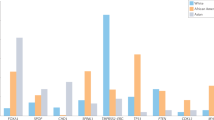
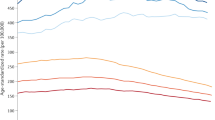
Abstract
It is becoming clear that some of the differences in cancer risk, incidence and survival among people of different racial and ethnic backgrounds can be attributed to biological factors. However, identifying these factors and exploiting them to help eliminate cancer disparities has proved challenging. With this in mind, we asked four scientists for their opinions on the most crucial advances, as well as the challenges and what the future holds for this important emerging area of research.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kolonel, L. N., Altshuler, D. & Henderson, B. E. The multiethnic cohort study: exploring genes, lifestyle and cancer risk. Nature Rev. Cancer 4, 519–527 (2004).
Kolonel, L. N. et al. A multiethnic cohort in Hawaii and Los Angeles: baseline characteristics. Am. J. Epidemiol. 151, 346–357 (2000).
Setiawan, V. W. et al. Breast cancer risk factors defined by estrogen and progesterone receptor status: the multiethnic cohort study. Am. J. Epidemiol. 169, 1251–1259 (2009).
Haiman, C. A. et al. Ethnic and racial differences in the smoking-related risk of lung cancer. N. Engl. J. Med. 354, 333–342 (2006).
Amundadottir, L. T. et al. A common variant associated with prostate cancer in European and African populations. Nature Genet. 38, 652–658 (2006).
Freedman, M. L. et al. Admixture mapping identifies 8q24 as a prostate cancer risk locus in African-American men. Proc. Natl Acad. Sci. USA 103, 14068–14073 (2006).
Haiman, C. A. et al. Multiple regions within 8q24 independently affect risk for prostate cancer. Nature Genet. 39, 638–644 (2007).
Haiman, C. A. et al. Genome-wide association study of prostate cancer in men of African ancestry identifies a susceptibility locus at 17q21. Nature Genet. 43, 570–573 (2011).
Haiman, C. A. et al. A common variant at the TERT-CLPTM1L locus is associated with estrogen receptor-negative breast cancer. Nature Genet. 43, 1210–1214 (2011).
Siegel, R., Naishadham, D. & Jemal, A. Cancer statistics, 2012. CA Cancer J. Clin. 62, 10–29 (2012).
Hoffman, R. M. et al. Racial and ethnic differences in advanced-stage prostate cancer: the Prostate Cancer Outcomes Study. J. Natl Cancer Inst. 93, 388–395 (2001).
Robbins, A. S., Whittemore, A. S. & Thom, D. H. Differences in socioeconomic status and survival among white and black men with prostate cancer. Am. J. Epidemiol. 151, 409–416 (2000).
Evans, S., Metcalfe, C., Ibrahim, F., Persad, R. & Ben-Shlomo, Y. Investigating Black-White differences in prostate cancer prognosis: a systematic review and meta-analysis. Int. J. Cancer 123, 430–435 (2008).
Devgan, S. A. et al. Genetic variation of 3 β-hydroxysteroid dehydrogenase type II in three racial/ethnic groups: implications for prostate cancer risk. Prostate 33, 9–12 (1997).
Guo, Y., Sigman, D. B., Borkowski, A. & Kyprianou, N. Racial differences in prostate cancer growth: apoptosis and cell proliferation in Caucasian and African-American patients. Prostate 42, 130–136 (2000).
Shuch, B. et al. Racial disparity of epidermal growth factor receptor expression in prostate cancer. J. Clin. Oncol. 22, 4725–4729 (2004).
Yeager, M. et al. Genome-wide association study of prostate cancer identifies a second risk locus at 8q24. Nature Genet. 39, 645–649 (2007).
Wallace, T. A. et al. Tumor immunobiological differences in prostate cancer between African-American and European-American men. Cancer Res. 68, 927–936 (2008).
Reams, R. R. et al. Microarray comparison of prostate tumor gene expression in African-American and Caucasian American males: a pilot project study. Infect. Agent Cancer 4, S3 (2009).
Eheman, C. et al. Annual Report to the Nation on the status of cancer, 1975–2008, featuring cancers associated with excess weight and lack of sufficient physical activity. Cancer 118, 2338–2366 (2012).
Dawood, S., et al. Trends in survival over the past two decades among white and black patients with newly diagnosed stage IV breast cancer. J. Clin. Oncol. 26, 4892–4898 (2008).
Sorlie, T. et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl Acad. Sci. USA 98, 10869–10874 (2001).
Sotiriou, C. et al. Breast cancer classification and prognosis based on gene expression profiles from a population-based study. Proc. Natl Acad. Sci. USA 100, 10393–10398 (2003).
Carey, L. A. et al. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA 295, 2492–2502 (2006).
Al-Hajj, M., Wicha, M. S., Benito-Hernandez, A., Morrison, S. J. & Clarke, M. F. Prospective identification of tumorigenic breast cancer cells. Proc. Natl Acad. Sci. USA 100, 3983–3988 (2003).
Nguyen, L. V., Vanner, R., Dirks, P. & Eaves, C. J. Cancer stem cells: an evolving concept. Nature Rev. Cancer 12, 133–143 (2012).
Dean, M., Fojo, T. & Bates, S. Tumor stem cells and drug resistance. Nature Rev. Cancer 5, 275–284 (2005).
Li, X. et al. Resistance of tumorigenic breast cancer cells to chemotherapy. J. Natl Cancer Inst. 100, 9672–9679 (2008).
Creighton, C. J. et al. Residual breast cancers after conventional therapy display mesenchymal as well as tumor-initiating features. Proc. Natl Acad. Sci. USA 106, 13820–13825 (2009).
Shimono, Y. et al. Downregulation of miRNA-200c links breast cancer stem cells with normal stem cells. Cell 138, 592–603 (2009).
Flegal, K. M. et al. Prevalence and trends in obesity among US adults, 1999–2008. JAMA 303, 235–241 (2010).
Grivennikov, S. I., Gretin, F. R. & Karin, M. Immunity, Inflammation, and Cancer. Cell 140, 883–899 (2010).
Liu, S. et al. Breast cancer stem cells are regulated by mesenchymal stem cells through cytokine networks. Cancer Res. 71, 614–624 (2011).
Luedi, P. P. et al. Computational and experimental identification of novel human imprinted genes. Genome Res. 17, 1723–1730 (2007).
Dolinoy, D. C., Huang, D. & Jirtle, R. L. Maternal nutrient supplementation counteracts bisphenol A-induced DNA hypomethylation in early development. Proc. Natl Acad. Sci. USA 104, 13056–13061 (2007).
Dolinoy, D. C., Weidman, J. R., Waterland, R. A. & Jirtle, R. L. Maternal genistein alters coat color and protects Avy mouse offspring from obesity by modifying the fetal epigenome. Environ. Health Perspect. 114, 567–572 (2006).
Jemal, A. et al. Global cancer statistics. CA Cancer J. Clin. 61, 69–90 (2011).
Parkin, D. M. The global health burden of infection-associated cancers in the year 2002. Int. J. Cancer 118, 3030–3044 (2006).
Hardy, J. & Singleton, A. Genomewide association studies and human disease. N. Engl. J. Med. 360, 1759–1768 (2009).
Chung, C. C. & Chanock, S. J. Current status of genome-wide association studies in cancer. Hum. Genet. 130, 59–78 (2011).
Hung, R. J. et al. A susceptibility locus for lung cancer maps to nicotinic acetylcholine receptor subunit genes on 15q25. Nature 452, 633–637 (2008).
Thorgeirsson, T. E. et al. A variant associated with nicotine dependence, lung cancer and peripheral arterial disease. Nature 452, 638–642 (2008).
Amos, C. I. et al. Genome-wide association scan of tag SNPs identifies a susceptibility locus for lung cancer at 15q25.1. Nature Genet. 40, 616–622 (2008).
Wu, C. et al. Genetic variants on chromosome 15q25 associated with lung cancer risk in Chinese populations. Cancer Res. 69, 5065–5072 (2009).
Miki, D. et al. Variation in TP63 is associated with lung adenocarcinoma susceptibility in Japanese and Korean populations. Nature Genet. 42, 893–896 (2010).
Cai, Q. et al. Replication and functional genomic analyses of the breast cancer susceptibility locus at 6q25.1 generalize its importance in women of chinese, Japanese, and European ancestry. Cancer Res. 71, 1344–1355 (2011).
Haiman, C. A. et al. Characterizing genetic risk at known prostate cancer susceptibility loci in African Americans. PLoS Genet. 7, e1001387 (2011).
Fu, J., Festen, E. A. & Wijmenga, C. Multi-ethnic studies in complex traits. Hum. Mol. Genet. 20, R206–R213 (2011).
Xu, J. et al. Prostate cancer risk associated loci in African Americans. Cancer Epidemiol. Biomarkers Prev. 18, 2145–2149 (2009).
Freedman, M. L. et al. Principles for the post-GWAS functional characterization of cancer risk loci. Nature Genet. 43, 513–518 (2011).
Altshuler, D. M. et al. Integrating common and rare genetic variation in diverse human populations. Nature 467, 52–58 (2010).
Gravel, S. et al. Demographic history and rare allele sharing among human populations. Proc. Natl Acad. Sci. USA 108, 11983–11988 (2011).
Zuk, O., Hechter, E., Sunyaev, S. R. & Lander, E. S. The mystery of missing heritability: genetic interactions create phantom heritability. Proc. Natl Acad. Sci. USA 109,1193–1198 (2012).
Royal, C. D. & Dunston, G. M. Changing the paradigm from 'race' to human genome variation. Nature Genet. 36, S5–S7 (2004).
Parra, E. J., Kittles, R. A. & Shriver, M. D. Implications of correlations between skin color and genetic ancestry for biomedical research. Nature Genet. 36, S54–S60 (2004).
Rosenberg, N. A. et al. Genome-wide association studies in diverse populations. Nature Rev. Genet. 11, 356–366 (2010).
Macilwain, C. Funding in 2011: East heats up as West cools down. Cell 144, 167–169 (2011).
Haiman, C. A. & Stram, D. O. Exploring genetic susceptibility to cancer in diverse populations. Curr. Opin. Genet. Dev. 20, 330–335 (2010).
Maher, B. Personal genomes: the case of the missing heritability. Nature 456, 18–21 (2008).
Manolio, T. A. et al. Finding the missing heritability of complex diseases. Nature 461, 747–753 (2009).
Eichler, E. E. et al. Missing heritability and strategies for finding the underlying causes of complex disease. Nature Rev. Genet. 11, 446–450 (2010).
Hughes, L. et al. Assessing the clinical role of genetic markers of early-onset prostate cancer among high-risk men enrolled in prostate cancer early detection. Cancer Epidemiol. Biomarkers Prev. 21, 53–60 (2012).
Lander, E. S. Initial impact of the sequencing of the human genome. Nature 470, 187–197 (2011).
Ding, L. et al. Somatic mutations affect key pathways in lung adenocarcinoma. Nature 455, 1069–1075 (2008).
The Cancer Genome Atlas Research Network. Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature 455, 1061–1068 (2008).
Parsons, D. W. et al. An integrated genomic analysis of human glioblastoma multiforme. Science 321, 1807–1812 (2008).
Berger, M. F. et al. The genomic complexity of primary human prostate cancer. Nature 470, 214–220 (2011).
Boise, L. H. et al. bcl-x, a bcl-2-related gene that functions as a dominant regulator of apoptotic cell death. Cell 74, 597–608 (1993).
Stephens, P. J. et al. Massive genomic rearrangement acquired in a single catastrophic event during cancer development. Cell 144, 27–40 (2011).
Levental, K. R. et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell 139, 891–906 (2009).
Zeggini, E. Next-generation association studies for complex traits. Nature Genet. 43, 287–288 (2011).
Baylin, S. B. & Jones, P. A. A decade of exploring the cancer epigenome - biological and translational implications. Nature Rev. Cancer 11, 726–734 (2011).
Acknowledgements
B.E.H. is supported by the US National Institutes of Health (NIH) grants CA148537, CA136792 and CA054281. N.H.L. is supported by the NIH grants CA120316 and DK056108. V.S. apologizes in advance to the authors of articles that were not cited owing to reference limitations. H.S. is funded by the National Key Basic Research Program Grant (2011CB503805) and the National Natural Science Foundation of China (30972541 and 30901233), NIH grant (U19 CA148127) and the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Rights and permissions
About this article
Cite this article
Henderson, B., Lee, N., Seewaldt, V. et al. The influence of race and ethnicity on the biology of cancer. Nat Rev Cancer 12, 648–653 (2012). https://doi.org/10.1038/nrc3341
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc3341
This article is cited by
-
Algorithmic fairness in artificial intelligence for medicine and healthcare
Nature Biomedical Engineering (2023)
-
DNMT3B −579 G>T Promoter Polymorphism and the Risk of Gastric Cancer in the West of Iran
Journal of Gastrointestinal Cancer (2018)
-
Effect of genetic ancestry to the risk of susceptibility to gastric cancer in a mixed population of the Brazilian Amazon
BMC Research Notes (2017)
-
Breast cancers from black women exhibit higher numbers of immunosuppressive macrophages with proliferative activity and of crown-like structures associated with lower survival compared to non-black Latinas and Caucasians
Breast Cancer Research and Treatment (2016)
-
Disparities in race/ethnicity and socioeconomic status: risk of mortality of breast cancer patients in the California Cancer Registry, 2000–2010
BMC Cancer (2013)