Key Points
-
Adenocarcinomas of the lung and pancreas are among the most common and most deadly cancers. They share two risk factors — smoking and high-fat diet — with cardiovascular disease.
-
The arachidonic acid (AA) cascade is important in cardiovascular disease, and the overexpression of the gene that encodes cyclooxygenase-2 (COX2) in pulmonary and pancreatic adenocarcinoma indicates that the AA-cascade is also involved in the development of these cancers.
-
A nicotine-derived nitrosamine, nitrosamine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK), causes adenocarcinomas of the lung and pancreas in laboratory animals, and is thought to be largely responsible for the development of these cancers in smokers.
-
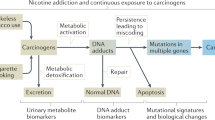
NNK has genotoxic effects on cells, such as the formation of DNA adducts and mutations in the RAS gene. These effects of NNK probably cause the development of cancers that express such gene mutations (∼30% of pulmonary and 50–90% of pancreatic adenocarcinomas).
-
NNK also has epigenetic effects on pulmonary and pancreatic cells by functioning as an agonist for β-adrenergic receptors. This reaction activates various signal-transduction pathways, and causes the release of AA followed by the formation of mitogenic AA metabolites. These β-adrenergic-receptor-mediated events activate transcription and cell proliferation, and are thought to cause pulmonary and pancreatic adenocarcinomas that do not express RAS mutations, as well as synergizing with the cancer-causing effects of mutated RAS.
-
β-Blockers, inhibitors of AA-metabolizing enzymes and a low-fat diet are already widely used for the treatment and prevention of cardiovascular disease. The data compiled in this review indicate that they will also be effective for the treatment and prevention of pulmonary and pancreatic adenocarcinomas.
-
Overexpression of COX2 in adenocarcinomas of the colon, prostate and breast, as well as recent reports that β-adrenergic signalling regulates growth of these cancers, indicate that adenocarcinomas at these organ sites might also be treated or prevented with β-blockers. They might also be treated with pharmacological or dietary inhibitors of the AA cascade.
Abstract
Adenocarcinoma of the lungs and pancreas are among the most common and most deadly smoking-associated cancers. Cigarette smoke contains various toxic chemicals, including a carcinogenic nitrosamine, nitrosamine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK). One of the most well-known features of NNK is the ability of its metabolites to bind to DNA and induce activating point mutations in the RAS gene. But NNK is also a β-adrenergic-receptor agonist that stimulates arachidonic acid release, leading to the formation of mitogenic metabolites that stimulate DNA synthesis and cell proliferation. NNK therefore contributes to tobacco-induced carcinogenesis by several mechanisms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
American Cancer Society. Cancer facts and figures. (American Cancer Society, Atlanta, Georgia, 2002).
Zheng, T. et al. Time trend and the age-period-cohort effect on the incidence of histologic types of lung cancer in Connecticut, 1960–1989. Cancer 74, 1556–1567 (1994).
Kelly, A., Blair, N. & Pechacek, T. F. Women and smoking: issues and opportunities. J. Womens Health Gender-Based Med. 10, 515–518 (2001).This reference summarizes the 2001 report of the Surgeon General on women and smoking. It documents that smoking-related lung cancer mortality in women in the United States has increased by about 600% since 1950.
Bunn, P. A. Jr, Vokes, E. E., Langer, C. J. & Schiller, J. H. An update on North American randomized studies in non-small cell lung cancer. Semin. Oncol. 25, 2–10 (1998).
Wynder, E. L. & Muscat, J. E. The changing epidemiology of smoking and lung cancer histology. Environ. Health Perspect. 103 (Suppl. 8), 143–148 (1995).
Levi, F., Franceschi, S., La Vecchia, C., Randimbison, L. & Te, V. C. Lung carcinoma trends by histologic type in Vaud and Neuchatel, Switzerland, 1974–1994. Cancer 79, 906–914 (1997).
Osann, K. E. Lung cancer in women: the importance of smoking, family history of cancer, and medical history of respiratory disease. Cancer Res. 51, 4893–4897 (1991).
Risch, H. A. et al. Are female smokers at higher risk for lung cancer than male smokers? A case–control analysis by histologic type. Am. J. Epidemiol. 138, 281–293 (1993).
Harris, R. E., Zang, E. A., Anderson, J. I. & Wynder, E. L. Race and sex differences in lung cancer risk associated with cigarette smoking. Int. J. Epidemiol. 22, 592–599 (1993).
Haugen, A. Women who smoke: are women more susceptible to tobacco-induced lung cancer? Carcinogenesis 23, 227–229 (2002).
Radzikowska, E., Roszkowski, K. & Glaz, P. Lung cancer in patients under 50 years old. Lung Cancer 33, 203–211 (2001).
Stellman, S. D. et al. Smoking and lung cancer risk in American and Japanese men: an international case–control study. Cancer Epidemiol. Biomarkers Prev. 10, 1193–1199 (2001).
Lee, P. N. Lung cancer and type of cigarette smoked. Inhal. Toxicol. 13, 951–976 (2001).
Chowdhury, P. & Rayford, P. L. Smoking and pancreatic disorders. Eur. J. Gastroenterol. Hepatol. 12, 869–877 (2000).
Gold, E. B. & Goldin, S. B. Epidemiology of and risk factors for pancreatic cancer. Surg. Oncol. Clin. N. Am. 7, 67–91 (1998).This epidemiological study identifies smoking and diets that are high in meat as risk factors for pancreatic cancer (see also reference 29).
Silverman, D. T. Risk factors for pancreatic cancer: a case–control study based on direct interviews. Teratog. Carcinog. Mutagen. 21, 7–25 (2001).
Duell, E. J. et al. A population-based, case–control study of polymorphisms in carcinogen-metabolizing genes, smoking, and pancreatic adenocarcinoma risk. J. Natl Cancer Inst. 94, 297–306 (2002).
Boyle, P. et al. Cigarette smoking and pancreas cancer: a case–control study of the search programme of the IARC. Int. J. Cancer 67, 63–71 (1996).
Hoffmann, D. et al. A study of tobacco carcinogenesis. LI. Relative potencies of tobacco-specific N-nitrosamines as inducers of lung tumours in A/J mice. Cancer Lett. 71, 25–30 (1993).
Schuller, H. M. et al. Pathobiology of lung tumors induced in hamsters by 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone and the modulating effect of hyperoxia. Cancer Res. 50, 1960–1965 (1990).
Schuller, H. M., Jorquera, R., Reichert, A. & Castonguay, A. Transplacental induction of pancreas tumors in hamsters by ethanol and the tobacco-specific nitrosamine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone. Cancer Res. 53, 2498–2501 (1993).
Hoffmann, D., Rivenson, A. & Hecht, S. S. The biological significance of tobacco-specific N-nitrosamines: smoking and adenocarcinoma of the lung. Crit. Rev. Toxicol. 26, 199–211 (1996).
Hecht, S. S. Recent studies on mechanisms of bioactivation and detoxification of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK), a tobacco-specific lung carcinogen. Crit. Rev. Toxicol. 26, 163–181 (1996).
Hecht, S. S. Carcinogen-derived biomarkers and lung cancer. Prev. Med. 25, 7–9 (1996).
Rivenson, A., Hoffmann, D., Prokopczyk, B., Amin, S. & Hecht, S. S. Induction of lung and exocrine pancreas tumors in F344 rats by tobacco-specific and Areca-derived N-nitrosamines. Cancer Res. 48, 6912–6917 (1988).
Hecht, S. S., Trushin, N., Castonguay, A. & Rivenson, A. Comparative tumorigenicity and DNA methylation in F344 rats by 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone and N-nitrosodimethylamine. Cancer Res. 46, 498–502 (1986).
Hoffmann, D., Castonguay, A., Rivenson, A. & Hecht, S. S. Comparative carcinogenicity and metabolism of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone and N′-nitrosonornicotine in Syrian golden hamsters. Cancer Res. 41, 2386–2393 (1981).
Hecht, S. S. Biochemistry, biology, and carcinogenicity of tobacco-specific N-nitrosamines. Chem. Res. Toxicol. 11, 559–603 (1998).
Hoffmann, D., Rivenson, A., Chung, F. L. & Hecht, S. S. Nicotine-derived N-nitrosamines (TSNA) and their relevance in tobacco carcinogenesis. Crit. Rev. Toxicol. 21, 305–311 (1991).
Hecht, S. S. & Hoffmann, D. N-nitroso compounds and tobacco-induced cancers in man. IARC Sci. Publ. 105, 54–61 (1991).
Brunnemann, K. D., Prokopczyk, B., Djordjevic, M. V. & Hoffmann, D. Formation and analysis of tobacco-specific N-nitrosamines. Crit. Rev. Toxicol. 26, 121–137 (1996).
Carmella, S. G., Borukhova, A., Desai, D. & Hecht, S. S. Evidence for endogenous formation of tobacco-specific nitrosamines in rats treated with tobacco alkaloids and sodium nitrite. Carcinogenesis 18, 587–592 (1997).
Correa, E., Joshi, P. A., Castonguay, A. & Schuller, H. M. The tobacco-specific nitrosamine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone is an active transplacental carcinogen in Syrian golden hamsters. Cancer Res. 50, 3435–3438 (1990).
Lackmann, G. M. et al. Metabolites of a tobacco-specific carcinogen in urine from newborns. J. Natl Cancer Inst. 91, 459–465 (1999).
Alavanja, M. C. et al. Lung cancer risk and red meat consumption among Iowa women. Lung Cancer 34, 37–46 (2001).
Alavanja, M. C., Brown, C. C., Swanson, C. & Brownson, R. C. Saturated fat intake and lung cancer risk among nonsmoking women in Missouri. J. Natl Cancer Inst. 85, 1906–1916 (1993).
Truninger, K. [Risk groups for pancreatic and bile duct carcinomas]. Schweiz Rundsch. Med. Prax. 89, 1299–1304 (2000).Describes, for the first time, the tumour-promoting effects of dietary fat on NNK-induced lung cancer in an animal model.
Howe, G. R. et al. A collaborative case–control study of nutrient intake and pancreatic cancer within the search programme. Int. J. Cancer 51, 365–372 (1992).
Hoffmann, D., Rivenson, A., Abbi, R. & Wynder, E. L. A study of tobacco carcinogenesis: effect of the fat content of the diet on the carcinogenic activity of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone in F344 rats. Cancer Res. 53, 2758–2761 (1993).
Veierod, M. B., Laake, P. & Thelle, D. S. Dietary fat intake and risk of lung cancer: a prospective study of 51,452 Norwegian men and women. Eur. J. Cancer Prev. 6, 540–549 (1997).This epidemiological study reports that dietary fat increases the risk of lung cancer in smokers.
Tucker, O. N. et al. Cyclooxygenase-2 expression is up-regulated in human pancreatic cancer. Cancer Res. 59, 987–990 (1999).Reports that cyclooxygenase is upregulated in human pancreatic cancer.
Molina, M. A., Sitja-Arnau, M., Lemoine, M. G., Frazier, M. L. & Sinicrope, F. A. Increased cyclooxygenase-2 expression in human pancreatic carcinomas and cell lines: growth inhibition by nonsteroidal anti-inflammatory drugs. Cancer Res. 59, 4356–4362 (1999).
Hida, T. et al. Increased expression of cyclooxygenase 2 occurs frequently in human lung cancers, specifically in adenocarcinomas. Cancer Res. 58, 3761–3764 (1998).Reports that, of the various types of lung cancer, adenocarcinoma are most likely to overexpress cyclooxygenase-2.
Rioux, N. & Castonguay, A. Recovery from 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone-induced immunosuppression in A/J mice by treatment with nonsteroidal anti-inflammatory drugs. J. Natl Cancer Inst. 89, 874–880 (1997).
Duperron, C. & Castonguay, A. Chemopreventive efficacies of aspirin and sulindac against lung tumorigenesis in A/J mice. Carcinogenesis 18, 1001–1006 (1997).The first report that documents a chemopreventive effect of a non-steroidal anti-inflammatory drug on NNK-induced lung cancer in an animal model.
Castonguay, A. & Rioux, N. Inhibition of lung tumourigenesis by sulindac: comparison of two experimental protocols. Carcinogenesis 18, 491–496 (1997).
Rioux, N. & Castonguay, A. Prevention of NNK-induced lung tumorigenesis in A/J mice by acetylsalicylic acid and NS-398. Cancer Res. 58, 5354–5360 (1998).Shows, for the first time, a cancer-preventive effect of a lipoxygenase inhibitor on NNK-induced lung cancer in an animal model.
Rioux, N. & Castonguay, A. Inhibitors of lipoxygenase: a new class of cancer chemopreventive agents. Carcinogenesis 19, 1393–1400 (1998).
Malkinson, A. M. et al. Inhibition of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone-induced mouse lung tumor formation by FGN-1 (sulindac sulfone). Carcinogenesis 19, 1353–1356 (1998).
Castonguay, A., Rioux, N., Duperron, C. & Jalbert, G. Inhibition of lung tumorigenesis by NSAIDS: a working hypothesis. Exp. Lung Res. 24, 605–615 (1998).
El-Bayoumy, K., Iatropoulos, M., Amin, S., Hoffmann, D. & Wynder, E. L. Increased expression of cyclooxygenase-2 in rat lung tumors induced by the tobacco-specific nitrosamine 4-(methylnitrosamino)-4-(3-pyridyl)-1-butanone: the impact of a high-fat diet. Cancer Res. 59, 1400–1403 (1999).
Harris, R. E., Schuller, H. M., Bell, J. L., Lu, P. Y. & Alshafie, G. A. Association of nonsteroidal anti-inflammatory drugs (NSAIDs) and anti-hypertensive medications (AHMs) with lung cancer among smokers and in a hamster model of NNK–induced lung cancer. Proc. Am. Assoc. Cancer Res. 42, 1424 (2001).
Schuller, H. M., Tithof, P. K., Williams, M. & Plummer, H. The tobacco-specific carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone is a β-adrenergic agonist and stimulates DNA synthesis in lung adenocarcinoma via β-adrenergic receptor-mediated release of arachidonic acid. Cancer Res. 59, 4510–4515 (1999).In vitro experiments that identify, for the first time, the tobacco nitrosamine NNK as an agonist for β-adrenergic receptors. This study also shows that binding of NNK to β-adrenergic receptors in human lung cancer cells causes the release of arachidonic acid, resulting in stimulation of DNA synthesis.
Schuller, H. M., Porter, B. & Riechert, A. β-Adrenergic modulation of NNK-induced lung carcinogenesis in hamsters. J. Cancer Res. Clin. Oncol. 126, 624–630 (2000).Reports that a β-blocker acts as a potent chemopreventive agent on NNK-induced lung cancer in an animal model. This study also showed that adrenaline and theophylline promoted the development of lung cancer.
Schuller, H. M. et al. Co-expression of β-adrenergic receptors and cyclooxygenase-2 in pulmonary adenocarcinoma. Int. J. Oncol. 19, 445–449 (2001).
Baek, S. J., Kim, K. S., Nixon, J. B., Wilson, L. C. & Eling, T. E. Cyclooxygenase inhibitors regulate the expression of a TGF-β superfamily member that has proapoptotic and antitumorigenic activities. Mol. Pharmacol. 59, 901–908 (2001).
Kim, K. S. et al. Expression and regulation of nonsteroidal anti-inflammatory drug-activated gene (NAG-1) in human and mouse tissue. Gastroenterology 122, 1388–1398 (2002).
Weddle, D. L., Tithoff, P., Williams, M. & Schuller, H. M. β-Adrenergic growth regulation of human cancer cell lines derived from pancreatic ductal carcinomas. Carcinogenesis 22, 473–479 (2001).In vitro studies describe, for the first time, that the growth of human pancreatic cancer cells is regulated by β-adrenergic receptors by the release of arachidonic acid and that NNK activates this pathway.
Schuller, H. M. et al. Inhibition of pancreatic carcinogenesis in hamsters by inhibitors of the AA-cascade or a β–blocker. Proc. Am. Assoc. Cancer Res. 43, 3201 (2002).
Hecht, S. S. et al. Quantitation of metabolites of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone after cessation of smokeless tobacco use. Cancer Res. 62, 129–134 (2002).Supports the hypothesis that β-adrenergic-receptor pathways are involved in NNK-induced lung carcinogenesis. The authors conclude that retention of NNAL in lungs of smokers and smokeless-tobacco users is best explained by the presence of β-adrenergic receptors.
Ruffolo, R. R. Jr, Bondinell, W. & Hieble, J. P. α- and β-adrenoceptors: from the gene to the clinic. 2. Structure–activity relationships and therapeutic applications. J. Med. Chem. 38, 3681–3716 (1995).
Hieble, J. P., Bondinell, W. E. & Ruffolo, R. R., Jr. α- and β-adrenoceptors: from the gene to the clinic. 1. Molecular biology and adrenoceptor subclassification. J. Med. Chem. 38, 3415–3444 (1995).
Pavoine, C., Magne, S., Sauvadet, A. & Pecker, F. Evidence for a β2-adrenergic/arachidonic acid pathway in ventricular cardiomyocytes. Regulation by the β1-adrenergic/cAMP pathway. J. Biol. Chem. 274, 628–637 (1999).
Borda, E. S., Tenenbaum, A., Sales, M. E., Rumi, L. & Sterin-Borda, L. Role of arachidonic acid metabolites in the action of a β-adrenergic agonist on human monocyte phagocytosis. Prostaglandins Leukot. Essent. Fatty Acids 58, 85–90 (1998).
Kan, H., Ruan, Y. & Malik, K. U. Signal transduction mechanism(s) involved in prostacyclin production elicited by acetylcholine in coronary endothelial cells of rabbit heart. J. Pharmacol. Exp. Ther. 282, 113–122 (1997).
Ruan, Y., Kan, H. & Malik, K. U. β-Adrenergic receptor stimulated prostacyclin synthesis in rabbit coronary endothelial cells is mediated by selective activation of phospholipase D: inhibition by adenosine 3′5′-cyclic monophosphate. J. Pharmacol. Exp. Ther. 281, 1038–1046 (1997).
Luttrell, L. M. et al. β-arrestin-dependent formation of β2 adrenergic receptor–Src protein kinase complexes. Science 283, 655–661 (1999).
Luttrell, L. M., Daaka, Y. & Lefkowitz, R. J. Regulation of tyrosine kinase cascades by G-protein-coupled receptors. Curr. Opin. Cell Biol. 11, 177–183 (1999).
Muyderman, H., Sinclair, J., Jardemark, K., Hansson, E. & Nilsson, M. Activation of β-adrenoceptors opens calcium-activated potassium channels in astroglial cells. Neurochem. Int. 38, 269–276 (2001).
Vossler, M. R. et al. cAMP activates MAP kinase and Elk-1 through a B-Raf- and Rap1-dependent pathway. Cell 89, 73–82 (1997).
Daniel, P. B., Walker, W. H. & Habener, J. F. Cyclic AMP signaling and gene regulation. Annu. Rev. Nutr. 18, 353–383 (1998).
Walker, W. H., Daniel, P. B. & Habener, J. F. Inducible cAMP early repressor ICER down-regulation of CREB gene expression in Sertoli cells. Mol. Cell Endocrinol. 143, 167–178 (1998).
Maudsley, S. et al. The β2-adrenergic receptor mediates extracellular signal-regulated kinase activation via assembly of a multi-receptor complex with the epidermal growth factor receptor. J. Biol. Chem. 275, 9572–9580 (2000).
Ahn, S., Maudsley, S., Luttrell, L. M., Lefkowitz, R. J. & Daaka, Y. Src-mediated tyrosine phosphorylation of dynamin is required for β2-adrenergic receptor internalization and mitogen-activated protein kinase signaling. J. Biol. Chem. 274, 1185–1188 (1999).
Biscardi, J. S., Belsches, A. P. & Parsons, S. J. Characterization of human epidermal growth factor receptor and c-Src interactions in human breast tumor cells. Mol. Carcinog. 21, 261–272 (1998).
Maa, M. C., Leu, T. H., McCarley, D. J., Schatzman, R. C. & Parsons, S. J. Potentiation of epidermal growth factor receptor-mediated oncogenesis by c-Src: implications for the etiology of multiple human cancers. Proc. Natl Acad. Sci. USA 92, 6981–6985 (1995).
Blake, R. A. et al. SU6656, a selective src family kinase inhibitor, used to probe growth factor signaling. Mol. Cell. Biol. 20, 9018–9027 (2000).
Chaturvedi, P., Reddy, M. V. & Reddy, E. P. Src kinases and not JAKs activate STATs during IL-3 induced myeloid cell proliferation. Oncogene 16, 1749–1758 (1998).
Garcia, R. et al. Constitutive activation of Stat3 in fibroblasts transformed by diverse oncoproteins and in breast carcinoma cells. Cell Growth Differ. 8, 1267–1276 (1997).
Smith, P. D. & Crompton, M. R. Expression of v-src in mammary epithelial cells induces transcription via STAT3. Biochem. J. 331, 381–385 (1998).
Darnell, J. E. Jr. STATs and gene regulation. Science 277, 1630–1635 (1997).
Mousavi, M., Hellstrom-Lindahl, E., Guan, Z. Z., Bednar, I. & Nordberg, A. Expression of nicotinic acetylcholine receptors in human and rat adrenal medulla. Life Sci. 70, 577–590 (2001).
Tachikawa, E. et al. Characterization of the functional subunit combination of nicotinic acetylcholine receptors in bovine adrenal chromaffin cells. Neurosci. Lett. 312, 161–164 (2001).
Ohta, S. et al. A comparative study of characteristics of current-type and conventional-type cationic bactericides. Biol. Pharm. Bull. 24, 1093–1096 (2001).
Utsunomiya, K. et al. Stimulation of catecholamine synthesis in cultured bovine adrenal medullary cells by leptin. J. Neurochem. 76, 926–934 (2001).
Li, Q. & Forsberg, E. J. Catecholamine secretion induced by nicotine is due to Ca2+ channel but not Na+ channel activation in porcine adrenal chromaffin cells. J. Pharmacol. Exp. Ther. 277, 1209–1214 (1996).
Masur, K., Niggemann, B., Zanker, K. S. & Entschladen, F. Norepinephrine-induced migration of SW 480 colon carcinoma cells is inhibited by β-blockers. Cancer Res. 61, 2866–2869 (2001).Evidence that noradrenaline binds to β-adrenergic receptors to induce the migration of colon cancer cells. These findings provide strong support for our theory that, in addition to adenocarcinomas in the lungs and pancreas, adenocarcinomas in the colon might benefit from therapy and prevention with β-blockers or inhibitors of AA-metabolizing enzymes.
Langenfeld, J. et al. Inhibited transformation of immortalized human bronchial epithelial cells by retinoic acid is linked to cyclin E down-regulation. Oncogene 13, 1983–1990 (1996).
Lonardo, F. et al. Evidence for the epidermal growth factor receptor as a target for lung cancer prevention. Clin. Cancer Res. 8, 54–60 (2002).
Hsieh, E. T., Shepherd, F. A. & Tsao, M. S. Co-expression of epidermal growth factor receptor and transforming growth factor-α is independent of RAS mutations in lung adenocarcinoma. Lung Cancer 29, 151–157 (2000).
Poch, B. et al. Epidermal growth factor induces cyclin D1 in human pancreatic carcinoma: evidence for a cyclin D1-dependent cell cycle progression. Pancreas 23, 280–287 (2001).
Murphy, L. O. et al. Pancreatic cancer cells require an EGF receptor-mediated autocrine pathway for proliferation in serum-free conditions. Br. J. Cancer 84, 926–935 (2001).
Tungekar, M. F. et al. Interleukin 4 receptor expression on human lung tumors and normal lung. Cancer Res. 51, 261–264 (1991).
Pai, R. et al. Prostaglandin E2 transactivates EGF receptor: a novel mechanism for promoting colon cancer growth and gastrointestinal hypertrophy. Nature Med. 8, 289–293 (2002).
Hecht, S. S. DNA adduct formation from tobacco-specific N-nitrosamines. Mutat. Res. 424, 127–142 (1999).
Hecht, S. S. Tobacco smoke carcinogens and lung cancer. J. Natl Cancer Inst. 91, 1194–1210 (1999).
Belinsky, S. A., Devereux, T. R., Maronpot, R. R., Stoner, G. D. & Anderson, M. W. Relationship between the formation of promutagenic adducts and the activation of the K-ras protooncogene in lung tumors from A/J mice treated with nitrosamines. Cancer Res. 49, 5305–5311 (1989).
Belinsky, S. A., Devereux, T. R. & Anderson, M. W. Role of DNA methylation in the activation of proto-oncogenes and the induction of pulmonary neoplasia by nitrosamines. Mutat. Res. 233, 105–116 (1990).
Belinsky, S. A., Foley, J. F., White, C. M., Anderson, M. W. & Maronpot, R. R. Dose-response relationship between O6-methylguanine formation in Clara cells and induction of pulmonary neoplasia in the rat by 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone. Cancer Res. 50, 3772–3780 (1990).
Peterson, L. A. & Hecht, S. S. O6-methylguanine is a critical determinant of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone tumorigenesis in A/J mouse lung. Cancer Res. 51, 5557–5564 (1991).
Staretz, M. E., Foiles, P. G., Miglietta, L. M. & Hecht, S. S. Evidence for an important role of DNA pyridyloxobutylation in rat lung carcinogenesis by 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone: effects of dose and phenethyl isothiocyanate. Cancer Res. 57, 259–266 (1997).
Chyczewski, L. et al. Morphological and molecular aspects of cancerogenesis in the lung. Folia Histochem. Cytobiol. 39, 149–152 (2001).
Kovalchuk, O. et al. K-ras codon 12 mutations detected with enriched PCR method in operable non-small cell lung cancer. Folia Histochem. Cytobiol. 39 (Suppl. 2), 68–69 (2001).
Vahakangas, K. H. et al. p53 and K-ras mutations in lung cancers from former and never-smoking women. Cancer Res. 61, 4350–4356 (2001).
van Laethem, J. L. Ki-ras oncogene mutations in chronic pancreatitis: which discriminating ability for malignant potential? Ann. NY Acad. Sci. 880, 210–218 (1999).
Oreffo, V. I., Lin, H. W., Padmanabhan, R. & Witschi, H. K-ras and p53 point mutations in 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone-induced hamster lung tumors. Carcinogenesis 14, 451–455 (1993).
Berger, D. H. et al. Mutational activation of K-ras in nonneoplastic exocrine pancreatic lesions in relation to cigarette smoking status. Cancer 85, 326–332 (1999).
Malats, N. et al. Ki-ras mutations in exocrine pancreatic cancer: association with clinico-pathological characteristics and with tobacco and alcohol consumption. PANK-ras I Project Investigators. Int. J. Cancer 70, 661–667 (1997).
Pabst, B. et al. Analysis of K-ras mutations in pancreatic tissue after fine needle aspirates. Anticancer Res. 19, 2481–2483 (1999).
Luttges, J. et al. The K-ras mutation pattern in pancreatic ductal adenocarcinoma usually is identical to that in associated normal, hyperplastic, and metaplastic ductal epithelium. Cancer 85, 1703–1710 (1999).
Wang, L., Spratt, T. E., Pegg, A. E. & Peterson, L. A. Synthesis of DNA oligonucleotides containing site-specifically incorporated O6-[4-oxo-4-(3-pyridyl)butyl]guanine and their reaction with O6-alkylguanine-DNA alkyltransferase. Chem. Res. Toxicol. 12, 127–131 (1999).
Wang, L. et al. Pyridyloxobutyl adduct O6-[4-oxo-4-(3-pyridyl)butyl]guanine is present in 4-(acetoxymethylnitrosamino)-1-(3-pyridyl)-1-butanone-treated DNA and is a substrate for O6-alkylguanine-DNA alkyltransferase. Chem. Res. Toxicol. 10, 562–567 (1997).
Kuller, L. H. Dietary fat and chronic diseases: epidemiologic overview. J. Am. Diet Assoc. 97, S9–S15 (1997).
Halimi, J. M. et al. The risk of hypertension in men: direct and indirect effects of chronic smoking. J. Hypertens. 20, 187–193 (2002).
Schaefer, E. J. Lipoproteins, nutrition, and heart disease. Am. J. Clin. Nutr. 75, 191–212 (2002).
Fitzpatrick, A. L., Daling, J. R., Furberg, C. D., Kronmal, R. A. & Weissfeld, J. L. Hypertension, heart rate, use of antihypertensives, and incident prostate cancer. Ann. Epidemiol. 11, 534–542 (2001).Epidemiolgical study reporting a protective effect of anti-hypertensive medication against prostate cancer and providing strong support for the hypothesis that, in addition to adenocarcinomas in the lungs and pancreas, adenocarcinomas of the prostate might benefit from treatment and prevention by β-blockers or inhibitors of AA-metabolizing enzymes.
Cakir, Y., Plummer, H. K. & Schuller, H. M. β-Adrenergic-arachidonic acid–GIRK channel associated regulation of human breast cancer cell lines. Proc. 7th Int. Eicosanoid Conf. 355 (2001).
Whelan, J. Antagonistic effects of dietary arachidonic acid and n-3 polyunsaturated fatty acids. J. Nutr. 126, S1086–S1091 (1996).
Whelan, J., Broughton, K. S., Surette, M. E. & Kinsella, J. E. Dietary arachidonic and linoleic acids: comparative effects on tissue lipids. Lipids 27, 85–88 (1992).
Whelan, J., Broughton, K. S. & Kinsella, J. E. The comparative effects of dietary α-linoleic acid and fish oil on 4- and 5-series leukotriene formation in vivo. Lipids 26, 119–126 (1991).
Petrik, M. B., McEntee, M. F., Chiu, C. H. & Whelan, J. Antagonism of arachidonic acid is linked to the antitumorigenic effect of dietary eicosapentaenoic acid in ApcMin/+ mice. J. Nutr. 130, 1153–1158 (2000).
Harris, R. E., Namboodiri, K., Stellman, S. D. & Wynder, E. L. Breast cancer and NSAID use: heterogeneity of effect in a case–control study. Prev. Med. 24, 119–120 (1995).
Rose, D. P. & Connolly, J. M. Dietary fat and breast cancer metastasis by human tumor xenografts. Breast Cancer Res. Treat. 46, 225–237 (1997).
Connolly, J. M., Liu, X. H. & Rose, D. P. Effects of dietary menhaden oil, soy, and a cyclooxygenase inhibitor on human breast cancer cell growth and metastasis in nude mice. Nutr. Cancer 29, 48–54 (1997).
Connolly, J. M., Coleman, M. & Rose, D. P. Effects of dietary fatty acids on DU145 human prostate cancer cell growth in athymic nude mice. Nutr. Cancer 29, 114–119 (1997).
Rose, D. P., Connolly, J. M. & Liu, X. H. Fatty acid regulation of breast cancer cell growth and invasion. Adv. Exp. Med. Biol. 422, 47–55 (1997).
Wu, A. H. et al. Previous lung disease and risk of lung cancer among lifetime nonsmoking women in the United States. Am. J. Epidemiol. 141, 1023–1032 (1995).
Mayne, S. T., Buenconsejo, J. & Janerich, D. T. Previous lung disease and risk of lung cancer among men and women nonsmokers. Am. J. Epidemiol. 149, 13–20 (1999).
Brenner, A. V. et al. Previous pulmonary diseases and risk of lung cancer in Gansu Province, China. Int. J. Epidemiol. 30, 118–124 (2001).
Author information
Authors and Affiliations
Related links
Related links
DATABASES
Cancer.gov
LocusLink
Medscape DrugInfo
OMIM
FURTHER INFORMATION
Center for Disease Control's tobacco information and prevention service
Glossary
- PANCREATIC DUCTAL/DUCTULAR EPITHELIUM
-
The pancreas contains a network of ducts that branch into smaller ductules, the inner lining of which are called ductal and ductular epithelium, respectively.
- DNA ADDUCT
-
DNA bound to a chemical compound.
- NITROSATION
-
Nitrosation is a chemical reaction that adds two nitrogen atoms and one oxygen atom — which comprise the 'nitroso group' — to another chemical.
- INTRATRACHEAL INSTILLATION
-
'Trachea' is the Latin word for windpipe. When agents such as chemicals or drugs are given by a tube inserted into the mouth through the larynx into the windpipe, this is called an intratracheal instillation. This form of drug administration is used to make sure that the drug goes directly into the lungs.
- N3 POLYUNSATURATED FATTY ACIDS
-
Fatty acids consist of a chain of carbon and hydrogen atoms with the acidic carboxyl group 'COOH' at the end. The term 'polyunsaturated' means that the carbon chain includes double bonds. The term 'N3' means that the first double bond starts at the third carbon atom on the side of the molecule that is opposite to the carboxyl group.
- CATECHOLAMINES
-
Adrenaline and noradrenaline (also known S-adrenaline) are produced by cells in the adrenal gland. They are called catecholamines because their chemical structure is characterized by the presence of a catechol ring.
Rights and permissions
About this article
Cite this article
Schuller, H. Mechanisms of smoking-related lung and pancreatic adenocarcinoma development. Nat Rev Cancer 2, 455–463 (2002). https://doi.org/10.1038/nrc824
Issue Date:
DOI: https://doi.org/10.1038/nrc824
This article is cited by
-
Association of genetic risk and lifestyle with pancreatic cancer and their age dependency: a large prospective cohort study in the UK Biobank
BMC Medicine (2023)
-
The role of ADRB2 gene polymorphisms in malignancies
Molecular Biology Reports (2021)
-
Excessive miR-25-3p maturation via N6-methyladenosine stimulated by cigarette smoke promotes pancreatic cancer progression
Nature Communications (2019)
-
Conversion of nornicotine to 6-hydroxy-nornicotine and 6-hydroxy-myosmine by Shinella sp. strain HZN7
Applied Microbiology and Biotechnology (2016)
-
Housing temperature-induced stress drives therapeutic resistance in murine tumour models through β2-adrenergic receptor activation
Nature Communications (2015)