Key Points
-
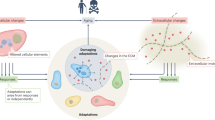
Although the term 'cellular senescence' was originally used to define the state of irreversible proliferative arrest by cultured cells that had reached their replicative limit, it is now widely used to describe states of cell cycle arrest in different in vivo biological settings, including age-associated loss of regenerative capacity, tumour suppression, inflammation, wound healing and embryogenesis.
-
A central problem in the senescence field is the lack of a uniform definition of cellular senescence, coupled with inconsistent application of biomarkers to identify and enumerate senescent cells in vivo.
-
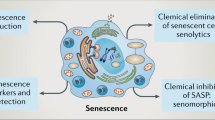
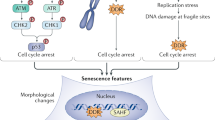
Although frequently used in various combinations to denote putatively senescent cells in vivo, senescence-associated biomarkers — such as robust expression of lysosomal β-galactosidase and the cyclin D-dependent kinase inhibitor p16INK4A, activation of the DNA-damage response, alterations in paracrine secretion and changes in heterochromatin — are individually nonspecific. There is no consensus on which amalgamation of these biomarkers describes the senescent state.
-
Many cancer-associated stress factors activate senescence biomarkers, supporting the roles of senescence-associated processes in tumour suppression. A commonly used senescence marker, the tumour suppressor p16INK4A, progressively increases during organismal ageing and may indeed restrict longevity in certain animal models, but it is unclear whether its expression marks the accumulation of senescent cells per se or, instead, accompanies proliferative arrest in response to many forms of cellular stress.
-
We advocate that 'cellular senescence' should be strictly defined as stress-induced proliferative arrest accompanied by the failure to re-enter the cell division cycle in response to mitogenic and oncogenic stimuli. More-specific descriptors could then be applied to characterize many of the diverse phenotypes that define complex and possibly distinct cellular states currently aggregated under the umbrella term 'senescence'.
Abstract
'Cellular senescence', a term originally defining the characteristics of cultured cells that exceed their replicative limit, has been broadened to describe durable states of proliferative arrest induced by disparate stress factors. Proposed relationships between cellular senescence, tumour suppression, loss of tissue regenerative capacity and ageing suffer from lack of uniform definition and consistently applied criteria. Here, we highlight caveats in interpreting the importance of suboptimal senescence-associated biomarkers, expressed either alone or in combination. We advocate that more-specific descriptors be substituted for the now broadly applied umbrella term 'senescence' in defining the suite of diverse physiological responses to cellular stress.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
16 July 2015
In the version of this article that was originally published, the figure permission credit line was missing from the legend of Figure 2. The following credit line has now been added to the online versions of the article: "Figure adapted with permission from REF. 71, Elsevier, and REF. 174, Wiley Periodicals.".
References
Hayflick, L. & Moorhead, P. S. The serial cultivation of human diploid cell strains. Exp. Cell Res. 25, 585–621 (1961).
Hayflick, L. The limited in vitro lifetime of human diploid cell strains. Exp. Cell Res. 37, 614–636 (1965).
Harley, C. B., Futcher, A. B. & Greider, C. W. Telomeres shorten during aging of human fibroblasts. Nature 345, 458–460 (1990).
Bodnar, A. G. et al. Extension of life-span by introduction of telomerase into normal human cells. Science 279, 349–352 (1998). This landmark paper provides direct evidence that experimental restoration of catalytically active telomerase in cultured human cells bypasses replicative senescence without causing oncogenic transformation.
Todaro, G. J. & Green, H. Quantitative studies of the growth of mouse embryo cells in culture and their development into established lines. J. Cell Biol. 17, 299–313 (1963).
Blasco, M. A. et al. Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 91, 25–34 (1997). This paper demonstrates that, although laboratory strains of mice exhibit long telomeres, knocking out the RNA subunit of telomerase and interbreeding telomerase-deficient mice for sequential generations unmasks the potential effects of telomere attrition in this species.
Harvey, D. M. & Levine, A. J. p53 alteration is a common event in the spontaneous immortalization of primary BALB/c murine embryo fibroblasts. Genes Dev. 5, 2375–2385 (1991).
Serrano, M., Lin, A. W., McCurrach, M. E., Beach, D. & Lowe, S. W. Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 88, 593–602 (1997). The first demonstration of OIS associated with increased expression of p16INK4A.
Kamijo, T. et al. Tumor suppression at the mouse INK4a locus mediated by the alternative reading frame product p19ARF. Cell 91, 649–659 (1997). This paper was the first to describe epistasis between ARF and p53, and unveiled the role of ARF as a tumour suppressor.
Wright, W. E. & Shay, J. W. Telomere dynamics in cancer progression and prevention: fundamental differences in human and mouse telomere biology. Nat. Med. 6, 849–851 (2000).
Sherr, C. J. & DePinho, R. A. Cellular senescence: mitotic clock or culture shock? Cell 102, 407–410 (2000).
Kuilman, T., Michaloglou, C., Mooi, W. J. & Peeper, D. S. The essence of senescence. Genes Dev. 24, 2463–2479 (2010).
Rodier, F. & Campisi, J. Four faces of cellular senescence. J. Cell Biol. 192, 547–556 (2011).
Campisi, J. Aging, cellular senescence, and cancer. Annu. Rev. Physiol. 75, 685–705 (2013).
Collado, M., Blasco, M. A. & Serrano, M. Cellular senescence in cancer and aging. Cell 130, 223–233 (2007).
Kim, W. Y. & Sharpless, N. E. The regulation of INK4/ARF in cancer and aging. Cell 127, 265–275 (2006).
Shay, J. W., Pereira-Smith, O. M. & Wright, W. E. A role for both RB and p53 in the regulation of human cellular senescence. Exp. Cell Res. 196, 33–39 (1991).
Beauséjour, C. M. et al. Reversal of human cellular senescence: roles of the p53 and p16 pathways. EMBO J. 22, 4212–4222 (2003). This paper reveals that low threshold levels of p16INK4A expression in certain senescent human cell lines facilitate their re-entry into the cell division cycle.
Sage, J., Miller, A. L., Pérez-Mancera, P. A., Wysocki, J. M. & Jacks, T. Acute mutation of retinoblastoma gene function is sufficient for cell cycle re-entry. Nature 424, 223–228 (2003).
Pardee, A. B. G1 events and regulation of cell proliferation. Science 246, 603–608 (1989).
Sherr, C. J. & Roberts, J. M. Inhibitors of mammalian G1 cyclin-dependent kinases. Genes Dev. 9, 1149–1163 (1995).
Blagosklonny, M. V. Geroconversion: irreversible step to cellular senescence. Cell Cycle 13, 3628–3635 (2014).
Blais, A., van Oevelen, C. J. C., Margueron, R., Acosta-Alvear, D. & Dynlacht, B. D. Retinoblastoma tumor suppressor protein-dependent methylation of histone H3 lysine 27 is associated with irreversible cell cycle exit. J. Cell Biol. 179, 1399–1412 (2007).
Burkhart, D. L. & Sage, J. Cellular mechanisms of tumour suppression by the retinoblastoma gene. Nat. Rev. Cancer 8, 671–682 (2008).
Indovina, P., Marcelli, E., Casini, N., Rizzo, V. & Giordano, A. Emerging roles of RB family: new defense mechanisms against tumor progression. J. Cell. Physiol. 228, 525–535 (2013).
Kareta, M. S. et al. Inhibition of pluripotency networks by the Rb tumor suppressor restricts reprogramming and tumorigenesis. Cell Stem Cell 16, 39–50 (2015).
Polyak, K. et al. P27Kip1, a cyclin–Cdk inhibitor, links transforming growth-factor-β and contact inhibition to cell-cycle arrest. Genes Dev. 8, 9–22 (1994).
Balciunaite, E. et al. Pocket protein complexes are recruited to distinct targets in quiescent and proliferating cells. Mol. Cell. Biol. 25, 8166–8178 (2005). This paper details elegant analyses using combined ChIP and DNA microarrays ('ChIP-on-ChIP') that define different mechanistic roles of RB and p130 in regulating E2F target genes in quiescent and proliferating cells.
El-Deiry, W. S. et al. WAF1, a potential mediator of p53 tumor suppression. Cell 75, 817–825 (1993).
Harper, J. W., Adami, G. R., Wei, N., Keyomarsi, K. & Elledge, S. J. The p21 Cdk-interacting protein Cip1 is a potent inhibitor of G1 cyclin-dependent kinases. Cell 75, 805–816 (1993).
Xiong, Y. et al. p21 is a universal inhibitor of cyclin kinases. Nature 366, 701–704 (1993).
Wang, Y. A., Elson, A. & Leder, P. Loss of p21 increases sensitivity to ionizing radiation and delays the onset of lymphoma in atm-deficient mice. Proc. Natl Acad. Sci. USA 94, 14590–14595 (1997).
Deng, C., Zhang, P., Harper, J. W., Elledge, S. J. & Leder, P. Mice lacking p21CIP1/WAF1 undergo normal development, but are defective in G1 checkpoint control. Cell 82, 675–684 (1995).
Martín-Caballero, J., Flores, J. M., García-Palencia, P. & Serrano, M. Tumor susceptibility of p21Waf1/Cip1-deficient mice. Cancer Res. 61, 6234–6238 (2001).
Pantoja, C. & Serrano, M. Murine fibroblasts lacking p21 undergo senescence and are resistant to transformation by oncogenic Ras. Oncogene 18, 4974–4982 (1999).
Di Leonardo, A., Linke, S. P., Clarkin, K. & Wahl, G. M. DNA damage triggers a prolonged p53-dependent G1 arrest and long-term induction of Cip1 in normal human fibroblasts. Genes Dev. 8, 2540–2551 (1994).
Sang, L., Coller, H. A. & Roberts, J. M. Control of the reversibility of cellular quiescence by the transcriptional repressor HES1. Science 321, 1095–1100 (2008).
Spencer, S. L. et al. The proliferation-quiescence decision is controlled by a bifurcation in CDK2 activity at mitotic exit. Cell 155, 369–383 (2013).
Krenning, L., Feringa, F. M., Shaltiel, I. A., van den Berg, J. & Medema, R. H. Transient activation of p53 in G2 phase is sufficient to induce senescence. Mol. Cell 55, 59–72 (2014).
Alcorta, D. A. et al. Involvement of the cyclin-dependent kinase inhibitor p16 (INK4a) in replicative senescence of normal human fibroblasts. Proc. Natl Acad. Sci. USA 93, 13742–13747 (1996).
Robles, S. J. & Adami, G. R. Agents that cause DNA double strand breaks lead to p16INK4a enrichment and the premature senescence of normal fibrolasts. Oncogene 16, 1113–1123 (1998).
Stein, G. H., Drullinger, L. F., Soulard, A. & Dulic, V. Differential roles for cyclin-dependent kinase inhibitors p21 and p16 in the mechanisms of senescence and differentiation in human fibroblasts. Mol. Cell. Biol. 19, 2109–2117 (1999).
Johmura, Y. et al. Necessary and sufficient role for a mitosis skip in senescence induction. Mol. Cell 55, 73–84 (2014).
Gire, V. & Dulic, V. Senescence from G2 arrest, revisited. Cell Cycle 14, 297–304 (2015).
Sherwood, S. W., Rush, D., Ellsworth, J. L. & Schimke, R. T. Defining cellular senescence in IMR-90 cells — a flow cytometric analysis. Proc. Natl Acad. Sci. USA 85, 9086–9090 (1988).
Mao, Z., Ke, Z., Gorbunova, V. & Seluanov, A. Replicatively senescent cells are arrested in G1 and G2 phases. Aging 4, 431–435 (2012).
Wagner, M. et al. Replicative senescence of human endothelial cells in vitro involves G1 arrest, polyploidization and senescence-associated apoptosis. Exp. Gerontol. 36, 1327–1347 (2001).
Takahashi, A. et al. Mitogenic signalling and the p16INK4a–Rb pathway cooperate to enforce irreversible cellular senescence. Nat. Cell Biol. 8, 1291–1297 (2006).
Aksoy, O. et al. The atypical E2F family member E2F7 couples the p53 and RB pathways during cellular senescence. Genes Dev. 26, 1546–1557 (2012).
Lanni, J. S. & Jacks, T. Characterization of the p53-dependent postmitotic checkpoint following spindle disruption. Mol. Cell. Biol. 18, 1055–1064 (1998).
Hayashi, M. T. & Karlseder, J. DNA damage associated with mitosis and cytokinesis failure. Oncogene 32, 4593–4601 (2013).
Coppe, J. P. et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 6, 2853–2868 (2008).
Rodier, F. et al. Persistent DNA damage signalling triggers senescence-associated inflammatory cytokine secretion. Nat. Cell Biol. 11, 973–979 (2009).
Narita, M. et al. Rb-mediated heterochromatin formation and silencing of E2F target genes during cellular senescence. Cell 113, 703–716 (2003). This paper provides the earliest description of RB-mediated SAHF formation and its effects in silencing E2F target genes.
Bartkova, J. et al. Oncogene-induced senescence is part of the tumorigenesis barrier imposed by DNA damage checkpoints. Nature 444, 633–637 (2006).
Di Micco, R. et al. Oncogene-induced senescence is a DNA damage response triggered by DNA hyper-replication. Nature 444, 638–642 (2006). References 55 and 56 illustrate the ability of oncogene-induced stress to activate DNA damage-dependent checkpoint responses that provide a barrier to tumour formation.
Rodier, F. et al. DNA-SCARS: distinct nuclear structures that sustain damage-induced senescence growth arrest and inflammatory cytokine secretion. J. Cell Sci. 124, 68–81 (2011).
Fumagalli, M., Rossiello, F., Mondello, C. & di Fagagna, F. D. Stable cellular senescence is associated with persistent DDR activation. PLoS ONE 9, e110969 (2014).
Dimri, G. P. et al. A biomarker that identifies senescent human-cells in culture and in aging skin in vivo. Proc. Natl Acad. Sci. USA 92, 9363–9367 (1995). This paper gives the first description of an assay for SA β -gal activity at pH 6.0, which is now widely used to denote senescent cells.
Itahana, K., Campisi, J. & Dimri, G. P. Methods to detect biomarkers of cellular senescence: the senescence-associated β-galactosidase assay. Methods Mol. Biol. 371, 21–31 (2007).
Kurz, D. J., Decary, S., Hong, Y. & Erusalimsky, J. D. Senescence-associated β-galactosidase reflects an increase in lysosomal mass during replicative ageing of human endothelial cells. J. Cell Sci. 113, 3613–3622 (2000).
Lee, B. Y. et al. Senescence-associated β-galactosidase is lysosomal β-galactosidase. Aging Cell 5, 187–195 (2006).
Coates, P. J., Lorimore, S. A., Rigat, B. A., Lane, D. P. & Wright, E. G. Induction of endogenous β-galactosidase by ionizing radiation complicates the analysis of p53–lacZ transgenic mice. Oncogene 20, 7096–7097 (2001).
Bursuker, I., Rhodes, J. M. & Goldman, R. β-galactosidase — an indicator of the maturational stage of mouse and human mononuclear phagocytes. J. Cell. Physiol. 112, 385–390 (1982).
Kopp, H. G., Hooper, A. T., Shmelkov, S. V. & Rafii, S. β-galactosidase staining on bone marrow. The osteoclast pitfall. Histol. Histopathol. 22, 971–976 (2007).
Young, A. R. J. & Narita, M. Connecting autophagy to senescence in pathophysiology. Curr. Opin. Cell Biol. 22, 234–240 (2010).
Dorr, J. R. et al. Synthetic lethal metabolic targeting of cellular senescence in cancer therapy. Nature 501, 421–425 (2013).
Ivanov, A. et al. Lysosome-mediated processing of chromatin in senescence. J. Cell Biol. 202, 129–143 (2013).
Serrano, M., Hannon, G. J. & Beach, D. A new regulatory motif in cell-cycle control causing specific-inhibition of cyclin-D/CDK4. Nature 366, 704–707 (1993). This paper describes the landmark discovery of p16INK4A and its role as a specific inhibitor of the cyclin D-dependent kinase CDK4.
Hannon, G. J. & Beach, D. p15INK4b is a potential effector of TGF-β-induced cell-cycle arrest. Nature 371, 257–261 (1994).
Quelle, D. E., Zindy, F., Ashmun, R. A. & Sherr, C. J. Alternative reading frames of the INK4a tumor-suppressor gene encode two unrelated proteins capable of inducing cell-cycle arrest. Cell 83, 993–1000 (1995). This paper describes the surprising discovery of a second protein (ARF) that is encoded by the mouse Cdkn2a gene.
Pomerantz, J. et al. The Ink4a tumor suppressor gene product, 19Arf, interacts with MDM2 and neutralizes MDM2's inhibition of p53. Cell 92, 713–723 (1998).
Zhang, Y., Xiong, Y. & Yarbrough, W. G. ARF promotes MDM2 degradation and stabilizes p53: ARF–INK4a locus deletion impairs both the Rb and p53 tumor suppression pathways. Cell 92, 725–734 (1998).
Kamijo, T. et al. Functional and physical interactions of the ARF tumor suppressor with p53 and Mdm2. Proc. Natl Acad. Sci. USA 95, 8292–8297 (1998).
Chandler, H. & Peters, G. Stressing the cell cycle in senescence and aging. Curr. Opin. Cell Biol. 25, 765–771 (2013).
Bignell, G. R. et al. Signatures of mutation and selection in the cancer genome. Nature 463, 893–898 (2010).
Beroukhim, R. et al. The landscape of somatic copy-number alteration across human cancers. Nature 463, 899–905 (2010).
Rheinwald, J. G. et al.A two-stage, p16INK4A- and p53-dependent keratinocyte senescence mechanism that limits replicative potential independent of telomere status. Mol. Cell. Biol. 22, 6930–6930 (2002).
Herbig, U., Jobling, W. A., Chen, B. P. C., Chen, D. J. & Sedivy, J. M. Telomere shortening triggers senescence of human cells through a pathway involving ATM, 53, and p21CIP1, but not p16INK4A. Mol. Cell 14, 501–513 (2004).
Yamakoshi, K. et al. Real-time in vivo imaging of p16Ink4a reveals cross talk with p53. J. Cell Biol. 186, 393–407 (2009).
Krimpenfort, P. et al. p15Ink4b is a critical tumour suppressor in the absence of p16Ink4a. Nature 448, 943–946 (2007).
Kuilman, T. et al. Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell 133, 1019–1031 (2008).
Cudejko, C. et al. p16INK4a deficiency promotes IL-4-induced polarization and inhibits proinflammatory signaling in macrophages. Blood 118, 2556–2566 (2011).
Wiedemeyer, R. et al. Feedback circuit among INK4 tumor suppressors constrains human glioblastoma development. Cancer Cell 13, 355–364 (2008).
Shapiro, G. I. et al. Reciprocal Rb inactivation and p16INK4 expression in primary lung cancers and cell lines. Cancer Res. 55, 505–509 (1995).
Khleif, S. N. et al. Inhibition of cyclin D–CDK4/CDK6 activity is associated with an E2F-mediated induction of cyclin kinase inhibitor activity. Proc. Natl Acad. Sci. USA 93, 4350–4354 (1996).
Nakao, Y. et al. Induction of p16 during immortalization by HPV 16 and 18 and not during malignant transformation. Br. J. Cancer 75, 1410–1416 (1997).
Witkiewicz, A. K., Knudsen, K. E., Dicker, A. P. & Knudsen, E. S. The meaning of p16ink4a expression in tumors: functional significance, clinical associations and future developments. Cell Cycle 10, 2497–2503 (2011).
Ohtani, N., Yamakoshi, K., Takahashi, A. & Hara, E. Real-time in vivo imaging of p16Ink4a gene expression: a new approach to study senescence stress signaling in living animals. Cell Div. 5, 1 (2010).
Baker, D. J. et al. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature 479, 232–236 (2011).
Demaria, M. et al. An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev. Cell 31, 722–733 (2014).
Burd, C. E. et al. Monitoring tumorigenesis and senescence in vivo with a p16INK4a-luciferase model. Cell 152, 340–351 (2013). References 80 and 89–92 describe the use of different p16 reporters in mice and demonstrate that increased expression of p16, together with other senescence markers such as SASP and SAβ-gal, marks the accumulation of various senescent cell types during inflammation, wound healing, ageing and tumorigenesis. Conditional removal of cells persistently expressing p16 can delay the onset of age-associated pathologies and attenuate already established age-associated disorders; by contrast, the transient appearance of p16- and SASP-positive cells can promote tissue repair.
Zindy, F., Quelle, D. E., Roussel, M. F. & Sherr, C. J. Expression of the p16INK4a tumor suppressor versus other INK4 family members during mouse development and aging. Oncogene 15, 203–211 (1997).
Krishnamurthy, J. et al. Ink4a/Arf expression is a biomarker of aging. J. Clin. Invest. 114, 1299–1307 (2004).
Waaijer, M. E. et al. The number of p16INK4a positive cells in human skin reflects biological age. Aging Cell 11, 722–725 (2012).
Sorrentino, J. A. et al. p16INK4a reporter mice reveal age-promoting effects of environmental toxicants. J. Clin. Invest. 124, 169–173 (2014).
Natarajan, E. et al. Co-expression of p16INK4A and laminin 5 γ2 by microinvasive and superficial squamous cell carcinomas in vivo and by migrating wound and senescent keratinocytes in culture. Am. J. Pathol. 163, 477–491 (2003).
Jun, J. I. & Lau, L. F. Cellular senescence controls fibrosis in wound healing. Aging 2, 627–631 (2010).
Jun, J. I. & Lau, L. F. The matricellular protein CCN1 induces fibroblast senescence and restricts fibrosis in cutaneous wound healing. Nat. Cell Biol. 12, 676–685 (2010).
Lemster, B. H. et al. Induction of CD56 and TCR-independent activation of T cells with aging. J. Immunol. 180, 1979–1990 (2008).
Liu, Y. et al. Expression of p16INK4a in peripheral blood T-cells is a biomarker of human aging. Aging Cell 8, 439–448 (2009).
Liu, Y. et al. Expression of p16INK4a prevents cancer and promotes aging in lymphocytes. Blood 117, 3257–3267 (2011).
Migliaccio, M., Raj, K., Menzel, O. & Rufer, N. Mechanisms that limit the in vitro proliferative potential of human CD8+ T lymphocytes. J. Immunol. 174, 3335–3343 (2005).
Erickson, S. et al. Involvement of the Ink4 proteins p16 and p15 in T-lymphocyte senescence. Oncogene 17, 595–602 (1998).
Schmitt, C. A. et al. A senescence program controlled by p53 and p16INK4a contributes to the outcome of cancer therapy. Cell 109, 335–346 (2002).
Sanoff, H. K. et al. Effect of cytotoxic chemotherapy on markers of molecular age in patients with breast cancer. J. Natl Cancer Inst. 106, dju057 (2014).
Nelson, J. A. E. et al. Expression of p16INK4a as a biomarker of T-cell aging in HIV-infected patients prior to and during antiretroviral therapy. Aging Cell 11, 916–918 (2012).
Signer, R. A. J., Montecino-Rodriguez, E., Witte, O. N. & Dorshkind, K. Aging and cancer resistance in lymphoid progenitors are linked processes conferred by p16Ink4a and Arf. Genes Dev. 22, 3115–3120 (2008).
Jaruga, E., Skierski, J., Radziszewska, E. & Sikora, E. Proliferation and apoptosis of human T cells during replicative senescence — a critical approach. Acta Biochim. Polon. 47, 293–300 (2000).
Shi, L. et al. KLRG1 impairs CD4+ T cell responses via p16ink4a and p27kip1 pathways: role in hepatitis B vaccine failure in individuals with hepatitis C virus infection. J. Immunol. 192, 649–657 (2014).
Di Mitri, D. et al. Reversible senescence in human CD4+CD45RA+CD27− memory T cells. J. Immunol. 187, 2093–2100 (2011).
Liu, Y. & Sharpless, N. E. Tumor suppressor mechanisms in immune aging. Curr. Opin. Immunol. 21, 431–439 (2009).
Akbar, A. N. & Henson, S. M. Are senescence and exhaustion intertwined or unrelated processes that compromise immunity? Nat. Rev. Immunol. 11, 289–295 (2011).
d'Adda di Fagagna, F. et al. A DNA damage checkpoint response in telomere-initiated senescence. Nature 426, 194–198 (2003).
Takai, H., Smogorzewska, A. & de Lange, T. DNA damage foci at dysfunctional telomeres. Curr. Biol. 13, 1549–1556 (2003).
Nakamura, A. J. et al. Both telomeric and non-telomeric DNA damage are determinants of mammalian cellular senescence. Epigenetics Chromatin 1, 6 (2008).
Halazonetis, T. D., Gorgoulis, V. G. & Bartek, J. An oncogene-induced DNA damage model for cancer development. Science 319, 1352–1355 (2008).
Bakkenist, C. J. & Kastan, M. B. DNA damage activates ATM through intermolecular autophosphorylation and dimer dissociation. Nature 421, 499–506 (2003).
Munro, J., Barr, N. I., Ireland, H., Morrison, V. & Parkinson, E. K. Histone deacetylase inhibitors induce a senescence-like state in human cells by a p16-dependent mechanism that is independent of a mitotic clock. Exp. Cell Res. 295, 525–538 (2004).
Kosar, M. et al. Senescence-associated heterochromatin foci are dispensable for cellular senescence, occur in a cell type- and insult-dependent manner, and follow expression of p16ink4a. Cell Cycle 10, 457–468 (2011).
Di Micco, R. et al. Interplay between oncogene-induced DNA damage response and heterochromatin in senescence and cancer. Nat. Cell Biol. 13, 292–302 (2011).
Kennedy, A. L. et al. Senescent mouse cells fail to overtly regulate the HIRA histone chaperone and do not form robust Senescence Associated Heterochromatin Foci. Cell Div. 5, 16 (2010).
Trimarchi, J. M. & Lees, J. A. Sibling rivalry in the E2F family. Nat. Rev. Mol. Cell Biol. 3, 11–20 (2002).
Chandra, T. et al. Independence of repressive histone marks and chromatin compaction during senescent heterochromatic layer formation. Mol. Cell 47, 203–214 (2012).
Chandra, T. & Narita, M. High-order chromatin structure and the epigenome in SAHFs. Nucleus 4, 23–28 (2013).
Chien, Y. C. et al. Control of the senescence-associated secretory phenotype by NF-κB promotes senescence and enhances chemosensitivity. Genes Dev. 25, 2125–2136 (2011).
Acosta, J. C. et al. A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nat. Cell Biol. 15, 978–990 (2013). This paper details an elegant study describing the paracrine spreading of SASP during OIS and control of SASP expression by inflammasome-mediated IL-1 expression.
Xue, W. et al. Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature 445, 656–660 (2007).
Krizhanovsky, V. et al. Senescence of activated stellate cells limits liver fibrosis. Cell 134, 657–667 (2008).
Lujambio, A. et al. Non-cell-autonomous tumor suppression by p53. Cell 153, 449–460 (2013). References 128–130 underscore how p53 activity in both tumour cells and non-cancer cells in the liver governs aspects of the innate immune response to regulate liver fibrosis and constrain tumorigenesis.
Krtolica, A., Parrinello, S., Lockett, S., Desprez, P. Y. & Campisi, J. Senescent fibroblasts promote epithelial cell growth and tumorigenesis: a link between cancer and aging. Proc. Natl Acad. Sci. USA 98, 12072–12077 (2001).
Kang, T. W. et al. Senescence surveillance of pre-malignant hepatocytes limits liver cancer development. Nature 479, 547–551 (2011).
Ye, J. et al. Human regulatory T cells induce T-lymphocyte senescence. Blood 120, 2021–2031 (2012).
Braumuller, H. et al. T-helper-1-cell cytokines drive cancer into senescence. Nature 494, 361–365 (2013).
Neves, J., Demaria, M., Campisi, J. & Jasper, H. Of flies, mice, and men: evolutionarily conserved tissue damage responses and aging. Dev. Cell 32, 9–18 (2015).
Wolf, J., Rose-John, S. & Garbers, C. Interleukin-6 and its receptors: a highly regulated and dynamic system. Cytokine 70, 11–20 (2014).
Ataie-Kachoie, P., Pourgholami, M. H., Richardson, D. & Morris, D. L. Gene of the month: Interleukin 6 (IL-6). J. Clin. Pathol. 67, 932–937 (2014).
Blackburn, E. H. Structure and function of telomeres. Nature 350, 569–573 (1991).
Lansdorp, P. M. Telomeres and disease. EMBO J. 28, 2532–2540 (2009).
Artandi, S. E. & DePinho, R. A. Telomeres and telomerase in cancer. Carcinogenesis 31, 9–18 (2010).
Shay, J. W. & Wright, W. E. Role of telomeres and telomerase in cancer. Semin. Cancer Biol. 21, 349–353 (2011).
Jaskelioff, M. et al. Telomerase reactivation reverses tissue degeneration in aged telomerase-deficient mice. Nature 469, 102–106 (2011). Following on earlier studies using cultured human cells (reference 4) and genetically engineered telomerase-deficient mice (reference 6), this study demonstrates that telomerase reactivation in adult mice reduces DNA damage and reverses degenerative phenotypes.
Armanios, M. Telomeres and age-related disease: how telomere biology informs clinical paradigms. J. Clin. Invest. 123, 996–1002 (2013).
Aubert, G., Hills, M. & Lansdorp, P. M. Telomere length measurement — caveats and a critical assessment of the available technologies and tools. Mutat. Res. 730, 59–67 (2012).
Hewitt, G. et al. Telomeres are favoured targets of a persistent DNA damage response in ageing and stress-induced senescence. Nat. Commun. 3, 708 (2012).
Rudolph, K. L. et al. Longevity, stress response, and cancer in aging telomerase-deficient mice. Cell 96, 701–712 (1999).
Collado, M. et al. Tumour biology — senescence in premalignant tumours. Nature 436, 642–642 (2005).
Munoz-Espin, D. et al. Programmed cell senescence during mammalian embryonic development. Cell 155, 1104–1118 (2013).
Storer, M. et al. Senescence is a developmental mechanism that contributes to embryonic growth and patterning. Cell 155, 1119–1130 (2013). References 148 and 149 provocatively conclude that senescent cells transiently appear during various stages of mouse embryonic development, when they function to promote tissue remodelling. Is this the evolutionary origin of damage-induced senescence in adult animals?
Huang, T. & Rivera-Pérez, J. A. Senescence-associated β-galactosidase activity marks the visceral endoderm of mouse embryos but is not indicative of senescence. Genesis 52, 300–308 (2014).
Li, C., Finkelstein, D. & Sherr, C. J. Arf tumor suppressor and miR-205 regulate cell adhesion and formation of extraembryonic endoderm from pluripotent stem cells. Proc. Natl Acad. Sci. USA 110, E1112–E1121 (2013).
Michaloglou, C. et al. BRAFE600-associated senescence-like cell cycle arrest of human naevi. Nature 436, 720–724 (2005).
Gray-Schopfer, V. C. et al. Cellular senescence in naevi and immortalisation in melanoma: a role for p16? Br. J. Cancer 95, 496–505 (2006).
Vredeveld, L. C. et al. Abrogation of BRAFV600E-induced senescence by PI3K pathway activation contributes to melanomagenesis. Genes Dev. 26, 1055–1069 (2012).
Damsky, W. et al. mTORC1 activation blocks BrafV600E-induced growth arrest but is insufficient for melanoma formation. Cancer Cell 27, 41–56 (2015).
Sharpless, N. E. & Depinho, R. A. How stem cells age and why this makes us grow old. Nat. Rev. Mol. Cell Biol. 8, 703–713 (2007).
Munoz-Espin, D. & Serrano, M. Cellular senescence: from physiology to pathology. Nat. Rev. Mol. Cell Biol. 15, 482–496 (2014).
Martin, N., Beach, D. & Gill, J. Ageing as developmental decay: insights from p16INK4a. Trends Mol. Med. 20, 667–674 (2014).
van Deursen, J. M. The role of senescent cells in ageing. Nature 509, 439–446 (2014).
Signer, R. A. J. & Morrison, S. J. Mechanisms that regulate stem cell aging and life span. Cell Stem Cell 12, 152–165 (2013).
Zhu, Y. et al. The Achilles heel of senescent cells: from transcriptome to senolytic drugs. Aging Cell http://dx.doi.org/10.1111/acel.12344 (2015).
González-Navarro, H. et al. p19ARF deficiency reduces macrophage and vascular smooth muscle cell apoptosis and aggravates atherosclerosis. J. Am. Coll. Cardiol. 55, 2258–2268 (2010).
Herbig, U., Ferreira, M., Condel, L., Carey, D. & Sedivy, J. M. Cellular senescence in aging primates. Science 311, 1257 (2006).
Ershler, W. B. & Keller, E. T. Age-associated increased interleukin-6 gene expression, late-life diseases, and frailty. Annu. Rev. Med. 51, 245–270 (2000).
Krishnamurthy, J. et al. p16INK4a induces an age-dependent decline in islet regenerative potential. Nature 443, 453–457 (2006).
Janzen, V. et al. Stem-cell ageing modified by the cyclin-dependent kinase inhibitor p16INK4a. Nature 443, 421–426 (2006).
Molofsky, A. V. et al. Increasing p16INK4a expression decreases forebrain progenitors and neurogenesis during ageing. Nature 443, 448–452 (2006).
Chen, H. et al. PDGF signalling controls age-dependent proliferation in pancreatic β-cells. Nature 478, 349–355 (2011).
Cosgrove, B. D. et al. Rejuvenation of the muscle stem cell population restores strength to injured aged muscles. Nat. Med. 20, 255–264 (2014).
Sousa-Victor, P. et al. Geriatric muscle stem cells switch reversible quiescence into senescence. Nature 506, 316–321 (2014).
Berent-Maoz, B., Montecino-Rodriguez, E., Signer, R. A. J. & Dorshkind, K. Fibroblast growth factor-7 partially reverses murine thymocyte progenitor aging by repression of Ink4a. Blood 119, 5715–5721 (2012).
Baker, D. J. et al. Opposing roles for p16Ink4a and p19Arf in senescence and ageing caused by BubR1 insufficiency. Nat. Cell Biol. 10, 825–836 (2008).
Jeck, W. R., Siebold, A. P. & Sharpless, N. E. Review: a meta-analysis of GWAS and age-associated diseases. Aging Cell 11, 727–731 (2012).
Sherr, C. J. Ink4–Arf locus in cancer and aging. Wiley Interdiscip. Rev. Dev. Biol. 1, 731–741 (2012).
LaPak, K. M. & Burd, C. E. The molecular balancing act of p16INK4a in cancer and aging. Mol. Cancer Res. 12, 167–183 (2014).
Henson, J. D., Neumann, A. A., Yeager, T. R. & Reddel, R. R. Alternative lengthening of telomeres in mammalian cells. Oncogene 21, 598–610 (2002).
O'Sullivan, R. J. & Almouzni, G. Assembly of telomeric chromatin to create ALTernative endings. Trends Cell Biol. 24, 675–685 (2014).
Cho, N. W., Dilley, R. L., Lampson, M. A. & Greenberg, R. A. Interchromosomal homology searches drive directional ALT telomere movement and synapsis. Cell 159, 108–121 (2014).
Boquoi, A. et al. Reversible cell cycle inhibition and premature aging features imposed by conditional expression of p16Ink4a. Aging Cell 14, 139–147 (2015).
Althubiti, M. et al. Characterization of novel markers of senescence and their prognostic potential in cancer. Cell Death Dis. 5, e1528 (2014).
Acknowledgements
The authors thank J. Sage, K. Dorshkind, C. Burd, A. Banito and J. Morris for comments and criticisms on the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- mTOR
-
A serine/threonine kinase incorporated into mTOR complex 1 (mTORC1) and mTORC2, which act as nutrient sensors and regulators of translation.
- APC/C
-
(Anaphase-promoting complex/cyclosome). A multi-subunit ubiquitin ligase complex that degrades cyclins A and B, depending on two alternative substrate selectivity factors, CDC20 and CDH1, that function during mitosis and G1 phase, respectively.
- Foci
-
A nonspecific term frequently used to designate discrete, punctate topological sites (for example, of chromosomal DNA damage or heterochromatinization), which can also be described as speckles, detected by microscopy and usually with the aid of fluorescence-based antibodies.
- CDKN2A
-
(Cyclin-dependent kinase inhibitor 2A; also known as the INK4A–ARF locus). Originally used to designate the gene encoding p16INK4A, the locus is now recognized to encode a second, unrelated alternative reading frame (ARF) protein as well.
- Ataxia telangiectasia mutated
-
(ATM). A serine/threonine kinase that acts as a sensor of DNA damage and that phosphorylates various substrates during the different phases of the cell cycle to activate checkpoint responses that arrest the growth of cells with DNA damage.
- γH2AX
-
A phosphorylated histone variant that decorates chromatin sites of DNA damage and is required for the assembly of repair proteins during the DNA-damage response.
- Heterochromatin protein 1
-
(HP1). A family of proteins that bind to trimethylated Lys9 on histone H3, which is important in gene silencing.
- Dyskeratosis congenita
-
A rare inherited disorder presenting with variable degenerative ageing phenotypes accompanied by reduced telomere maintenance and shortened lifespan; it is most commonly triggered by mutations affecting X-linked DKC1, which encodes the telomerase cofactor dyskerin.
Rights and permissions
About this article
Cite this article
Sharpless, N., Sherr, C. Forging a signature of in vivo senescence. Nat Rev Cancer 15, 397–408 (2015). https://doi.org/10.1038/nrc3960
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc3960
This article is cited by
-
Glial senescence enhances α-synuclein pathology owing to its insufficient clearance caused by autophagy dysfunction
Cell Death Discovery (2024)
-
METTL3 promotes cellular senescence of colorectal cancer via modulation of CDKN2B transcription and mRNA stability
Oncogene (2024)
-
Mallory-Denk bodies and hepatocellular senescence: a causal relationship?
Virchows Archiv (2024)
-
Oncogene-induced senescence in meningiomas—an immunohistochemical study
Journal of Neuro-Oncology (2024)
-
Targeting therapy-induced senescence as a novel strategy to combat chemotherapy-induced peripheral neuropathy
Supportive Care in Cancer (2024)