Key Points
-
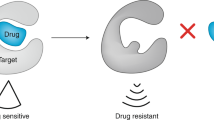
Multidrug resistance of cancer cells is a potentially surmountable obstacle to effective chemotherapy of cancer.
-
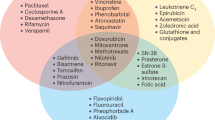
ATP-binding cassette (ABC) transporters, including MDR1 (ABCB1), MRP1 (ABCC1) and ABCG2, can confer multidrug resistance to cancer cells in vitro.
-
MRP2 (ABCC2), MRP3 (ABCC3), MRP4 (ABCC4), MRP5 (ABCC5), ABCA2 and BSEP (ABCB11) are capable of transporting drugs; future studies are needed to determine a role in drug resistance.
-
ABC transporters such as MDR1 and MRP1 are expressed in many human cancers, including leukaemias and some solid tumours; in some studies, expression of these transporters has been shown to correlate with response to therapy and survival.
-
Inhibitors of ABC transporters such as MDR1/P-glycoprotein have been tested in clinical trials with a suggestion of benefit, especially in acute myelogenous leukaemia.
-
Interpretation of clinical trials using inhibitors of MDR1/P-glycoprotein has been confounded by their effects on the pharmacokinetics of anticancer drugs.
-
Development of inhibitors of ABC transporters should focus on potency and specificity to minimize unexpected pharmacokinetic effects.
-
Efficacy should be confirmed using surrogate assays.
-
Normal tissues might be protected from toxicity by gene transfer of drug-resistance genes.
-
Prevention of ABC transporter induction in cancer cells might help to avert drug resistance.
Abstract
Chemotherapeutics are the most effective treatment for metastatic tumours. However, the ability of cancer cells to become simultaneously resistant to different drugs — a trait known as multidrug resistance — remains a significant impediment to successful chemotherapy. Three decades of multidrug-resistance research have identified a myriad of ways in which cancer cells can elude chemotherapy, and it has become apparent that resistance exists against every effective drug, even our newest agents. Therefore, the ability to predict and circumvent drug resistance is likely to improve chemotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gorre, M. E. et al. Clinical resistance to STI-571 cancer therapy caused by BCR–ABL gene mutation or amplification. Science 293, 876–880 (2001).
Pluen, A. et al. Role of tumor–host interactions in interstitial diffusion of macromolecules: cranial vs. subcutaneous tumors. Proc. Natl Acad. Sci. USA 98, 4628–4633 (2001).
Jain, R. K. Delivery of molecular and cellular medicine to solid tumors. Adv. Drug Deliv. Rev. 46, 149–168 (2001).
Jain, R. K. Normalizing tumor vasculature with anti-angiogenic therapy: a new paradigm for combination therapy. Nature Med. 7, 987–989 (2001).
Green, S. K., Frankel, A. & Kerbel, R. S. Adhesion-dependent multicellular drug resistance. Anticancer Drug Des. 14, 153–168 (1999).
Durand, R. E. & Olive, P. L. Resistance of tumor cells to chemo- and radiotherapy modulated by the three-dimensional architecture of solid tumors and spheroids. Methods Cell Biol. 64, 211–233 (2001).
Dean, M., Rzhetsky, A. & Alliknets, R. The human ATP-binding cassette (ABC) transporter superfamily. Genome Res. 11, 1156–1166 (2001).Most recent update of the ABC transporter family, emphasizing genetic disorders that result from disruption of ABC genes.
Ambudkar, S. V. et al. Biochemical, cellular, and pharmacological aspects of the multidrug transporter. Annu. Rev. Pharmacol. Toxicol. 39, 361–398 (1999).
Shen, D. W., Goldenberg, S., Pastan, I. & Gottesman, M. M. Decreased accumulation of [14C]carboplatin in human cisplatin-resistant cells results from reduced energy-dependent uptake. J. Cell Physiol. 183, 108–116 (2000).
Shen, D., Pastan, I. & Gottesman, M. M. Cross-resistance to methotrexate and metals in human cisplatin-resistant cell lines results from a pleiotropic defect in accumulation of these compounds associated with reduced plasma membrane binding proteins. Cancer Res. 58, 268–275 (1998).
Schuetz, E. G., Beck, W. T. & Schuetz, J. D. Modulators and substrates of P-glycoprotein and cytochrome P4503A coordinately up-regulate these proteins in human colon carcinoma cells. Mol. Pharmacol. 49, 311–318 (1996).
Synold, T. W., Dussault, I. & Forman, B. M. The orphan nuclear receptor SXR coordinately regulates drug metabolism and efflux. Nature Med. 7, 584–590 (2001).This paper provides direct evidence for coordination of drug-detoxifying systems through activation of a single transcription factor, SXR.
Liu, Y. Y., Han, T. Y., Giuliano, A. E. & Cabot, M. C. Ceramide glycosylation potentiates cellular multidrug resistance. FASEB J. 15, 719–730 (2001).
Juliano, R.L. & Ling, V. A surface glycoprotein modulating drug permeability in Chinese hamster ovary cell mutants. Biochim. Biophys. Acta 455, 152–162 (1976).
Ueda, K., Cardarelli, C., Gottesman, M. M. & Pastan, I. Expression of a full-length cDNA for the human MDR1 gene confers resistance to colchicine, doxorubicin, and vinblastine. Proc. Natl Acad. Sci. USA 84, 3004–3008 (1987).
Chen, C.J. et al. Internal duplication and homology with bacterial transport proteins in Mdr-1 (P-glycoprotein) gene from multidrug-resistant human cells. Cell 47, 371–380 (1986).Description of the sequence of the MDR1 (P-glycoprotein) cDNA and its homology to two bacterial transporters, thereby defining the first members of the ABC transporter family.
Senior, A. E. & Bhagat, S. P-glycoprotein shows strong catalytic cooperativity between the two nucleotide sites. Biochemistry 37, 831–836 (1998).
Ramachandra, M. et al. Human P-glycoprotein exhibits reduced affinity for substrates during a catalytic transition state. Biochemistry 37, 5010–5019 (1998).
Sauna, Z. E. & Ambudkar, S. V. Evidence for a requirement for ATP hydrolysis at two distinct steps during a single turnover of the catalytic cycle of human P-glycoprotein. Proc. Natl Acad. Sci. USA 97, 2515–2520 (2000).
Cole, S.P.C. et al. Overexpression of a transporter gene in a multidrug-resistant human lung cancer cell line. Science 258, 1650–1654 (1992).
Loe, D. W., Deeley, R. G. & Cole, S. P. Characterization of vincristine transport by the M(r) 190,000 multidrug resistance protein (MRP): evidence for cotransport with reduced glutathione. Cancer Res. 58, 5130–5136 (1998).
Jedlitschky, G. et al. Transport of glutathione, glucuronate, and sulfate conjugates by the MRP gene-encoded conjugate export pump. Cancer Res. 56, 988–994 (1996).Observations that led to the conclusion that MRP was a broad-specificity organic anion transporter.
Muller, M. et al. Overexpression of the gene encoding the multidrug resistance-associated protein results in increased ATP-dependent glutathione S-conjugate transport. Proc. Natl Acad. Sci. USA 91, 13033–13037 (1994).
Borst, P., Evers, R., Kool, M. & Wijnholds, J. A family of drug transporters: the multidrug resistance-associated proteins. J. Natl Cancer Inst. 92, 1295–1302 (2000).
Miyake, K. et al. Molecular cloning of cDNAs which are highly overexpressed in mitoxantrone-resistant cells: demonstration of homology to ABC transport genes. Cancer Res. 59, 8–13 (1999).
Doyle, L. A. et al. A multidrug resistance transporter from human MCF-7 breast cancer cells. Proc. Natl Acad. Sci. USA 95, 15665–15670 (1998).
Allikmets, R., Schriml, L. M., Hutchinson, A., Romano-Spica, V. & Dean, M. A human placenta-specific ATP-binding cassette gene (ABCP) on chromosome 4q22 that is involved in multidrug resistance. Cancer Res. 58, 5337–5339 (1998).
Honjo, Y. et al. Acquired mutations in the MXR/BCRP/ABCP gene alter substrate specificity in MXR/BCRP/ABCP-overexpressing cells. Cancer Res. (in the press).
Komatani, H. et al. Identification of breast cancer resistant protein/mitoxantrone resistance/placenta-specific, ATP-binding cassette transporter as a transporter of NB-506 and J-107088, topoisomerase I inhibitors with an indolocarbazole structure. Cancer Res. 61, 2827–2832 (2001).
Childs, S., Yeh, R. L., Hui, D. & Ling, V. Taxol resistance mediated by transfection of the liver-specific sister gene of P-glycoprotein. Cancer Res. 58, 4160–4167 (1998).
Smit, J. J. et al. Homozygous disruption of the murine Mdr2 P-glycoprotein gene leads to a complete absence of phospholipid from bile and to liver disease. Cell 75, 451–462 (1993).
Zhou, Y., Gottesman, M. M. & Pastan, I. Studies of human MDR1–MDR2 chimeras demonstrate the functional exchangeability of a major transmembrane segment of the multidrug transporter and phosphatidylcholine flippase. Mol. Cell Biol. 19, 1450–1459 (1999).
Borst, P., Zelcer, N. & van Helvoort, A. ABC transporters in lipid transport. Biochim. Biophys. Acta 1486, 128–144 (2000).
Laing, N. M. et al. Amplification of the ATP-binding cassette 2 transporter gene is functionally linked with enhanced efflux of estramustine in ovarian carcinoma cells. Cancer Res. 58, 1332–1337 (1998).
Vulevic, B. et al. Cloning and characterization of human adenosine 5′-triphosphate-binding cassette, sub-family A, transporter 2 (ABCA2). Cancer Res. 61, 3339–3347 (2001).
Scheffer, G. L., Schroeijers, A. B., Izquierdo, M. A., Wiemer, E. A. & Scheper, R. J. Lung resistance-related protein/major vault protein and vaults in multidrug-resistant cancer. Curr. Opin. Oncol. 12, 550–556 (2000).
Kool, M. et al. MRP3, an organic anion transporter able to transport anti-cancer drugs. Proc. Natl Acad. Sci. USA 96, 6914–6919 (1999).
Belinsky, M. G. & Kruh, G. D. MOAT-E (ARA) is a full-length MRP/cMOAT subfamily transporter expressed in kidney and liver. Br. J. Cancer 80, 1342–1349 (1999).
Chen, Z. S., Lee, K. & Kruh, G. D. Transport of cyclic nucleotides and estradiol 17-β-D-glucuronide by multidrug resistance protein 4: resistance to 6-mercaptopurine and 6–thioguanine. J. Biol. Chem. 276, 33747–33754 (2001).
Schuetz, J. D. et al. MRP4: a previously unidentified factor in resistance to nucleoside-based antiviral drugs. Nature Med. 5, 1048–1051 (1999).
Wijnholds, J. et al. Multidrug-resistance protein 5 is a multispecific organic anion transporter able to transport nucleotide analogs. Proc. Natl Acad. Sci. USA 97, 7476–7481 (2000).
Jedlitschky, G., Burchell, B. & Keppler, D. The multidrug resistance protein 5 functions as an ATP-dependent export pump for cyclic nucleotides. J. Biol. Chem. 275, 30069–30074 (2000).
Struk, B. et al. Mutations of the gene encoding the transmembrane transporter protein ABC-C6 cause pseudoxanthoma elasticum. J. Mol. Med. 78, 282–286 (2000).
Bergen, A. A. et al. Mutations in ABCC6 cause pseudoxanthoma elasticum. Nature Genet. 25, 228–231 (2000).
Ringpfeil, F., Lebwohl, M. G., Christiano, A. M. & Uitto, J. Pseudoxanthoma elasticum: mutations in the MRP6 gene encoding a transmembrane ATP-binding cassette (ABC) transporter. Proc. Natl Acad. Sci. USA 97, 6001–6006 (2000).
Le Saux, O. et al. Mutations in a gene encoding an ABC transporter cause pseudoxanthoma elasticum. Nature Genet. 25, 223–227 (2000).
Gerloff, T. et al. The sister of P-glycoprotein represents the canalicular bile salt export pump of mammalian liver. J. Biol. Chem. 273, 10046–10050 (1998).
Lecureur, V. et al. Cloning and expression of murine sister of P-glycoprotein reveals a more discriminating transporter than MDR1/P-glycoprotein. Mol. Pharmacol. 57, 24–35 (2000).
Schinkel, A. H., Wagenaar, E., Mol, C. A. & van Deemter, L. P-glycoprotein in the blood–brain barrier of mice influences the brain penetration and pharmacological activity of many drugs. J. Clin. Invest. 97, 2517–2524 (1996).Follow-up study to the landmark observation that disruption of the PGPs failed to evoke a disease phenotype, but did render mice extraordinarily sensitive to ivermectin. This implicated a role for PGP in the blood–brain barrier.
Xie, R., Hammarlund-Udenaes, M., de Boer, A. G. & de Lange, E. C. The role of P-glycoprotein in blood–brain barrier transport of morphine: transcortical microdialysis studies in Mdr1a (−/−) and Mdr1a (+/+) mice. Br. J. Pharmacol. 128, 563–568 (1999).
Rao, V. V. et al. Choroid plexus epithelial expression of MDR1 P glycoprotein and multidrug resistance-associated protein contribute to the blood-cerebrospinal-fluid drug-permeability barrier. Proc. Natl Acad. Sci. USA 96, 3900–3905 (1999).Description of MRP and PGP localization in the epithelium of choroid plexus, and prediction of distinctive roles in the blood–cerebrospinal-fluid barrier.
Cordon-Cardo, C. et al. Expression of the multidrug resistance gene product (P-glycoprotein) in human normal and tumor tissues. J. Histochem. Cytochem. 38, 1277–1287 (1990).
Jonker, J. W. et al. Role of breast cancer resistance protein in the bioavailability and fetal penetration of topotecan. J. Natl Cancer Inst. 92, 1651–1656 (2000).
Maliepaard, M. et al. Subcellular localization and distribution of the breast cancer resistance protein transporter in normal human tissues. Cancer Res. 61, 3458–3464 (2001).
St-Pierre, M. V. et al. Expression of members of the multidrug resistance protein family in human term placenta. Am. J. Physiol. 279, R1495–R1503 (2000). | PubMed |
Schinkel, A. H. et al. Normal viability and altered pharmacokinetics in mice lacking Mdr1-type (drug-transporting) P-glycoproteins. Proc. Natl Acad. Sci. USA 94, 4028–4033 (1997).Clear demonstration that PGP affects the tissue distribution and retention of drugs other than anticancer agents.
Scheffer, G. L. et al. Specific detection of multidrug resistance proteins MRP1, MRP2, MRP3, MRP5, and MDR3 P-glycoprotein with a panel of monoclonal antibodies. Cancer Res. 60, 5269–5277 (2000).
Kool, M., van der Linden, M., de Haas, M., Baas, F. & Borst, P. Expression of human MRP6, a homologue of the multidrug resistance protein gene MRP1, in tissues and cancer cells. Cancer Res. 59, 175–182 (1999).
Konig, J., Nies, A. T., Cui, Y., Leier, I. & Keppler, D. Conjugate export pumps of the multidrug resistance protein (MRP) family: localization, substrate specificity, and MRP2-mediated drug resistance. Biochim. Biophys. Acta 1461, 377–394 (1999).
Paulusma, C. C. et al. Congenital jaundice in rats with a mutation in a multidrug resistance-associated protein gene. Science 271, 1126–1128 (1996).
Ito, K. et al. Molecular cloning of canalicular multispecific organic anion transporter defective in EHBR. Am. J. Physiol. 272, G16–G22 (1997).
Kartenbeck, J., Leuschner, U., Mayer, R. & Keppler, D. Absence of the canalicular isoform of the MRP gene-encoded conjugate export pump from the hepatocytes in Dubin–Johnson syndrome. Hepatology 23, 1061–1066 (1996).
Paulusma, C. C. et al. A mutation in the human canalicular multispecific organic anion transporter gene causes the Dubin–Johnson syndrome. Hepatology 25, 1539–1542 (1997).
Strautnieks, S. S. et al. A gene encoding a liver-specific ABC transporter is mutated in progressive familial intrahepatic cholestasis. Nature Genet. 20, 233–238 (1998).
Wang, R. et al. Targeted inactivation of sister of P-glycoprotein gene (Spgp) in mice results in nonprogressive but persistent intrahepatic cholestasis. Proc. Natl Acad. Sci. USA 98, 2011–2016 (2001).
Ruetz, S. & Gros, P. Phosphatidylcholine translocase: a physiological role for the Mdr2 gene. Cell 77, 1071–1081 (1994).First physiological role for an MDR gene is convincingly described.
de Vree, J. M. et al. Mutations in the MDR3 gene cause progressive familial intrahepatic cholestasis. Proc. Natl Acad. Sci. USA 95, 282–287 (1998).
Mayer, U. et al. Full blockage of intestinal P-glycoprotein and extensive inhibition of blood–brain barrier P-glycoprotein by oral treatment of mice with PSC-833. J. Clin. Invest. 100, 2430–2436 (1997).
Greiner, B. et al. The role of intestinal P-glycoprotein in the interaction of digoxin and rifampin. J. Clin. Invest. 104, 147–153 (1999).
Lown, K. S. et al. Role of intestinal P-glycoprotein (Mdr1) in interpatient variation in the oral bioavailability of cyclosporine. Clin. Pharmacol. Ther. 62, 248–260 (1997).
Sparreboom, A. et al. Limited oral bioavailability and active epithelial excretion of paclitaxel (Taxol) caused by P-glycoprotein in the intestine. Proc. Natl Acad. Sci. USA 94, 2031–2035 (1997).
Evers, R. et al. Basolateral localization and export activity of the human multidrug resistance-associated protein in polarized pig kidney cells. J. Clin. Invest. 97, 1211–1218 (1996).
Wijnholds, J. et al. Increased sensitivity to anticancer drugs and decreased inflammatory response in mice lacking the multidrug resistance-associated protein. Nature Med. 3, 1275–1279 (1997).
Dietrich, C. G., de Waart, D. R., Ottenhoff, R., Schoots, I. G. & Elferink, R. P. Increased bioavailability of the food-derived carcinogen 2-amino-1-methyl-6-phenylimidazo[4,5-b] pyridine in Mrp2-deficient rats. Mol. Pharmacol. 59, 974–980 (2001).
Fojo, A.T. et al. Expression of a multidrug resistance gene in human tumors and tissues. Proc. Natl Acad. Sci. USA 84, 265–269 (1987).
Goldstein, L. J. et al. Expression of a multidrug resistance gene in human cancers. J. Natl Cancer Inst. 81, 116–124 (1989).
van den Heuvel-Eibrink, M. M., Sonneveld, P. & Pieters, R. The prognostic significance of membrane transport-associated multidrug resistance (MDR) proteins in leukemia. Int. J. Clin. Pharmacol. Ther. 38, 94–110 (2000).
Beck, W.T. et al. Methods to detect P-glycoprotein-associated multidrug resistance in patients' tumors: consensus recommendations. Cancer Res. 56, 3010–3020 (1996).
Hipfner, D. R. et al. Epitope mapping of monoclonal antibodies specific for the 190-kDa multidrug resistance protein (MRP). Br. J. Cancer 78, 1134–1140 (1998).
Nooter, K. et al. Expression of the multidrug resistance-associated protein (MRP) gene in human cancers. Clin. Cancer Res. 1, 1301–1310 (1995).
Leith, C. P. et al. Frequency and clinical significance of the expression of the multidrug resistance proteins MDR1/P-glycoprotein, MRP1, and LRP in acute myeloid leukemia: a Southwest Oncology Group Study. Blood 94, 1086–1099 (1999).Assay of leukaemic cells obtained from 351 patients with acute myelogenous leukaemia, and confirmation that PGP expression and function correlate with a decreased complete remission rate and an increased rate of resistant disease.
Han, K. et al. Expression of functional markers in acute nonlymphoblastic leukemia. Acta Haematol. 104, 174–180 (2000).
Dorr, R. et al. Phase I/II study of the P-glycoprotein modulator PSC 833 in patients with acute myeloid leukemia. J. Clin. Oncol. 19, 1589–1599 (2001).
van der Kolk, D. M. et al. P-glycoprotein and multidrug resistance protein activities in relation to treatment outcome in acute myeloid leukemia. Clin. Cancer Res. 6, 3205–3214 (2000).
Legrand, O., Simonin, G., Beauchamp-Nicoud, A., Zittoun, R. & Marie, J. P. Simultaneous activity of MRP1 and PGP is correlated with in vitro resistance to daunorubicin and with in vivo resistance in adult acute myeloid leukemia. Blood 94, 1046–1056 (1999).
Michieli, M. et al. P-glycoprotein, lung resistance-related protein and multidrug resistance associated protein in de novo acute non-lymphocytic leukaemias: biological and clinical implications. Br. J. Haematol. 104, 328–335 (1999).
Broxterman, H. J. et al. Do P-glycoprotein and major vault protein (MVP/LRP) expression correlate with in vitro daunorubicin resistance in acute myeloid leukemia? Leukemia 13, 258–265 (1999).
Tidefelt, U. et al. P-glycoprotein inhibitor valspodar (PSC-833) increases the intracellular concentrations of daunorubicin in vivo in patients with P-glycoprotein-positive acute myeloid leukemia. J. Clin. Oncol. 18, 1837–1844 (2000).
Burger, H. et al. Expression of the multidrug resistance-associated protein (MRP) in acute and chronic leukemias. Leukemia 8, 990–997 (1994).
Filipits, M. et al. Multidrug resistance-associated protein in acute myeloid leukemia: no impact on treatment outcome. Clin. Cancer Res. 3, 1419–1425 (1997).
Ross, D. D., Karp, J. E., Chen, T. T. & Doyle, L. A. Expression of breast cancer resistance protein in blast cells from patients with acute leukemia. Blood 96, 365–368 (2000).
Trock, B. J., Leonessa, F. & Clarke, R. Multidrug resistance in breast cancer: a meta-analysis of MDR1/GP170 expression and its possible functional significance. J. Natl Cancer Inst. 89, 917–931 (1997).Meta-analysis showing PGP expression in approximately 40% of breast cancer samples, and a correlation with decreased treatment response.
Vecchio, S. D. et al. In vivo detection of multidrug-resistant (MDR1) phenotype by 99m sestamibi scan in untreated breast cancer patients. Eur. J. Nucl. Med. 24, 150–159 (1997).Study showing an inverse correlation between MRK-16 binding to breast cancer samples and 99mTc-sestamibi retention in breast cancers.
Sun, S. S. et al. Expression of mediated P-glycoprotein multidrug resistance related to Tc-99m MIBI scintimammography results. Cancer Lett. 153, 95–100 (2000).
Kao, C. H. et al. P-glycoprotein and multidrug resistance-related protein expressions in relation to technetium-99m methoxyisobutylisonitrile scintimammography findings. Cancer Res. 61, 1412–1414 (2001).
Dexter, D. W. et al. Quantitative reverse transcriptase-polymerase chain reaction measured expression of MDR1 and MRP in primary breast carcinoma. Clin. Cancer Res. 4, 1533–1542 (1998).
Filipits, M. et al. MRP and MDR1 gene expression in primary breast carcinomas. Clin. Cancer Res. 2, 1231–1237 (1996).
Nooter, K. et al. The prognostic significance of expression of the multidrug resistance-associated protein (MRP) in primary breast cancer. Br. J. Cancer 76, 486–493 (1997).
Oka, M. et al. The clinical role of MDR1 gene expression in human lung cancer. Anticancer Res. 17, 721–724 (1997).
Savaraj, N. et al. Multidrug-resistant gene expression in small-cell lung cancer. Am. J. Clin. Oncol. 20, 398–403 (1997).
Young, L. C. et al. Expression of multidrug resistance protein-related genes in lung cancer: correlation with drug response. Clin. Cancer Res. 5, 673–680 (1999).
Nooter, K. et al. Expression of the multidrug resistance-associated protein (MRP) gene in primary non-small-cell lung cancer. Ann. Oncol. 7, 75–81 (1996).
Wright, S. R. et al. Immunohistochemical detection of multidrug resistance protein in human lung cancer and normal lung. Clin. Cancer Res. 4, 2279–2289 (1998).
Chan, H. S. L., Thorner, P. S., Haddad, G. & Ling, V. Immunohistochemical detection of P-glycoprotein: prognostic correlation in soft tissue sarcoma of childhood. J. Clin. Oncol. 8, 689–704 (1990).
Chan, H. S., Grogan, T. M., Haddad, G., DeBoer, G. & Ling, V. P-glycoprotein expression: critical determinant in the response to osteosarcoma chemotherapy. J. Natl Cancer Inst. 89, 1706–1715 (1997).
Perri, T. et al. Effect of P-glycoprotein expression on outcome in the Ewing family of tumors. Pediatr. Hematol. Oncol. 18, 325–334 (2001).
Baldini, N. et al. Expression of P-glycoprotein in high-grade osteosarcomas in relation to clinical outcome. N. Engl. J. Med. 333, 1380–1385 (1995).
Coley, H. M. et al. Incidence of P-glycoprotein overexpression and multidrug resistance (MDR) reversal in adult soft tissue sarcoma. Eur. J. Cancer 36, 881–888 (2000).
Wunder, J. S. et al. MDR1 gene expression and outcome in osteosarcoma: a prospective, multicenter study. J. Clin. Oncol. 18, 2685–2694 (2000).
Kuttesch, J. F. et al. P-glycoprotein expression at diagnosis may not be a primary mechanism of therapeutic failure in childhood rhabdomyosarcoma. J. Clin. Oncol. 14, 886–900 (1996).
Ferry, D. R., Traunecker, H. & Kerr, D. J. Clinical trials of P-glycoprotein reversal in solid tumours. Eur. J. Cancer 32A, 1070–1081 (1996).
Kerr, D. J. et al. The effect of verapamil on the pharmacokinetics of adriamycin. Cancer Chemother. Pharmacol. 18, 239–242 (1986).
Fisher, G. A. & Sikic, B. I. Clinical studies with modulators of multidrug resistance. Hematol Oncol Clin North Am 9, 363–382 (1995).
Bradshaw, D. M. & Arceci, R. J. Clinical relevance of transmembrane drug efflux as a mechanism of multidrug resistance. J. Clin. Oncol. 16, 3674–3690 (1998).
Fisher, G. A., Lum, B. L., Hausdorff, J. & Sikic, B. I. Pharmacological considerations in the modulation of multidrug resistance. Eur. J. Cancer 32A, 1082–1088 (1996).
Chaudhary, P. M. & Roninson, I. B. Expression and activity of P-glycoprotein, a multidrug efflux pump, in human hematopoietic stem cells. Cell 66, 85–94 (1991).Early paper that highlights the role of P-glycoprotein in normal cells.
Chico, I. et al. Phase I study of infusional paclitaxel in combination with the P-glycoprotein antagonist PSC- 833. J. Clin. Oncol. 19, 832–842 (2001).
Advani, R. et al. Treatment of refractory and relapsed acute myelogenous leukemia with combination chemotherapy plus the multidrug resistance modulator PSC-833 (Valspodar). Blood 93, 787–795 (1999).
Kornblau, S. M. et al. Phase I study of mitoxantrone plus etoposide with multidrug blockade by SDZ PSC-833 in relapsed or refractory acute myelogenous leukemia. J. Clin. Oncol. 15, 1796–1802 (1997).
List, A. F. et al. Phase I/II trial of cyclosporine as a chemotherapy-resistance modifier in acute leukemia. J. Clin. Oncol. 11, 1652–1660 (1993).
Lee, E. J. et al. Parallel phase I studies of daunorubicin given with cytarabine and etoposide with or without the multidrug resistance modulator PSC-833 in previously untreated patients 60 years of age or older with acute myeloid leukemia: results of cancer and leukemia group B study 9420. J. Clin. Oncol. 17, 2831–2839 (1999).
Solary, E. et al. Combination of quinine as a potential reversing agent with mitoxantrone and cytarabine for the treatment of acute leukemias: a randomized multicenter study. Blood 88, 1198–1205 (1996).
List, A. F. et al. Benefit of cyclosporine (CsA) modulation of drug resistance in patients with poor-risk acute myeloid leukemia: a Southwest Oncology Group (SWOG) Study. Blood 98, 3212–3220 (2001).These long-term results indicate a survival advantage of treatment of acute myelogenous leukaemia with daunomycin in combination with the PGP antagonist cyclosporin A.
Sonneveld, P. et al. Modulation of multidrug-resistant multiple myeloma by cyclosporin. Lancet 340, 255–259 (1992).
Millward, M. J. et al. Oral verapamil with chemotherapy for advanced non-small cell lung cancer: a randomised study. Br. J. Cancer 67, 1031–1035 (1993).
Fracasso, P. M. et al. Phase I study of paclitaxel in combination with a multidrug resistance modulator, PSC-833 (Valspodar), in refractory malignancies. J. Clin. Oncol. 18, 1124 (2000).
Fracasso, P. M. et al. Phase II study of paclitaxel and valspodar (PSC-833) in refractory ovarian carcinoma: a gynecologic oncology group study. J. Clin. Oncol. 19, 2975–2982 (2001).
Bohme, M., Buchler, M., Muller, M. & Keppler, D. Differential inhibition by cyclosporins of primary-active ATP-dependent transporters in the hepatocyte canalicular membrane. FEBS Lett. 333, 193–196 (1993).
Lum, B. L. et al. Alteration of etoposide pharmacokinetics and pharmacodynamics by cyclosporine in a phase I trial to modulate multidrug resistance. J. Clin. Oncol. 10, 1635–1642 (1992).
Bartlett, N. L. et al. Phase I trial of doxorubicin with cyclosporine as a modulator of multidrug resistance. J. Clin. Oncol. 12, 835–842 (1994).
Rowinsky, E. K. et al. Phase I and pharmacokinetic study of paclitaxel in combination with biricodar, a novel agent that reverses multidrug resistance conferred by overexpression of both MDR1 and MRP. J. Clin. Oncol. 16, 2964–2976 (1998).
Wilson, W. H. et al. Phase I and pharmacokinetic study of the multidrug resistance modulator dexverapamil with EPOCH chemotherapy. J. Clin. Oncol. 13, 1985–1994 (1995).
Sparreboom, A. et al. Clinical pharmacokinetics of doxorubicin in combination with GF120918, a potent inhibitor of MDR1 P-glycoprotein. Anticancer Drugs 10, 719–728 (1999).
Advani, R. et al. A phase I trial of doxorubicin, paclitaxel, and valspodar (PSC- 833), a modulator of multidrug resistance. Clin. Cancer Res. 7, 1221–1229 (2001).
Kang, M. H. et al. The P-glycoprotein antagonist PSC- 833 increases the plasma concentrations of 6α-hydroxypaclitaxel, a major metabolite of paclitaxel. Clin. Cancer Res. 7, 1610–1617 (2001).
Solary, E. et al. Sufficient levels of quinine in the serum circumvent the multidrug resistance of the human leukemic cell line K562/ADM. Cancer 68, 1714–1719 (1991).
Minami, H. et al. Phase I study of intravenous PSC-833 and doxorubicin: reversal of multidrug resistance. Jpn. J. Cancer Res. 92, 220–230 (2001).
Robey, R. et al. Efflux of rhodamine from CD56+ cells as a surrogate marker for reversal of P-glycoprotein-mediated drug efflux by PSC-833. Blood 93, 306–314 (1999).
Witherspoon, S. M. et al. Flow cytometric assay of modulation of P-glycoprotein function in whole blood by the multidrug resistance inhibitor GG918. Clin. Cancer Res. 2, 7–12 (1996).
Luker, G.D., Facasso, P.M., Dobkin, J. & Piwnica-Worms, D. Modulation of the multidrug resistance P-glycoprotein: detection with technetium-99m-sestamibi in vivo. J. Nucl. Med. 38, 369–372 (1997).Promising study showing the ability of PSC-833 to increase liver retention of 99mTc-sestamibi as a surrogate for PGP inhibition.
Chen, C. C. et al. Detection of in vivo P-glycoprotein inhibition by PSC-833 using Tc-99m sestamibi. Clin. Cancer Res. 3, 545–552 (1997).
Peck, R. A. et al. Phase I and pharmacokinetic study of the novel MDR1 and MRP1 inhibitor biricodar administered alone and in combination with doxorubicin. J. Clin. Oncol. 19, 3130–3141 (2001).
Hendrikse, N. H. et al. 99mTc-sestamibi is a substrate for P-glycoprotein and the multidrug resistance-associated protein. Br. J. Cancer 77, 353–358 (1998).
Chen, W. S. et al. Effects of MDR1 and MDR3 P-glycoproteins, MRP1, and BCRP/MXR/ABCP on the transport of (99m)Tc-tetrofosmin. Biochem. Pharmacol. 60, 413–426 (2000).
Beketic-Oreskovic, L., Duran, G. E., Chen, G., Dumontet, C. & Sikic, B. I. Decreased mutation rate for cellular resistance to doxorubicin and suppression of Mdr1 gene activation by the cyclosporin PSC-833. J. Natl Cancer Inst. 87, 1593–1602 (1995).Cells exposed to doxorubicin in the presence of PSC-833 develop resistance less frequently, supporting an argument for early intervention with PGP inhibitors.
Abolhoda, A. et al. Rapid activation of MDR1 gene expression in human metastatic sarcoma after in vivo exposure to doxorubicin. Clin. Cancer Res. 5, 3352–3356 (1999).
Lee, J. S. et al. Rhodamine efflux patterns predict P-glycoprotein substrates in the National Cancer Institute Drug Screen. Mol. Pharmacol. 46, 627–638 (1994).
Sorrentino, B. P. et al. Selection of drug-resistant bone marrow cells in vivo after retroviral transfer of human MDR1. Science 257, 99–103 (1992).First use of MDR1 in a gene therapy setting, providing a means to increase dose intensity by protecting bone-marrow cells from chemotherapy toxicity.
Flasshove, M. et al. Ex vivo expansion and selection of human CD34+ peripheral blood progenitor cells after introduction of a mutated dihydrofolate reductase cDNA via retroviral gene transfer. Blood 85, 566–574 (1995).
Moscow, J. A. et al. Engraftment of MDR1 and NeoR gene-transduced hematopoietic cells after breast cancer chemotherapy. Blood 94, 52–61 (1999).
Bunting, K. D., Galipeau, J., Topham, D., Benaim, E. & Sorrentino, B. P. Transduction of murine bone marrow cells with an MDR1 vector enables ex vivo stem cell expansion, but these expanded grafts cause a myeloproliferative syndrome in transplanted mice. Blood 92, 2269–2279 (1998).
Dano, K. Active outward transport of daunomycin in resistant Ehrlich ascites tumor cells. Biochim. Biophys. Acta 323, 466–483 (1973).
Abraham, J. et al. A phase I study of the novel P-glycoprotein (PGP) antagonist, XR9576 in combination with vinorelbine. Proc. Am. Soc. Clin. Oncol. 20, 287 (2001).
Dantzig, A. H. et al. Reversal of P-glycoprotein-mediated multidrug resistance by a potent cyclopropyldibenzosuberane modulator, LY335979. Cancer Res. 56, 4171–4179 (1996).
Starling, J. J. et al. Pharmacological characterization of LY335979: a potent cyclopropyldibenzosuberane modulator of P-glycoprotein. Adv. Enzyme Regul. 37, 335–347 (1997).
Hyafil, F., Vergely, C., Du Vignaud, P. & Grand-Perret, T. In vitro and in vivo reversal of multidrug resistance by GF120918, an acridonecarboxamide derivative. Cancer Res. 53, 4595–4602 (1993).
de Bruin, M., Miyake, K., Litman, T., Robey, R. & Bates, S. E. Reversal of resistance by GF120918 in cell lines expressing the ABC half-transporter, MXR. Cancer Lett. 146, 117–126 (1999).
Lowe, S. W., Ruley, H. E., Jacks, T. & Housman, D. E. p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 74, 957–967 (1993).
Schneider, J. et al. Expression of LRP and MDR1 in locally advanced breast cancer preditcs axillary node invasion at the time of rescue mastectomy after induction chemotherapy. Breast Cancer Res. 3, 183–191 (2001).
Tolcher, A. W. et al. Phase I crossover study of paclitaxel with r-verapamil in patients with metastatic breast cancer. J. Clin. Oncol. 14, 1173–1184 (1996).
Mechetner, E. et al. Levels of multidrug resistance (MDR1) P-glycoprotein expression by human breast cancer correclate with in vitro resistance to taxol and doxorubicin. Clin. Cancer Res. 4, 389–398 (1998).
Izquierdo, M. A. et al. Drug resistance-associated marker LRP for prediction of response to chemotherapy and prognoses in advanced ovarian carcinoma. J. Natl Cancer Inst. 87, 1230–1237 (1995).
Schneider, J., Jimenez, E., Marenbach, K., Marx, D. & Meden, H. Co-expression of the MDR1 gene and HSP27 in human ovarian cancer. Anticancer Res. 18, 2967–2971 (1998).
Baekelandt, M. M., Holm, R., Nesland, J. M., Troupe, C. G. & Kristensen, G. B. P-glycoprotein expression is a marker for chemotherapy resistance and prognosis in advanced ovarian cancer. Anticancer Res. 20, 1061–1067 (2000).
Fedeli, L. et al. Pharmacokinetics of vincristine in cancer patients treated with nifedipine. Cancer 64, 1805–1811 (1989).
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
DATABASES
progressive familial intrahepatic cholestasis type-3
FURTHER INFORMATION
Glossary
- MULTIDRUG RESISTANCE
-
Simultaneous resistance to several structurally unrelated drugs that do not have a common mechanism of action.
- VINCA ALKALOIDS
-
A family of natural-product anticancer drugs, extracted from the periwinkle family, that depolymerize microtubules. Examples include vincristine and vinblastine.
- ANTHRACYCLINES
-
Semi-synthetic anticancer derivatives of anthraquinone that intercalate into DNA and inhibit DNA topoisomerase II. Examples include daunorubicin and doxorubicin.
- CYTOCHROME P450
-
A group of enzymes that are located on the endoplasmic reticulum, and are involved in drug metabolism and detoxification. They are primarily expressed in the liver and small intestine.
- MULTIFACTORIAL MULTIDRUG RESISTANCE
-
Multidrug resistance caused by several different mechanisms of resistance that operate simultaneously.
- FLIPPASE
-
A transport system that moves substrates from one leaflet of the membrane bilayer to the other leaflet.
- BILIRUBIN
-
A breakdown product of haemoglobin that is processed by the liver, where it is conjugated to glucuronic acid and excreted in the bile. Accumulation of bilirubin in the blood and tissues can lead to jaundice and neurological damage.
- CANALICULAR MEMBRANES
-
Surface of the hepatocyte that faces the biliary canaliculus, through which bile is excreted.
- MYELOSUPPRESSION
-
Temporary inhibition of bone-marrow production, caused by chemotherapy-mediated cytotoxicity to blood-cell precursors.
- TAXANES
-
A family of natural-product and semi-synthetic agents, including paclitaxel, which was originally isolated from the bark of the yew tree. Their mechanism of action includes stabilization of microtubules and inhibition of mitosis.
- CEREBELLAR ATAXIA
-
Difficulty in walking caused by impaired cerebellar function.
- MYELODYSPLASTIC SYNDROME
-
A disorder of haematopoietic cells that often leads to acute leukaemia.
- CD56+ CELLS
-
A subset of circulating lymphocytes, known as natural killer cells, that express the CD56 antigen. They are used to test drug effectiveness in clinical studies because they express high levels of MDR1/P-glycoprotein.
Rights and permissions
About this article
Cite this article
Gottesman, M., Fojo, T. & Bates, S. Multidrug resistance in cancer: role of ATP–dependent transporters. Nat Rev Cancer 2, 48–58 (2002). https://doi.org/10.1038/nrc706
Issue Date:
DOI: https://doi.org/10.1038/nrc706
This article is cited by
-
Polymer lipid hybrid nanoparticles encapsulated with Emodin combined with DOX reverse multidrug resistance of breast cancer via IL-6/JAK2/STAT3 signaling pathway
Cancer Nanotechnology (2024)
-
ABCB1 overexpression through locus amplification represents an actionable target to combat paclitaxel resistance in pancreatic cancer cells
Journal of Experimental & Clinical Cancer Research (2024)
-
Curcumin Loaded onto Folic acid Carbon dots as a Potent drug Delivery System for Antibacterial and Anticancer Applications
Journal of Cluster Science (2024)
-
Study of pathogenic T-helper cell subsets in Asian Indian patients with Takayasu arteritis
Immunologic Research (2024)
-
Generation and characterization of a zebrafish knockout model of abcb4, a homolog of the human multidrug efflux transporter P-glycoprotein
Human Genomics (2023)