Key Points
Summary
-
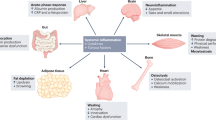
Patients with cancer cachexia show a progressive loss of body weight, which is mainly due to loss of fat and skeletal muscle. Survival of cancer patients is directly related to the total weight loss and also the rate of weight loss.
-
Although anorexia occurs in cancer patients, the reduction in food intake alone is unable to explain the metabolic changes that are seen in cachexia. Nutritional supplementation and pharmacological manipulation of appetite are unable to restore loss of lean body mass.
-
Resting energy expenditure is increased in patients with lung and pancreatic cancer, but not in gastric and colorectal cancer. Increased energy expenditure might be related to the upregulation of uncoupling proteins (UCPs) — particularly UCP3 in skeletal muscle.
-
Loss of adipose tissue arises predominantly from an increase in lipolysis. Lipolysis is induced by a tumour product, lipid-mobilizing factor (LMF), which acts through a β3-adrenoceptor.
-
Loss of skeletal muscle arises from a fall in protein synthesis and an increase in protein degradation. The decreased protein synthesis could arise from the inactivity of the patient, coupled with a reduction in the supply or balance of amino acids due to acute-phase protein production. Increased protein degradation seems to be mainly due to an increased expression of the components of the ubiquitin-proteasome proteolytic pathway in skeletal muscle.
-
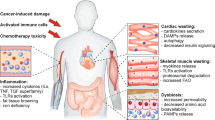
Tissue catabolism in cachexia is partially mediated by cytokines such as tumour necrosis factor-α (TNF-α) or interleukin (IL)-1 and IL-6. Tumour catabolic products such as LMF and proteolysis-inducing factor (PIF) directly stimulate tissue breakdown and are also correlated with human cancer cachexia.
-
Therapy has been mainly targeted at TNF-α and PIF. Agents that are directed solely at TNF-α have not shown clinical activity so far. Anticatabolic agents, such as eicosapentaenoic acid (EPA), effectively downregulate the increased expression of the ubiquitin-proteasome pathway in skeletal muscle and are clinically effective in restoring loss of lean body mass in cachectic cancer patients, especially in combination with a protein and energy-dense supplement.
-
Future therapy will consist of a combination of anabolic and anticatabolic agents.
Abstract
Cachexia — the massive (up to 80%) loss of both adipose tissue and skeletal muscle mass — is a significant factor in the poor performance status and high mortality rate of cancer patients. Although this metabolic defect has been known since cancer was first studied, it is only recently that major advances have been made in the identification of catabolic factors that act to destroy host tissues during the cachectic process. Although anorexia is frequently present, depression of food intake alone seems not to be responsible for the wasting of body tissues, as nutritional supplementation or pharmacological manipulation of appetite is unable to reverse the catabolic process — particularly with respect to skeletal muscle. However, recent clinical studies in cancer patients have shown that nutritional supplementation can be effective when combined with agents that attenuate the action of tumour factors and modifiers of the central effects on appetite might also show promise.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
DeWys, W. D. et al. Prognostic effect of weight loss prior to chemotherapy in cancer patients. Am. J. Med. 69, 491–497 (1980).
Fearon, K. C. H. The mechanisms and treatment of weight loss in cancer. Proc. Nutr. Soc. 51, 251–265 (1992).
Wigmore, S. J., Plester, C. E., Richardson, R. A. & Fearon, K. C. H. Changes in nutritional status associated with unresectable pancreatic cancer. Br. J. Cancer 75, 106–109 (1997).
Kritchevsky, S. B. et al. Changes in plasma lipid and lipoprotein cholesterol and weight prior to diagnosis of cancer. Cancer Res. 51, 3198–3203 (1991).
Grosvenor, M., Balcavage, L. & Chlebowski, R. T. Symptoms potentially influencing weight loss in a cancer population. Cancer 63, 330–338 (1989).
Costa, G. et al. Weight loss and cachexia in lung cancer. Nutr. Cancer 2, 98–103 (1980).
Evans, W. K. et al. Limited impact of total parenteral nutrition on nutritional status during treatment for small cell lung cancer. Cancer Res. 45, 3347–3353 (1985).
Popiela, T., Lucchi, R. & Giongo, F. Methylprednisolone as palliative therapy for female terminal cancer patients. Eur. J. Cancer Clin. Oncol. 25, 1823–1829 (1989).
Maltoni, M. et al. High-dose progestins for the treatment of cancer anorexia-cachexia syndrome. A systematic review of randomised clinical trials. Ann. Oncol. 12, 289–300 (2001).
Loprinzi, C. L., Schaid, D. J., Dose, A. M., Burnham, N. L. & Jensen, M. D. Body-composition changes in patients who gain weight while receiving megestrol acetate. J. Clin. Oncol. 11, 152–154 (1993).
Simons, J. P. F. H. A. et al. Effects of medroxyprogesterone acetate on food intake, body composition, and resting energy expenditure in patients with advanced, nonhormone-sensitive cancer. Cancer 82, 553–560 (1998).
Rowland, K. M. Jr et al. Randomized double-blind placebo-controlled trial of cisplatin and etoposide plus megestrol acetate/placebo in extensive small-cell lung cancer: a North Central Cancer Treatment Group study. J. Clin. Oncol. 14, 135–141 (1996).
Maltoni, M. et al. Medroxyprogesterone acetate reduces the in vitro production of cytokines and serotonin involved in anorexia/cachexia and emesis of peripheral blood mononuclear cells of cancer patients. Eur. J. Cancer 33, 602–607 (1997).
Bing, C., Taylor, S., Tisdale, M. J. & Williams, G. Cachexia in MAC16 adenocarcinoma: suppression of hunger despite normal regulation of leptin, insulin and hypothalamic neuropeptide Y. J. Neurochem. 79, 1004–1012 (2001).
Marks, D. L., Ling, N. & Cone, R. D. Role of the central melanocortin system in cachexia. Cancer Res. 61, 1432–1438 (2001).Demonstrates that cachexia induced by lipopolysaccharide administration and by tumour growth is ameliorated by blockage of the hypothalamic melanocortin-4 receptor.
Kardinal, C. G. et al. A controlled trial of cyproheptadine in cancer patients with anorexia and/or cachexia. Cancer 65, 2657–2661 (1990).
Fredrix, E. W. H. M. et al. Energy balance in relation to cancer cachexia. Clin. Nutr. 9, 319–324 (1990).
Falconer, J. S., Fearon, K. C. H., Plester, C. E., Ross, J. A. & Carter, D. C. Cytokines, the acute-phase response and resting energy expenditure in cachectic patients with pancreatic cancer. Ann. Surg. 219, 325–331 (1994).
McMillan, D. C. et al. Longitudinal study of body cell mass depletion and the inflammatory response in cancer patients. Nutr. Cancer 31, 101–105 (1998).
Falconer, J. S. et al. Acute-phase protein response and survival duration of patients with pancreatic cancer. Cancer 75, 2077–2082 (1995).
Clapham, J. C. et al. Mice overexpressing human uncoupling protein-3 in skeletal muscle are hyperphagic and lean. Nature 406, 415–418 (2000).
Bing, C. et al. Increased gene expression of brown fat uncoupling protein (UCP)1 and skeletal muscle and UCP2 and UCP3 in MAC16-induced cancer cachexia. Cancer Res. 60, 2405–2410 (2000).
Busquets, S. et al. Hyperlipemia: a role in regulating UCP3 gene expression in skeletal muscle during cancer cachexia? FEBS Lett. 505, 255–258 (2001).
Collins, P., Bing, C., McCulloch, P. & Williams, G. Muscle UCP-3 mRNA levels are elevated in weight loss associated with gastrointestinal adenocarcinoma in humans. Br. J. Cancer 86, 372–375 (2002).
Busquets, S. et al. In the rat, tumor necrosis factor-α administration results in an increase in both UCP2 and UCP3 mRNA in skeletal muscle: a possible mechanism for cytokine-induced thermogenesis? FEBS Lett. 440, 348–350 (1998).
Bing, C. et al. Expression of uncoupling proteins-1, -2 and-3 mRNA is induced by an adenocarcinoma-derived lipid-mobilizing factor. Br. J. Cancer 86, 612–618 (2002).
Cabrero, A. et al. Down-regulation of uncoupling protein-3 and-2 by thiazolidinediones in C2C12 myotubes. FEBS Lett. 484, 37–42 (2000).
Eden, E., Edstrom, S., Bennegard, K., Schersten, T. & Lundholm, K. Glucose flux in relation to energy expenditure in malnourished patients with and without cancer during periods of fasting and feeding. Cancer Res. 44, 1718–1724 (1984).
Kaibara, A. et al. Leptin produces anorexia and weight loss without inducing an acute phase response or protein wasting. Am. J. Physiol. 274, R1518–R1525 (1998).
Drott, C., Persson, H. & Lundholm, K. Cardiovascular and metabolic response to adrenaline infusion in weight-losing patients with and without cancer. Clin. Physiol. 9, 427–439 (1989).
Hyltander, A., Daneryd, P., Sandstrom, R., Korner, U. & Lundholm, K. Beta-adrenoceptor activity and resting energy metabolism in weight losing cancer patients. Eur. J. Cancer 36, 330–334 (2000).
Russell, S. T., Hirai, K. & Tisdale, M. J. Role of β3-adrenergic receptors in the action of a tumour lipid mobilizing factor. Br. J. Cancer 86, 424–428 (2002).
Norton, J. A., Stein, T. P. & Brennan, M. F. Whole body protein synthesis and turnover in normal man and malnourished patients with and without cancer. Ann. Surg. 194, 123–128 (1981).
Lundholm, K., Bennegard, K., Eden, E. & Rennie, M. J. Efflux of 3-methylhistidine from the leg of cancer patients who experience weight loss. Cancer Res. 42, 4807–4811 (1982).
Lundholm, K., Bylund, A. C., Holm, J. & Schersten, T. Skeletal muscle metabolism in patients with malignant tumour. Eur. J. Cancer 12, 465–473 (1976).
Warren, R. S., Jeevanandam, M. & Brennan, M. F. Protein synthesis in the tumor-influenced hepatocyte. Surgery 98, 275–281 (1985).
Lecker, S. V., Solomon, V., Mitch, W. E. & Goldberg, A. L. Muscle protein breakdown and critical role of the ubiquitin-proteasome pathway in normal and disease states. J. Nutr. 129, 227S–237S (1999).
Goll, D. E., Thompson, V. F., Taylor, R. G. & Christiansen, J. A. Role of the calpain system in muscle growth. Biochimie 74, 225–237 (1992).
Lowell, B. B., Ruderman, N. B. & Goodman, M. N. Evidence that lysosomes are not involved in the degradation of myofibrilar proteins in rat skeletal muscle. Biochem. J. 234, 237–240 (1986).
Hasselgren, P. O. & Fischer, J. E. Muscle cachexia: current concepts of intracellular mechanisms and molecular regulation. Ann. Surg. 233, 9–17 (2001).
Jagoe, R. T., Redfern, C. P. F., Roberts, R. G., Gibson, G. J. & Goodship, T. H. J. Skeletal muscle mRNA levels for cathepsin B, but not components of the ubiquitin-proteasome pathway are increased in patients with lung cancer referred for thoracotomy. Clin. Sci. 102, 353–361 (2002).
Kisselev, A. F., Akopian, T. N., Castillo, V. & Goldberg, A. L. Proteasome active sites allosterically regulate each other, suggesting a cyclical bite-chew mechanism for protein breakdown. Mol. Cell 4, 395–402 (1999).
Tanaka, K. Molecular biology of the proteasome. Biochem. Biophys. Res. Commun. 247, 537–541 (1998).
Hasselgren, P.-O., Wray, C. & Mammen, J. Molecular regulation of muscle cachexia: it may be more than the proteasome. Biochem. Biophys. Res. Commun. 290, 1–10 (2002).
Temparis, S. et al. Increasd ATP-ubiquitin-dependent proteolysis in skeletal muscle of tumor-bearing rats. Cancer Res. 54, 5568–5573 (1994).
Lorite, M. J., Thompson, M. G., Drake, J. L., Carling, G. & Tisdale, M. J. Mechanism of muscle protein degradation induced by a cancer cachectic factor. Br. J. Cancer 78, 850–856 (1998).
Bodine, S. C. et al. Identification of ubiquitin ligases required for skeletal muscle atrophy. Science 294, 1704–1708 (2001).
Gomes, M. D., Lecker, S. H., Jagoe, R. T., Navon, A. & Goldberg, A. L. Atrogin-1, a muscle specific F-box protein highly expressed during muscle atrophy. Proc. Natl Acad. Sci. USA 98, 14440–14445 (2001).References 47 and 48 provide evidence that ubiquitin-protein ligases (E3) might be rate-limiting for proteasome proteolysis in skeletal muscle.
Bossola, M. et al. Increased muscle ubiquitin mRNA levels in gastric cancer . Am J Physiol Regul Integr Comp Physiol 280, R1518–R1523 (2001).
Williams, A., Sun, X., Fischer, J. E. & Hasselgren, P.-O. The expression of genes in the ubiquitin-proteasome proteolytic pathway is increased in skeletal muscle from patients with cancer. Surgery 126, 744–750 (1999).The first report of increased expression of genes in the ubiquitin-proteasome proteolytic pathway in muscle tissue from patients with cancer.
Mahony, S. M. & Tisdale, M. J. Induction of weight loss and metabolic alterations by human recombinant tumour necrosis factor. Br. J. Cancer 58, 345–351 (1988).
Strassman, G., Fong, M., Kenney, J. S. & Jacob, C. O. Evidence for the involvement of IL-6 in experimental cancer cachexia. J. Clin. Invest. 89, 1681–1684 (1992).
Espat, N. J. et al. Ciliary neurotrophic factor is catabolic and shares with IL-6 the capacity to induce an acute phase response. Am. J. Physiol. 271, R185–R190 (1996).
Langstein, H. et al. The role of γ-interferon and tumor necrosis factor-α in an experimental rat model of cancer cachexia. Cancer Res. 51, 2302–2306 (1991).
Hirai, K., Hussey, H. J., Barber, M. D., Price, S. A. & Tisdale, M. J. Biological evaluation of a lipid-mobilizing factor isolated from the urine of cancer patients. Cancer Res. 58, 2359–2365 (1998).
Todorov, P. et al. Characterization of a cancer cachectic factor. Nature 379, 739–742 (1996).This is the first report on the isolation and characterization of a 24-kDa glycoprotein that is produced by cachexia-inducing mouse and human tumours, which initiates protein catabolism in skeletal muscle.
Oliff, A. et al. Tumors secreting human TNF/cachectin induce cachexia in mice. Cell 50, 555–561 (1987).
Strassmann, G. et al. Mechanisms of experimental cancer cachexia. Local involvement of IL-1 in colon-26 tumor. J. Immunol. 150, 2341–2346 (1993).
Henderson, J. T., Mullen, B. J. M. & Roder, J. C. Physiological effects of CNTF-induced wasting. Cytokine 8, 784–793 (1996).
Matthys, P. et al. Anti-interferon-γ antibody treatment, growth of Lewis lung tumours in mice and tumour-associated cachexia. Eur. J. Cancer 27, 182–186 (1991).
Beck, S. A. & Tisdale, M. J. Production of lipolytic and proteolytic factors by a murine tumor-producing cachexia in the host. Cancer Res. 47, 5919–5923 (1987).
Karayiannakis, A. J. et al. Serum levels of tumor necrosis factor-α and nutritional status in pancreatic cancer patients. Anticancer Res. 21, 1355–1358 (2001).
Maltoni, M. et al. Serum levels of tumour necrosis factor and other cytokines do not correlate with weight loss and anorexia in cancer patients. Support. Care Cancer 5, 130–135 (1997).
Scott, H. R., McMillan, D. C., Crilly, A., McArdle, C. S. & Milroy, R. The relationship between weight loss and interleukin-6 in non-small-cell lung cancer. Br. J. Cancer 73, 1560–1562 (1996).Provides evidence of increased IL-6 in weight-losing cancer patients that might be related to weight loss and the APR.
Moradi, M. M. et al. Serum and ascitic fluid levels of interleukin-1, interleukin-6 and tumor necrosis factor-alpha in patients with ovarian epithelial cancer. Cancer 72, 2433–2437 (1993).
Lorite, M. J. et al. Activation of ATP-ubiquitin-dependent proteolysis in skeletal muscle in vivo and murine myoblasts in vitro by a proteolysis-inducing factor (PIF). Br. J. Cancer 85, 297–302 (2001).
Wigmore, S. J. et al. Characteristics of patients with pancreatic cancer expressing a novel cancer cachectic factor. Br. J. Surg. 87, 53–58 (2000).
Hauner, H., Petruschke, T., Russ, M., Rohrig, K. & Eckel, J. Effects of tumor necrosis factor alpha (TNFα) on glucose transport and lipid metabolism of newly-differentiated human fat cells in cell culture. Diabetologica 38, 764–771 (1995).
Li, Y. P. et al. Skeletal muscle myocytes undergo protein loss and reactive oxygen-mediated NF-κB activation in response to tumor necrosis factor α. FASEB J. 12, 871–880 (1998).Demonstrates that TNF-α is capable of directly inducing protein degradation in vitro through the ubiquitin-proteasome pathway.
Ebisui, C. et al. Interleukin-6 induces proteolysis by activating intracellular proteases (cathepsins B and L, proteasome) in C2C12 myotubes. Clin. Sci. 89, 431–439 (1995).
Llovera, M. et al. Different cytokines modulate ubiquitin gene expression in rat skeletal muscle. Cancer Lett. 133, 83–87 (1998).
Watchorn, T. M., Waddell, I. D., Dowidar, N. & Ross, J. A. Proteolysis-inducing factor regulates hepatic gene expression via the transcription factors NF-κB and STAT3. FASEB J. 15, 562–564 (2001).Suggests that PIF, in addition to inducing protein degradation in skeletal muscle, might be responsible for APP production in liver via activation of cytokine production.
Cangiano, C. et al. Effects of administration of oral branched-chain amino acids on anorexia and caloric intake in cancer patients. J. Natl Cancer Inst. 88, 550–552 (1996).
Lissoni, P. et al. Is there a role for melatonin in the treatment of neoplastic cachexia? Eur. J. Cancer 32A, 1340–1343 (1996).
Lissoni, P. et al. Inhibition of tumor necrosis factor-alpha secretion by pentoxifylline in advanced cancer patients with abnormally high blood levels of tumor necrosis factor α J. Biol. Regul. Homeost. Agents 7, 73–75(1993).
Goldberg, R. M. et al. Pentoxifylline for treatment of cancer anorexia and cachexia? A randomised, double-blind, placebo-controlled trial. J. Clin. Oncol. 13, 2856–2859 (1995).
Bruera, E. et al. Thalidomide in patients with cachexia due to terminal cancer. Preliminary report. Ann. Oncol. 10, 857–859 (1999).
Reyes-Teran, G. et al. Effects of thalidomide on HIV-associated wasting syndrome: a randomized, double-blind, placebo-controlled trial. AIDS 10, 1501–1507 (1996).
McMillam, D. C., Gorman, P. O., Fearon, K. C. H. & McArdle, C. S. A pilot study of megestrol acetate and ibuprofen in the treatment of cachexia in gastrointestinal patients. Br. J. Cancer 76, 788–790 (1997).
Whitehouse, A. S., Smith, H. J., Drake, J. L. & Tisdale, M. J. Mechanism of attenuation of skeletal muscle protein catabolism in cancer cachexia by eicosapentaenoic acid. Cancer Res. 61, 3604–3609 (2001).Provides a mechanistic interpretation of the effect of EPA on protein degradation in skeletal muscle by downregulating the increased expression of the ubiquitin-proteasome proteolytic pathway that accompanies cachexia.
Wigmore, S. J. et al. The effect of polyunsaturated fatty acids on the progress of cachexia in patients with pancreatic cancer. Nutrition 12, S27–S30 (1996).
Wigmore, S. J., Barber, M. D., Ross, J. A., Tisdale, M. J. & Fearon, K. C. H. Effect of oral eicosapentaenoic acid on weight loss in patients with pancreatic cancer. Nutr. Cancer 36, 177–184 (2000).
Barber, M. D., Ross, J. A., Voss, A. C., Tisdale, M. J. & Fearon, K. C. H. The effect of an oral nutritional supplement enriched with fish oil on weight-loss in patients with pancreatic cancer. Br. J. Cancer 81, 80–86 (1999).Combination of fish oil with an energy-dense nutritional supplement was found to increase body weight in cachectic cancer patients. The increase in body weight was solely due to an increase in lean body mass.
May, P. E., Barker, A., D'Olimpio, J. T., Hourihane, A. & Abumrad, N. N. Reversal of cancer-related wasting using oral supplementation with a combination of β-hydroxy-β-methylbutyrate, arginine and glutamine. Am. J. Surg. 183, 471–479 (2002).This is the first clinical trial demonstrating the ability of the leucine metabolite, β-hydroxy-β-methylbutyrate, to increase fat-free mass in cachectic cancer patients.
Ostaszewski, P. et al. The leucine metabolite 3-hydroxy-3-methylbutyrate (HMB) modifies protein turnover in muscles of the laboratory rats and domestic chickens in vitro. J. Anim. Physiol. Anim. Nutr. 84, 1–8 (2000).
Barber, M. D., Fearon, K. C. H., Tisdale, M. J., McMillan, D. C. & Ross, J. A. Effect of a fish oil-enriched nutritional supplement on metabolic mediators in patients with pancreatic cancer cachexia. Nutr. Cancer 40, 118–124 (2001).
Hussey, H. J. & Tisdale, M. J. Effect of a cachectic factor on carbohydrate metabolism and attenuation by eicosapentaenoic acid. Br. J. Cancer 80, 1231–1235 (1999).
Smith, H. J., Lorite, M. J. & Tisdale, M. J. Effect of a cancer cachectic factor on protein synthesis/degradation in murine C2C12 myoblasts: modulation by eicosapentaenoic acid. Cancer Res. 59, 5507–5513 (1999).
Tan, C. & Waldmann, T. A. Proteasome inhibitor PS-341, a potential therapeutic agent for adult T-cell leukaemia. Cancer Res. 62, 1083–1086 (2002).
Spataro, V., Norbury, C & Harris, A. L. The ubiquitin-proteasome pathway in cancer. Br. J. Cancer 77, 448–455 (1998).
Thompson, M. P., Cooper, S. T., Parry, B. R. & Tuckey, J. A. Increased expression of the mRNA for the hormone-sensitive lipase in adipose tissue of cancer patients. Biochim. Biophys. Acta 1180, 236–242 (1993).
Pisa, P., Stenke, L., Bernell, P., Hanssom, M. & Hast, R. Tumor necrosis factor-α and interferon-α in serum of multiple myeloma patients. Anticancer Res. 10, 817–820 (1990).
Todorov, P. T. et al. Purification and characterization of a tumor lipid-mobilizing factor. Cancer Res. 58, 2353–2358 (1998).
Strang, P. The effect of megestrol acetate on anorexia, weight loss and cachexia in cancer and AIDS patients. Anticancer Res. 17, 657–664 (1997).
Author information
Authors and Affiliations
Related links
Related links
DATABASES
Cancer.gov
LocusLink
OMIM
FURTHER INFORMATION
Biomedica cachexia information page
Glossary
- CARCASS LIPID
-
The total fat content of the body when the water has been removed.
- BROWN ADIPOSE TISSUE
-
(BAT). A special type of adipose tissue, the sole function of which is to burn off excess fat and generate heat. It is found in the interscapular region and is most important in the neonate. No BAT has been detected in adult humans, although brown adipocytes might be present in white adipose tissue.
- LIPOLYSIS
-
The process by which triglycerides, which are stored in adipose tissue, are broken down into glycerol and non-esterified fatty acids.
- ASTHENIA
-
Muscle weakness.
- KARNOVSKY INDEX
-
A physician-scored performance scale ranging from 0 to 100 that determines the level of patient activity. A score of 0 means that the patient is totally inactive, a score of 50 means that the patient is just able to get out of bed, and a score of 100 means that the patient is able to function normally.
Rights and permissions
About this article
Cite this article
Tisdale, M. Cachexia in cancer patients. Nat Rev Cancer 2, 862–871 (2002). https://doi.org/10.1038/nrc927
Issue Date:
DOI: https://doi.org/10.1038/nrc927
This article is cited by
-
The time-course of cancer cachexia onset reveals biphasic transcriptional disruptions in female skeletal muscle distinct from males
BMC Genomics (2023)
-
Nomogram for predicting the overall survival of underweight patients with colorectal cancer: a clinical study
BMC Gastroenterology (2023)
-
Investigating pathogenic SNP of PKCι in HCV-induced hepatocellular carcinoma
Scientific Reports (2023)
-
A large-scale study integrating nutritional indicators and clinicopathological parameters to evaluate prognosis, follow-up, and postoperative chemotherapy decisions in rectal cancer patients
Supportive Care in Cancer (2023)
-
A novel prognostic model based on pretreatment serum albumin and ECOG PS for primary CNS lymphoma: an international, multi-center study
Journal of Neuro-Oncology (2023)