Key Points
-
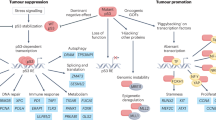
MDM2 and MDMX are RING domain proteins that exert their oncogenic effects primarily by inhibiting the p53 tumour suppressor protein.
-
Each protein is overexpressed in diverse tumour types by mechanisms including gene amplification and post-translational stabilization; this is generally more frequent in tumours with a wild-type TP53 allele.
-
Despite their similar structures, only MDM2 has intrinsic E3 ubiquitin ligase activity. Although MDM2 alone can inhibit p53, its RING-dependent heterodimerization with MDMX has an important role in p53 inhibition.
-
Both MDM2 and MDMX interact with multiple other partners. Aberrant interactions with these partners may also affect gene expression and genome stability.
-
Structure-based drug design has yielded several MDM antagonists that block MDM–p53 interactions, leading to p53 activation. At least one agent has progressed to clinical trials.
-
Systems biology studies are providing the rationale for using MDM protein antagonists in combination with both approved and experimental pathway-targeted anticancer drugs.
Abstract
The MDM2 and MDMX (also known as HDMX and MDM4) proteins are deregulated in many human cancers and exert their oncogenic activity predominantly by inhibiting the p53 tumour suppressor. However, the MDM proteins modulate and respond to many other signalling networks in which they are embedded. Recent mechanistic studies and animal models have demonstrated how functional interactions in these networks are crucial for maintaining normal tissue homeostasis, and for determining responses to oncogenic and therapeutic challenges. This Review highlights the progress made and pitfalls encountered as the field continues to search for MDM-targeted antitumour agents.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Bieging, K. T. & Attardi, L. D. Deconstructing p53 transcriptional networks in tumor suppression. Trends Cell Biol. 22, 97–106 (2012).
Momand, J., Zambetti, G. P., Olson, D. C., George, D. & Levine, A. J. The mdm-2 oncogene product forms a complex with the p53 protein and inhibits p53-mediated transactivation. Cell 69, 1237–1245 (1992). The first demonstration that MDM2 could inhibit p53 activity.
Shibagaki, I. et al. p53 mutation, murine double minute 2 amplification, and human papillomavirus infection are frequently involved but not associated with each other in esophageal squamous cell carcinoma. Clin. Cancer Res. 1, 769–773 (1995).
Forslund, A. et al. MDM2 gene amplification is correlated to tumor progression but not to the presence of SNP309 or TP53 mutational status in primary colorectal cancers. Mol. Cancer Res. 6, 205–211 (2008).
Ito, M. et al. Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature 455, 1061–1068 (2008).
Lam, S. et al. Role of Mdm4 in drug sensitivity of breast cancer cells. Oncogene 29, 2415–2426 (2010).
Mejia-Guerrero, S. et al. Characterization of the 12q15 MDM2 and 12q13-14 CDK4 amplicons and clinical correlations in osteosarcoma. Genes Chromosomes Cancer 49, 518–525 (2010).
Ito, M. et al. Comprehensive mapping of p53 pathway alterations reveals an apparent role for both SNP309 and MDM2 amplification in sarcomagenesis. Clin. Cancer Res. 17, 416–426 (2011).
Gilkes, D. M. et al. Regulation of MDMX expression by mitogenic signaling. Mol. Cell. Biol. 28, 1999–2010 (2008).
Gembarska, A. et al. MDM4 is a key therapeutic target in cutaneous melanoma. Nature Med. 18, 1239–1247 (2012). The first report showing that MDMX is associated with aggressive melanoma in vivo and can be targeted with a dual MDM2 and MDMX inhibitor peptide.
Kussie, P. H. et al. Structure of the MDM2 oncoprotein bound to the p53 tumor suppressor transactivation domain. Science 274, 948–953 (1996). The structure that described the hydrophobic p53-binding pocket of MDM2, on which many drug discovery efforts are now based.
Laurie, N. A. et al. Inactivation of the p53 pathway in retinoblastoma. Nature 444, 61–66 (2006).
Barak, Y., Gottlieb, E., Juven-Gershon, T. & Oren, M. Regulation of mdm2 expression by p53: alternative promoters produce transcripts with nonidentical translation potential. Genes Dev. 8, 1739–1749 (1994).
Phillips, A. et al. HDMX-L is expressed from a functional p53-responsive promoter in the first intron of the HDMX gene and participates in an autoregulatory feedback loop to control p53 activity. J. Biol. Chem. 285, 29111–29127 (2010).
Fang, S., Jensen, J. P., Ludwig, R. L., Vousden, K. H. & Weissman, A. M. Mdm2 is a RING finger-dependent ubiquitin protein ligase for itself and p53. J. Biol. Chem. 275, 8945–8951 (2000). This paper revealed that MDM2 controls the stability of both itself and p53, and that the RING domain was required for these functions.
Linares, L. K., Hengstermann, A., Ciechanover, A., Muller, S. & Scheffner, M. HdmX stimulates Hdm2-mediated ubiquitination and degradation of p53. Proc. Natl Acad. Sci. USA 100, 12009–12014 (2003).
Huang, L. et al. The p53 inhibitors MDM2/MDMX complex is required for control of p53 activity in vivo. Proc. Natl Acad. Sci. USA 108, 12001–12006 (2011).
Pant, V., Xiong, S., Iwakuma, T., Quintas-Cardama, A. & Lozano, G. Heterodimerization of Mdm2 and Mdm4 is critical for regulating p53 activity during embryogenesis but dispensable for p53 and Mdm2 stability. Proc. Natl Acad. Sci. USA 108, 11995–12000 (2011).
Plechanovova, A., Jaffray, E. G., Tatham, M. H., Naismith, J. H. & Hay, R. T. Structure of a RING E3 ligase and ubiquitin-loaded E2 primed for catalysis. Nature 489, 115–120 (2012).
Poyurovsky, M. V. et al. The Mdm2 RING domain C-terminus is required for supramolecular assembly and ubiquitin ligase activity. EMBO J. 26, 90–101 (2007).
Uldrijan, S., Pannekoek, W. J. & Vousden, K. H. An essential function of the extreme C-terminus of MDM2 can be provided by MDMX. EMBO J. 26, 102–112 (2007).
Pishas, K. I. et al. Nutlin-3a is a potential therapeutic for ewing sarcoma. Clin. Cancer Res. 17, 494–504 (2011).
McEvoy, J. et al. Analysis of MDM2 and MDM4 single nucleotide polymorphisms, mRNA splicing and protein expression in retinoblastoma. PLoS ONE 7, e42739 (2012).
Francoz, S. et al. Mdm4 and Mdm2 cooperate to inhibit p53 activity in proliferating and quiescent cells in vivo. Proc. Natl Acad. Sci. USA 103, 3232–3237 (2006).
Ries, S. et al. Opposing effects of Ras on p53: transcriptional activation of mdm2 and induction of p19ARF. Cell 103, 321–330 (2000).
Alt, J. R., Greiner, T. C., Cleveland, J. L. & Eischen, C. M. Mdm2 haplo-insufficiency profoundly inhibits Myc-induced lymphomagenesis. EMBO J. 22, 1442–1450 (2003).
Terzian, T. et al. Haploinsufficiency of Mdm2 and Mdm4 in tumorigenesis and development. Mol. Cell. Biol. 27, 5479–5485 (2007).
De Clercq, S. et al. Widespread overexpression of epitope-tagged Mdm4 does not accelerate tumor formation in vivo. Mol. Cell. Biol. 30, 5394–5405 (2010).
Zhou, J. X. et al. IFN regulatory factor 8 regulates MDM2 in germinal center B cells. J. Immunol. 183, 3188–3194 (2009).
Tarocchi, M. et al. Carcinogen-induced hepatic tumors in KLF6+/− mice recapitulate aggressive human hepatocellular carcinoma associated with p53 pathway deregulation. Hepatology 54, 522–531 (2011).
Zhang, X. et al. Transcription factor NFAT1 activates the mdm2 oncogene independent of p53. J. Biol. Chem. 287, 30468–30476 (2012).
Mayo, L. D. & Donner, D. B. A phosphatidylinositol 3-kinase/Akt pathway promotes translocation of Mdm2 from the cytoplasm to the nucleus. Proc. Natl Acad. Sci. USA 98, 11598–11603 (2001).
Lopez-Pajares, V., Kim, M. M. & Yuan, Z. M. Phosphorylation of MDMX mediated by Akt leads to stabilization and induces 14-3-3 binding. J. Biol. Chem. 283, 13707–13713 (2008).
Chen, L., Li, C., Pan, Y. & Chen, J. Regulation of p53-MDMX interaction by casein kinase 1 α. Mol. Cell. Biol. 25, 6509–6520 (2005).
Mayo, L. D., Turchi, J. J. & Berberich, S. J. Mdm-2 phosphorylation by DNA-dependent protein kinase prevents interaction with p53. Cancer Res. 57, 5013–5016 (1997).
Zuckerman, V. et al. c-Abl phosphorylates Hdmx and regulates its interaction with p53. J. Biol. Chem. 284, 4031–4039 (2009).
Meek, D. W. & Hupp, T. R. The regulation of MDM2 by multisite phosphorylation-opportunities for molecular-based intervention to target tumours? Semin. Cancer Biol. 20, 19–28 (2010).
Maya, R. et al. ATM-dependent phosphorylation of Mdm2 on serine 395: role in p53 activation by DNA damage. Genes Dev. 15, 1067–1077 (2001).
Chen, L., Gilkes, D. M., Pan, Y., Lane, W. S. & Chen, J. ATM and Chk2-dependent phosphorylation of MDMX contribute to p53 activation after DNA damage. EMBO J. 24, 3411–3422 (2005). This paper revealed the correlation between phosphorylation and degradation of MDMX and activation of p53.
Pereg, Y. et al. Differential roles of ATM- and Chk2-mediated phosphorylations of Hdmx in response to DNA damage. Mol. Cell. Biol. 26, 6819–6831 (2006).
Cheng, Q., Chen, L., Li, Z., Lane, W. S. & Chen, J. ATM activates p53 by regulating MDM2 oligomerization and E3 processivity. EMBO J. 28, 3857–3867 (2009).
Jackson, J. G., Post, S. M. & Lozano, G. Regulation of tissue- and stimulus-specific cell fate decisions by p53 in vivo. J. Pathol. 223, 127–136 (2011).
Lu, X., Nguyen, T. A., Zhang, X. & Donehower, L. A. The Wip1 phosphatase and Mdm2: cracking the “Wip” on p53 stability. Cell Cycle 7, 164–168 (2008).
Zhang, X. et al. Phosphorylation and degradation of MdmX is inhibited by Wip1 phosphatase in the DNA damage response. Cancer Res. 69, 7960–7968 (2009).
Winter, M. et al. Protein kinase CK1δ phosphorylates key sites in the acidic domain of murine double-minute clone 2 protein (MDM2) that regulate p53 turnover. Biochemistry 43, 16356–16364 (2004).
Wang, Y. V., Leblanc, M., Wade, M., Jochemsen, A. G. & Wahl, G. M. Increased radioresistance and accelerated B cell lymphomas in mice with Mdmx mutations that prevent modifications by DNA-damage-activated kinases. Cancer Cell 16, 33–43 (2009). The first report that MDMX phosphorylation is crucial for the response to ionizing radiation in vivo and that MDMX cooperates with another oncogene, MYC , to accelerate cancer.
Gannon, H. S., Woda, B. A. & Jones, S. N. ATM phosphorylation of Mdm2 Ser394 regulates the amplitude and duration of the DNA damage response in mice. Cancer Cell 21, 668–679 (2012).
Lin, C. P., Choi, Y. J., Hicks, G. G. & He, L. The emerging functions of the p53-miRNA network in stem cell biology. Cell Cycle 11, 2063–2072 (2012).
Lujambio, A. & Lowe, S. W. The microcosmos of cancer. Nature 482, 347–355 (2012).
Pichiorri, F. et al. Downregulation of p53-inducible microRNAs 192, 194, and 215 impairs the p53/MDM2 autoregulatory loop in multiple myeloma development. Cancer Cell 18, 367–381 (2010).
Markey, M. & Berberich, S. J. Full-length hdmX transcripts decrease following genotoxic stress. Oncogene 27, 6657–6666 (2008).
Mandke, P. et al. MicroRNA-34a modulates MDM4 expression via a target site in the open reading frame. PLoS ONE 7, e42034 (2012).
Wynendaele, J. et al. An illegitimate microRNA target site within the 3′ UTR of MDM4 affects ovarian cancer progression and chemosensitivity. Cancer Res. 70, 9641–9649 (2010).
Concepcion, C. P. et al. Intact p53-dependent responses in miR-34-deficient mice. PLoS Genet. 8, e1002797 (2012). This article forces a re-evaluation of the contexts in which miR-34 contributes to the p53 response in vivo.
Forte, E. et al. The Epstein-Barr virus (EBV)-induced tumor suppressor microRNA MiR-34a is growth promoting in EBV-infected B cells. J. Virol. 86, 6889–6898 (2012).
Forte, E. & Luftig, M. A. MDM2-dependent inhibition of p53 is required for Epstein-Barr virus B-cell growth transformation and infected-cell survival. J. Virol. 83, 2491–2499 (2009).
Lee, H. R. et al. Kaposi's sarcoma-associated herpesvirus viral interferon regulatory factor 4 targets MDM2 to deregulate the p53 tumor suppressor pathway. J. Virol. 83, 6739–6747 (2009).
Wang, P. et al. Elevated Mdm2 expression induces chromosomal instability and confers a survival and growth advantage to B cells. Oncogene 27, 1590–1598 (2008).
Dharel, N. et al. MDM2 promoter SNP309 is associated with the risk of hepatocellular carcinoma in patients with chronic hepatitis C. Clin. Cancer Res. 12, 4867–4871 (2006).
Sarek, G. et al. Reactivation of the p53 pathway as a treatment modality for KSHV-induced lymphomas. J. Clin. Invest. 117, 1019–1028 (2007).
Marine, J. C. & Lozano, G. Mdm2-mediated ubiquitylation: p53 and beyond. Cell Death Differ. 17, 93–102 (2010).
Fakharzadeh, S. S., Trusko, S. P. & George, D. L. Tumorigenic potential associated with enhanced expression of a gene that is amplified in a mouse tumor cell line. EMBO J. 10, 1565–1569 (1991).
Danovi, D. et al. Amplification of Mdmx (or Mdm4) directly contributes to tumor formation by inhibiting p53 tumor suppressor activity. Mol. Cell. Biol. 24, 5835–5843 (2004).
Lenos, K. et al. Oncogenic functions of hMDMX in in vitro transformation of primary human fibroblasts and embryonic retinoblasts. Mol. Cancer 10, 111 (2011).
Lundgren, K. et al. Targeted expression of MDM2 uncouples S phase from mitosis and inhibits mammary gland development independent of p53. Genes Dev. 11, 714–725 (1997).
Jones, S. N., Hancock, A. R., Vogel, H., Donehower, L. A. & Bradley, A. Overexpression of Mdm2 in mice reveals a p53-independent role for Mdm2 in tumorigenesis. Proc. Natl Acad. Sci. USA 95, 15608–15612 (1998).
Post, S. M. et al. A high-frequency regulatory polymorphism in the p53 pathway accelerates tumor development. Cancer Cell 18, 220–230 (2010). In vivo evidence that an MDM2 SNP is associated with increased cancer incidence in women is causally associated with tumorigenesis.
Xiong, S. et al. Spontaneous tumorigenesis in mice overexpressing the p53-negative regulator Mdm4. Cancer Res. 70, 7148–7154 (2010).
Park, S. S. et al. Insertion of Myc into Igh accelerates peritoneal plasmacytomas in mice. Cancer Res. 65, 7644–7652 (2005).
Miller, K. R., Kelley, K., Tuttle, R. & Berberich, S. J. HdmX overexpression inhibits oncogene induced cellular senescence. Cell Cycle 9, 3376–3382 (2010).
Hsu, B. et al. Evidence that c-myc mediated apoptosis does not require wild-type p53 during lymphomagenesis. Oncogene 11, 175–179 (1995).
Catalogue of Somatic Mutations in Cancer. Wellcome Trust Sanger Institute [online]
Slack, A., Lozano, G. & Shohet, J. M. MDM2 as MYCN transcriptional target: implications for neuroblastoma pathogenesis. Cancer Lett. 228, 21–27 (2005).
Mayo, L. D., Dixon, J. E., Durden, D. L., Tonks, N. K. & Donner, D. B. PTEN protects p53 from Mdm2 and sensitizes cancer cells to chemotherapy. J. Biol. Chem. 277, 5484–5489 (2002).
Cipriano, R., Patton, J. T., Mayo, L. D. & Jackson, M. W. Inactivation of p53 signaling by p73 or PTEN ablation results in a transformed phenotype that remains susceptible to Nutlin-3 mediated apoptosis. Cell Cycle 9, 1373–1379 (2010).
Tabe, Y. et al. MDM2 antagonist nutlin-3 displays antiproliferative and proapoptotic activity in mantle cell lymphoma. Clin. Cancer Res. 15, 933–942 (2009).
Brown, C. J., Lain, S., Verma, C. S., Fersht, A. R. & Lane, D. P. Awakening guardian angels: drugging the p53 pathway. Nature Rev. Cancer 9, 862–873 (2009).
Martins, C. P., Brown-Swigart, L. & Evan, G. I. Modeling the therapeutic efficacy of p53 restoration in tumors. Cell 127, 1323–1334 (2006).
Ventura, A. et al. Restoration of p53 function leads to tumour regression in vivo. Nature 445, 661–665 (2007).
Xue, W. et al. Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature 445, 656–660 (2007).
Ringshausen, I., O'Shea, C. C., Finch, A. J., Swigart, L. B. & Evan, G. I. Mdm2 is critically and continuously required to suppress lethal p53 activity in vivo. Cancer Cell 10, 501–514 (2006).
Bondar, T. & Medzhitov, R. p53-mediated hematopoietic stem and progenitor cell competition. Cell Stem Cell 6, 309–322 (2010).
Wang, Y. V. et al. Fine-tuning p53 activity through C-terminal modification significantly contributes to HSC homeostasis and mouse radiosensitivity. Genes Dev. 25, 1426–1438 (2011).
Tovar, C. et al. Small-molecule MDM2 antagonists reveal aberrant p53 signaling in cancer: implications for therapy. Proc. Natl Acad. Sci. USA 103, 1888–1893 (2006).
Garcia, D. et al. Validation of MdmX as a therapeutic target for reactivating p53 in tumors. Genes Dev. 25, 1746–1757 (2011).
Lane, D. P., Cheok, C. F. & Lain, S. p53-based cancer therapy. Cold Spring Harb. Perspect. Biol. 2, a001222 (2010).
Kole, R., Krainer, A. R. & Altman, S. RNA therapeutics: beyond RNA interference and antisense oligonucleotides. Nature Rev. Drug Discov. 11, 125–140 (2012).
Wang, H., Ma, X., Ren, S., Buolamwini, J. K. & Yan, C. A small-molecule inhibitor of MDMX activates p53 and induces apoptosis. Mol. Cancer Ther. 10, 69–79 (2011).
Kapitzky, L. et al. Cross-species chemogenomic profiling reveals evolutionarily conserved drug mode of action. Mol. Syst. Biol. 6, 451 (2010).
Pereg, Y. et al. Phosphorylation of Hdmx mediates its Hdm2- and ATM-dependent degradation in response to DNA damage. Proc. Natl Acad. Sci. USA 102, 5056–5061 (2005).
Whitesell, L. & Lindquist, S. L. HSP90 and the chaperoning of cancer. Nature Rev. Cancer 5, 761–772 (2005).
Vaseva, A. V., Yallowitz, A. R., Marchenko, N. D., Xu, S. & Moll, U. M. Blockade of Hsp90 by 17AAG antagonizes MDMX and synergizes with Nutlin to induce p53-mediated apoptosis in solid tumors. Cell Death Dis. 2, e156 (2011).
Wang, X. et al. Three-dimensional reconstruction of protein networks provides insight into human genetic disease. Nature Biotechnol. 30, 159–164 (2012).
Yildirim, M. A., Goh, K. I., Cusick, M. E., Barabasi, A. L. & Vidal, M. Drug-target network. Nature Biotechnol. 25, 1119–1126 (2007).
Wells, J. A. & McClendon, C. L. Reaching for high-hanging fruit in drug discovery at protein-protein interfaces. Nature 450, 1001–1009 (2007).
Vassilev, L. T. et al. In vivo activation of the p53 pathway by small-molecule antagonists of MDM2. Science 303, 844–848 (2004). This paper describes the first small-molecule MDM2 antagonist that inhibits the p53–MDM2 interaction and activates p53 in a non-genotoxic manner.
Ding, K. et al. Structure-based design of spiro-oxindoles as potent, specific small-molecule inhibitors of the MDM2-p53 interaction. J. Med. Chem. 49, 3432–3435 (2006).
Ray-Coquard, I. et al. Effect of the MDM2 antagonist RG7112 on the P53 pathway in patients with MDM2-amplified, well-differentiated or dedifferentiated liposarcoma: an exploratory proof-of-mechanism study. Lancet Oncol. 13, 1133–1140 (2012).
Patton, J. T. et al. Levels of HdmX expression dictate the sensitivity of normal and transformed cells to Nutlin-3. Cancer Res. 66, 3169–3176 (2006).
Wade, M., Rodewald, L. W., Espinosa, J. M. & Wahl, G. M. BH3 activation blocks Hdmx suppression of apoptosis and cooperates with Nutlin to induce cell death. Cell Cycle 7, 1973–1982 (2008).
Hu, B., Gilkes, D. M., Farooqi, B., Sebti, S. M. & Chen, J. MDMX overexpression prevents p53 activation by the MDM2 inhibitor Nutlin. J. Biol. Chem. 281, 33030–33035 (2006).
Wade, M., Wong, E. T., Tang, M., Stommel, J. M. & Wahl, G. M. Hdmx modulates the outcome of p53 activation in human tumor cells. J. Biol. Chem. 281, 33036–33044 (2006).
Graves, B. et al. Activation of the p53 pathway by small-molecule-induced MDM2 and MDMX dimerization. Proc. Natl Acad. Sci. USA 109, 11788–11793 (2012).
Brennan, R. C. et al. Targeting the p53 pathway in retinoblastoma with subconjunctival Nutlin-3a. Cancer Res. 71, 4205–4213 (2011).
Reed, D. et al. Identification and characterization of the first small molecule inhibitor of MDMX. J. Biol. Chem. 285, 10786–10796 (2010).
Bista, M. et al. On the mechanism of action of SJ-172550 in inhibiting the interaction of MDM4 and p53. PLoS ONE 7, e37518 (2012).
Montes de Oca Luna, R., Wagner, D. S. & Lozano, G. Rescue of early embryonic lethality in mdm2-deficient mice by deletion of p53. Nature 378, 203–206 (1995).
Parant, J. et al. Rescue of embryonic lethality in Mdm4-null mice by loss of Trp53 suggests a nonoverlapping pathway with MDM2 to regulate p53. Nature Genet. 29, 92–95 (2001). References 107 and 108 demonstrate that deletions of Mdm2 and Mdmx cause embryonic lethality that can be rescued by Trp53 deletion, thus providing genetic evidence that MDM2 and MDM4 are crucial p53 repressors with non-overlapping functions.
Xiong, S., Van Pelt, C. S., Elizondo-Fraire, A. C., Liu, G. & Lozano, G. Synergistic roles of Mdm2 and Mdm4 for p53 inhibition in central nervous system development. Proc. Natl Acad. Sci. USA 103, 3226–3231 (2006).
Bernal, F. et al. A stapled p53 helix overcomes HDMX-mediated suppression of p53. Cancer Cell 18, 411–422 (2010).
Hu, B., Gilkes, D. M. & Chen, J. Efficient p53 activation and apoptosis by simultaneous disruption of binding to MDM2 and MDMX. Cancer Res. 67, 8810–8817 (2007).
Pazgier, M. et al. Structural basis for high-affinity peptide inhibition of p53 interactions with MDM2 and MDMX. Proc. Natl Acad. Sci. USA 106, 4665–4670 (2009).
Verdine, G. L. & Hilinski, G. J. Stapled peptides for intracellular drug targets. Methods Enzymol. 503, 3–33 (2012).
Aranovich, A. et al. Differences in the mechanisms of proapoptotic BH3 proteins binding to Bcl-XL and Bcl-2 quantified in live MCF-7 cells. Mol. Cell 45, 754–763 (2012).
Yang, Y. et al. Small molecule inhibitors of HDM2 ubiquitin ligase activity stabilize and activate p53 in cells. Cancer Cell 7, 547–559 (2005).
Roxburgh, P. et al. Small molecules that bind the Mdm2 RING stabilize and activate p53. Carcinogenesis 33, 791–798 (2012).
Herman, A. G. et al. Discovery of Mdm2-MdmX E3 ligase inhibitors using a cell-based ubiquitination assay. Cancer Discov. 1, 312–325 (2011).
Shloush, J. et al. Structural and functional comparison of the RING domains of two p53 E3 ligases, Mdm2 and Pirh2. J. Biol. Chem. 286, 4796–4808 (2011).
Saville, M. K. et al. Regulation of p53 by the ubiquitin-conjugating enzymes UbcH5B/C in vivo. J. Biol. Chem. 279, 42169–42181 (2004).
Ye, Y. & Rape, M. Building ubiquitin chains: E2 enzymes at work. Nature Rev. Mol. Cell Biol. 10, 755–764 (2009).
Deshaies, R. J. & Joazeiro, C. A. RING domain E3 ubiquitin ligases. Annu. Rev. Biochem. 78, 399–434 (2009).
Cohen, P. & Tcherpakov, M. Will the ubiquitin system furnish as many drug targets as protein kinases? Cell 143, 686–693 (2010).
Al-Lazikani, B., Banerji, U. & Workman, P. Combinatorial drug therapy for cancer in the post-genomic era. Nature Biotechnol. 30, 679–692 (2012).
Wade, M. & Wahl, G. M. Targeting Mdm2 and Mdmx in cancer therapy: better living through medicinal chemistry? Mol. Cancer Res. 7, 1–11 (2009).
Feng, J. et al. Stabilization of Mdm2 via decreased ubiquitination is mediated by protein kinase B/Akt-dependent phosphorylation. J. Biol. Chem. 279, 35510–35517 (2004).
Jackson, M. W. et al. Hdm2 nuclear export, regulated by insulin-like growth factor-I/MAPK/p90Rsk signaling, mediates the transformation of human cells. J. Biol. Chem. 281, 16814–16820 (2006).
Zhu, N., Gu, L., Li, F. & Zhou, M. Inhibition of the Akt/survivin pathway synergizes the antileukemia effect of nutlin-3 in acute lymphoblastic leukemia cells. Mol. Cancer Ther. 7, 1101–1109 (2008).
Zhang, W. et al. Blockade of mitogen-activated protein kinase/extracellular signal-regulated kinase kinase and murine double minute synergistically induces Apoptosis in acute myeloid leukemia via BH3-only proteins Puma and Bim. Cancer Res. 70, 2424–2434 (2010).
Barretina, J. et al. The Cancer Cell Line Encyclopedia enables predictive modelling of anticancer drug sensitivity. Nature 483, 603–607 (2012).
Garnett, M. J. et al. Systematic identification of genomic markers of drug sensitivity in cancer cells. Nature 483, 570–575 (2012).
Sullivan, K. D. et al. ATM and MET kinases are synthetic lethal with nongenotoxic activation of p53. Nature Chem. Biol. 8, 646–654 (2012).
Moumen, A., Patane, S., Porras, A., Dono, R. & Maina, F. Met acts on Mdm2 via mTOR to signal cell survival during development. Development 134, 1443–1451 (2007).
Cheok, C. F., Verma, C. S., Baselga, J. & Lane, D. P. Translating p53 into the clinic. Nature Rev. Clin. Oncol. 8, 25–37 (2011).
Bouska, A. & Eischen, C. M. Murine double minute 2: p53-independent roads lead to genome instability or death. Trends Biochem. Sci. 34, 279–286 (2009).
Wade, M., Wang, Y. V. & Wahl, G. M. The p53 orchestra: Mdm2 and Mdmx set the tone. Trends Cell Biol. 20, 299–309 (2010).
Chymkowitch, P., Le May, N., Charneau, P., Compe, E. & Egly, J. M. The phosphorylation of the androgen receptor by TFIIH directs the ubiquitin/proteasome process. EMBO J. 30, 468–479 (2011).
Kim, K. et al. MDM2 regulates estrogen receptor α and estrogen responsiveness in breast cancer cells. J. Mol. Endocrinol. 46, 67–79 (2011).
Lin, H. K., Wang, L., Hu, Y. C., Altuwaijri, S. & Chang, C. Phosphorylation-dependent ubiquitylation and degradation of androgen receptor by Akt require Mdm2 E3 ligase. EMBO J. 21, 4037–4048 (2002).
Linn, D. E. et al. Differential regulation of androgen receptor by PIM-1 kinases via phosphorylation-dependent recruitment of distinct ubiquitin E3 ligases. J. Biol. Chem. 287, 22959–22968 (2012).
Sanchez, M., Picard, N., Sauve, K. & Tremblay, A. Coordinate regulation of estrogen receptor β degradation by Mdm2 and CREB-binding protein in response to growth signals. Oncogene 20 Feb 2012 (doi:10.1038/onc.2012.19).
Saji, S. et al. MDM2 enhances the function of estrogen receptor α in human breast cancer cells. Biochem. Biophys. Res. Commun. 281, 259–265 (2001).
Tang, Y. A. et al. MDM2 overexpression deregulates the transcriptional control of RB/E2F leading to DNA methyltransferase 3A overexpression in lung cancer. Clin. Cancer Res. 18, 4325–4333 (2012).
Chen, L. et al. MDM2 recruitment of lysine methyltransferases regulates p53 transcriptional output. EMBO J. 29, 2538–2552 (2010).
Mungamuri, S. K. et al. p53-mediated heterochromatin reorganization regulates its cell fate decisions. Nature Struct. Mol. Biol. 19, 478–484 (2012).
Onder, T. T. et al. Chromatin-modifying enzymes as modulators of reprogramming. Nature 483, 598–602 (2012).
Peters, A. H. et al. Loss of the Suv39h histone methyltransferases impairs mammalian heterochromatin and genome stability. Cell 107, 323–337 (2001).
Bouska, A., Lushnikova, T., Plaza, S. & Eischen, C. M. Mdm2 promotes genetic instability and transformation independent of p53. Mol. Cell. Biol. 28, 4862–4874 (2008).
Kadakia, M., Brown, T. L., McGorry, M. M. & Berberich, S. J. MdmX inhibits Smad transactivation. Oncogene 21, 8776–8785 (2002).
Wunderlich, M., Ghosh, M., Weghorst, K. & Berberich, S. J. MdmX represses E2F1 transactivation. Cell Cycle 3, 472–478 (2004).
Matijasevic, Z., Krzywicka-Racka, A., Sluder, G. & Jones, S. N. MdmX regulates transformation and chromosomal stability in p53-deficient cells. Cell Cycle 7, 2967–2973 (2008).
Busuttil, V. et al. NF-κB inhibits T-cell activation-induced, p73-dependent cell death by induction of MDM2. Proc. Natl Acad. Sci. USA 107, 18061–18066 (2010).
Chen, J. et al. A restricted cell population propagates glioblastoma growth after chemotherapy. Nature 488, 522–526 (2012).
Rao, S. K., Edwards, J., Joshi, A. D., Siu, I. M. & Riggins, G. J. A survey of glioblastoma genomic amplifications and deletions. J. Neurooncol 96, 169–179 (2010).
Minamino, T. et al. A crucial role for adipose tissue p53 in the regulation of insulin resistance. Nature Med. 15, 1082–1087 (2009).
Tomita, K. et al. p53/p66Shc-mediated signaling contributes to the progression of non-alcoholic steatohepatitis in humans and mice. J. Hepatol 57, 837–843 (2012).
Hallenborg, P. et al. Mdm2 controls CREB-dependent transactivation and initiation of adipocyte differentiation. Cell Death Differ. 19, 1381–1389 (2012).
Crago, A. M. & Singer, S. Clinical and molecular approaches to well differentiated and dedifferentiated liposarcoma. Curr. Opin. Oncol. 23, 373–378 (2011).
Issaeva, N. et al. Small molecule RITA binds to p53, blocks p53-HDM-2 interaction and activates p53 function in tumors. Nature Med. 10, 1321–1328 (2004).
Nieves-Neira, W. et al. DNA protein cross-links produced by NSC 652287, a novel thiophene derivative active against human renal cancer cells. Mol. Pharmacol. 56, 478–484 (1999).
Rivera, M. I. et al. Selective toxicity of the tricyclic thiophene NSC 652287 in renal carcinoma cell lines: differential accumulation and metabolism. Biochem. Pharmacol. 57, 1283–1295 (1999).
Di Conza, G. et al. IGF-1R/MDM2 relationship confers enhanced sensitivity to RITA in Ewing sarcoma cells. Mol. Cancer Ther. 11, 1247–1256 (2012).
Kojima, K., Burks, J. K., Arts, J. & Andreeff, M. The novel tryptamine derivative JNJ-26854165 induces wild-type p53- and E2F1-mediated apoptosis in acute myeloid and lymphoid leukemias. Mol. Cancer Ther. 9, 2545–2557 (2010).
Christophorou, M. A., Ringshausen, I., Finch, A. J., Swigart, L. B. & Evan, G. I. The pathological response to DNA damage does not contribute to p53-mediated tumour suppression. Nature 443, 214–217 (2006).
Valentin-Vega, Y. A., Okano, H. & Lozano, G. The intestinal epithelium compensates for p53-mediated cell death and guarantees organismal survival. Cell Death Differ. 15, 1772–1781 (2008).
Valentin-Vega, Y. A., Box, N., Terzian, T. & Lozano, G. Mdm4 loss in the intestinal epithelium leads to compartmentalized cell death but no tissue abnormalities. Differentiation 77, 442–449 (2009).
Boesten, L. S. et al. Mdm2, but not Mdm4, protects terminally differentiated smooth muscle cells from p53-mediated caspase-3-independent cell death. Cell Death Differ. 13, 2089–2098 (2006).
Maetens, M. et al. Distinct roles of Mdm2 and Mdm4 in red cell production. Blood 109, 2630–2633 (2007).
Grier, J. D., Xiong, S., Elizondo-Fraire, A. C., Parant, J. M. & Lozano, G. Tissue-specific differences of p53 inhibition by Mdm2 and Mdm4. Mol. Cell. Biol. 26, 192–198 (2006).
Xiong, S., Van Pelt, C. S., Elizondo-Fraire, A. C., Fernandez-Garcia, B. & Lozano, G. Loss of Mdm4 results in p53-dependent dilated cardiomyopathy. Circulation 115, 2925–2930 (2007).
Macias, E. et al. An ARF-independent c-MYC-activated tumor suppression pathway mediated by ribosomal protein-Mdm2 Interaction. Cancer Cell 18, 231–243 (2010).
Itahana, K. et al. Targeted inactivation of Mdm2 RING finger E3 ubiquitin ligase activity in the mouse reveals mechanistic insights into p53 regulation. Cancer Cell 12, 355–366 (2007).
Cheng, X. et al. Activation of murine double minute 2 by Akt in mammary epithelium delays mammary involution and accelerates mammary tumorigenesis. Cancer Res. 70, 7684–7689 (2010).
Popowicz, G. M. et al. Structures of low molecular weight inhibitors bound to MDMX and MDM2 reveal new approaches for p53-MDMX/MDM2 antagonist drug discovery. Cell Cycle 9, 1104–1111 (2010).
Rew, Y. et al. Structure-based design of novel inhibitors of the MDM2-p53 interaction. J. Med. Chem. 55, 4936–4954 (2012).
Liu, M. et al. D-peptide inhibitors of the p53-MDM2 interaction for targeted molecular therapy of malignant neoplasms. Proc. Natl Acad. Sci. USA 107, 14321–14326 (2010).
Acknowledgements
Studies relevant to the topics discussed here were supported by grants from the US National Institutes of Health (R01-CA61449 and R03-MH089489-01), Cancer Center Support Grant CA014195 and a sanofi-aventis sponsored research grant awarded to G.M.W.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
DATABASES
National Cancer Institute Drug Dictionary
NCI Drug Dictionary
FURTHER INFORMATION
Glossary
- Antagonists
-
Chemical substances that interfere with or inhibit the physiological activity of other biological entities such as proteins or enzymes.
- miRNAs
-
Derived from an RNA polymerase II transcribed precursor, miRNAs are a class of non-protein coding mRNA that reduces the expression of cellular proteins through various mechanisms.
- Hemizygous
-
A genetic status in which one allelic copy of a gene is deleted or otherwise inactivated.
- Haploinsufficiency
-
A genetic status in which a single wild-type copy of an allelic pair is present, but the level of expression of the product is insufficient to give wild-type function.
- Myeloablation
-
The depletion of bone marrow cells.
- Neoadjuvant therapy
-
Administration of therapeutic agents to reduce tumour volume before giving a primary treatment such as surgery.
- IC50
-
The half-maximal inhibitory concentration, which is the concentration of a compound causing 50% inhibition of biological or biochemical function.
Rights and permissions
About this article
Cite this article
Wade, M., Li, YC. & Wahl, G. MDM2, MDMX and p53 in oncogenesis and cancer therapy. Nat Rev Cancer 13, 83–96 (2013). https://doi.org/10.1038/nrc3430
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc3430
This article is cited by
-
Pharmacological reactivation of p53 in the era of precision anticancer medicine
Nature Reviews Clinical Oncology (2024)
-
Research progress on antitumor mechanisms and molecular targets of Inula sesquiterpene lactones
Chinese Medicine (2023)
-
Advances in blood–brain barrier-crossing nanomedicine for anti-glioma
Cancer Nanotechnology (2023)
-
Unmasking the biological function and regulatory mechanism of NOC2L: a novel inhibitor of histone acetyltransferase
Journal of Translational Medicine (2023)
-
Precise pancreatic cancer therapy through targeted degradation of mutant p53 protein by cerium oxide nanoparticles
Journal of Nanobiotechnology (2023)