Key Points
-
Germline mutations that inactivate the VHL tumour suppressor gene cause a variety of tumours including clear cell renal carcinomas, haemangioblastomas and pheochromocytomas. VHL mutations are also common in sporadic clear cell renal carcinomas and haemangioblastomas.
-
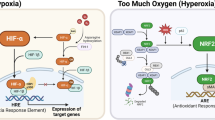
The product of VHL has multiple functions, including directing the polyubiquitylation of hypoxia-inducible factor-α (HIFα). Recognition by VHL requires that the HIFα subunit be modified by O2-dependent prolyl hydroxylase (Phd) family members.
-
Inappropriate accumulation of HIFα, and especially the HIF2α subunit, has a causal role in VHL−/− renal carcinomas and its involvement is suspected in VHL−/− haemangioblastomas. Genotype–phenotype correlations suggest that these two tumours differ with respect to the level of HIFα activation required for tumorigenesis.
-
Hypomorphic VHL, hypomorphic PHD2 and hypermorphic HIF2α mutations have been linked to familial polycythaemia.
-
VHL binds to microtubules and is required for maintenance of a specialized structure called the primary cilium. Loss of this activity probably contributes to the development of visceral cysts in VHL disease.
-
Pheochromocytomas are intra-adrenal paragangliomas (sympathetic nervous system tumours). The genes linked to familial paraganglioma, including VHL, NF1, RET and succinate dehydrogenase subunit genes, encode proteins that regulate neuronal apoptosis in response to loss of growth factors such as nerve growth factor.
Abstract
The von Hippel–Lindau disease is caused by inactivating germline mutations of the VHL tumour suppressor gene and is associated with an increased risk of a variety of tumours in an allele-specific manner. The role of the heterodimeric transcription factor hypoxia-inducible factor (HIF) in the pathogenesis of VHL-defective tumours has been more firmly established during the past 5 years. In addition, there is now a greater appreciation of HIF-independent VHL functions that are relevant to tumour development, including maintenance of the primary cilium, regulation of extracellular matrix formation and turnover, and modulation of cell death in certain cell types following growth factor withdrawal or in response to other forms of stress.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Maxwell, P. et al. The von Hippel–Lindau gene product is necessary for oxgyen-dependent proteolysis of hypoxia-inducible factor α subunits. Nature 399, 271–275 (1999).
Tanimoto, K., Makino, Y., Pereira, T. & Poellinger, L. Mechanism of regulation of the hypoxia-inducible factor-1α by the von Hippel–Lindau tumor suppressor protein. EMBO J. 19, 4298–4309 (2000).
Cockman, M. et al. Hypoxia inducible factor-α binding and ubiquitylation by the von Hippel-Lindau tumor suppressor protein. J. Biol. Chem. 275, 25733–25741 (2000).
Kamura, T. et al. Activation of HIF1α ubiquitination by a reconstituted von Hippel–Lindau tumor suppressor complex. Proc. Natl Acad. Sci. USA 97, 10430–10435 (2000).
Ohh, M. et al. Ubiquitination of HIF requires direct binding to the von Hippel-Lindau protein β domain. Nature Cell Biol. 2, 423–427 (2000).
Ivan, M. et al. HIFα targeted for VHL-mediated destruction by proline hydroxylation: implications for O2 sensing. Science 292, 464–468 (2001).
Jaakkola, P. et al. Targeting of HIF-α to the von Hippel–Lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science 292, 468–472 (2001).
Yu, F., White, S., Zhao, Q. & Lee, F. HIF-1α binding to VHL is regulated by stimulus-sensitive proline hydroxylation. Proc. Natl Acad. Sci. USA 98, 9630–9635 (2001).
Epstein, A. et al. C. elegans EGL-9 and mammalian homologs define a family of dioxygenases that regulate HIF by prolyl hydroxylation. Cell 107, 43–54 (2001).
Bruick, R. & McKnight, S. A conserved family of prolyl-4-hydroxylases that modify HIF. Science 294, 1337–1340 (2001).
Ivan, M. et al. Biochemical purification and pharmacological inhibition of a mammalian prolyl hydroxylase acting on hypoxia-inducible factor. Proc. Natl Acad. Sci. USA 99, 13459–13464 (2002).
Masson, N., Willam, C., Maxwell, P., Pugh, C. & Ratcliffe, P. Independent function of two destruction domains in hypoxia-inducible factor-α chains activated by prolyl hydroylation. EMBO J. 20, 5197–5206 (2001).
Kaelin Jr, W. G. von Hippel–Lindau disease. Annu. Rev. Pathol. Mech. Dis. 2, 145–173 (2007).
Maynard, M. A. et al. Multiple splice variants of the human HIF-3 α locus are targets of the von Hippel–Lindau E3 ubiquitin ligase complex. J. Biol. Chem. 278, 11032–11040 (2003).
Makino, Y., Kanopka, A., Wilson, W. J., Tanaka, H. & Poellinger, L. Inhibitory PAS domain protein (IPAS) is a hypoxia-inducible splicing variant of the hypoxia-inducible factor-3α locus. J. Biol. Chem. 277, 32405–32408 (2002).
Makino, Y. et al. Inhibitory PAS domain protein is a negative regulator of hypoxia-inducible gene expression. Nature 414, 550–554 (2001).
Hirsila, M., Koivunen, P., Gunzler, V., Kivirikko, K. I. & Myllyharju, J. Characterization of the human prolyl 4-hydroxylases that modify the hypoxia-inducible factor. J. Biol. Chem. 278, 30772–30780 (2003).
Koivunen, P., Hirsila, M., Kivirikko, K. I. & Myllyharju, J. The length of peptide substrates has a marked effect on hydroxylation by the hypoxia-inducible factor prolyl 4-hydroxylases. J. Biol. Chem. 281, 28712–28720 (2006).
Kaelin, W. G. Jr & Ratcliffe, P. J. Oxygen sensing by metazoans: the central role of the HIF hydroxylase pathway. Mol. Cell 30, 393–402 (2008).
Berra, E. et al. HIF prolyl-hydroxylase 2 is the key oxygen sensor setting low steady-state levels of HIF-1α in normoxia. EMBO J. 22, 4082–4090 (2003). First paper suggesting that PHD2 (EGLN1) is the primary HIF prolyl hydroxylase under normal conditions.
Takeda, K. et al. Placental but not heart defects are associated with elevated hypoxia-inducible factor α levels in mice lacking prolyl hydroxylase domain protein 2. Mol. Cell Biol. 26, 8336–8346 (2006).
Takeda, K. et al. Regulation of adult erythropoiesis by prolyl hydroxylase domain proteins. Blood 111, 3229–3235 (2008).
Minamishima, Y. A. et al. Somatic inactivation of the PHD2 prolyl hydroxylase causes polycythemia and congestive heart failure. Blood 111, 3236–3244 (2008).
Percy, M. J. et al. A family with erythrocytosis establishes a role for prolyl hydroxylase domain protein 2 in oxygen homeostasis. Proc. Natl Acad. Sci. USA 103, 654–659 (2006). First report linking EGLN1 (PHD2) mutations to familial polycythaemia.
Marxsen, J. H. et al. Hypoxia-inducible factor-1 (HIF-1) promotes its degradation by induction of HIF-α-prolyl-4-hydroxylases. Biochem. J. 381, 761–767 (2004).
Aprelikova, O. et al. Regulation of HIF prolyl hydroxylases by hypoxia-inducible factors. J. Cell Biochem. 92, 491–501 (2004).
Cioffi, C. L., Liu, X. Q., Kosinski, P. A., Garay, M. & Bowen, B. R. Differential regulation of HIF-1 α prolyl-4-hydroxylase genes by hypoxia in human cardiovascular cells. Biochem. Biophys. Res. Commun. 303, 947–953 (2003).
Appelhoff, R. J. et al. Differential function of the prolyl hydroxylases PHD1, PHD2, and PHD3 in the regulation of hypoxia-inducible factor. J. Biol. Chem. 279, 38458–38465 (2004).
Ginouves, A., Ilc, K., Macias, N., Pouyssegur, J. & Berra, E. PHDs overactivation during chronic hypoxia “desensitizes” HIFα and protects cells from necrosis. Proc. Natl Acad. Sci. USA 105, 4745–4750 (2008).
Ang, S. O. et al. Disruption of oxygen homeostasis underlies congenital Chuvash polycythemia. Nature Genet. 32, 614–621 (2002).
Bento, M. C. et al. Congenital polycythemia with homozygous and heterozygous mutations of von Hippel–Lindau gene: five new Caucasian patients. Haematologica 90, 128–129 (2005).
Hickey, M. M., Lam, J. C., Bezman, N. A., Rathmell, W. K. & Simon, M. C. von Hippel–Lindau mutation in mice recapitulates Chuvash polycythemia via hypoxia-inducible factor-2α signaling and splenic erythropoiesis. J. Clin. Invest. 117, 3879–3889 (2007).
Lando, D. et al. FIH-1 is a an asparaginyl hydroxylase that regulates the transcriptional activity of hypoxia inducible factor. Genes Dev. 16, 1466–1471 (2002).
Hewitson, K. S. et al. Hypoxia-inducible factor (HIF) asparagine hydroxylase is iIdentical to factor inhibiting HIF (FIH) and is related to the cupin structural family. J. Biol. Chem. 277, 26351–26355 (2002).
Dayan, F., Roux., D., Brahimi-Horn, M. C., Pouyssegur, J. & Mazure, N. M. The oxygen sensor factor-inhibiting hypoxia-inducible factor-1 controls expression of distinct genes through the bifunctional transcriptional character of hypoxia-inducible factor-1α. Cancer Res. 66, 3688–3698 (2006).
Koivunen, P., Hirsila, M., Gunzler, V., Kivirikko, K. I. & Myllyharju, J. Catalytic properties of the asparaginyl hydroxylase (FIH) in the oxygen sensing pathway are distinct from those of its prolyl 4-hydroxylases. J. Biol. Chem. 279, 9899–9904 (2004). References 35 and 36 provided the first evidence that FIH1 might remain active at intermediate levels of hypoxia sufficient to inactivate the Phd family members.
Davidowitz, E., Schoenfeld, A. & Burk, R. VHL induces renal cell differentiation and growth arrest through integration of cell-cell and cell-extracellular matrix signaling. Mol. Cell. Biol. 21, 865–874 (2001).
Pause, A., Lee, S., Lonergan, K. M. & Klausner, R. D. The von Hippel–Lindau tumor suppressor gene is required for cell cycle exit upon serum withdrawal. Proc. Natl Acad. Sci. USA 95, 993–998 (1998).
Iliopoulos, O., Kibel, A., Gray, S. & Kaelin, W. G. Tumor suppression by the human von Hippel–Lindau gene product. Nature Med. 1, 822–826 (1995).
Gnarra, J. R. et al. Post-transcriptional regulation of vascular endothelial growth factor mRNA by the VHL tumor suppressor gene product. Proc. Natl Acad. Sci. USA 93, 10589–10594 (1996).
Lieubeau-Teillet, B. et al. von Hippel–Lindau gene-mediated growth suppression and induction of differentiation in renal cell carcinoma cells grown as multicellular tumor spheroids. Cancer Res. 58, 4957–4962 (1998).
Kondo, K., Klco, J., Nakamura, E., Lechpammer, M. & Kaelin, W. G. Inhibition of HIF is necessary for tumor suppression by the von Hippel–Lindau protein. Cancer Cell 1, 237–246 (2002).
Maranchie, J. K. et al. The contribution of VHL substrate binding and HIF1-α to the phenotype of VHL loss in renal cell carcinoma. Cancer Cell 1, 247–255 (2002).
Raval, R. R. et al. Contrasting properties of hypoxia-inducible factor 1 (HIF-1) and HIF-2 in von Hippel–Lindau-associated renal cell carcinoma. Mol. Cell. Biol. 25, 5675–5686 (2005).
Kondo, K., Kim, W. Y., Lechpammer, M. & Kaelin, W. G. Jr. Inhibition of HIF2α is sufficient to suppress pVHL-defective tumor growth. PLoS Biol. 1, E83 (2003).
Zimmer, M., Doucette, D., Siddiqui, N. & Iliopoulos, O. Inhibition of hypoxia-inducible factor is sufficient for growth suppression of VHL−/− tumors. Mol. Cancer Res. 2, 89–95 (2004).
Hughes, M. D., Kapllani, E., Alexander, A. E., Burk, R. D. & Schoenfeld, A. R. HIF-2α downregulation in the absence of functional VHL is not sufficient for renal cell differentiation. Cancer Cell Int. 7, 13 (2007).
Li, L. et al. Hypoxia-inducible factor linked to differential kidney cancer risk seen with type 2A and type 2B VHL mutations. Mol. Cell. Biol. 27, 5381–5392 (2007).
Clifford, S. et al. Contrasting effects on HIF-1α regulation by disease-causing pVHL mutations correlate with patterns of tumourigenesis in von Hippel–Lindau disease. Hum. Mol. Genet. 10, 1029–1038 (2001).
Hoffman, M. et al. von Hippel–Lindau protein mutants linked to type 2C VHL disease preserve the ability to downregulate HIF. Hum. Mol. Genet. 10, 1019–1027 (2001).
Hu, C. J. et al. Differential regulation of the transcriptional activities if hypoxia-inducible factor 1α (HIF-1α) and HIF-2α in stem cells. Mol. Cell. Biol. 26, 3514–3526 (2006).
Haase, V., Glickman, J., Socolovsky, M. & Jaenisch, R. Vascular tumors in livers with targeted inactivation of the von Hippel–Lindau tumor suppressor. Proc. Natl Acad. Sci. USA 98, 1583–1588 (2001).
Ma, W. et al. Hepatic vascular tumors, angiectasis in multiple organs, and impaired spermatogenesis in mice with conditional inactivation of the VHL gene. Cancer Res. 63, 5320–5328 (2003).
Kim, W. Y. et al. Failure to prolyl hydroxylate hypoxia-inducible factor α phenocopies VHL inactivation in vivo. EMBO J. 25, 4650–4662 (2006).
Rankin, E. B. et al. Inactivation of the arylhydrocarbon receptor nuclear translocator (Arnt) suppresses von Hippel–Lindau disease-associated vascular tumors in mice. Mol. Cell. Biol. 25, 3163–3172 (2005). References 54 and 55 provided in vivo evidence supporting a crucial role of HIF in the development of pathology in mice lacking VHL.
Rankin, E. B. et al. Hypoxia-inducible factor-2 (HIF-2) regulates hepatic erythropoietin in vivo. J. Clin. Invest. 117, 1068–1077 (2007).
Rankin, E. B. et al. Hypoxia-inducible factor-2 regulates vascular tumorigenesis in mice. Oncogene 19 May 2008 (doi: 10.1038/onc.2008.160).
Rankin, E. B., Tomaszewski, J. E. & Haase, V. H. Renal cyst development in mice with conditional inactivation of the von Hippel–Lindau tumor suppressor. Cancer Res. 66, 2576–2583 (2006).
Lei, L. et al. Hypoxia inducible factor-dependent degeneration, failure, and malignant transformation of the heart in the absence of the von Hippel–Lindau protein. Mol. Cell Biol. 19 Feb 2008 (doi:10.1128/MCB.01580-07).
Bracken, C. P. et al. Cell-specific regulation of hypoxia-inducible factor (HIF)-1α and HIF-2α stabilization and transactivation in a graded oxygen environment. J. Biol. Chem. 281, 22575–22585 (2006).
Yan, Q., Bartz, S., Mao, M., Li, L. & Kaelin, W. G. Jr. The hypoxia-inducible factor 2α N-terminal and C-terminal transactivation domains cooperate to promote renal tumorigenesis in vivo. Mol. Cell Biol. 27, 2092–2102 (2007). References 60 and 61 highlight the potential interplay of the two HIFα transactivation domains with respect to gene expression and tumorigenesis
Kong, X. et al. Histone deacetylase inhibitors induce VHL and ubiquitin-independent proteasomal degradation of hypoxia-inducible factor 1α. Mol. Cell. Biol. 26, 2019–2028 (2006).
Kong, X., Alvarez-Castelao, B., Lin, Z., Castano, J. G. & Caro, J. Constitutive/hypoxic degradation of HIF-α proteins by the proteasome is independent of von Hippel Lindau protein ubiquitylation and the transactivation activity of the protein. J. Biol. Chem. 282, 15498–15505 (2007).
Liu, Y. V. et al. RACK1 competes with HSP90 for binding to HIF-1α and is required for O2-independent and HSP90 inhibitor-induced degradation of HIF-1α. Mol. Cell 25, 207–217 (2007).
Isaacs, J. S. et al. Hsp90 regulates a von Hippel Lindau-independent hypoxia-inducible factor-1 α-degradative pathway. J. Biol. Chem. 277, 29936–29944 (2002).
Qian, D. et al. Class II histone deacetylases are associated with VHL-independent regulation of hypoxia-inducible factor 1α. Cancer Res. 66, 8814–8821 (2006).
Covello, K. L. et al. HIF-2α regulates Oct-4: effects of hypoxia on stem cell function, embryonic development, and tumor growth. Genes Dev. 20, 557–570 (2006). This paper illuminated a rather direct link between hypoxia, HIF and stem cell behaviour
Gordan, J. D., Bertout, J. A., Hu, C. J., Diehl, J. A. & Simon, M. C. HIF-2α promotes hypoxic cell proliferation by enhancing c-myc transcriptional activity. Cancer Cell 11, 335–347 (2007).
Zhang, H. et al. HIF-1 inhibits mitochondrial biogenesis and cellular respiration in VHL-deficient renal cell carcinoma by repression of C-MYC activity. Cancer Cell 11, 407–420 (2007).
Papandreou, I., Cairns, R. A., Fontana, L., Lim, A. L. & Denko, N. C. HIF-1 mediates adaptation to hypoxia by actively downregulating mitochondrial oxygen consumption. Cell. Metab. 3, 187–197 (2006).
Kim, J. W., Tchernyshyov, I., Semenza, G. L. & Dang, C. V. HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell. Metab. 3, 177–185 (2006). References 69–71 provide a mechanistic explanation for the Pasteur effect, with high levels of HIF inhibiting entry of pyruvate into the Krebs cycle
Chintalapudi, M. R. et al. Cyr61/CCN1 and CTGF/CCN2 mediate the proangiogenic activity of VHL-mutant renal carcinoma cells. Carcinogenesis 29, 696–703 (2008).
Petrella, B. L., Lohi, J. & Brinckerhoff, C. E. Identification of membrane type-1 matrix metalloproteinase as a target of hypoxia-inducible factor-2 α in von Hippel–Lindau renal cell carcinoma. Oncogene 24, 1043–1052 (2005).
Erler, J. T. et al. Lysyl oxidase is essential for hypoxia-induced metastasis. Nature 440, 1222–1226 (2006).
Staller, P. et al. Chemokine receptor CXCR4 downregulated by von Hippel–Lindau tumour suppressor pVHL. Nature 425, 307–311 (2003).
Zagzag, D. et al. Stromal cell-derived factor-1α and CXCR4 expression in hemangioblastoma and clear cell-renal cell carcinoma: von Hippel–Lindau loss-of-function induces expression of a ligand and its receptor. Cancer Res. 65, 6178–6188 (2005).
Franovic, A. et al. Translational up-regulation of the EGFR by tumor hypoxia provides a nonmutational explanation for its overexpression in human cancer. Proc. Natl Acad. Sci. USA 104, 13092–13097 (2007).
Krishnamachary, B. et al. Hypoxia-inducible factor-1-dependent repression of E-cadherin in von Hippel–Lindau tumor suppressor-null renal cell carcinoma mediated by TCF3, ZFHX1A, and ZFHX1B. Cancer Res. 66, 2725–2731 (2006).
Esteban, M. A. et al. Regulation of E-cadherin expression by VHL and hypoxia-inducible factor. Cancer Res. 66, 3567–3575 (2006).
Evans, A. J. et al. VHL promotes E2 box-dependent E-cadherin transcription by HIF-mediated regulation of SIP1 and snail. Mol. Cell. Biol. 27, 157–169 (2007).
Koochekpour, S. et al. The von Hippel–Lindau tumor suppressor gene inhibits hepatocyte growth factor/scatter factor-induced invasion and branching morphogenesis in renal carcinoma cells. Mol. Cell. Biol. 19, 5902–5912 (1999).
Pennacchietti, S. et al. Hypoxia promotes invasive growth by transcriptional activation of the met protooncogene. Cancer Cell 3, 347–361 (2003).
Hara, S. et al. Hypoxia enhances c-Met/HGF receptor expression and signaling by activating HIF-1α in human salivary gland cancer cells. Oral Oncol. 42, 593–598 (2006).
Hayashi, M. et al. Up-regulation of c-met protooncogene product expression through hypoxia-inducible factor-1α is involved in trophoblast invasion under low-oxygen tension. Endocrinology 146, 4682–4689 (2005).
Nakaigawa, N. et al. Inactivation of von Hippel–Lindau gene induces constitutive phosphorylation of MET protein in clear cell renal carcinoma. Cancer Res. 66, 3699–3705 (2006).
Peruzzi, B., Athauda, G. & Bottaro, D. P. The von Hippel–Lindau tumor suppressor gene product represses oncogenic β-catenin signaling in renal carcinoma cells. Proc. Natl Acad. Sci. USA 103, 14531–14536 (2006).
Isaacs, J. S. et al. HIF overexpression correlates with biallelic loss of fumarate hydratase in renal cancer: novel role of fumarate in regulation of HIF stability. Cancer Cell 8, 143–153 (2005). This paper showed that inactivation of fumarate hydratase, as seen in some papillary renal cancers, leads to accumulation of fumarate, which then inhibits PHD2 function.
Ryan, H., Lo, J. & Johnson, R. HIF-1α is required for solid tumor formation and embryonic vascularization. EMBO J. 17, 3005–3015 (1998).
Carmeliet, P. et al. Role of HIF-1α in hypoxia-mediated apoptosis, cell proliferation and tumour angiogenesis. Nature 394, 485–490 (1998).
Maxwell, P. et al. Hypoxia-inducible factor-1 modulates gene expression in solid tumors and influences both angiogenesis and tumor growth. Proc. Natl Acad. Sci. USA 94, 8104–8109 (1997).
Blouw, B. et al. The hypoxic response of tumors is dependent on their microenvironment. Cancer Cell 4, 133–146 (2003).
Mack, F. A. et al. Loss of pVHL is sufficient to cause HIF dysregulation in primary cells but does not promote tumor growth. Cancer Cell 3, 75–88 (2003).
Semenza, G. L. Targeting HIF-1 for cancer therapy. Nature Rev. Cancer 3, 721–732 (2003).
Yoder, B. K. Role of primary cilia in the pathogenesis of polycystic kidney disease. J. Am. Soc. Nephrol. 18, 1381–1388 (2007).
Singla, V. & Reiter, J. F. The primary cilium as the cell's antenna: signaling at a sensory organelle. Science 313, 629–633 (2006).
Lutz, M. S. & Burk, R. D. Primary cilium formation requires von Hippel–Lindau gene function in renal-derived cells. Cancer Res. 66, 6903–6907 (2006).
Esteban, M. A., Harten, S. K., Tran, M. G. & Maxwell, P. H. Formation of primary cilia in the renal epithelium is regulated by the von Hippel–Lindau tumor suppressor protein. J. Am. Soc. Nephrol. 17, 1801–1806 (2006).
Thoma, C. R. et al. pVHL and GSK3β are components of a primary cilium-maintenance signalling network. Nature Cell Biol. 9, 588–595 (2007).
Schermer, B. et al. The von Hippel–Lindau tumor suppressor protein controls ciliogenesis by orienting microtubule growth. J. Cell Biol. 175, 547–554 (2006). References 96–99 were the first to report that VHL loss leads to loss of the primary cilium.
Lolkema, M. P. et al. Allele-specific regulation of primary cilia function by the von Hippel–Lindau tumor suppressor. Eur. J. Hum. Genet. 16, 73–78 (2008).
Hergovich, A., Lisztwan, J., Barry, R., Ballschmieter, P. & Krek, W. Regulation of microtubule stability by the von Hippel–Lindau tumour suppressor protein pVHL. Nature Cell Biol. 5, 64–70 (2003).
Lolkema, M. P. et al. The von Hippel–Lindau tumour suppressor interacts with microtubules through kinesin-2. FEBS Lett. 581, 4571–4576 (2007).
Frew, I. J. et al. pVHL and PTEN tumour suppressor proteins cooperatively suppress kidney cyst formation. EMBO J. 27, 1747–1757 (2008).
Frew, I. J. et al. Combined VHLH and PTEN mutation causes genital tract cystadenoma and squamous metaplasia. Mol. Cell. Biol. 28, 4536–4548 (2008).
Bishop, T. et al. Genetic analysis of pathways regulated by the von Hippel–Lindau tumor suppressor in Caenorhabditis elegans. PLoS Biol. 2, e289 (2004). This paper provides defintive genetic evidence for a HIF-independent role of VHL in control of the extracellular matrix.
Grosfeld, A. et al. Interaction of hydroxylated collagen IV with the von Hippel–Lindau tumor suppressor. J. Biol. Chem. 282, 13264–13269 (2007).
Kurban, G. et al. Collagen matrix assembly is driven by the interaction of von Hippel-Lindau tumor suppressor protein with hydroxylated collagen IV α 2. Oncogene 27, 1004–1012 (2008).
Ohh, M. et al. The von Hippel-Lindau tumor suppressor protein is required for proper assembly of an extracellular fibronectin matrix. Mol. Cell 1, 959–968 (1998).
Tang, N., Mack, F., Haase, V. H., Simon, M. C. & Johnson, R. S. pVHL function is essential for endothelial extracellular matrix deposition. Mol. Cell Biol. 26, 2519–2530 (2006).
Kurban, G., Hudon, V., Duplan, E., Ohh, M. & Pause, A. Characterization of a von Hippel Lindau pathway involved in extracellular matrix remodeling, cell invasion, and angiogenesis. Cancer Res. 66, 1313–1319 (2006).
Stickle, N. H. et al. pVHL modification by NEDD8 is required for fibronectin matrix assembly and suppression of tumor development. Mol. Cell Biol. 24, 3251–3261 (2004).
Russell, R. C. & Ohh, M. NEDD8 acts as a 'molecular switch' defining the functional selectivity of VHL. EMBO Rep. 9, 486–491 (2008).
Schoenfeld, A., Davidowitz, E. & Burk, R. Endoplasmic reticulum/cytosolic localization of von Hippel–Lindau gene products is mediated by a 64-amino acid region. Int. J. Cancer 91, 457–467 (2001).
Esteban-Barragan, M. et al. Role of the von Hippel–Lindau tumor suppressor gene in the formation of β1-integrin fibrillar adhesions. Cancer Res. 62, 2929–2936 (2002).
Nakamura, E. & Kaelin, W. G. Jr. Recent insights into the molecular pathogenesis of pheochromocytoma and paraganglioma. Endocr. Pathol. 17, 97–106 (2006).
Vogel, K. S., Brannan, C. I., Jenkins, N. A., Copeland, N. G. & Parada, L. F. Loss of neurofibromin results in neurotrophin-independent survival of embryonic sensory and sympathetic neurons. Cell 82, 733–742 (1995).
Lee, S. et al. Neuronal apoptosis linked to EglN3 prolyl hydroxylase and familial pheochromocytoma genes: Developmental culling and cancer. Cancer Cell 8, 155–167 (2005). This paper placed PHD3 (EGLN3) on a neuronal survival pathway that is crucial during embyrogenesis and linked disruptions of this pathway to various familial paraganglioma genes
Okuda, H. et al. The von Hippel–Lindau tumor suppressor protein mediates ubiquitination of activated atypical protein kinase C. J. Biol. Chem. 276, 43611–43617 (2001).
Rantanen, K. et al. Prolyl hydroxylase PHD3 activates oxygen-dependent protein aggregation. Mol. Biol. Cell 19, 2231–2240 (2008).
Schlisio, S. et al. The kinesin KIF1Bβ acts downstream from EglN3 to induce apoptosis and is a potential 1p36 tumor suppressor. Genes Dev. 22, 884–893 (2008). This paper showed that KIF1Bβ is necessary and sufficient for PHD3-induced apoptosis.
Bishop, T. et al. Abnormal sympathoadrenal development and systemic hypotension in PHD3−/− mice. Mol. Cell. Biol. 28, 3386–3400 (2008).
Munirajan, A. K. et al. KIF1B functions as a haploinsufficient tumor suppressor gene mapped to chromosome 1p36.2 by inducing apoptotic cell death. J. Biol. Chem. 283, 24426–24434 (2008). References 120 and 122 showed that KIF1Bβ has the properties of a haploinsufficient tumour suppressor.
Yang, H. et al. pVHL acts as an adaptor to promote the inhibitory phosphorylation of the NF-κB agonist Card9 by CK2. Mol. Cell 28, 15–27 (2007).
Roe, J. S. et al. p53 stabilization and transactivation by a von Hippel–Lindau protein. Mol. Cell 22, 395–405 (2006).
Mukhopadhyay, D., Knebelmann, B., Cohen, H., Ananth, S. & Sukhatme, V. The von Hippel–Lindau tumor suppressor gene product interacts with Sp1 to repress vascular endothelial growth factor promoter activity. Mol. Cell. Biol. 17, 5629–5639 (1997).
Cohen, H. et al. An important von Hippel–Lindau tumor suppressor domain mediates Sp1-binding and self-association. Biochem. Biophys. Res. Commun. 266, 43–50 (1999).
Rafty, L. A. & Khachigian, L. M. von Hippel-Lindau tumor suppressor protein represses platelet-derived growth factor B-chain gene expression via the Sp1 binding element in the proximal PDGF-B promoter. J. Cell Biochem. 85, 490–495 (2002).
Iliopoulos, O., Jiang, C., Levy, A. P., Kaelin, W. G. & Goldberg, M. A. Negative regulation of hypoxia-inducible denes by the von Hippel–Lindau protein. Proc. Natl. Acad. Sci. USA 93, 10595–10599 (1996).
Datta, K. et al. Role of elongin-binding domain of von Hippel Lindau gene product on HuR-mediated VPF/VEGF mRNA stability in renal cell carcinoma. Oncogene 24, 7850–7858 (2005).
Yuen, J. S. et al. The VHL tumor suppressor inhibits expression of the IGF1R and its loss induces IGF1R upregulation in human clear cell renal carcinoma. Oncogene 26, 6499–6508 (2007).
Kuznetsova, A. V. et al. von Hippel–Lindau protein binds hyperphosphorylated large subunit of RNA polymerase II through a proline hydroxylation motif and targets it for ubiquitination. Proc. Natl Acad. Sci. USA 100, 2706–2711 (2003).
Mikhaylova, O. et al. The von Hippel–Lindau tumor suppressor protein and Egl-9-Type proline hydroxylases regulate the large subunit of RNA polymerase II in response to oxidative stress. Mol. Cell Biol. 28, 2701–2717 (2008).
Motzer, R. J. et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N. Engl. J. Med. 356, 115–124 (2007).
Escudier, B. et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N. Engl. J. Med. 356, 125–134 (2007).
Rixe, O. et al. Axitinib treatment in patients with cytokine-refractory metastatic renal-cell cancer: a phase II study. Lancet Oncol. 8, 975–984 (2007).
Escudier, B. et al. Bevacizumab plus interferon alfa-2a for treatment of metastatic renal cell carcinoma: a randomised, double-blind phase III trial. Lancet 370, 2103–2111 (2007).
Brugarolas, J. B., Vazquez, F., Reddy, A., Sellers, W. R. & Kaelin, W. G. Jr. TSC2 regulates VEGF through mTOR-dependent and -independent pathways. Cancer Cell 4, 147–158 (2003).
Arsham, A. M., Howell, J. J. & Simon, M. C. A novel hypoxia-inducible factor-independent hypoxic response regulating mammalian target of rapamycin and its targets. J. Biol. Chem. 278, 29655–29660 (2003).
Zhong, H. et al. Modulation of hypoxia-inducible factor 1α expression by the epidermal growth factor/phosphatidylinositol 3-kinase/PTEN/AKT/FRAP pathway in human prostate cancer cells: implications for tumor angiogenesis and therapeutics. Cancer Res. 60, 1541–1545 (2000).
Hudson, C. C. et al. Regulation of hypoxia-inducible factor 1α expression and function by the mammalian target of rapamycin. Mol. Cell. Biol. 22, 7004–7014 (2002).
Thomas, G. V. et al. Hypoxia-inducible factor determines sensitivity to inhibitors of mTOR in kidney cancer. Nature Med. 12, 122–127 (2006).
Phung, T. L. et al. Endothelial Akt signaling is rate-limiting for rapamycin inhibition of mouse mammary tumor progression. Cancer Res. 67, 5070–5075 (2007).
Chan, J., Bayliss, P. E., Wood, J. M. & Roberts, T. M. Dissection of angiogenic signaling in zebrafish using a chemical genetic approach. Cancer Cell 1, 257–267 (2002).
Hudes, G. et al. A phase 3, randomized, 3-arm study of temsirolimus (TEMSR) or interferon-α (IFN) or the combination of TEMSR + IFN in the treatment of first-line, poor-risk patients with advanced renal cell carcinoma (adv RCC). J. Clin. Oncol. 24, abstract LBA4 (2006).
Motzer, R. J. et al. RAD001 vs placebo in patients with metastatic renal cell carcinoma (RCC) after progression on VEGFr-TKI therapy: Results from a randomized, double-blind, multicenter Phase-III study. J. Clin. Oncol. 26, abstract LBA5026 (2008).
Acknowledgements
W.G.K. is supported by the Doris Duke Charitable Foundation as a Distinguished Clinical Scientist and by the National Institutes of Health, the Howard Hughes Medical Institute and the Breast Cancer Research Foundation.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
W.G.K. owns equity in, and consults for, Fibrogen, Inc. (South San Francisco, California, USA). Fibrogen, Inc. is developing drugs targeting prolyl hydroxylases.
Related links
Related links
DATABASES
OMIM
FURTHER INFORMATION
Rights and permissions
About this article
Cite this article
Kaelin Jr, W. The von Hippel–Lindau tumour suppressor protein: O2 sensing and cancer. Nat Rev Cancer 8, 865–873 (2008). https://doi.org/10.1038/nrc2502
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc2502
This article is cited by
-
The nerve cells in gastrointestinal cancers: from molecular mechanisms to clinical intervention
Oncogene (2024)
-
Signaling pathways in cancer metabolism: mechanisms and therapeutic targets
Signal Transduction and Targeted Therapy (2023)
-
Biology and therapeutic targeting of vascular endothelial growth factor A
Nature Reviews Molecular Cell Biology (2023)
-
PIN1 and CDK1 cooperatively govern pVHL stability and suppressive functions
Cell Death & Differentiation (2023)
-
Final efficacy and safety results and biomarker analysis of a phase 2 study of cabozantinib in Japanese patients with advanced renal cell carcinoma
International Journal of Clinical Oncology (2023)