Key Points
-
Bone metastases are a common complication of cancer and are generally incurable. They cause considerable pain, pathological bone fractures and hypercalcaemia. Up to 50% of patients prescribed anti-resorptive drugs to treat bone metastases develop new bone metastases, skeletal complications and disease progression, emphasizing the need for new therapies.
-
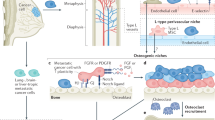
Tumour invasion into bone is associated with osteoclast and osteoblast recruitment. Osteoclasts secrete acid, collagenases and proteases that demineralize the bone matrix and degrade matricellular proteins. Macrophage colony stimulating factor and receptor activator of NF-κB ligand (RANKL) are important growth factors that support osteoclastogenesis, and they are primarily produced by osteoblasts. Osteoprotegerin is an endogenous decoy receptor of RANKL that inhibits osteoclastogenesis.
-
Bone marrow mesenchymal stem cells are directed along the osteoblast lineage through local factors, such as transforming growth factor-β (TGFβ), bone morphogenetic proteins (BMPs) and WNT proteins. These pathways lead to the expression of three key transcriptional regulators of osteoblast function, including RUNX2. The osteoblast-stimulating activity of metastatic tumour cells is thought to be due to the ability of these cells to express many of the factors that can drive osteoblast formation.
-
Osteoblasts and bone marrow stromal cells may attract metastatic tumour cells to bone and provide a niche through protein interactions that include integrins, such as α4β1–vascular cell adhesion molecule 1; chemokines, such as CXCL12–CXCR4; BMPs; Notch; nestin; and osteopontin. These mechanisms are similar to the physiological recruitment of haematopoietic stem cells.
-
The invasion and growth of metastatic tumour cells in the bone involves the modulation of a large number of genes and proteins that include matrix metalloproteinases, parathyroid hormone-related protein, TGFβ, interleukin-6, Jagged 1–Notch, GLI2, RUNX2, hypoxia-induced growth factor 1α, calcium and the calcium-sensing receptor.
-
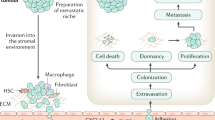
Beyond the effects on osteoclasts and osteoblasts, tumours in the bone microenvironment recruit and modulate the function of platelets, myeloid cells, immune cells and nerve cells, and induce the formation of new blood vessels. These changes all help to ensure the growth and survival of metastatic tumour cells in bone and represent important therapeutic targets.
-
Drugs, such as bisphosphonates or RANKL antibodies, that target osteoclastogenesis decrease the incidence of skeletal complications and are the current standard of care for patients with bone metastases. These anti-resorptive agents might also have direct antitumour effects.
-
Advances in our understanding of the basic biology of bone remodelling, biomechanics and haematopoiesis, coupled with the advances in cancer genetics and tumour imaging should yield new therapeutic targets and insights into cancer metastasis in bone.
Abstract
When cancer metastasizes to bone, considerable pain and deregulated bone remodelling occurs, greatly diminishing the possibility of cure. Metastasizing tumour cells mobilize and sculpt the bone microenvironment to enhance tumour growth and to promote bone invasion. Understanding the crucial components of the bone microenvironment that influence tumour localization, along with the tumour-derived factors that modulate cellular and protein matrix components of bone to favour tumour expansion and invasion, is central to the pathophysiology of bone metastases. Basic findings of tumour–bone interactions have uncovered numerous therapeutic opportunities that focus on the bone microenvironment to prevent and treat bone metastases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Roodman, G. D. Mechanisms of bone metastasis. N. Engl. J. Med. 350, 1655–1664 (2004).
Coleman, R. E. Metastatic bone disease: clinical features, pathophysiology and treatment strategies. Cancer Treat. Rev. 27, 165–176 (2001).
Mundy, G. R. Metastasis to bone: causes, consequences and therapeutic opportunities. Nature Rev. Cancer 2, 584–593 (2002).
Fukutomi, M. et al. Increased incidence of bone metastases in hepatocellular carcinoma. Eur. J. Gastroenterol. Hepatol. 13, 1083–1088 (2001).
Guise, T. A. et al. Basic mechanisms responsible for osteolytic and osteoblastic bone metastases. Clin. Cancer Res. 12, 6213s–6216s (2006).
Kingsley, L. A., Fournier, P. G., Chirgwin, J. M. & Guise, T. A. Molecular biology of bone metastasis. Mol. Cancer Ther. 6, 2609–2617 (2007).
Lipton, A. et al. Normalization of bone markers is associated with improved survival in patients with bone metastases from solid tumors and elevated bone resorption receiving zoledronic acid. Cancer 113, 193–201 (2008).
Aft, R. et al. Effect of zoledronic acid on disseminated tumour cells in women with locally advanced breast cancer: an open label, randomised, phase 2 trial. Lancet Oncol. 11, 421–428 (2010).
Pantel, K. et al. Detection and clinical implications of early systemic tumor cell dissemination in breast cancer. Clin. Cancer Res. 9, 6326–6334 (2003).
Braun, S. et al. Lack of effect of adjuvant chemotherapy on the elimination of single dormant tumor cells in bone marrow of high-risk breast cancer patients. J. Clin. Oncol. 18, 80–86 (2000).
Hirbe, A., Morgan, E. A., Uluçkan, Ö . & Weilbaecher, K. Skeletal complications of breast cancer therapies. Clin. Cancer Res. 12, 6309s–6314s (2006).
Body, J. J. et al. Effects of denosumab in patients with bone metastases, with and without previous bisphosphonate exposure. J. Bone Miner. Res. 25, 440–446 (2009).
Fizazi, K. et al. Randomized phase II trial of denosumab in patients with bone metastases from prostate cancer, breast cancer, or other neoplasms after intravenous bisphosphonates. J. Clin. Oncol. 27, 1564–1571 (2009).
Guise, T. A. et al. Evidence for a causal role of parathyroid hormone-related protein in the pathogenesis of human breast cancer-mediated osteolysis. J. Clin. Invest. 98, 1544–1549 (1996).
Teitelbaum, S. L. & Ross, F. P. Genetic regulation of osteoclast development and function. Nature Rev. Genet. 4, 638–649 (2003).
Dougall, W. C. et al. RANK is essential for osteoclast and lymph node development. Genes Dev. 13, 2412–2424 (1999).
Suda, T. et al. Modulation of osteoclast differentiation and function by the new members of the tumor necrosis factor receptor and ligand families. Endocr. Rev. 20, 345–357 (1999).
Tang, Y. et al. TGF-β1-induced migration of bone mesenchymal stem cells couples bone resorption with formation. Nature Med. 15, 757–765 (2009).
Komori, T. et al. Targeted disruption of Cbfa1 results in a complete lack of bone formation owing to maturational arrest of osteoblasts. Cell 89, 755–764 (1997).
Nakashima, K. et al. The novel zinc finger-containing transcription factor osterix is required for osteoblast differentiation and bone formation. Cell 108, 17–29 (2002).
Yang, X. et al. ATF4 is a substrate of RSK2 and an essential regulator of osteoblast biology; implication for Coffin-Lowry Syndrome. J. Biol. Chem. 279, 47109–47114 (2004).
Xiao, G. et al. Cooperative interactions between activating transcription factor 4 and Runx2/Cbfa1 stimulate osteoblast-specific osteocalcin gene expression. J. Biol. Chem. 280, 30689–30696 (2005).
Kaplan, R. N. et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438, 820–827 (2005).
Hiratsuka, S. et al. The S100A8-serum amyloid A3-TLR4 paracrine cascade establishes a pre-metastatic phase. Nature Cell Biol. 10, 1349–1355 (2008).
Purhonen, S. et al. Bone marrow-derived circulating endothelial precursors do not contribute to vascular endothelium and are not needed for tumor growth. Proc. Natl Acad. Sci. USA 105, 6620–6625 (2008).
Kelly, T. et al. Expression of heparanase by primary breast tumors promotes bone resorption in the absence of detectable bone metastases. Cancer Res. 65, 5778–5784 (2005).
Pazolli, E. et al. Senescent stromal-derived osteopontin promotes preneoplastic cell growth. Cancer Res. 69, 1230–1239 (2009).
McAllister, S. S. et al. Systemic endocrine instigation of indolent tumor growth requires osteopontin. Cell 133, 994–1005 (2008).
Anborgh, P. H., Mutrie, J. C., Tuck, A. B. & Chambers, A. F. Role of the metastasis-promoting protein osteopontin in the tumour microenvironment. J. Cell. Mol. Med. 14, 2037–2044 (2006).
Lynch C. C. et al. MMP-7 promotes prostate cancer-induced osteolysis via the solubilization of RANKL. Cancer Cell 7, 485–496 (2005). Microarray analysis identified MMP7 as being upregulated at the tumor–bone interface in mice with prostate cancer. Osteoclast-produced MMP7 processed RANKL to a soluble form that promoted osteoclast activation. MMP7-deficient mice had reduced prostate tumour-induced osteolysis and RANKL processing.
Li, X. et al. A destructive cascade mediated by CCL2 facilitates prostate cancer growth in bone. Cancer Res. 69, 1685–1692 (2009).
Lehr, J. E. & Pienta, K. J. Preferential adhesion of prostate cancer cells to a human bone marrow endothelial cell line. J. Natl Cancer Inst. 90, 118–123 (1998).
Brenner, S. et al. CXCR4-transgene expression significantly improves marrow engraftment of cultured hematopoietic stem cells. Stem Cells 22, 1128–1133 (2004).
Kahn, J. et al. Overexpression of CXCR4 on human CD34+ progenitors increases their proliferation, migration, and NOD/SCID repopulation. Blood 103, 2942–2949 (2004).
Kang, Y. et al. A multigenic program mediating breast cancer metastasis to bone. Cancer Cell 3, 537–549 (2003). This study shows the multigenic nature of tumour tropism to bone: tumour cells must express a 'toolbox' of genes that act on the bone microenvironment in a cooperative way to promote bone metastases.
Yoneda, T. Cellular and molecular basis of preferential metastasis of breast cancer to bone. J. Orthop. Sci. 5, 75–81 (2000).
Stier, S. et al. Osteopontin is a hematopoietic stem cell niche component that negatively regulates stem cell pool size. J. Exp. Med. 201, 1781–1791 (2005).
Christopher, M. J., Liu, F., Hilton, M. J., Long, F. & Link, D. C. Suppression of CXCL12 production by bone marrow osteoblasts is a common and critical pathway for cytokine-induced mobilization. Blood 114, 1331–1339 (2009).
Calvi, L. M. et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature 425, 841–846 (2003).
Zhang, J. et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nature 425, 836–841 (2003).
Papayannopoulou, T. Mechanisms of stem-/progenitor-cell mobilization: the anti-VLA-4 paradigm. Semin. Hematol. 37, 11–18 (2000).
Hidalgo, A., Peired, A. J., Weiss, L. A., Katayama, Y. & Frenette, P. S. The integrin αMβ2 anchors hematopoietic progenitors in the bone marrow during enforced mobilization. Blood 104, 993–1001 (2004).
Mendez-Ferrer, S. et al. Mesenchymal and haematopoietic stem cells form a unique bone marrow niche. Nature 466, 829–834 (2010).
Kollet, O. et al. Osteoclasts degrade endosteal components and promote mobilization of hematopoietic progenitor cells. Nature Med. 12, 657–664 (2006).
Mendez-Ferrer, S. & Frenette, P. S. Hematopoietic stem cell trafficking: regulated adhesion and attraction to bone marrow microenvironment. Ann. N. Y Acad. Sci. 1116, 392–413 (2007).
Shiozawa, Y. et al. Prostate cancer metastases target the hematopoietic stem cell niche to establish footholds in marrow. J. Clin. Invest. 121, 1298–1312 (2011). Metastatic prostate cancer cells directly compete for occupancy of the HSC niche and once there begin a process of either evicting HSCs into the peripheral blood or driving HSCs into progenitor cell pools.
Muller, A. et al. Involvement of chemokine receptors in breast cancer metastasis. Nature 410, 50–56 (2001).
Sun, Y. X. et al. Expression and activation of αvβ3 integrins by SDF-1/CXC12 increases the aggressiveness of prostate cancer cells. Prostate 67, 61–73 (2007).
Sun, Y. X. et al. Skeletal localization and neutralization of the SDF-1(CXCL12)/CXCR4 axis blocks prostate cancer metastasis and growth in osseous sites in vivo. J. Bone Miner. Res. 20, 318–329 (2005).
Smith, M. C. et al. CXCR4 regulates growth of both primary and metastatic breast cancer. Cancer Res. 64, 8604–8612 (2004).
Chinni, S. R. et al. CXCL12/CXCR4 signaling activates Akt-1 and MMP-9 expression in prostate cancer cells: the role of bone microenvironment-associated CXCL12. Prostate 66, 32–48 (2006).
Lapteva, N., Yang, A. G., Sanders, D. E., Strube, R. W. & Chen, S. Y. CXCR4 knockdown by small interfering RNA abrogates breast tumor growth in vivo. Cancer Gene Ther. 12, 84–89 (2005).
Sun, Y. X. et al. Expression of CXCR4 and CXCL12 (SDF-1) in human prostate cancers (PCa) in vivo. J. Cell. Biochem. 89, 462–473 (2003).
Orimo, A. et al. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121, 335–348 (2005).
Clezardin, P. Integrins in bone metastasis formation and potential therapeutic implications. Curr. Cancer Drug Targets 9, 801–806 (2009).
Schneider, J. G., Amend, S. R. & Weilbaecher, K. N. Integrins and bone metastasis: integrating tumor cell and stromal cell interactions. Bone 48, 54–65 (2010).
Korah, R., Boots, M. & Wieder, R. Integrin α5β1 promotes survival of growth-arrested breast cancer cells: an in vitro paradigm for breast cancer dormancy in bone marrow. Cancer Res. 64, 4514–4522 (2004).
Liesveld, J. L., Dipersio, J. F. & Abboud, C. N. Integrins and adhesive receptors in normal and leukemic CD34+ progenitor cells: potential regulatory checkpoints for cellular traffic. Leuk. Lymphoma 14, 19–28 (1994).
Lang, S. H., Clarke, N. W., George, N. J. & Testa, N. G. Primary prostatic epithelial cell binding to human bone marrow stroma and the role of α2β1 integrin. Clin. Exp. Metastasis 15, 218–227 (1997).
Hall, C. L. et al. Type I collagen receptor (α2β1) signaling promotes prostate cancer invasion through RhoC GTPase. Neoplasia 10, 797–803 (2008).
Hall, C. L., Dai, J., van Golen, K. L., Keller, E. T. & Long, M. W. Type I collagen receptor (α2β1) signaling promotes the growth of human prostate cancer cells within the bone. Cancer Res. 66, 8648–8654 (2006).
Mori, Y. et al. Anti-α4 integrin antibody suppresses the development of multiple myeloma and associated osteoclastic osteolysis. Blood 104, 2149–2154 (2004).
Matsuura, N. et al. Induction of experimental bone metastasis in mice by transfection of integrin α4β1 into tumor cells. Am. J. Pathol. 148, 55–61 (1996).
Michigami, T. et al. Cell-cell contact between marrow stromal cells and myeloma cells via VCAM-1 and α4β1-integrin enhances production of osteoclast-stimulating activity. Blood 96, 1953–1960 (2000).
Parmo-Cabanas, M. et al. Integrin α4β1 involvement in stromal cell-derived factor-1α-promoted myeloma cell transendothelial migration and adhesion: role of cAMP and the actin cytoskeleton in adhesion. Exp. Cell Res. 294, 571–580 (2004).
Coleman, R. E. et al. Predictive value of bone resorption and formation markers in cancer patients with bone metastases receiving the bisphosphonate zoledronic acid. J. Clin. Oncol. 23, 4925–4935 (2005).
Henderson, M. A. et al. Parathyroid hormone-related protein localization in breast cancers predict improved prognosis. Cancer Res. 66, 2250–2256 (2006).
Yin, J. J. et al. TGF-β signaling blockade inhibits PTHrP secretion by breast cancer cells and bone metastases development. J. Clin. Invest. 103, 197–206 (1999).
Lu, X. et al. ADAMTS1 and MMP1 proteolytically engage EGF-like ligands in an osteolytic signaling cascade for bone metastasis. Genes Dev. 23, 1882–1894 (2009). This is the first preclinical evidence that blocking TGFβ signalling decreases the development and progression of breast cancer bone metastases.
Nannuru, K. C. et al. Matrix metalloproteinase (MMP)-13 regulates mammary tumor-induced osteolysis by activating MMP9 and transforming growth factor-β signaling at the tumor-bone interface. Cancer Res. 70, 3494–3504 (2010).
Sethi, N., Dai, X., Winter, C. G. & Kang, Y. Tumor-derived JAGGED1 promotes osteolytic bone metastasis of breast cancer by engaging notch signaling in bone cells. Cancer Cell 19, 192–205 (2011). The tumour-derived Notch ligand Jagged1 promotes tumour growth in bone by stimulating IL-6 release from osteoblasts and subsequent osteoclastogenesis. Therefore, secretase inhibitors of the Notch pathway, could prove to be valuable therapeutic drugs for bone metastases.
Sterling, J. A. et al. The hedgehog signaling molecule Gli2 induces parathyroid hormone-related peptide expression and osteolysis in metastatic human breast cancer cells. Cancer Res. 66, 7548–7553 (2006).
Alexaki, V. I. et al. GLI2-mediated melanoma invasion and metastasis. J. Natl Cancer Inst. 102, 1148–1159 (2010).
Pratap, J. et al. Ectopic runx2 expression in mammary epithelial cells disrupts formation of normal acini structure: imimplications for breast cancer progression. Cancer Res. 69, 6807–6814 (2009).
Javed, A. et al. Impaired intranuclear trafficking of Runx2 (AML3/CBFA1) transcription factors in breast cancer cells inhibits osteolysis in vivo. Proc. Natl Acad. Sci. USA 102, 1454–1459 (2005).
Hiraga, T., Kizaka-Kondoh, S., Hirota, K., Hiraoka, M. & Yoneda, T. Hypoxia and hypoxia-inducible factor-1 expression enhance osteolytic bone metastases of breast cancer. Cancer Res. 67, 4157–4163 (2007).
Dunn, L. K. et al. Hypoxia and TGF-β drive breast cancer bone metastases through parallel signaling pathways in tumor cells and the bone microenvironment. PLoS ONE 4, e6896 (2009). Hypoxia and TGFβ signalling in parallel drive the development of tumour bone metastases and regulate a common set of tumour genes ( VEGF and CXCR4 ). Small-molecule inhibitors, by acting on both tumour cells and the bone microenvironment, additively decrease tumour burden, while improving skeletal quality.
Sanders, J. L. et al. Extracellular calcium-sensing receptor expression and its potential role in regulating parathyroid hormone-related peptide secretion in human breast cancer cell lines. Endocrinology 141, 4357–4364 (2000).
Yamaguchi, T. et al. Expression of extracellular calcium-sensing receptor in human osteoblastic MG-63 cell line. Am. J. Physiol. Cell Physiol. 280, C382–C393 (2001).
Yano, S. et al. Calcium-sensing receptor activation stimulates parathyroid hormone-related protein secretion in prostate cancer cells: role of epidermal growth factor receptor transactivation. Bone 35, 664–672 (2004).
Mamillapalli, R., VanHouten, J., Zawalich, W. & Wysolmerski, J. Switching of G-protein usage by the calcium-sensing receptor reverses its effect on parathyroid hormone-related protein secretion in normal versus malignant breast cells. J. Biol. Chem. 283, 24435–24447 (2008).
Liao, J., Schneider, A., Datta, N. S. & McCauley, L. K. Extracellular calcium as a candidate mediator of prostate cancer skeletal metastasis. Cancer Res. 66, 9065–9073 (2006).
Saidak, Z. et al. Extracellular calcium promotes the migration of breast cancer cells through the activation of the calcium sensing receptor. Exp. Cell Res. 315, 2072–2080 (2009).
Letz, S. et al. Novel activating mutations of the calcium-sensing receptor: the calcilytic NPS-2143 mitigates excessive signal transduction of mutant receptors. J. Clin. Endocrinol. Metab. 95, E229–E233 (2010).
Sangaletti, S. et al. Macrophage-derived SPARC bridges tumor cell-extracellular matrix interactions toward metastasis. Cancer Res. 68, 9050–9059 (2008).
Shevde, L. A., Das, S., Clark, D. W. & Samant, R. S. Osteopontin: an effector and an effect of tumor metastasis. Curr. Mol. Med. 10, 71–81 (2010).
Contie, S., Voorzanger-Rousselot, N., Litvin, J., Clezardin, P. & Garnero, P. Increased expression and serum levels of the stromal cell-secreted protein periostin in breast cancer bone metastases. Int. J. Cancer 128, 352–360 (2010).
Bellahcene, A., Castronovo, V., Ogbureke, K. U., Fisher, L. W. & Fedarko, N. S. Small integrin-binding ligand N-linked glycoproteins (SIBLINGs): multifunctional proteins in cancer. Nature Rev. Cancer 8, 212–226 (2008).
Sharp, J. A., Waltham, M., Williams, E. D., Henderson, M. A. & Thompson, E. W. Transfection of MDA-MB-231 human breast carcinoma cells with bone sialoprotein (BSP) stimulates migration and invasion in vitro and growth of primary and secondary tumors in nude mice. Clin. Exp. Metastasis 21, 19–29 (2004).
Carlinfante, G. et al. Differential expression of osteopontin and bone sialoprotein in bone metastasis of breast and prostate carcinoma. Clin. Exp. Metastasis 20, 437–444 (2003).
Chaplet, M. et al. Expression of dentin sialophosphoprotein in human prostate cancer and its correlation with tumor aggressiveness. Int. J. Cancer 118, 850–856 (2006).
Bucciarelli, E. et al. Low dentin matrix protein 1 expression correlates with skeletal metastases development in breast cancer patients and enhances cell migratory capacity in vitro. Breast Cancer Res. Treat 105, 95–104 (2007).
Kelly, T., Suva, L. J., Nicks, K. M., MacLeod, V. & Sanderson, R. D. Tumor-derived syndecan-1 mediates distal cross-talk with bone that enhances osteoclastogenesis. J. Bone Miner. Res. 25, 1295–1304 (2010).
Araki, K. et al. Decorin suppresses bone metastasis in a breast cancer cell line. Oncology 77, 92–99 (2009).
Ikushima, H. & Miyazono, K. TGFβ signalling: a complex web in cancer progression. Nature Rev. Cancer 10, 415–424 (2010).
Kang, Y. et al. Breast cancer bone metastasis mediated by the Smad tumor suppressor pathway. Proc. Natl Acad. Sci. USA 102, 13909–13914 (2005).
Javelaud, D. et al. Stable overexpression of Smad7 in human melanoma cells impairs bone metastasis. Cancer Res. 67, 2317–2324 (2007).
Korpal, M., Yan, J., Lu, X., Xu, S., Lerit, D. A. & Kang, Y. Imaging transforming growth factor-β signaling dynamics and therapeutic response in breast cancer bone metastasis. Nature Med. 15, 960–966 (2009). This is the first direct evidence that a major source of active TGFβ signalling to tumour cells is that released from mineralized bone matrix as a consequence of osteoclastic bone resorption.
Schneider, A. et al. Bone turnover mediates preferential localization of prostate cancer in the skeleton. Endocrinology 146, 1727–1736 (2005).
Ooi, L. L. et al. Vitamin D deficiency promotes human breast cancer growth in a murine model of bone metastasis. Cancer Res. 70, 1835–1844 (2010). Vitamin D deficiency working at least in part via alterations in the bone microenvironment promotes the growth of human breast cancer cells in bone.
Hirbe, A. C. et al. Granulocyte colony-stimulating factor enhances bone tumor growth in mice in an osteoclast-dependent manner. Blood 109, 3424–3431 (2007).
Park, B. K. et al. NF-κB in breast cancer cells promotes osteolytic bone metastasis by inducing osteoclastogenesis via GM-CSF. Nature Med. 13, 62–69 (2007).
Hirbe, A. C. et al. Disruption of CXCR4 enhances osteoclastogenesis and tumor growth in bone. Proc. Natl Acad. Sci. USA 104, 14062–14067 (2007).
Takayama, S. et al. The relationship between bone metastasis from human breast cancer and integrin αvβ3 expression. Anticancer Res. 25, 79–83 (2005).
Boissier, S. et al. Bisphosphonates inhibit breast and prostate carcinoma cell invasion, an early event in the formation of bone metastases. Cancer Res. 60, 2949–2954 (2000).
Croucher, P. I. et al. Osteoprotegerin inhibits the development of osteolytic bone disease in multiple myeloma. Blood 98, 3534–3540 (2001).
Gao, L. et al. HTLV-1 Tax transgenic mice develop spontaneous osteolytic bone metastases prevented by osteoclast inhibition. Blood 106, 4294–4302 (2005). This paper describes a spontaneous animal model of hypercalcaemia and tumour-associated osteolysis and demonstrates that the viral oncogene Tax is responsible for cellular transformation and induces the expression of osteoclast-activating factors resulting in bone loss and hypercalcaemia.
Desai, B., Rogers, M. J. & Chellaiah, M. A. Mechanisms of osteopontin and CD44 as metastatic principles in prostate cancer cells. Mol. Cancer 6, 18 (2007).
Nemoto, H. et al. Osteopontin deficiency reduces experimental tumor cell metastasis to bone and soft tissues. J. Bone Miner. Res. 16, 652–659 (2001).
Bakewell, S. J. et al. Platelet and osteoclast β3 integrins are critical for bone metastasis. Proc. Natl Acad. Sci. USA 100, 14205–14210 (2003).
Rachkovsky, M. et al. Melanoma x macrophage hybrids with enhanced metastatic potential. Clin. Exp. Metastasis 16, 299–312 (1998).
Pawelek, J. M. & Chakraborty, A. K. Fusion of tumour cells with bone marrow-derived cells: a unifying explanation for metastasis. Nature Rev. Cancer 8, 377–386 (2008).
Andersen, T. L. et al. Osteoclast nuclei of myeloma patients show chromosome translocations specific for the myeloma cell clone: a new type of cancer-host partnership? J. Pathol. 211, 10–17 (2007).
Vignery, A. Macrophage fusion: are somatic and cancer cells possible partners? Trends Cell Biol. 15, 188–193 (2005).
Lu, X. & Kang, Y. Efficient acquisition of dual metastasis organotropism to bone and lung through stable spontaneous fusion between MDA-MB-231 variants. Proc. Natl Acad. Sci. USA 106, 9385–9390 (2009).
Abe, M. et al. Osteoclasts enhance myeloma cell growth and survival via cell-cell contact: a vicious cycle between bone destruction and myeloma expansion. Blood 104, 2484–2491 (2004).
Gonzalez-Suarez, E. et al. RANK ligand mediates progestin-induced mammary epithelial proliferation and carcinogenesis. Nature 468, 103–107 (2010). RANKL and RANK are essential for mammary gland development in mice and mediate progesterone-induced proliferation in the mammary gland. This paper highlights the bone-independent role of RANKL that may provide new targeted approaches for breast cancer therapy.
Clarke, N. W., McClure, J. & George, N. J. Morphometric evidence for bone resorption and replacement in prostate cancer. Br. J. Urol. 68, 74–80 (1991).
Roudier, M. P. et al. Histopathological assessment of prostate cancer bone osteoblastic metastases. J. Urol. 180, 1154–1160 (2008).
Koeneman, K. S., Yeung, F. & Chung, L. W. Osteomimetic properties of prostate cancer cells: a hypothesis supporting the predilection of prostate cancer metastasis and growth in the bone environment. Prostate 39, 246–261 (1999).
Dudley, A. C. et al. Calcification of multipotent prostate tumor endothelium. Cancer Cell 14, 201–211 (2008).
Odero-Marah, V. A. et al. Receptor activator of NF-κB Ligand (RANKL) expression is associated with epithelial to mesenchymal transition in human prostate cancer cells. Cell Res. 18, 858–870 (2008).
Brubaker, K. D., Vessella, R. L, Brown, L. G. & Corey, E. Prostate cancer expression of runt-domain transcription factor Runx2, a key regulator of osteoblast differentiation and function. Prostate 56, 13–22 (2003).
Leong, D. T. et al. Cancer-related ectopic expression of the bone-related transcription factor RUNX2 in non-osseous metastatic tumor cells is linked to cell proliferation and motility. Breast Cancer Res. 12, R89 (2010).
Akech, J. et al. Runx2 association with progression of prostate cancer in patients: mechanisms mediating bone osteolysis and osteoblastic metastatic lesions. Oncogene 29, 811–812 (2010). RUNX2, traditionally known to be osteoblast specific, was found to be highly expressed in metastatic prostate cancer cells and associated with tumour growth in bone. This highlights the osteomimicry of tumour cells that metastasize to bone and provides a new target to consider for therapeutic development.
Chackal-Roy, M., Niemeyer, C., Moore, M. & Zetter, B. R. Stimulation of human prostatic carcinoma cell growth by factors present in human bone marrow. J. Clin. Invest. 84, 43–50 (1989).
Gleave, M., Hsieh, J. T., Gao, C. A., von Eschenbach, A. C. & Chung, L. W. Acceleration of human prostate cancer growth in vivo by factors produced by prostate and bone fibroblasts. Cancer Res. 51, 3753–3761 (1991).
Cooper, C. R. et al. Stromal factors involved in prostate carcinoma metastasis to bone. Cancer 97, 739–747 (2003).
De Palma, M. & Naldini, L. Role of haematopoietic cells and endothelial progenitors in tumour angiogenesis. Biochim. Biophys. Acta 1766, 159–166 (2006).
Stockmann, C. et al. Deletion of vascular endothelial growth factor in myeloid cells accelerates tumorigenesis. Nature 456, 814–818 (2008).
Silva, R., D'Amico, G., Hodivala-Dilke, K. M. & Reynolds, L. E. Integrins: the keys to unlocking angiogenesis. Arterioscler. Thromb. Vasc. Biol. 28, 1703–1713 (2008).
Prantl, L. et al. Adipose tissue-derived stem cells promote prostate tumor growth. Prostate 70, 1709–1715 (2010).
Klopp, A. H. et al. Mesenchymal stem cells promote mammosphere formation and decrease E-cadherin in normal and malignant breast cells. PLoS ONE 5, e12180 (2010).
Studeny, M. et al. Mesenchymal stem cells: potential precursors for tumor stroma and targeted-delivery vehicles for anticancer agents. J. Natl Cancer Inst. 96, 1593–1603 (2004).
Bianco, P., Robey, P. G., Saggio, I. & Riminucci, M. “Mesenchymal” stem cells in human bone marrow (skeletal stem cells): a critical discussion of their nature, identity, and significance in incurable skeletal disease. Hum. Gene Ther. 21, 1057–1066 (2010).
Yang, L., Edwards, C. M. & Mundy, G. R. Gr-1+CD11b+ myeloid-derived suppressor cells: formidable partners in tumor metastasis. J. Bone Miner. Res. 25, 1701–1706 (2010).
Bingle, L., Brown, N. J. & Lewis, C. E. The role of tumour-associated macrophages in tumour progression: implications for new anticancer therapies. J. Pathol. 196, 254–265 (2002).
Dirkx, A. E., Oude Egbrink, M. G., Wagstaff, J. & Griffioen, A. W. Monocyte/macrophage infiltration in tumors: modulators of angiogenesis. J. Leukoc. Biol. 80, 1183–1196 (2006).
Gabrilovich, D. I. et al. The terminology issue for myeloid-derived suppressor cells. Cancer Res. 67, 425; author reply 426 (2007).
Vaira, S. et al. RelB is the NF-κB subunit downstream of NIK responsible for osteoclast differentiation. Proc. Natl Acad. Sci. USA 105, 3897–3902 (2008).
Melani, C., Sangaletti, S., Barazzetta, F. M., Werb, Z. & Colombo, M. P. Amino-biphosphonate-mediated MMP-9 inhibition breaks the tumor-bone marrow axis responsible for myeloid-derived suppressor cell expansion and macrophage infiltration in tumor stroma. Cancer Res. 67, 11438–11446 (2007).
Hirbe, A. C. et al. The bisphosphonate zoledronic acid decreases tumor growth in bone in mice with defective osteoclasts. Bone 44, 908–916 (2009).
Dunn, G. P., Koebel, C. M. & Schreiber, R. D. Interferons, immunity and cancer immunoediting. Nature Rev. Immunol. 6, 836–848 (2006).
Takayanagi, H. Osteoimmunology and the effects of the immune system on bone. Nature Rev. Rheumatol. 5, 667–676 (2009).
Sato, K. et al. Th17 functions as an osteoclastogenic helper T cell subset that links T cell activation and bone destruction. J. Exp. Med. 203, 2673–2682 (2006).
Gay, L. J. & Felding-Habermann, B. Contribution of platelets to tumour metastasis. Nature Rev. Cancer 11, 123–134 (2011).
Boucharaba, A. et al. Platelet-derived lysophosphatidic acid supports the progression of osteolytic bone metastases in breast cancer. J. Clin. Invest. 114, 1714–1725 (2004). This paper shows that lysophosphatidic acid derived from activated platelets can promote tumour growth and osteolysis in a mouse model of bone metastases.
Uluçkan, Ö. et al. APT102, a novel adpase, cooperates with aspirin to disrupt bone metastasis in mice. J. Cell. Biochem. 104, 1311–1323 (2008).
Li, X. et al. Inhibitory effects of megakaryocytic cells in prostate cancer skeletal metastasis. J. Bone Miner. Res. 26, 125–134 (2010).
Beeton, C. A., Bord, S., Ireland, D. & Compston, J. E. Osteoclast formation and bone resorption are inhibited by megakaryocytes. Bone 39, 985–990 (2006).
Chollet, M. E. et al. Evidence of a colocalisation of osteoprotegerin (OPG) with von Willebrand factor (VWF) in platelets and megakaryocytes α granules. Studies from normal and grey platelets. Br. J. Haematol. 148, 805–807 (2009).
Lemieux, J. M., Horowitz, M. C. & Kacena, M. A. Involvement of integrins α3β1 and α5β1 and glycoprotein IIb in megakaryocyte-induced osteoblast proliferation. J. Cell. Biochem. 109, 927–932 (2010).
Bernasconi, S. et al. Effects of granulocyte-monocyte colony-stimulating factor (GM-CSF) on expression of adhesion molecules and production of cytokines in blood monocytes and ovarian cancer-associated macrophages. Int. J. Cancer 60, 300–307 (1995).
Zaslavsky, A. et al. Platelet-derived thrombospondin-1 is a critical negative regulator and potential biomarker of angiogenesis. Blood 115, 4605–4613 (2010).
Fidler, I. J. Metastasis: guantitative analysis of distribution and fate of tumor embolilabeled with 125 I-5-iodo-2′-deoxyuridine. J. Natl Cancer Inst. 45, 773–782 (1970).
Luzzi, K. J. et al. Multistep nature of metastatic inefficiency: dormancy of solitary cells after successful extravasation and limited survival of early micrometastases. Am. J. Pathol. 153, 865–873 (1998).
Braun, S. et al. Cytokeratin-positive cells in the bone marrow and survival of patients with stage I, II, or III breast cancer. N. Engl. J. Med. 342, 525–533 (2000).
Damiano, J. S., Cress, A. E., Hazlehurst, L. A., Shtil, A. A. & Dalton, W. S. Cell adhesion mediated drug resistance (CAM-DR): role of integrins and resistance to apoptosis in human myeloma cell lines. Blood 93, 1658–1667 (1999).
Morgan, T. M. et al. Disseminated tumor cells in prostate cancer patients after radical prostatectomy and without evidence of disease predicts biochemical recurrence. Clin. Cancer Res. 15, 677–683 (2009).
Diel, I. J. et al. Micrometastatic breast cancer cells in bone marrow at primary surgery: prognostic value in comparison with nodal status. J. Natl Cancer Inst. 88, 1652–1658 (1996).
Coleman, R. E. et al. Advancing treatment for metastatic bone cancer: consensus recommendations from the Second Cambridge Conference. Clin. Cancer Res. 14, 6387–6395 (2008).
Watson, M. A. et al. Isolation and molecular profiling of bone marrow micrometastases identifies TWIST1 as a marker of early tumor relapse in breast cancer patients. Clin. Cancer Res. 13, 5001–5009 (2007).
Zhang, X. H. et al. Latent bone metastasis in breast cancer tied to Src-dependent survival signals. Cancer Cell 16, 67–78 (2009).
Rack, B. et al. Effect of zoledronate on persisting isolated tumour cells in patients with early breast cancer. Anticancer Res. 30, 1807–1813 (2010).
Shiozawa, Y. et al. GAS6/AXL axis regulates prostate cancer invasion, proliferation, and survival in the bone marrow niche. Neoplasia 12, 116–127 (2010).
Rogers, M. J. New insights into the molecular mechanisms of action of bisphosphonates. Curr. Pharm. Des. 9, 2643–2658 (2003).
Gnant, M. et al. Endocrine therapy plus zoledronic acid in premenopausal breast cancer. N. Engl. J. Med. 360, 679–691 (2009).
Stopeck, A. T. et al. Denosumab compared with zoledronic acid for the treatment of bone metastases in patients with advanced breast cancer: a randomized, double-blind study. J. Clin. Oncol. 28, 5132–5139 (2010).
Jensen, A. B. et al. The cathepsin K inhibitor odanacatib suppresses bone resorption in women with breast cancer and established bone metastases: results of a 4-week, double-blind, randomized, controlled trial. Clin. Breast Cancer 10, 452–458 (2010).
Rucci, N., Susa, M. & Teti, A. Inhibition of protein kinase c-Src as a therapeutic approach for cancer and bone metastases. Anticancer Agents Med. Chem. 8, 342–349 (2008).
Desgrosellier, J. S. & Cheresh, D. A. Integrins in cancer: biological implications and therapeutic opportunities. Nature Rev. Cancer 10, 9–22 (2009).
Korpal, M. & Kang, Y. Targeting the transforming growth factor-β signalling pathway in metastatic cancer. Eur. J. Cancer 46, 1232–1240 (2010).
Balooch, G. et al. TGF-β regulates the mechanical properties and composition of bone matrix. Proc. Natl Acad. Sci. USA 102, 18813–18818 (2005).
Mohammad, K. S. et al. Pharmacologic inhibition of the TGF-β type I receptor kinase has anabolic and anti-catabolic effects on bone. PLoS ONE 4, e5275 (2009).
Edwards, J. R. et al. Inhibition of TGF-β signaling by 1D11 antibody treatment increases bone mass and quality in vivo. J. Bone Miner. Res. 25, 2419–2426 (2010).
Buijs, J. T. et al. Bone morphogenetic protein 7 in the development and treatment of bone metastases from breast cancer. Cancer Res. 67, 8742–8751 (2007).
Buijs, J. T. et al. BMP7, a putative regulator of epithelial homeostasis in the human prostate, is a potent inhibitor of prostate cancer bone metastasis in vivo. Am. J. Pathol. 171, 1047–1057 (2007). In normal epithelial cells, BMP7 and TGFβ prevent EMT. Loss of BMP7 expression in tumours results in reduced E-cadherin expression, EMT and metastases to bone. This model re-emphasizes the 'double-edged sword' properties and the complexity of TGFβ superfamily members in cancer biology.
Gautschi, O. P., Frey, S. P. & Zellweger, R. Bone morphogenetic proteins in clinical applications. ANZ J. Surg. 77, 626–631 (2007).
Prud'homme, G. J. Pathobiology of transforming growth factor β in cancer, fibrosis and immunologic disease, and therapeutic considerations. Lab. Invest. 87, 1077–1091 (2007).
Nelson, J. B. et al. Identification of endothelin-1 in the pathophysiology of metastatic adenocarcinoma of the prostate. Nature Med. 1, 944–949 (1995).
Yin, J. J. et al. A causal role for endothelin-1 in the pathogenesis of osteoblastic bone metastases. Proc. Natl Acad. Sci. USA 100, 10954–10959 (2003). This is the first preclinical evidence that endothelin receptor blockade reduces the development and progression of osteoblastic bone metastases.
Carducci, M. A. & Jimeno, A. Targeting bone metastasis in prostate cancer with endothelin receptor antagonists. Clin. Cancer Res. 12, 6296s–6300s (2006).
Clines, G. A. et al. Dickkopf homolog 1 mediates endothelin-1-stimulated new bone formation. Mol. Endocrinol. 21, 486–498 (2007).
Shepard, D. R. & Dreicer, R. Zibotentan for the treatment of castrate-resistant prostate cancer. Expert Opin. Investig Drugs 19, 899–908 (2010).
Nervi, B. et al. Chemosensitization of acute myeloid leukemia (AML) following mobilization by the CXCR4 antagonist AMD3100. Blood 113, 6206–6214 (2009).
Azab, A. K. et al. CXCR4 inhibitor AMD3100 disrupts the interaction of multiple myeloma cells with the bone marrow microenvironment and enhances their sensitivity to therapy. Blood 113, 4341–4351 (2009).
Peyruchaud, O., Serre, C. M., NicAmhlaoibh, R., Fournier, P. & Clezardin, P. Angiostatin inhibits bone metastasis formation in nude mice through a direct anti-osteoclastic activity. J. Biol. Chem. 278, 45826–45832 (2003).
Twardowski, P. W. et al. Biologic markers of angiogenesis: circulating endothelial cells in patients with advanced malignancies treated on phase I protocol with metronomic chemotherapy and celecoxib. Cancer Invest. 26, 53–59 (2008).
Merz, M., Komljenovic, D., Zwick, S., Semmler, W. & Bauerle, T. Sorafenib tosylate and paclitaxel induce anti-angiogenic, anti-tumour and anti-resorptive effects in experimental breast cancer bone metastases. Eur. J. Cancer 47, 277–286 (2010).
Gao, D. et al. Endothelial progenitor cells control the angiogenic switch in mouse lung metastasis. Science 319, 195–198 (2008).
Mellick, A. S. et al. Using the transcription factor inhibitor of DNA binding 1 to selectively target endothelial progenitor cells offers novel strategies to inhibit tumor angiogenesis and growth. Cancer Res. 70, 7273–7282 (2010).
Kai, L. et al. Targeting prostate cancer angiogenesis through metastasis-associated protein 1 (MTA1). Prostate 71, 268–280 (2011).
Meuser, T. et al. Symptoms during cancer pain treatment following WHO-guidelines: a longitudinal follow-up study of symptom prevalence, severity and etiology. Pain 93, 247–257 (2001).
Goblirsch, M. J., Zwolak, P. P. & Clohisy, D. R. Biology of bone cancer pain. Clin. Cancer Res. 12, 6231s–6235s (2006).
Honore, P. et al. Osteoprotegerin blocks bone cancer-induced skeletal destruction, skeletal pain and pain-related neurochemical reorganization of the spinal cord. Nature Med. 6, 521–528 (2000). Bone pain that is associated with bone metastasis is difficult to treat. This report demonstrates that osteoclastic resorption at the site of a tumour deposit in bone recruits pain fibres and changes the spinal chord neurochemical composition to promote bone pain. Blockade of osteoclastic resorption reduced pain, spinal chord reorganziation and nerve recruitment associated with bone tumours.
Nakanishi, M. et al. Acid activation of Trpv1 leads to an up-regulation of calcitonin gene-related peptide expression in dorsal root ganglion neurons via the CaMK-CREB cascade: a potential mechanism of inflammatory pain. Mol. Biol. Cell 21, 2568–2577 (2010).
McEwan, A. J. Use of radionuclides for the palliation of bone metastases. Semin. Radiat. Oncol. 10, 103–114 (2000).
Ding, L. et al. Genome remodelling in a basal-like breast cancer metastasis and xenograft. Nature 464, 999–1005 (2010).
Hall, C. L, Kang, S., MacDougald, O. A. & Keller, E. T. Role of Wnts in prostate cancer bone metastases. J. Cell. Biochem. 97, 661–672 (2006).
Voorzanger-Rousselot, N. et al. Increased Dickkopf-1 expression in breast cancer bone metastases. Br. J. Cancer 97, 964–970 (2007).
Tian, E. et al. The role of the Wnt-signaling antagonist DKK1 in the development of osteolytic lesions in multiple myeloma. N. Engl. J. Med. 349, 2483–2494 (2003). This paper shows that the osteoblast inhibitor DKK1 is expressed by myeloma cells and is detected at higher levels in patients with myeloma who have osteolytic lesions. The discovery that myeloma cells express osteoblast inhibitors explains why bone loss is so severe in this disease.
Dai, J. et al. Prostate cancer induces bone metastasis through Wnt-induced bone morphogenetic protein-dependent and independent mechanisms. Cancer Res. 65, 8274–8285 (2005).
Giuliani, N. et al. Production of Wnt inhibitors by myeloma cells: potential effects on canonical Wnt pathway in the bone microenvironment. Cancer Res. 67, 7665–7674 (2007).
Hjertner, O. et al. Hepatocyte growth factor (HGF) induces interleukin-11 secretion from osteoblasts: a possible role for HGF in myeloma-associated osteolytic bone disease. Blood 94, 3883–3888 (1999).
Oshima, T. et al. Myeloma cells suppress bone formation by secreting a soluble Wnt inhibitor, sFRP-2. Blood 106, 3160–3165 (2005).
Bonewald, L. F. The amazing osteocyte. J. Bone Miner. Res. 26, 229–238 (2011).
Hauge, E. M., Qvesel, D., Eriksen, E. F., Mosekilde, L. & Melsen, F. Cancellous bone remodeling occurs in specialized compartments lined by cells expressing osteoblastic markers. J. Bone Miner. Res. 16, 1575–1582 (2001).
Pettit, A. R., Chang, M. K., Hume, D. A. & Raggatt, L. J. Osteal macrophages: a new twist on coupling during bone dynamics. Bone 43, 976–982 (2008).
Baud'huin, M. et al. Interleukin-34 is expressed by giant cell tumours of bone and plays a key role in RANKL-induced osteoclastogenesis. J. Pathol. 221, 77–86 (2010).
Catena, R. et al. PDGFR signalling blockade in marrow stroma impairs lung cancer bone metastasis. Cancer Res. 71, 164–174 (2011).
Acknowledgements
Dedicated in memory of G. Mundy. The authors would like to thank P. Ross, M. Tomasson, C. Hall, D. Novack, S. Amend, J. Schneider, M. Hurchla and C. Winkeler for feedback on this manuscript. The authors are grateful to K. Pienta (University of Michigan School of Medicine, USA) for providing the gross autopsy specimen from vertebral body involved with prostate cancer, V. Reichert and J. Burkett (Washington University Medical School, St. Louis, USA) for providing the radiological images of bone metastases, R. Aft (Washington University School of Medicine, USA) for providing the photograph of bone marrow disseminated tumour cells, and D. Novack (Washington University School of Medicine) for providing the histological slide from bone metastasis biospy. K.N.W. is supported by the US National Institutes of Health (NIH) (R01-CA52152 and P01-CA100730) and the US Department of Defense (W81XWH-01-1-360). T.A.G. is supported by R01CA69158, R01DK065837, R01DK067333, U01CA143057, V-Foundation and Indiana Economic Development Grant. L.K.M. is supported by the US National Institutes of Health (NIH) (PO1-CA093900, RO1- DK53904), the US Department of Defense (W81XWH-08-1-0037) and Centocor Inc. The authors regret that there are many other important studies that they were unable to include owing to space limitations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
L.K.M. declares research funding from Centrocor Inc. and stock in Amgen. Educational seminar provided to Amgen employees. T.A.G. is a consultant for Novartis and Amgen. K.N.W. declares no competing financial interests.
Related links
Rights and permissions
About this article
Cite this article
Weilbaecher, K., Guise, T. & McCauley, L. Cancer to bone: a fatal attraction. Nat Rev Cancer 11, 411–425 (2011). https://doi.org/10.1038/nrc3055
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc3055
This article is cited by
-
Tumor-derived cell-free DNA and circulating tumor cells: partners or rivals in metastasis formation?
Clinical and Experimental Medicine (2024)
-
Cancer aggravation due to persistent pain signals with the increased expression of pain-related mediators in sensory neurons of tumor-bearing mice
Molecular Brain (2023)
-
Phototherapy techniques for the management of musculoskeletal disorders: strategies and recent advances
Biomaterials Research (2023)
-
Progressive resolution optimizer (PRO) predominates over photon optimizer (PO) in sparing of spinal cord for spine SABR VMAT plans
BMC Cancer (2023)
-
Conditional knockout of PDK1 in osteoclasts suppressed osteoclastogenesis and ameliorated prostate cancer-induced osteolysis in murine model
European Journal of Medical Research (2023)