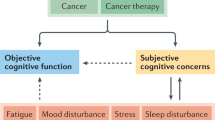
Key Points
-
Evidence for chemotherapy-induced cognitive changes comes from studies that have used neuropsychological testing, imaging (structural and functional magnetic resonance imaging (MRI) and positron emission tomography (PET)) and electrophysiological (electroencephalogram) assessments. Emerging data from animal studies also support the effect of chemotherapy on cognitive function.
-
Most chemotherapy agents administered systemically do not cross the blood–brain barrier in significant doses; however, the amount that enters the brain can be modified by genetic variability in blood–brain barrier transporters. In addition, recent data from animal studies suggest that very small doses of common chemotherapy agents can cause cell death and reduced cell division in brain structures crucial for cognition, even at doses that do not effectively kill tumour cells.
-
Chemotherapy might cause cognitive changes through DNA damage caused directly by the cytotoxic agents or through increases in oxidative stress. Many chemotherapeutic agents also cause the shortening of telomeres, thereby accelerating cell ageing. Genetic variability in DNA-repair genes might influence the extent of, and recovery from, chemotherapy-associated DNA damage.
-
Chemotherapy-induced cognitive changes might also be related to the neurotoxic effects of cytokine deregulation. Cytokine deregulation and inflammation can also lead to increased oxidative stress, which could establish a cycle of increased DNA damage that triggers additional cytokine release.
-
Variability in genes that regulate neural repair and/or plasticity, such as apolipoprotein E (APOE) and brain-derived neurotrophic factor (BDNF), and neurotransmission, such as catechol-O-methyltransferase (COMT), might increase the vulnerability of an individual to chemotherapy-induced cognitive changes.
-
Changes in levels of oestrogen and testosterone are associated with cognitive decline, and can be influenced by chemotherapy (for example, chemotherapy-induced menopause) or hormonal treatments, such as tamoxifen or aromatase inhibitors for breast cancer or androgen ablation for prostate cancer.
-
The effects of chemotherapy-associated cardiovascular toxicity and alterations in neuroendocrine function on cognitive function require investigation.
Abstract
The mechanism(s) for chemotherapy-induced cognitive changes are largely unknown; however, several candidate mechanisms have been identified. We suggest that shared genetic risk factors for the development of cancer and cognitive problems, including low-efficiency efflux pumps, deficits in DNA-repair mechanisms and/or a deregulated immune response, coupled with the effect of chemotherapy on these systems, might contribute to cognitive decline in patients after chemotherapy. Furthermore, the genetically modulated reduction of capacity for neural repair and neurotransmitter activity, as well as reduced antioxidant capacity associated with treatment-induced reduction in oestrogen and testosterone levels, might interact with these mechanisms and/or have independent effects on cognitive function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Silberfarb, P. M. Chemotherapy and cognitive defects in cancer patients. Annu. Rev. Med. 34, 35–46 (1983).
Wieneke, M. H. & Dienst, E. R. Neuropsychological assessment of cognitive functioning following chemotherapy for breast cancer. Psychooncology 4, 61–66 (1995).
van Dam, F. S. et al. Impairment of cognitive function in women receiving adjuvant treatment for high-risk breast cancer: high-dose versus standard-dose chemotherapy. [comment]. J. Natl Cancer Inst. 90, 210–218 (1998).
Schagen, S. B. et al. Cognitive deficits after postoperative adjuvant chemotherapy for breast carcinoma. Cancer 85, 640–650 (1999).
Brezden, C. B. et al. Cognitive function in breast cancer patients receiving adjuvant chemotherapy. J. Clin. Oncol. 18, 2695–2701 (2000).
Ahles, T. A. et al. Neuropsychological impact of standard-dose chemotherapy in long-term survivors of breast cancer and lymphoma. J. Clin. Oncol. 20, 485–493 (2002).
Tchen, M. et al. Cognitive function, fatigue, and menopausal symptoms in women receiving adjuvant chemotherapy for breast cancer. J. Clin. Oncol. 21, 4175–4183 (2003).
Castellon, S. A. et al. Neurocognitive performance in breast cancer survivors exposed to adjuvant chemotherapy and tamoxifen. J. Clin. Exp. Neuropsychol. 26, 955–969 (2004).
Wefel, J. S. et al. The cognitive sequelae of standard-dose adjuvant chemotherapy in women with breast carcinoma: results of a prospective, randomized, longitudinal trial. Cancer 100, 2292–2299 (2004).
Schilling, V. et al. The effects of adjuvant chemotherapy on cognition in women with breast cancer — preliminary results of an observational study. The Breast 14, 142–150 (2005).
Jenkins, V. et al. A 3-year prospective study of the effects of adjuvant treatments on cognition in women with early stage breast cancer. Br. J. Cancer 94, 828–834 (2006).
Donovan, K. A. et al. Cognitive functioning after adjuvant chemotherapy and/or radiotherapy for early stage breast carcinoma. Cancer 104, 2499–2507 (2005).
Tannock, I. F., Ahles, T. A., Ganz, P. A. & van Dam, F. S. Cognitive impairment associated with chemotherapy for cancer: report of a workshop. J. Clin. Oncol. 22, 2233–2239 (2004). This paper provides an overview of a consensus conference that included most of the researchers from around the world who were conducting research on chemotherapy-induced cognitive changes.
Ahles, T. A. & Saykin, A. J. Breast cancer chemotherapy-related cognitive dysfunction. Clin. Breast Cancer Suppl. 3, S84–S90 (2002).
Ferguson, R. J. & Ahles, T. A. Low neuropsychologic performance among adult cancer survivors treated with chemotherapy. Curr. Neurol. Neurosci. Rep. 3, 215–222 (2003).
Anderson-Hanley, C., Sherman, M. L., Riggs, R. Agocha, V. V. & Compas, B. E. Neuropsychological effects of treatments for adults with cancer: A meta-analysis and review of the literature. J. Int. Neuropsychol. Soc. 9, 967–982 (2003).
Saykin, A. J., Ahles, T. A. & McDonald, B. C. Mechanisms of chemotherapy-induced cognitive disorders: neuropsychological, pathophysiological and neuroimaging perspectives. Sem. Clin. Neuropsych. 8, 201–216 (2003).
Stemmer, S. et al. White matter changes in patients with breast cancer treated with high-dose chemotherapy and autologous bone marrow support. Am. J. Neuroradiol. 15, 1267–1273 (1994).
Saykin, A. J. et al. Altered brain activation following systemic chemotherapy for breast cancer: interim analysis from a prospective study. J. Int. Neuropsychol. Soc. 12, 131 (2006).
Kreukels, B. P. et al. Electrophysiological correlates of information processing in breast-cancer patients treated with chemotherapy. Breast Cancer Res. Treat. 94, 53–61 (2005).
Kreukels, B. P. et al. Effects of high-dose and conventional-dose adjuvant chemotherapy on long-term cognitive sequelae in patients with breast cancer: an electrophysiologic study. Clin. Breast Cancer 7, 67–78 (2006).
Wefel, J. S. et al. Chemobrain in breast carcinoma? A prologue. Cancer 101, 466–475 (2004).
Meyers, C. A., Albitar M. & Estey, E. Cognitive impairment, fatigue, and cytokine levels in patients with acute myelogenous leukemia or myelodysplastic syndrome. Cancer 104, 788–793 (2005).
Ahles, T. A. et al. Psychological and neuropsychological functioning of patients with limited small-cell lung cancer treated with chemotherapy and radiation therapy with or without warfarin, a study for Cancer and Leukemia Group B. J. Clin. Oncol. 16, 1954–1960 (1998).
Wagner, L. I. et al. Trajectory of cognitive impairment during breast cancer treatment: a prospective analysis. J. Clin. Oncol. Suppl. 24, 8500 (2006).
Heflin, L. H. et al. Cancer as a risk factor for long-term cognitive deficits and dementia. J. Natl Cancer Inst. 97, 854–856 (2005).
Verstappen C. C. P., Heimans, J. J., Hoekman, K. & Postma T. J. Neurotoxic complications of chemotherapy in patients with cancer: Clinical signs and optimal management. Drugs 63, 1549–1563 (2003).
Troy, L. et al. Cisplatin-based therapy: A neurological and neuropsychological review. Psychooncology 9, 29–39 (2000).
Ginos J. Z. et al. [13N]cisplatin PET to assess pharmacokinetics of intro-arterial versus intravenous chemotherapy for malignant brain tumors. J. Nucl. Med. 28, 1844–1852 (1987).
Mitsuki, S. et al. Pharmacokinetics of 11C-labelled BCNU and SARCNU in gliomas studied by PET. J.Neurooncol. 10, 47–55 (1991).
Gangloff, A. et al. Estimation of paclitaxel biodistribution and uptake in human-derived xenografts in vivo with 18F-Fluoropaclitaxel. J. Nucl. Med. 46, 1866–1871 (2005).
Dietrich, J., Han, R., Yang, Y., Mayer-Proschel, M. & Noble, M. CNS progenitor cells and oligodendrocytes are targets of chemotherapeutic agents in vitro and in vivo. J. Biol. 5, 22 [Epub ahead of print] (2006). This is an excellent paper describing in vitro and in vivo (mice) studies showing that common chemotherapy agents caused increased cell death and decreased cell division in the subventricular zone and in the dentate gyrus of the hippocampus, and in the corpus callosum. These effects were seen with doses that were not effective in causing cell death in tumor cell lines.
Jamroziak, K. & Robak, T. Pharmacogenomics of MDR1/ABCB1 gene: the influence on risk and clinical outcome of haematological malignancies. Hematology 9, 91–105 (2004).
Hoffmeyer, S. et al. Functional polymorphisms of the human multidrug-resistance gene: multiple sequence variations and correlation of one allele with P-glycoprotein expression and activity in vivo. Proc. Natl Acad. Sci. USA 97, 3473–3478 (2000).
Kreb, R. Implications of genetic polymorphisms in drug transporters for pharmacotherapy. Cancer Lett. 234, 4–33 (2006). This manuscript describes evidence for genetic variability in drug transporters and their influence on drug disposition and clinical response.
Muramatsu, T. et al. Age-related differences in vincristine toxicity and biodistribution in wild-type and transporter-deficient mice. Oncol. Res. 14, 331–343 (2004).
Uhr, M., Holsboer, F. & Muller, M. B. Penetration of endogenous steroid hormones corticosterone, cortisol, aldosterone and progesterone into the brain is enhanced in mice deficient for both mdr1a and mdr1b p-glycoproteins. J. Neuroendocrinol. 14, 753–759 (2002).
Rolig, R. L. & McKinnon, P. J. Linking DNA damage and neurodegeneration. Trends Neurosci. 23, 417–424 (2000). This paper provides an overview of the evidence linking DNA damage to neurodegeneration and cognitive function.
Goode, E. L., Ulrich, C. M. & Potter, J. D. Polymorphisms in DNA repair genes and associations with cancer risk. Cancer Epidemiol. Biomarkers Prev. 11, 1513–1530 (2002).
Caldecott, K. W. DNA single-strand breaks and neurodegeneration. DNA Repair 3, 875–882 (2004).
Abner, C. W. & McKinnon, P. J. The DNA double-strand break response in the nervous system. DNA Repair 3, 1141–1147 (2004).
Fishel, M. L., Vasko, M. R. & Kelley, M. R. DNA repair in neurons: so if they don't divide what's to repair? Mutat. Res. 614, 24–36 (2007). This paper reviews the relevance of DNA-repair pathways to DNA damage in post-mitotic neurons, and the impact of DNA damage on neuronal survival and brain ageing. Additionally, these authors relate DNA repair to neurotoxicity associated with chemotherapy, including cognitive side effects and peripheral neuropathy.
Sedletska, Y., Giraud-Panis, M.-J. & Malinge, J.-M. Cisplatin is a DNA-damaging antitumour coumpound triggering multifactorial biochemical responses in cancer cells: Importance of apoptotic pathways. Curr. Med. Chem. Anticancer Agents 5, 251–265 (2005).
Blasiak J. et al. Basal, oxidative and alkylative DNA damage, DNA repair efficacy and mutagen sensitivity in breast cancer. Mutat. Res. 554, 139–148 (2004). Using the comet assay, these investigators showed greater DNA damage and lower DNA repair efficacy in patients with breast cancer, both before and after chemotherapy.
Nadin S. B., Vargas-Roig, L. M., Drago, G., Ibarra, J. & Ciocca, D. R. DNA damage and repair in peripheral blood lymphocytes from healthy individuals and cancer patients: a pilot study on the implications in response to chemotherapy. Cancer Lett. 239, 84–87 (2006).
Wardell, T. M. et al. Changes in human mitochondrial genome after treatment of malignant disease. Mutat. Res. 525, 19–27 (2003).
Weijl, N. I. et al. Non-protein bound iron release during chemotherapy in cancer patients. Clin. Sci. 106, 475–484 (2004).
Kaya, E. et al. Oxidant/antioxidant parameters and their relationship with chemotherapy in Hodgkin's lymphoma. J. Int. Med. Res. 33, 687–692 (2005).
Papageorgiou, M. et al. Cancer chemotherapy reduces plasma total antioxidant capacity in children with malignancies. Leukemia Res. 29, 11–16 (2005).
Kennedy, D. D., Ladas, E. J., Rheingold, S. R., Blumberg, J. & Kelly, K. M. Antioxidant status decreases in children with acute lymphoblastic leukemia during the first six months of chemotherapy treatment. Ped. Blood Cancer 44, 378–385 (2005).
Mariani, E., Polidori, M. C., Cherubini, A. & Mecocci, P. Oxidative stress in brain aging, neurodegenerative and vascular diseases: an overview. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 827, 65–75 (2005).
Migliore, L. et al. Oxidative DNA damage in peripheral leukocytes of mild cognitive impairment and AD patients. Neurobiol. Aging 26, 567–573 (2005). The results of this study showed that patients with mild cognitive impairment, a condition characterized by a relatively isolated impairment in memory in the context of normal functioning in other areas, had higher levels of DNA damage compared with older adults without mild cognitive impairment.
Keller, J. N. et al. Evidence of increased oxidative damage in subjects with mild cognitive impairment. Neurology 64, 1152–1156 (2005). This study showed that patients who had been diagnosed with mild cognitive impairment had elevated levels of DNA damage in the brain at autopsy, suggesting a relationship between DNA damage seen peripherally in this population and DNA damage in the CNS.
Harrison, J. F. et al. Oxidative stress-induced apoptosis in neurons correlates with mitcochondrial DNA base exicision repair pathway imbalance. Nucleic Acids Res. 33, 4660–4671 (2005).
Vajdovich, P. et al. Redox status in dogs with non-hodgkin lymphomas. An ESR study. Cancer Lett. 224, 339–346 (2005).
Hung, R., Hall, J., Brennan, P. & Boffetta, P. Genetic polymorphisms in the base excision repair pathway and cancer risk: A HuGE review. Am. J. Epidemiol. 162, 925–942 (2005).
von Zglinicki, T. & Martin-Ruiz, C. M. Telomeres as biomarkers for ageing and age-related diseases. Curr. Mol. Med. 5, 197–203 (2005).
Vasa-Nicotera, M. et al. Mapping of a major locus that determines telomere length in humans. Am. J. Hum. Genet. 76, 147–151 (2005).
Schroder, C. P. et al. Telomere length in breast cancer patients before and after chemotherapy with or without stem cell transplation. Br. J. Cancer 84, 1348–1353 (2001).
Lahav, M. et al. Nonmyeloablative conditioning does not prevent telomere shortening after allogeneic stem cell transplantation. Transplantation 80, 969–976 (2005).
Maccormick, R. E. Possible acceleration of aging by adjuvant chemotherapy: a cause of early onset frailty? Med. Hypotheses 67, 212–215 (2006). This manuscript reviews the evidence that morbidity associated with chemotherapy might be related to acceleration of the ageing process.
Flanary, B. E. & Streit, W. J. Progressive telomere shortening occurs in cultured rat microglia, but not astrocytes. Glia 45, 75–88 (2004).
Wilson, C. J., Finch, C. E. & Cohen H. J. Cytokines and cognition — the case for head-to-toe inflammatory paradigm. J. Am. Geriatr. Soc. 50, 2041–2056 (2002).
Tonelli, L. H., Postolache, T. T. & Sternberg, E. M. Inflammatory genes and neural activity: involvement of immune genes in synaptic function and behavior. Front. Biosci. 10, 675–680 (2005).
Cleeland, C. S. et al. Are the symptoms of cancer and cancer treatment due to a shared biologic mechanism? A cytokine-immunologic model of cancer symptoms. Cancer 97, 2919–2925 (2003).
Kelley, K. W. et al. Cytokine-induced sickness behavior. Brain Behav. Immun. 17, S112–S118 (2003)
Maier, S. F. & Watkins L. R. Immune-to-central nervous system communication and its role in modulating pain and cognition: Implications for cancer and cancer treatments. Brain Behav. Immun. 17, S125–S131 (2003).
Reichenberg, A. et al. Cytokine-associated emotional and cognitive disturbances in humans. Arch. Gen. Psych. 58, 445–452 (2001).
Krabbe, K. S. et al. Low-dose endotoxemia and human neuropsychological functions. Brain Behav. Immun. 19, 453–460 (2005).
Trask, P. C., Esper, P., Riba, M. & Redman, B. Psychiatric side effects of interferon therapy: prevalence, proposed mechanisms, and future directions. J. Clin. Oncol. 18, 2316–2326 (2000).
Scheibel, R. S., Valentine, A. D., O'Brien, S. & Meyers, C. A. Cognitive dysfunction and depression during treatment with interferon-α and chemotherapy. J.Neuropsych. Clin. Neurosci. 16, 185–191 (2004).
Capuron, L., Ravaud, A. & Dantzer, R. Timing and specificity of the cognitive changes induced by interleukin-2 and interferon-α treatments in cancer patients. Psychosom. Med. 63, 376–386 (2001).
Tsavaris, N., Kosmas, C., Vadiaka, M., Kanelopoulos, P. & Boulamatsis, D. Immune changes in patients with advanced breast cancer undergoing chemotherapy with taxanes. Br. J. Cancer 87, 21–27 (2002).
Pusztai, L. et al. Changes in plasma levels of inflammatory cytokines in response to paclitaxel chemotherapy. Cytokine 25, 94–102 (2004).
Penson, R. T. et al. Chtokines IL-1β, IL-2, IL-6, IL-8, MCP-1, GM-CSF and TNFα in patients with epithelial ovarian cancer and their relationship to treatment with paclitaxel. Int. J. Gynecol. Cancer 10, 33–41 (2000).
Callado-Hidalgo, A., Bower, J. E., Ganz, P. A., Cole, S. W. & Irwin, M. R. Inflammatory biomarkers for persistent fatigue in breast cancer survivors. Clin. Cancer Res. 12, 2759–2766
Bower, J. E., Ganz, P. A., Aziz, N., Fahey, J. L. Fatigue and proinflammatory cytokine activity in breast cancer survivors. Psychosom. Med. 64, 604–611 (2002).
Balkwill, F. & Mantovani, A. Inflammation and cancer: back to Virchow? Lancet 357, 539–545 (2001).
de Visser, K. E., Eichten, A. & Coussens, L. M. Paradoxical roles of the immune system during cancer development. Nature Rev. Cancer 6, 24–37 (2006). This paper presents evidence in support of a role for chronic inflammation in enhancing the predisposition to develop cancer. Furthermore, the authors present evidence that genetic polymorphisms that regulate immune function can affect cancer risk.
McGeer, P. L. & McGeer, E. G. Polymorphisms in inflammatory genes and risk of Alzheimer Disease. Arch. Neurol. 58, 1790–1792 (2001).
Morley, K. I. & Montgomery, G. W. The genetics of cognitive processes: candidate genes in humans and animals. Behav. Genet. 31, 511–531 (2001).
Chen, Y. et al. Motor and cognitive deficits in apolipoprotein E-deficient mice after closed head injury. Neuroscience 80, 1255–1262 (1997).
Hartman, R. E. et al. Apolipoprotein E4 influences amyloid deposition but not cell loss after traumatic brain injury in a mouse model of Alzheimer's disease. J. Neurosci. 22, 10083–10087 (2002).
Sabo, T. et al. Susceptibility of transgenic mice expressing human apolipoprotein E to closed head injury: the allele E3 is neuroprotective whereas E4 increases fatalities. Neuroscience 101, 879–884 (2000).
Laws, S. M. et al. APOE-epsilon4 and APOE-491A polymorphisms in individuals with subjective memory loss. Mol. Psych. 7, 768–775 (2002).
Nathoo, N. et al. Genetic vulnerability following traumatic brain injury: the role of apolipoprotein E. Mol. Pathol. 56, 132–136 (2003).
Ahles, T. A., Saykin, A. J. & Noll, W. W. et al. The relationship of APOE genotype to neuropsychological performance in long-term cancer survivors treated with standard dose chemotherapy. Psychooncology 12, 612–619 (2003) This is the first study to show an association between APOE genotype and cognitive functioning in long-term cancer survivors.
Lind, J. et al. Reduced hippocampal volume in non-demented carriers of the apolipoprotein E ɛ4: relation to chronological age and recognition memory. Neurosci. Lett. 396, 23–27 (2006).
Pang, P. T. & Lu, B. Regulation of late-phase LTP and long-term memory in normal and ageing hippocampus: role of secreted proteins tPA and BDNF. Age. Res. Rev. 3, 407–430 (2004).
Savitz, J., Solms, M. & Ramesar, R. The molecular genetics of cognition: dopamine, COMT, and BDNF. Genes Brain Behav. 5, 311–328 (2006). This paper provides an excellent review of the research examining the relationship between genetic variability and cognitive functioning with an emphasis on dopamine receptor genes, catechol-O-methyltransferase ( COMT ) and brain-derived neurotrophic factor ( BDNF).
Egan, M. F. et al. The BDNF val66met polymorphism affects activity-dependent secretion of BDNF and human memory and hippocampal function. Cell 112, 257–269 (2003).
Hariri, A. R. et al. Brain-derived neurotrophic factor val66met polymorphism affects human memory-related hippocampal activity and predicts memory performance. J. Neurosci. 23, 6690–6694 (2003).
Pezawas, L. et al. The brain-derived neurotrophic factor val66met polymorphism and variation in human cortical morphology. J. Neurosci. 24, 10099–10102 (2004).
McAllister, T. W. et al. Cognitive effects of cytotoxic cancer chemotherapy: predisposing risk factors and potential treatments. Curr. Psych. Rep. 6, 364–371 (2004).
Weinberger, D. R. et al. Prefrontal neurons and the genetics of schizophrenia. Biol. Psychiatry 50, 825–844 (2001).
Malhotra, A. K. et al. A functional polymorphism in the COMT gene and performance on a test of prefrontal cognition. Am. J. Psych. 159, 652–654 (2001).
Zec, R. F. & Trivedi, M. A. The effects of estrogen replacement therapy on neuropsychological functioning in postmenopausal women with and without dementia: a critical and theoretical review. Neuropsychol. Rev. 12, 65–109 (2002).
Bender, D. M., Paraska, K. K., Sereika, S. M., Ryan, C. M. & Berga, S. L. Cognitive function and reproductive hormones in adjuvant therapy for breast cancer: a critical review. J. Pain Sympt. Manage. 21, 407–424 (2001).
Jenkins, V. A., Bloomfield, D. J., Shilling, V. M. & Edginton, T. L. Does neoadjuvant hormone therapy for early prostate cancer affect cognition? Results from a pilot study. Br. J. Urol. 96, 48–53 (2005).
Unfer, T. C. et al. Influence of hormone replacement therapy on blood antioxidant enzymes in menopausal women. Clin. Chim. Acta 369, 73–77 (2006).
Chisu, V., Lepore, M. G., Zedda, M. & Farina, V. Testosterone induces neuroprotection from oxidative stress. Effects on catalase activity and 3-Nitro-L-styrosine incorporation into α-tubulin in a mouse neuroblastoma cell line. Arch. Ital. Biol. 144, 63–73 (2006).
Lee, D.-C., Im, J.-A., Kim, J.-H., Lee, H.-R. & Shim, J.-Y. Effect of long-term hormone therapy on telomere length in postmenopausal women. Yonsei Med. J. 46, 471–479 (2005).
Theodoulou M. & Seidman, A. D. Cardiac effects of adjuvant therapy for early breast cancer. Semin. Oncol. 30, 730–739 (2003).
Miller, D. B. & O' Callaghan, J. P. Aging, stress and the hippocampus. Age. Res. Rev. 4, 123–140 (2005).
Hukovic, N. & Brown E. S. Effects of prescription corticosteroids on mood and cognition. Adv. Psychosom. Med. 24, 161–167 (2003).
Lee, G. D. et al. Transient improvement in cognitive function and synaptic plasticity in rats following cancer chemotherapy. Clin. Cancer Res. 12, 198–205 (2006).
Winocur, G., Vardy, J, Bims, M. A., Kerr, L. & Tannock, I. The effects of the anti-cancer drugs, methotrexate and 5-fluorouracil, on cognitive function in mice. Pharmacol. Biochem. Behav. 85, 66–75 (2006). These investigators developed an animal model of chemotherapy-induced cognitive deficits and showed deficits in memory and learning tasks in mice treated with chemotherapy that were similar to deficits seen in breast cancer survivors.
Acknowledgements
The authors are supported by grants from the Office of Cancer Survivorship of the US National Cancer Institute and a National Institutes of Health Roadmap U54 Grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Magnetic resonance imaging
-
A noninvasive technique that produces high-resolution, computerized images of internal body tissues. Structural MRI enables abnormalities of brain structure to be evaluated and volumetric measurements of specific structures to be made. Functional MRI enables activation patterns in various brain areas in response to the performance of cognitive or motor tasks to be examined.
- P-300
-
The P-300 is a neural-evoked potential component of the EEG. The P-300 is an event-related potential that is triggered approximately 300 milliseconds after the presentation of an unexpected or novel stimulus.
- Encephalopathy
-
Encephalopathy refers to alterations in brain structure and/or function that can have several causes, including infection, exposure to toxic chemicals (for example, chemotherapy), poor nutrition or lack of oxygen or blood flow to the brain. The primary symptoms of encephalopathy are alterations in mental status.
- Leukoencephalopathy
-
Alterations of the white matter of the brain owing to infection or exposure to toxic chemicals.
- Ototoxicity
-
Toxicity associated with organs or nerves involved with hearing or balance.
- Cerebellar symptoms
-
The cerebellum is an area of the brain that is important for the integration of sensory input and motor output. Disorders of the cerebellum include symptoms associated with equilibrium, posture, motor learning and fine motor control.
- Positron emission tomography
-
A nuclear medicine imaging technique that produces a three-dimensional image of functional or metabolic processes in the body by scanning for a radioactive isotope (for example, a radiolabelled chemotherapy agent) that has been injected into the bloodstream.
- Ataxia telangiectasia
-
A disorder caused by a mutation of the ataxia telangiectasia mutated (ATM) gene, which is important for DNA repair. Ataxia telangiectasia causes progressive immunological and neurological problems, including cognitive symptoms, and people with ataxia telangiectasia have a significantly increased risk of cancer.
- Xeroderma pigmentosum
-
A genetic DNA-repair disorder in which the body is unable to repair DNA damage or mutations caused by ultraviolet light.
- Glia
-
A group of non-neuronal cells in the brain that provide support and nutrition, form myelin and influence signal transmission in the nervous system.
- Sickness behaviour
-
Physiological response to infection that includes symptoms such as decreased activity level, fatigue, decreased motivation and cognitive problems.
- Vagus nerve
-
The vagus nerve is the only cranial nerve that extends from the brainstem to all the organs in the abdomen.
- Excitotoxicity
-
The process by which neurons are damaged or killed through the over-activation of receptors for the excitatory neurotransmitter glutamate.
- Long-term potentiation
-
Long-lasting increase in the functioning of a synapse, which is thought to be related to neural plasticity and the cellular basis for learning and memory.
Rights and permissions
About this article
Cite this article
Ahles, T., Saykin, A. Candidate mechanisms for chemotherapy-induced cognitive changes. Nat Rev Cancer 7, 192–201 (2007). https://doi.org/10.1038/nrc2073
Issue Date:
DOI: https://doi.org/10.1038/nrc2073
This article is cited by
-
The neurobiology of SARS-CoV-2 infection
Nature Reviews Neuroscience (2024)
-
Nrf2 Signaling Pathway: a Potential Therapeutic Target in Combating Oxidative Stress and Neurotoxicity in Chemotherapy-Induced Cognitive Impairment
Molecular Neurobiology (2024)
-
Patient-reported neurocognitive function in adult survivors of childhood and adolescent osteosarcoma and Ewing sarcoma
Journal of Cancer Survivorship (2023)
-
Cognitive function is mediated by deficit accumulation in older, long-term breast cancer survivors
Journal of Cancer Survivorship (2023)
-
A randomised controlled trial evaluating two cognitive rehabilitation approaches for cancer survivors with perceived cognitive impairment
Journal of Cancer Survivorship (2023)