes apart. Annu. Rev. Genet. 31, 635–662 (1997).
Woods, C. G. DNA repair disorders. Arch. Dis. Child. 78, 178–184 (1998).
Auerbach, A. D. & Verlander, P. C. Disorders of DNA replication and repair. Curr. Opin. Pediatr. 9, 600–616 (1997).
Denekamp, J. Cell kinetics and radiation biology. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 49, 357–380 (1986).
Sinclair, W. K. in Time and Dose Relationships in Radiation Biology as Applied to Radiotherapy. BNL Reports 50203 (C-57), 97–107 (1969).
Sinclair, W. K. N-ethylmaleimide and the cyclic response to X-rays of synchronous Chinese hamster cells. Radiat. Res. 55, 41–57 (1973).
Komarova, E. A. et al. Stress-induced secretion of growth inhibitors: a novel tumor suppressor function of p53. Oncogene 17, 1089–1096 (1998).
Takai, H. et al. Chk2-deficient mice exhibit radioresistance and defective p53-mediated transcription. EMBO J. 21, 5195–5205 (2002).
Bunz, F. et al. Disruption of p53 in human cancer cells alters the responses to therapeutic agents. J. Clin. Invest. 104, 263–269 (1999).
Hawkins, D. S., Demers, G. W. & Galloway, D. A. Inactivation of p53 enhances sensitivity to multiple chemotherapeutic agents. Cancer Res. 56, 892–898 (1996). Inactivation of p53 might cause cell sensitization to treatment with anticancer agents.
Palacios, C., Gutierrez del Arroyo, A., Silva, A. & Collins, M. K. The role of p53 in death of IL-3-dependent cells in response to cytotoxic drugs. Oncogene 19, 3556–3559 (2000).
Offer, H. et al. p53 modulates base excision repair activity in a cell cycle-specific manner after genotoxic stress. Cancer Res. 61, 88–96 (2001).
Pardo, F. S. et al. Transfection of rat embryo cells with mutant p53 increases the intrinsic radiation resistance. Radiat. Res. 140, 180–185 (1994).
Wang, L. et al. Gamma-ray-induced cell killing and chromosome abnormalities in the bone marrow of p53-deficient mice. Radiat. Res. 146, 259–266 (1996).
DiBiase, S. J., Guan, J., Curran, W. J., Jr & Iliakis, G. Repair of DNA double-strand breaks and radiosensitivity to killing in an isogenic group of p53 mutant cell lines. Int. J. Radiat. Oncol. Biol. Phys. 45, 743–751 (1999).
Gudkov, A. V. Converting p53 from a killer into a healer. Nature Med. 8, 1196–1198 (2002).
Seo, Y. R., Kelley, M. & Smith, M. L. Selenomethionine regulation of p53 by a ref-1-dependent redox mechanism. Proc. Natl Acad. Sci. USA 99, 13969–13971 (2002).
Schultheiss, T. E., Kun, L. E., Ang, K. K. & Stephens, L. C. Radiation response of the central nervous system. Int. J. Radiat. Oncol. Biol. Phys. 31, 1093–1112 (1995).
Michel, C. Radiation embryology. Experientia 45, 69–77 (1989).
Chang, B. D. et al. A senescence-like phenotype distinguishes tumor cells that undergo terminal proliferation arrest after exposure to anticancer agents. Cancer Res. 59, 3761–3767 (1999).
Chang, B. D. et al. p21Waf1/Cip1/Sdi1-induced growth arrest is associated with depletion of mitosis-control proteins and leads to abnormal mitosis and endoreduplication in recovering cells. Oncogene 19, 2165–2170 (2000).
Macip, S. et al. Inhibition of p21-mediated ROS accumulation can rescue p21-induced senescence. EMBO J. 21, 2180–2188 (2002). References 37–39 show that senescence-like permanent growth arrest can be an important type of tumour-cell response to treatment with DNA-damaging agents.
Wang, J. Y., Naderi, S. & Chen, T. T. Role of retinoblastoma tumor suppressor protein in DNA damage response. Acta Oncol. 40, 689–695 (2001). The role of RB is essential for the establishment of irreversible growth arrest in response to IR: inactivation of RB allows the cell to resume proliferation after prolonged p53-dependent growth arrest following severe DNA damage.
Song, S. & Lambert, P. F. Different responses of epidermal and hair follicular cells to radiation correlate with distinct patterns of p53 and p21 induction. Am. J. Pathol. 155, 1121–1127 (1999).
Komarova, E. A. et al. Transgenic mice with p53-responsive lacZ: p53 activity varies dramatically during normal development and determines radiation and drug sensitivity in vivo. EMBO J. 16, 1391–1400 (1997).
Komarova, E. A., Christov, K., Faerman, A. I. & Gudkov, A. V. Different impact of p53 and p21 on the radiation response of mouse tissues. Oncogene 19, 3791–3798 (2000).
Lowe, S. W., Schmitt, E. M., Smith, S. W., Osborne, B. A. & Jacks, T. p53 is required for radiation-induced apoptosis in mouse thymocytes. Nature 362, 847–849 (1993).
MacCallum, D. E. et al. The p53 response to ionising radiation in adult and developing murine tissues. Oncogene 13, 2575–2587 (1996).
Westphal, C. H. et al. Atm and p53 cooperate in apoptosis and suppression of tumorigenesis, but not in resistance to acute radiation toxicity. Nature Genet. 16, 397–401 (1997). References 41–46 illustrate the role of p53-dependent apoptosis in normal tissue damage by IR, indicating that p53 might be a determinant of the side effects of cancer treatment.
Komarov, P. G. et al. A chemical inhibitor of p53 that protects mice from the side effects of cancer therapy. Science 285, 1733–1737 (1999). Identification (by chemical library screening) and in vivo and in vitro testing of a p53 inhibitor as an approach to reduce IR-induced tissue damage.
Hall, P. A., McKee, P. H., Menage, H. D., Dover, R. & Lane, D. P. High levels of p53 protein in UV-irradiated normal human skin. Oncogene 8, 203–207 (1993).
Campbell, C., Quinn, A. G., Angus, B., Farr, P. M. & Rees, J. L. Wavelength specific patterns of p53 induction in human skin following exposure to UV radiation. Cancer Res. 53, 2697–2699 (1993).
Merritt, A. J. et al. The role of p53 in spontaneous and radiation-induced apoptosis in the gastrointestinal tract of normal and p53-deficient mice. Cancer Res. 54, 614–617 (1994).
Clarke, A. R., Gledhill, S., Hooper, M. L., Bird, C. C. & Wyllie, A. H. p53 dependence of early apoptotic and proliferative responses within the mouse intestinal epithelium following gamma-irradiation. Oncogene 9, 1767–1773 (1994). References 50 and 51 describe the p53 dependence of IR-induced damage to the epithelium of the small intestine.
Midgley, C. A. et al. Coupling between gamma irradiation, p53 induction and the apoptotic response depends upon cell type in vivo. J. Cell Sci. 108, 1843–1848 (1995).
Rogel, A., Popliker, M., Webb, C. G. & Oren, M. p53 cellular tumor antigen: analysis of mRNA levels in normal adult tissues, embryos, and tumors. Mol. Cell. Biol. 5, 2851–2855 (1985).
Burns, T. F., Bernhard, E. J. & El-Deiry, W. S. Tissue specific expression of p53 target genes suggests a key role for KILLER/DR5 in p53-dependent apoptosis in vivo. Oncogene 20, 4601–4612 (2001).
Haks, M. C., Krimpenfort, P., van den Brakel, J. H. & Kruisbeek, A. M. Pre-TCR signaling and inactivation of p53 induces crucial cell survival pathways in pre-T cells. Immunity 11, 91–101 (1999).
Guidos, C. J. et al. V(D)J recombination activates a p53-dependent DNA damage checkpoint in SCID lymphocyte precursors. Genes Dev. 10, 2038–2054 (1996).
Newmeyer, D. D. & Green, D. R. Surviving the cytochrome seas. Neuron 21, 653–655 (1998).
Li, F. et al. Cell-specific induction of apoptosis by microinjection of cytochrome c. Bcl-xL has activity independent of cytochrome c release. J. Biol. Chem. 272, 30299–30305 (1997).
Deshmukh, M. & Johnson, E. M. Jr. Evidence of a novel event during neuronal death: development of competence-to-die in response to cytoplasmic cytochrome c. Neuron 21, 695–705 (1998).
Westphal, C. H. et al. Loss of Atm radiosensitizes multiple p53 null tissues. Cancer Res. 58, 5637–5639 (1998).
Paris, F. et al. Endothelial apoptosis as the primary lesion initiating intestinal radiation damage in mice. Science 293, 293–297 (2001). Tissue failure in response to IR can result from the selective damage of the most sensitive tissue component (in this case, the endothelium), leading to a chain reaction of pathological consequences.
Chow, B. M., Li, Y. Q. & Wong, C. S. Radiation-induced apoptosis in the adult central nervous system is p53- dependent. Cell Death Differ. 7, 712–720 (2000).
Weichselbaum, R. R., Epstein, J. & Little, J. B. In vitro radiosensitivity of human dipliod fibroblasts derived from patients with unusual clinical responses to radiation. Radiology 121, 479–482 (1976).
Barcellos-Hoff, M. H. Radiation-induced transforming growth factor beta and subsequent extracellular matrix reorganization in murine mammary gland. Cancer Res. 53, 3880–3886 (1993).
Flanders, K. C. et al. Mice lacking Smad3 are protected against cutaneous injury induced by ionizing radiation. Am. J. Pathol. 160, 1057–1068 (2002).
Johnstone, R. W., Ruefli, A. A. & Lowe, S. W. Apoptosis: a link between cancer genetics and chemotherapy. Cell 108, 153–164 (2002).
Schmitt, C. A. & Lowe, S. W. Bcl-2 mediates chemoresistance in matched pairs of primary E(mu)-myc lymphomas in vivo. Blood Cells Mol. Dis. 27, 206–216 (2001).
Cordon-Cardo, C., Dalbagni, G., Sarkis, A. S. & Reuter, V. E. Genetic alterations associated with bladder cancer. Important Adv. Oncol. [vol?] 71–83 (1994).
Falette, N. et al. Prognostic value of p53 gene mutations in a large series of node-negative breast cancer patients. Cancer Res. 58, 1451–1455 (1998).
Molina, R. et al. p53 oncoprotein as a prognostic indicator in patients with breast cancer. Anticancer Res. 18, 507–511 (1998).
Brown, J. M. & Wouters, B. G. Apoptosis: mediator or mode of cell killing by anticancer agents? Drug Resist. Update 4, 135–136 (2001).
Nieder, C., Petersen, S., Petersen, C. & Thames, H. D. The challenge of p53 as prognostic and predictive factor in gliomas. Cancer Treat. Rev. 26, 67–73 (2000).
Sierra, A. et al. Bcl-2 with loss of apoptosis allows accumulation of genetic alterations: a pathway to metastatic progression in human breast cancer. Int. J. Cancer 89, 142–147 (2000).
Deveraux, Q. L. et al. IAPs block apoptotic events induced by caspase-8 and cytochrome c by direct inhibition of distinct caspases. EMBO J. 17, 2215–2223 (1998).
Beere, H. M. et al. Heat-shock protein 70 inhibits apoptosis by preventing recruitment of procaspase-9 to the Apaf-1 apoptosome. Nature Cell Biol. 2, 469–475 (2000).
Lowe, S. W. et al. p53 status and the efficacy of cancer therapy in vivo. Science 266, 807–810 (1994). This paper had a very strong impact in the field by establishing the concept of p53-dependent apoptosis as the mechanism of treatment sensitivity of tumours.
Malumbres, M. & Barbacid, M. To cycle or not to cycle: a critical decision in cancer. Nature Rev. Cancer 1, 222–231 (2001).
Roninson, I. B., Broude, E. V. & Chang, B. D. If not apoptosis, then what? Treatment-induced senescence and mitotic catastrophe in tumor cells. Drug Resist. Update 4, 303–313 (2001).
Hendry, J. H. & West, C. M. Apoptosis and mitotic cell death: their relative contributions to normal-tissue and tumour radiation response. Int. J. Radiat. Biol. 71, 709–719 (1997).
Ianzini, F. & Mackey, M. A. Delayed DNA damage associated with mitotic catastrophe following X-irradiation of HeLa S3 cells. Mutagenesis 13, 337–344 (1998).
Abend, M., Gilbertz, K. P., Rhein, A. & van Beuningen, D. Early and late G2 arrest of cells undergoing radiation-induced apoptosis or micronucleation. Cell Prolif. 29, 101–113 (1996).
Dini, L., Coppola, S., Ruzittu, M. T. & Ghibelli, L. Multiple pathways for apoptotic nuclear fragmentation. Exp. Cell Res. 223, 340–347 (1996).
Schmitt, C. A. et al. A senescence program controlled by p53 and p16INK4a contributes to the outcome of cancer therapy. Cell 109, 335–346 (2002). Blocking the apoptotic programme in tumours of immunocyte origin causes a switch to an alternative type of p53-mediated response to treatment in vivo — senescence-like permanent arrest.
Slichenmyer, W. J., Nelson, W. G., Slebos, R. J. & Kastan, M. B. Loss of a p53-associated G1 checkpoint does not decrease cell survival following DNA damage. Cancer Res. 53, 4164–4168 (1993).
Brachman, D. G. et al. p53 mutation does not correlate with radiosensitivity in 24 head and neck cancer cell lines. Cancer Res. 53, 3667–3669 (1993). References 84 and 85 show that wild-type p53 in the tumour is not necessarily associated with higher sensitivity to treatment.
Graeber, T. G. et al. Hypoxia-mediated selection of cells with diminished apoptotic potential in solid tumours. Nature 379, 88–91 (1996).
Gurova, K. V. et al. Apoptosis inhibitor as a suppressor of tumor progression: expression of Bcl-2 eliminates selective advantages for p53-deficient cells in the tumor. Cancer Biol. Ther. 1, 39–44; discussion 45–46 (2002).
Gurova, K. & Gudkov, A. V. Paradoxical role of apoptosis in cancer progression. J. Cell Biochem. 88, 128–137 (2003).
Komarova, E. A. & Gudkov, A. V. Could p53 be a target for therapeutic suppression? Semin. Cancer Biol. 8, 389–400 (1998).
Funk, J. O., Samuel, T. & Weber, H. O. in Cell Cycle Checkpoints and Cancer (ed. Blagosklonny, M. V.) 64–77 (Eurekah, Austin, Texas, 2001).
Khanna, K. K., Lavin, M. F., Jackson, S. P. & Mulhern, T. D. ATM, a central controller of cellular responses to DNA damage. Cell Death Differ. 8, 1052–1065 (2001).
Tuteja, N. & Tuteja, R. Unraveling DNA repair in human: molecular mechanisms and consequences of repair defect. Crit. Rev. Biochem. Mol. Biol. 36, 261–290 (2001).
Rosen, E. M., Fan, S., Rockwell, S. & Goldberg, I. D. The molecular and cellular basis of radiosensitivity: implications for understanding how normal tissues and tumors respond to therapeutic radiation. Cancer Invest. 17, 56–72 (1999).
Bernstein, C., Bernstein, H., Payne, C. M. & Garewal, H. DNA repair/pro-apoptotic dual-role proteins in five major DNA repair pathways: fail-safe protection against carcinogenesis. Mutat. Res. 511, 145–178 (2002).
Prives, C. & Hall, P. A. The p53 pathway. J. Pathol. 187, 112–126 (1999).
Vousden, K. H. & Lu, X. Live or let die: the cell's response to p53. Nature Rev. Cancer 2, 594–604 (2002).
Ford, J. M. & Hanawalt, P. C. Expression of wild-type p53 is required for efficient global genomic nucleotide excision repair in UV-irradiated human fibroblasts. J. Biol. Chem. 272, 28073–28080 (1997). References 34 and 97 indicate a direct involvement of p53 in DNA repair.
Levine, A. J. p53, the cellular gatekeeper for growth and division. Cell 88, 323–331 (1997).
Schuler, M. & Green, D. R. Mechanisms of p53-dependent apoptosis. Biochem. Soc. Trans. 29, 684–688 (2001).
Li, H., Zhu, H., Xu, C. J. & Yuan, J. Cleavage of BID by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell 94, 491–501 (1998).
Sheard, M. A. Ionizing radiation as a response-enhancing agent for CD95-mediated apoptosis. Int. J. Cancer 96, 213–220 (2001).
Embree-Ku, M., Venturini, D. & Boekelheide, K. Fas is involved in the p53-dependent apoptotic response to ionizing radiation in mouse testis. Biol. Reprod. 66, 1456–1461 (2002).
McGowan, C. H. Checking in on Cds1 (Chk2): a checkpoint kinase and tumor suppressor. Bioessays 24, 502–511 (2002).
Albrechtsen, N. et al. Maintenance of genomic integrity by p53: complementary roles for activated and non-activated p53. Oncogene 18, 7706–7717 (1999).
Troelstra, C. et al. Molecular cloning of the human DNA excision repair gene ERCC-6. Mol. Cell. Biol. 10, 5806–5813 (1990).
Buchhop, S. et al. Interaction of p53 with the human Rad51 protein. Nucleic Acids Res. 25, 3868–3874 (1997).
Basu, S. & Kolesnick, R. Stress signals for apoptosis: ceramide and c-Jun kinase. Oncogene 17, 3277–3285 (1998).
Kolesnick, R. The therapeutic potential of modulating the ceramide/sphingomyelin pathway. J. Clin. Invest. 110, 3–8 (2002).
Aladjem, M. I. et al. ES cells do not activate p53-dependent stress responses and undergo p53-independent apoptosis in response to DNA damage. Curr. Biol. 8, 145–155 (1998).
Norimura, T., Nomoto, S., Katsuki, M., Gondo, Y. & Kondo, S. p53-dependent apoptosis suppresses radiation-induced teratogenesis. Nature Med. 2, 577–580 (1996). Loss of p53 in mice is associated with a marked increase in the teratogenic effect of IR, presumably because genetically damaged cells are no longer eliminated by p53-mediated apoptosis. These observations allowed David Lane to name p53 “a guardian of babies”.
Brash, D. E. Cellular proofreading. Nature Med. 2, 525–526 (1996).
Hall, P. A. & Lane, D. P. Tumor suppressors: a developing role for p53? Curr. Biol. 7, R144–R147 (1997).
Vos, O. Effects and consequences of prenatal irradiation. Boll. Soc. Ital. Biol. Sper. 65, 481–500 (1989).
Hendry, J. H., Cai, W. B., Roberts, S. A. & Potten, C. S. p53 deficiency sensitizes clonogenic cells to irradiation in the large but not the small intestine. Radiat. Res. 148, 254–259 (1997).
Hasegawa, M., Zhang, Y., Niibe, H., Terry, N. H. & Meistrich, M. L. Resistance of differentiating spermatogonia to radiation-induced apoptosis and loss in p53-deficient mice. Radiat. Res. 149, 263–270 (1998).
Tron, V. A. et al. p53-regulated apoptosis is differentiation dependent in ultraviolet B- irradiated mouse keratinocytes. Am. J. Pathol. 153, 579–585 (1998).
Frenkel, J. et al. Accentuated apoptosis in normally developing p53 knockout mouse embryos following genotoxic stress. Oncogene 18, 2901–2907 (1999).
Uberti, D. et al. Epithelial cells of different organs exhibit distinct patterns of p53-dependent and p53-independent apoptosis following DNA insult. Exp. Cell Res. 252, 123–133 (1999).
Hendry, J. H., Adeeko, A., Potten, C. S. & Morris, I. D. p53 deficiency produces fewer regenerating spermatogenic tubules after irradiation. Int. J. Radiat. Biol. 70, 677–682 (1996).
Rojas, A. & Denekamp, J. Modifiers of radiosensitivity. Experientia 45, 41–52 (1989).
Jordan, B. F. et al. Insulin increases the sensitivity of tumors to irradiation: involvement of an increase in tumor oxygenation mediated by a nitric oxide-dependent decrease of the tumor cells oxygen consumption. Cancer Res. 62, 3555–3561 (2002).
Neta, R., Douches, S. & Oppenheim, J. J. Interleukin 1 is a radioprotector. J. Immunol. 136, 2483–2485 (1986).
Zsebo, K. M. et al. Radioprotection of mice by recombinant rat stem cell factor. Proc. Natl Acad. Sci. USA 89, 9464–9468 (1992).
Neta, R., Stiefel, S. M., Finkelman, F., Herrmann, S. & Ali, N. IL-12 protects bone marrow from and sensitizes intestinal tract to ionizing radiation. J. Immunol. 153, 4230–4237 (1994).
King, D. P. & Takahashi, J. S. Molecular genetics of circadian rhythms in mammals. Annu. Rev. Neurosci. 23, 713–742 (2000).
Haus, E. Chronobiology of the mammalian response to ionizing radiation potential applications in oncology. Chronobiol. Int. 19, 77–100 (2002).
Acknowledgements
We thank all members of our laboratory and collaborators for their impact in formation of the authors' view on the problem of radiation response in vivo. The authors' experimental research is supported by grants from the National Cancer Institute and Quark Biotech, Inc. (to A.V.G.).
Author information
Authors and Affiliations
Corresponding author
Glossary
- IONIZING RADIATION
-
(IR). Corpuscular or electromagnetic radiation that is capable of producing ions, directly or indirectly, in its passage through matter. The absorbed dose of ionizing radiation is measured as the gray (Gy, 1 joule of energy absorbed by 1 kilogram of material).
- GAMMA-RAYS AND X-RAYS
-
The distinction between gamma-rays and X-rays lies in their origin: gamma-rays originate from excited and unstable nuclei, whereas X-rays are produced by electron energy transitions within the atom or through the deceleration of high-kinetic energy electrons.
- RADIATION RESISTANCE (SENSITIVITY)
-
A relative term, the exact meaning of which depends on the subject and context. At the level of the organism, it is related to the ability to survive after exposure to ionizing radiation. At the tissue level, it describes the ability of the tissue or organ to maintain structural and functional integrity after irradiation. At the cellular level, it might either reflect the cell's ability to continue normal proliferation (form colonies in colony assays) or to retain viability (determined by cell-viability assays).
- GENOTOXIC STRESS
-
Different types of DNA damage that are recognized by stress-response sensor mechanisms and activate DNA repair, including double-stranded and single-stranded DNA breaks and covalent modifications of DNA.
- CELL-CYCLE CHECKPOINTS
-
A series of control mechanisms that induce growth arrest and act at several specific stages of the cell cycle. These prevent cells from entering into a new phase until they have successfully completed the previous one.
- MITOTIC CATASTROPHE
-
A series of pathological events that occur after aberrant mitosis and usually result in cell death. Such a mitosis does not produce proper chromosome segregation and cell division, but leads to the formation of large non-viable cells with several nuclei, containing fractions of broken chromosomes. It can be followed by apoptosis.
- CLONOGENIC POTENTIAL
-
The ability of the cell to propagate and to serve as a progenitor of a clone. Estimated by clonogenic or colony assays in vivo and in vitro.
- ATAXIA TELANGIECTASIA
-
Ataxia telangiectasia (AT) is an inherited syndrome that is characterized by cerebellar ataxia, telangiectases (dilated small blood vessels that appear in specific locations), immunodeficiency, radiosensitivity and predisposition to lymphatic leukaemias and other malignancies. It is associated with defects in the ATM gene, which encodes a 370-kDa protein — a member of the phosphatidylinositol 3-kinases superfamily — that mediates activation of DNA repair and p53 pathways.
- REACTIVE OXYGEN SPECIES
-
(ROS). Highly reactive chemical radicals that are generated as products of oxygen degradation.
- BCL2
-
BCL2 is an anti-apoptotic protein that is a member of a large family of proteins, consisting of anti-apoptotic and pro-apoptotic members. These proteins regulate the release of cytochrome c from mitochondria, leading to activation of the proteolytic caspase cascade and apoptosis.
- MYELOABLATION
-
Depletion of bone-marrow haematopoietic cells, resulting in subsequent reduction of peripheral-blood cell counts.
- BYSTANDER EFFECT
-
Indirect cytotoxicity of radiation or drugs to unaffected healthy cells that results from changes in tissue microenvironment caused by treatment-induced damage to neighbouring cells.
- OLIGODENDROCYTES
-
The non-neural cells of ectodermal origin that form part of the adventitial structure of the central nervous system.
- FIBROSIS
-
A common pathological consequence of tissue irradiation that develops a long time after treatment (months and years in radiation-treated cancer patients). It is characterized by abnormal proliferation of different cell components within tissue and results in substitution of epithelia for connective tissue.
- CYTOKINES
-
Hormone-like proteins that are produced by stimulated cells and serve as intercellular messengers. Cytokines are pleiotropic: they act on multiple cells via high-affinity membrane receptors. Signalling by the cytokine–receptor complex leads to stimulation of a diverse range of cell functions, including production of cytokines, growth and proliferation, differentiation, motility, apoptosis and growth inhibition.
- HYPOTHALAMIC SUPRACHIASMATIC NUCLEUS
-
The part of the brain that directs behavioural and physiological oscillations in the entire mammalian organism.
- REPLICATIVE SENESCENCE
-
Permanent growth arrest that cells undergo in culture (and presumably in tissues) after their telomeres become critically short as a result of multiple cell divisions.
- THERAPEUTIC INDEX
-
The ratio between therapeutic effect and the damage caused by treatment.
Rights and permissions
About this article
Cite this article
Gudkov, A., Komarova, E. The role of p53 in determining sensitivity to radiotherapy. Nat Rev Cancer 3, 117–129 (2003). https://doi.org/10.1038/nrc992
Issue Date:
DOI: https://doi.org/10.1038/nrc992
This article is cited by
-
Translating p53-based therapies for cancer into the clinic
Nature Reviews Cancer (2024)
-
Advances in radiotherapy and immunity in hepatocellular carcinoma
Journal of Translational Medicine (2023)
-
Efficacy of curcumin for amelioration of radiotherapy-induced oral mucositis: a preliminary randomized controlled clinical trial
BMC Cancer (2023)
-
Accurate interpretation of p53 immunohistochemical patterns is a surrogate biomarker for TP53 alterations in large B-cell lymphoma
BMC Cancer (2023)
-
p53 and p21 dynamics encode single-cell DNA damage levels, fine-tuning proliferation and shaping population heterogeneity
Communications Biology (2023)