Key Points
-
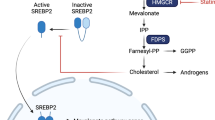
Statins function in the mevalonate pathway as small-molecule inhibitors of 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase, which lowers cholesterol. These agents are effective in preventing cardiovascular disease (CVD), largely because of this effect.
-
Large randomized controlled trials (RCTs) that analysed the effects of statins and other lipid-lowering agents (for example, fibrates, nicotinic acid and cholestyramine) to prevent CVD included safety monitoring to address whether statins increased cancer incidence and cancer mortality. Ironically, these results were the first to suggest that statins can prevent cancer.
-
In addition to the HMG-CoA-dependent effects, statins have important cholesterol/HMG-CoA-independent effects, such as effects on lymphocyte-function-associated antigen 1 (LFA1), that are thought to contribute to potential cancer prevention.
-
Important work in preclinical models of colorectal and breast cancer, and melanoma, indicates that statin anticancer effects involve the inhibition of geranylgeranylation, primarily of Rho proteins.
-
Secondary results of the CVD RCTs, as well as observational and preclinical studies, indicate that statins have a strong potential for preventing colorectal cancer and melanoma.
-
The beneficial effects of statins on inhibiting carcinogenesis could involve their effects on important disease pathways including inflammation, immunomodulation and angiogenesis.
-
Statins are broad-spectrum agents. Current research is revealing important new statin targets (such as LFA1, Rho isoforms, and post-prenylation enzymes) leading to the development of more-specifically targeted agents for cancer prevention.
Abstract
Randomized controlled trials for preventing cardiovascular disease indicated that statins had provocative and unexpected benefits for reducing colorectal cancer and melanoma. These findings have led to the intensive study of statins in cancer prevention, including recent, large population-based studies showing statin-associated reductions in overall, colorectal and prostate cancer. Understanding the complex cellular effects (for example, on angiogenesis and inflammation) and the underlying molecular mechanisms of statins (for example, 3-hydroxy-3-methylglutaryl coenzyme-A (HMG-CoA) reductase-dependent processes that involve geranylgeranylation of Rho proteins, and HMG-CoA-independent processes that involve lymphocyte-function-associated antigen 1) will advance the development of molecularly targeted agents for preventing cancer. This understanding might also help the development of drugs for other ageing-related diseases with interrelated molecular pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Oliver, M. F. Might treatment of hypercholesterolaemia increase non-cardiac mortality? Lancet 337, 1529–1531 (1991).
Newman, T. B. & Hulley, S. B. Carcinogenicity of lipid-lowering drugs. JAMA 275, 55–60 (1996).
Ridker, P. M. et al. Inflammation, pravastatin, and the risk of coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events (CARE) investigators. Circulation 98, 839–844 (1998). Important early report indicating that statins can have cholesterol-independent effects — such as anti-inflammatory effects that are proving to be important in CVD, cancer and other disease settings — in addition to well-known HMG-CoA reductase-dependent and cholesterol-dependent effects.
Gotto, A. M. Jr et al. Relation between baseline and on-treatment lipid parameters and first acute major coronary events in the Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TexCAPS). Circulation 101, 477–484 (2000).
Ehrenstein, M. R., Jury, E. C. & Mauri, C. Statins for atherosclerosis — as good as it gets? N. Engl. J. Med. 352, 73–75 (2005).
Chan, K. K. W., Oza, A. M. & Siu, L. L. The statins as anticancer agents. Clin. Cancer Res. 9, 10–19 (2003).
Wong, W. W. L., Dimitroulakos, J., Minden, M. D. & Penn, L. Z. HMB-CoA reductase inhibitors and the malignant cell: the statin family of drugs as triggers of tumor-specific apoptosis. Leukemia 16, 508–519 (2002).
Edwards, P. A. & Ericsson, J. Sterols and isoprenoids: signaling molecules derived from the cholesterol biosynthetic pathway. Annu. Rev. Biochem. 68, 157–185 (1999).
Houten, S. M., Frenkel, J. & Waterham, H. R. Isoprenoid biosynthesis in hereditary periodic fever syndromes and inflammation. Cell. Mol. Life Sci. 60, 1118–1134 (2003).
Mo, H. & Elson, C. E. Studies of the isoprenoid-mediated inhibition of mevalonate synthesis applied to cancer chemotherapy and chemoprevention. Exp. Biol. Med. 229, 567–585 (2004).
Simpson, K. J., Dugan, A. S. & Mercurio, A. M. Functional analysis of the contribution of RhoA and RhoC GTPases to invasive breast carcinoma. Cancer Res. 64, 8694–8701 (2004).
Kusama, T. et al. Inhibition of epidermal growth factor-induced RhoA translocation and invasion of human pancreatic cancer cells by 3-hydroxy-3-methylglutaryl-coenzyme A reductase inhibitors. Cancer Res. 61, 4885–4891 (2001).
Malliri, A. & Collard, J. G. Role of Rho-family proteins in cell adhesion and cancer. Curr. Opin. Cell Biol. 15, 583–589 (2003).
Zugaza, J. L., Caloca, M. J. & Bustelo X. R. Inverted signaling hierarchy between RAS and RAC in T-lymphocytes. Oncogene 23, 5823–5833 (2004).
Mazieres, J. et al. Geranylgeranylated, but not farnesylated, RhoB suppresses Ras transformation of NIH-3T3 cells. Exp. Cell. Res. 304, 354–364 (2005).
Singh, A. et al. Rac1b, a tumor associated, constitutively active Rac1 splice variant, promotes cellular transformation. Oncogene 23, 9369–9380 (2004).
Gerber, J. G. et al. Effect of Efavirenz on the pharmacokinetics of simvastatin, atorvastatin, and pravastatin: results of AIDS clinical trials group 5108 study. J. Acquir. Immune Defic. Syndr. 39, 307–312 (2005).
Buemi, M. et al. Pro-apoptotic effect of fluvastatin on human smooth muscle cells. Eur. J. Pharmacol. 370, 201–203 (1999).
Weitz-Schmidt, G. et al. Statins selectively inhibit leukocyte function antigen-1 by binding to a novel regulatory integrin site. Nature Med. 7, 687–692 (2001). First clear example of an HMG-CoA reductase-independent effect of statins and a mechanism (binding to LFA1) for this effect. This work shows that lovastatin directly binds to the L-site in the I-domain of the integrin LFA1, which has an important role in leukocyte migration and T-cell activation. These findings indicate that a new class of anti-inflammatory agents could be developed (see reference 122).
Katano, H., Pesnicak, L. & Cohen, J. I. Simvastatin induces apoptosis of Epstein-Barr virus (EBV)-transformed lymphoblastoid cell lines and delays development of EBV lymphomas. Proc. Natl Acad. Sci. USA 101, 4960–4965 (2004).
Rao, S. et al. Lovastatin-mediated G1 arrest is through inhibition of the proteasome, independent of hydroxymethyl glutaryl-CoA reductase. Proc. Natl Acad. Sci. USA 96, 7797–7802 (1999).
Ukomadu, C. & Dutta, A. p21-dependent inhibition of colon cancer cell growth by mevastatin is independent of inhibition of G1 cyclin-dependent kinases. J. Biol. Chem. 278, 43586–43594 (2003).
Veillard, N. R. & Mach, F. Statins: the new aspirin? Cell. Mol. Life Sci. 59, 1771–1786 (2002).
Youssef, S. et al. The HMG-CoA reductase inhibitor, atorvastatin, promotes a Th2 bias and reverses paralysis in central nervous system autoimmune disease. Nature 420, 78–84 (2002).
Hilgendorff, A. et al. Statins differ in their ability to block NF–κB activation in human blood monocytes. Intl J. Clin. Pharmacol. Ther. 41, 397–401 (2003).
Hakamada-Taguchi, R. et al. Inhibition of hydroxymethylglutaryl-coenzyme a reductase reduces Th1 development and promotes Th2 development. Circ. Res. 93, 948–956 (2003).
Tilkin-Mariame, A. F. et al. Geranylgeranyl transferase inhibition stimulates antimelanoma immune response through MHC class I and costimulatory molecule expression. FASEB J. 29 Jun 2005 (10.1096/fj.04-3482fje).
Weis, M., Heeschen, C., Glassford, A. J. & Cooke, J. P. Statins have biphasic effects on angiogenesis. Circulation 105, 739–745 (2002).
Sata, M. et al. Statins augment collateral growth in response to ischemia but they do not promote cancer and atherosclerosis. Hypertension 43, 1214–1220 (2004). Recent study of ischaemia and cancer in the same animal indicating that dose is not central to the anti-angiogenic effects of statins in carcinogenesis. This report alleviated concerns that clinically relevant doses of statins would promote tumour angiogenesis during treatment.
Kureishi, Y. et al. The HMG-CoA reductase inhibitor simvastatin activates the protein kinase Akt and promotes angiogenesis in normocholesterolemic animals. Nature Med. 6, 1004–1010 (2000).
Parmar, K. M. et al. Statins exert endothelial atheroprotective effects via the KLF2 transcription factor. J. Biol. Chem. 280, 26714–26719 (2005).
Vincent, L. et al. Cerivastatin, an inhibitor of 3-hydroxy-3-methylglutaryl coenzyme a reductase, inhibits endothelial cell proliferation induced by angiogenic factors in vitro and angiogenesis in in vivo models. Arterioscler. Thromb. Vasc. Biol. 22, 623–629 (2002).
Skaletz-Rorowski, A. et al. HMG-CoA reductase inhibitors promote cholesterol-dependent Akt/PKB translocation to membrane domains in endothelial cells. Cardiovasc. Res. 57, 253–264 (2003).
Skaletz-Rorowski, A. & Walsh, K. Statin therapy and angiogenesis. Curr. Opin. Lipidol. 14, 599–603 (2003).
Urbich, C., Dernbach, E., Zeiher, A. M. & Dimmeler, S. Double-edged role of statins in angiogenesis signaling. Circ. Res. 90, 737–744 (2002).
Wu, J., Wong, W. W. L., Khosravi, F., Minden, M. D. & Penn, L. Z. Blocking the Raf–MEK–ERK pathway sensitizes acute myelogenous leukemia cells to lovastatin-induced apoptosis. Cancer Res. 64, 6461–6468 (2004).
Gniadecki, R. Depletion of membrane cholesterol causes ligand-independent activation of Fas and apoptosis. Biochem. Biophys. Res. Commun. 320, 165–169 (2004).
Shibata, M. A., Ito, Y., Morimoto, J. & Otsuki, Y. Lovastatin inhibits tumor growth and lung metastasis in mouse mammary carcinoma model: a p53-independent mitochondrial-mediated apoptotic mechanism. Carcinogenesis 25, 1887–1898 (2004).
Macaulay, R. J., Wang, W., Dimitroulakos, J., Becker, L. E. & Yeger, H. Lovastatin-induced apoptosis of human medulloblastoma cell lines in vitro. J. Neurooncol. 42, 1–11 (1999).
Marcelli, M. et al. Caspase-7 is activated during lovastatin-induced apoptosis of the prostate cancer cell line LNCaP. Cancer Res. 58, 76–83 (1998).
Cafforio, P., Dammacco, F., Gernone, A. & Silvestris, F. Statins activate the mitochondrial pathway of apoptosis in human lymphoblasts and myeloma cells. Carcinogenesis 26, 883–891 (2005).
Crick, D. C., Andres, D. A., Danesi, R., Macchia, M. & Waechter, C. J. Geranylgeraniol overcomes the block of cell proliferation by lovastatin in C6 glioma cells. J. Neurochem. 70, 2397–2405 (1998).
Jakobisiak, M., Bruno, S., Skierski, J. S. & Darzynkiewicz, Z. Cell cycle-specific effects of lovastatin. Proc. Natl Acad. Sci. USA 88, 3628–3632 (1991).
Park, C., Lee, I. & Kang, W. K. Lovastatin-induced E2F-1 modulation and its effect on prostate cancer cell death. Carcinogenesis 22, 1727–1731 (2001).
Maltese, W. A. & Sheridan, K. M. Differentiation of neuroblastoma cells induced by an inhibitor of mevalonate synthesis: relation of neurite outgrowth and acetylcholinesterase activity to changes in cell proliferation and blocked isoprenoid synthesis. J. Cell. Physiol. 125, 540–558 (1985).
Hentosh, P., Yuh, S. H., Elson, C. E. & Peffley, D. M. Sterol-independent regulation of 3-hydroxy-3-methylglutaryl coenzyme A reductase in tumor cells. Mol. Carcinogen. 32, 154–166 (2001).
Attoub, S. et al. Leptin promotes invasiveness of kidney and colonic epithelial cells via phosphoinositide 3-kinase-, Rho-, and Rac-dependent signaling pathways. FASEB J. 14, 2329–2338 (2000).
Agarwal, B. et al. Mechanism of lovastatin-induced apoptosis in intestinal epithelial cells. Carcinogenesis 23, 521–528 (2002).
Erlinger, T. P., Platz, E. A., Rifai, N. & Helzlsouer, K. J. C-reactive protein and the risk of incident colorectal cancer. JAMA 291, 585–590 (2004).
Narisawa, T. et al. Chemoprevention by pravastatin, a 3-hydroxy-3-methylglutaryl-coenzyme A reductase inhibitor, of N-methyl-N-nitrosourea-induced colon carcinogenesis in F344 rats. Jpn. J. Cancer Res. 87, 798–804 (1996).
Agarwal, B. et al. Lovastatin augments sulindac-induced apoptosis in colon cancer cells and potentiates chemopreventive effects of sulindac. Gastroenterology 117, 838–847 (1999). Important, early preclinical study showing that statins reduced aberrant crypt foci, a clonal precursor of colorectal neoplasia, at the level of geranylgeranylation, and were synergistic with the NSAID sulindac in preventing aberrant crypt foci (see reference 48).
Rao, C. V., Newmark, H. L. & Reddy, B. S. Chemopreventive effect of farnesol and lanosterol on colon carcinogenesis. Cancer Detect. Prev. 26, 419–425 (2002).
Broitman, S. A., Wilkinson, J. T., Cerda, S. & Branch, S. K. Effects of monoterpenes and mevinolin on murine colon tumor CT-26 in vitro and its hepatic “metastases” in vivo. Adv. Exp. Med. Biol. 401, 111–130 (1996).
Swamy, M. V., Cooma, I., Reddy, B. S. & Rao, C. V. Lamin B, caspase-3 activity, and apoptosis induction by a combination of HMG-CoA reductase inhibitor and COX-2 inhibitors: a novel approach in developing effective chemopreventive regimens. Int. J. Oncol. 20, 753–759 (2002).
Feleszko, W. et al. Synergistic interaction between highly specific cyclooxygenase-2 inhibitor, MF-tricyclic and lovastatin in murine colorectal cancer cell lines. Oncol. Rep. 9, 879–885 (2002).
Reddy, B. S. et al. Synergistic effects of the combination of low doses of aspirin or celecoxib with Lipitor against colon carcinogenesis: a promising chemoprevention strategy. Proc. Amer. Assoc. Cancer. Res. 46, LB–4 (2005).
Solomon, S. D. et al. Cardiovascular risk associated with celecoxib in a clinical trial for colorectal adenoma prevention. N. Engl. J. Med. 352, 1071–1080 (2005).
Mueck, A. O., Seeger, H. & Wallwiener, D. Effect of statins combined with estradiol on the proliferation of human receptor-positive and receptor-negative breast cancer cells. Menopause 10, 332–336 (2003).
Seeger, H., Wallwiener, D. & Mueck, A. O. Statins can inhibit proliferation of human breast cancer cells in vitro. Exp. Clin. Endocrinol. Diabetes 111, 47–48 (2003).
Fritz, G., Brachetti, C., Bahlmann, F., Schmidt, M. & Kaina, B. Rho GTPases in human breast tumours: expression and mutation analyses and correlation with clinical parameters. Br. J. Cancer 87, 635–644 (2002).
Denoyelle, C. et al. Cerivastatin, an inhibitor of HMG–CoA reductase, inhibits the signaling pathways involved in the invasiveness and metastatic properties of highly invasive breast cancer cell lines: an in vitro study. Carcinogenesis 22, 1139–1148 (2001).
Wilde, C. & Aktories, K. The Rho–ADP–ribosylating C3 exoenzyme from Clostridium botulinum and related C3-like transferases. Toxicon. 39, 1647–1660 (2001).
Denoyelle, C. et al. Molecular mechanism of the anti-cancer activity of cerivastatin, an inhibitor of HMG-CoA reductase, on aggressive human breast cancer cells. Cell. Signal. 15, 327–338 (2003). An important preclinical study showing that cervistatin prevented RHOA prenylation and thereby inhibited RHOA–ROCK signalling. These findings support the idea that RHOA is an important target of statins in breast cancer prevention.
Silva, J., Beckedorf, A. & Bieberich, E. Osteoblast-derived oxysterol is a migration-inducing factor for human breast cancer cells. J. Biol. Chem. 278, 25376–25385 (2003).
Bourguignon, L. Y., Singleton, P. A., Zhu, H. & Diedrich, F. Hyaluronan-mediated CD44 interaction with RhoGEF and Rho kinase promotes Grb2-associated binder-1 phosphorylation and phosphatidylinositol 3-kinase signaling leading to cytokine (macrophage–colony stimulating factor) production and breast tumor progression. J. Biol. Chem. 278, 29420–29434 (2003).
Kumar, B. et al. Mevastatin induces degeneration and decreases viability of cAMP-induced differentiated neuroblastoma cells in culture by inhibiting proteasome activity, and mevalonic acid lactone prevents these effects. J. Neurosci. Res. 68, 627–635 (2002).
Murray, S. S., Tu, K. N., Young, K. L. & Murray, E. J. The effects of lovastatin on proteasome activities in highly purified rabbit 20 S proteasome preparations and mouse MC3T3-E1 osteoblastic cells. Metabolism 51, 1153–1160 (2002).
Wojcik, C. et al. Lovastatin and simvastatin are modulators of the proteasome. Intl J. Biochem. Cell. Biol. 32, 957–965 (2000).
Alonso, D. F. et al. Reduction of mouse mammary tumor formation and metastasis by lovastatin, an inhibitor of the mevalonate pathway of cholesterol synthesis. Breast Cancer Res. Treat. 50, 83–93 (1998).
Farina, H. G., Bublik, D. R., Alonso, D. F. & Gomez, D. E. Lovastatin alters cytoskeleton organization and inhibits experimental metastasis of mammary carcinoma cells. Clin. Exp. Metastasis 19, 551–559 (2002).
Esserman, L. et al. Breast cancer inhibition by statins. Proc. Am. Soc. Clin. Oncol. 23, 97 (2004).
Collisson, E. A., Carranza, D. C., Chen, I. Y. & Kolodney, M. S. Isoprenylation is necessary for the full invasive potential of RhoA overexpression in human melanoma cells. J. Invest. Dermatol. 119, 1172–1176 (2002).
Shellman, Y. G. et al. Lovastatin-induced apoptosis in human melanoma cell lines. Melanoma Res. 15, 83–89 (2005).
Collisson, E. A. et al. Atorvastatin prevents RhoC isoprenylation, invasion, and metastasis in human melanoma cells. Mol. Cancer Ther. 2, 941–948 (2003). A study providing a biological explanation for the significant secondary finding of a clinical trial, which showed that a statin reduced melanoma risk (see reference 104). This paper shows that RHOC is expressed in melanoma samples and that atorvastatin attenuated RHOC signalling and inhibited invasion and metastasis.
Busca, R. et al. Inhibition of Rho is required for cAMP-induced melanoma cell differentiation. Mol. Biol. Cell 9, 1367–1378 (1998).
Nakajima, M. et al. Effect of Wf-536, a novel ROCK inhibitor, against metastasis of B16 melanoma. Cancer Chemother. Pharmacol. 52, 319–324 (2003).
He, L., Mo, H., Hadisusilo, S., Qureshi, A. A. & Elson, C. E. Isoprenoids suppress the growth of murine B16 melanomas in vitro and in vivo. J. Nutr. 127, 668–674 (1997).
Lluria-Prevatt, M. et al. Effects of perillyl alcohol on melanoma in the TPras mouse model. Cancer Epidemiol. Biomarkers Prev. 11, 573–579 (2002).
Blais, L., Desgagne, A. & LeLorier, J. 3-Hydroxy-3-methylglutaryl coenzyme A reductase inhibitors and the risk of cancer: a nested case-control study. Arch. Intern. Med. 160, 2363–2368 (2000).
Graaf, M. R., Beiderbeck, A. B., Egberts, A. C., Richel, D. J. & Guchelaar, H. J. The risk of cancer in users of statins. J. Clin. Oncol. 22, 2388–2394 (2004). A large prospective observational study showing a significant reduction in overall cancer risk (and non-significant reductions in colorectal and prostate cancer) associated with statins. This study is therefore an important contribution to the current focus on statins for cancer prevention.
Kaye, J. A. & Jick, H. Statin use and cancer risk in the General Practice Research Database. Br. J. Cancer 90, 635–637 (2004).
Kaye, J. A., Meier, C. R., Walker, A. M. & Jick, H. Statin use, hyperlipidaemia, and the risk of breast cancer. Br. J. Cancer 86, 1436–1439 (2002).
Friis, S. et al. Cancer risk among statin users: a population-based cohort study. Int. J. Cancer 114, 643–647 (2005).
Platz, E. A. et al. Cholesterol-lowering drugs including statins and the risk of prostate cancer in a large prospective cohort study. Proc. Am. Assoc. Cancer Res. 46, 1034 (2005).
Shannon, J et al. Statins and prostate cancer risk: a case control study. Am. J. Epidemiol. 162, 318–325 (2005).
Singal, R., Khurana, V., Caldito, G. & Fort, C. Statins and prostate cancer risk: a case control study. Am. J. Epidemiol. 162 318–325 (2005).
Shibata, M. A. et al. Comparative effects of lovastatin on mammary and prostate oncogenesis in transgenic mouse models. Carcinogenesis 24, 453–459 (2003).
Poynter, J. N. et al. Statins and the risk of colorectal cancer. N. Engl. J. Med. 352, 2184–2192 (2005). Large population-based case–control study showing the strong inverse association between colorectal cancer and long-term statin use, solidifying the potential importance of statins for preventing colorectal cancer.
Wei, J. T. et al. Reported use of 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitors was not associated with reduced recurrence of colorectal adenomas. Cancer Epidemiol. Biomarkers Prev. 14, 1026–1027 (2005).
Beck, P., Wysowski, D. K., Downey, W. & Butler-Jones, D. Statin use and the risk of breast cancer. J. Clin. Epidemiol. 56, 280–285 (2003).
Cauley, J. A. et al. Lipid-lowering drug use and breast cancer in older women: a prospective study. J. Womens Health (Larchmt) 12, 749–756 (2003).
Coogan, P. F. et al. Statin use and the risk of breast and prostate cancer. Epidemiology 13, 262–267 (2002).
Boudreau, D. M. et al. The association between 3-hydroxy-3-methylglutaryl conenzyme A inhibitor use and breast carcinoma risk among postmenopausal women: a case-control study. Cancer 100, 2308–2316 (2004).
Dellavalle, R. P., Nicholas, M. K. & Schilling, L. M. Melanoma chemoprevention: a role for statins or fibrates? Am. J. Ther. 10, 203–210 (2003).
Khurana, V., Kochhar, R., Bejjanki, H. R. & Fort, C. Statins reduce the incidence of lung cancer: a study of half a million U. S. veterans. Proc. Am. Soc. Clin. Oncol. 16 (Suppl.), 1006 (2005).
Khurana, V., Barkin, J. S., Caldito, G. & Fort, C. Statins reduce the risk of pancreatic cancer in humans: half a million US veterans' case control study. Gastroenterol. 128 (Suppl.), A62–A62 (2005).
Khurana, V., Chalasani, R., Caldito, G. & Fort, C. Statins reduce the incidence of esophageal cancer: a study of half a million US veterans. Gastroenterol. 128 (Suppl.), A93–A93 (2005).
Zhang, Y. et al. Prior medical conditions and medication use and risk of non-Hodgkin lymphoma in Connecticut United States women. Cancer Causes Control 15, 419–428 (2004).
Fortuny, J. et al. Statin use and risk of lymphoma. Results from the European case-control study Epilymph. 9th International Conference on Malignant Lymphoma, Lugano, Switzerland, 8 June, 2005.
Duncan, R.E. et al. Statins and cancer development. Cancer Epidemiol. Biomarkers Prev. 14, 1897–1898 (2005).
Strandberg, T. E. et al. Mortality and incidence of cancer during 10-year follow-up of the Scandinavian Simvastatin Survival Study (4S). Lancet 364, 771–777 (2004). Longest follow-up analysis of any RCT (in CVD prevention), showing a significant overall mortality benefit of statins and non-significant benefits in cancer risk and mortality over 10 years. This analysis supports the feasibility of statins for long-term cancer prevention.
Shepherd, J. et al. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. West of Scotland Coronary Prevention Study Group. N. Engl. J. Med. 333, 1301–1307 (1995).
Sacks, F. M. et al. The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events Trial investigators. N. Engl. J. Med. 335, 1001–1009 (1996).
The Long-Term Intervention with Pravastatin in Ischaemic Disease (LIPID) Study Group. Prevention of cardiovascular events and death with pravastatin in patients with coronary heart disease and a broad range of initial cholesterol levels. N. Engl. J. Med. 339, 1349–1357 (1998).
Downs, J. R. et al. Primary prevention of acute coronary events with lovastatin in men and women with average cholesterol levels: results of AFCAPS/TexCAPS. Air Force/Texas Coronary Atherosclerosis Prevention Study. JAMA 279, 1615–1622 (1998). First RCT showing a secondary benefit of statins in cancer risk reduction. This benefit, a significant reduction in melanoma, helped overcome suspicions that statins had adverse cancer effects and promotes belief in the potential of statins to prevent melanoma and other cancers.
Heart Protection Study Collaborative Group. MRC/BHF heart protection study of cholesterol lowering with simvastatin in 20,536 high-risk individuals: a randomised placebo-controlled trial. Lancet 360, 7–22 (2002).
Shepherd, J. et al. Pravastatin in elderly individuals at risk of vascular disease (PROSPER): a randomised controlled trial. Lancet 360, 1623–1630 (2002).
Pfeffer, M. A. et al. Safety and tolerability of pravastatin in long-term clinical trials: Prospective Pravastatin Pooling (PPP) project. Circulation 105, 2341–2346 (2002).
Hebert, P. R., Gaziano, J. M., Chan, K. S. & Hennekens, C. H. Cholesterol lowering with statin drugs, risk of stroke, and total mortality. An overview of randomized trials. JAMA 278, 313–321 (1997).
Bjerre, L. M. & LeLorier, J. Do statins cause cancer? A meta-analysis of large randomized clinical trials. Am. J. Med. 110, 716–723 (2001).
Guallar, E. & Goodman, S. N. Statins and cancer: a case of meta-uncertainty. Am. J. Med. 110, 738–740 (2001).
Weitz-Schmidt, G. Statins as anti-inflammatory agents. Trends Pharm. Sci. 23, 482–486 (2002).
Jakobisiak, M. & Golab, J. Potential antitumor effects of statins. Intl J. Oncol. 23, 1055–1069 (2003).
Graaf, M. R., Richel, D. J., van Noorden, C. J. F. & Guchelaar, H. H. J. Effects of statin and farnesyltransferase inhibitors on the development and progression of cancer. Cancer Treat. Rev. 30, 609–641 (2004).
Caraglia, M. et al. Isoprenylation of intracellular proteins as a new target for the therapy of human neoplasms: preclinical and clinical implications. Curr. Drug Targets 6, 301–323 (2005).
Rowinsky, E. K, Windle J. J. & Von Hoff, D. D. Ras protein farnesyltransferase: a strategic target for anticancer therapeutic development. J. Clin. Oncol. 17, 3631–3652 (1999).
Liao, J. K. et al. Pleiotropic effects of statins. Annu. Rev. Pharmacol. Toxicol. 45, 89–118 (2005).
Pillé, J. Y. et al. Anti-RhoA and anti-RhoC siRNAs inhibit the proliferation and invasiveness of MDA-MB-231 breast cancer cells in vitro and in vivo. Mol. Ther. 11, 267–274 (2005).
Wang, C. et al. Identification of FBL2 as a geranylgeranylated cellular protein required for hepatitis C virus RNA replication. Mol. Cell 18, 425–434 (2005).
Winter-Vann, A. M. et al. A small-molecule inhibitor of isoprenylcysteine carboxyl methyltransferase with antitumor activity in cancer cells. Proc. Natl Acad. Sci. USA 102, 4336–4341 (2005). An important recent study by this investigative group, highlighting the potential importance of a post-prenylation enzyme target for potential anticancer drugs.
Anderson, J. L., Henriksen, B. S., Gibbs, R. A. & Hrycyna, C. A. The isoprenoid substrate specificity of isoprenylcysteine carboxylmethyltransferase: development of novel inhibitors. J. Biol. Chem. 280, 29454–29461 (2005).
Weitz-Schmidt, G., Welzenbach, K., Dawson, J. & Kallen, J. Improved lymphocyte function-associated antigen-1 (LFA-1) inhibition by statin derivatives: molecular basis determined by x- X-ray analysis and monitoring of LFA-1 conformational changes in vitro and ex vivo. J. Biol. Chem. 279, 46764–46771 (2004).
Subdo, J. et al. Non-steroidal anti-inflammatory drugs and the risk of oral cancer: a nested case-control study. Lancet 366, 1359–1366 (2005).
Mantha, A. J. et al. Targeting the mevalonate pathway inhibits the function of the epidermal growth factor receptor. Clin. Cancer Res. 11, 2398–2407 (2005).
Hawk, E. & Viner, J. L. Statins and cancer — beyond the “one drug, one disease” model. N. Engl. J. Med. 352, 2238–2239 (2005).
Topol, E. J. Intensive statin therapy — a sea change in cardiovascular prevention. N. Engl. J. Med. 350, 1562–1564 (2004).
Lippman, S. M. & Hong, W. K. Cancer prevention science and practice. Cancer Res. 62, 5119–5125 (2002).
Acknowledgements
This work was supported in part by the University of Texas M.D. Anderson Cancer Center support grants from the National Cancer Institute, National Institutes of Health Department of Health and Human Services.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- CHOLESTEROL
-
A lipid ringed sterol used by the body for the production of hormones, vitamin D and cell membranes; high levels in the blood stream are a marker for heart disease.
- CARDIOVASCULAR DISEASE
-
Disease caused by atherosclerosis of the coronary arteries.
- MEVALONATE
-
A fatty acid formed from HMG-CoA by HMG-CoA reductase, and an essential intermediate in the biosynthesis of cholesterol or geranylgeranyl pyrophosphate, which leads to the isoprenylation of the small G-proteins.
- RANDOMIZED CONTROLLED TRIAL
-
A study of individuals who are randomized to a therapy, which is used to evaluate the effect of a therapy versus a placebo. Resistant to bias from unmeasured risk factors as these should be distributed equally in both groups.
- OBSERVATIONAL STUDIES
-
Studies in which data on risk factors and disease outcomes are collected to detect associations between risk factors and disease. Individuals are not randomized, and choose their own exposure to risk factors, so bias can occur and might be undetected.
- POPULATION-BASED STUDY
-
A type of observational study in which the entire population of a geographical area (county, state or country) is studied for risk factors, and disease outcomes are recorded as they occur, to detect associations between risk factors and the incidence of disease. Minimizes selection bias.
- ANGIOGENESIS
-
The generation of new blood vessels, particularly arterial supply vessels. Can occur after trauma, ischaemic (lack of oxygen) injury or during the growth of a tumour.
- NEURODEGENERATION
-
A group of neurological diseases, affecting the central nervous system, that involve the loss of neurons. These diseases include Alzheimer disease and Parkinson disease.
- AGE-RELATED MACULAR DEGENERATION
-
A disease that blurs the central, high-resolution vision of the eye by damaging the macula. It is the main cause of central vision loss in Americans who are 50 or more years old.
- OSTEOPOROSIS
-
A condition that is characterized by a decrease in bone mass as well as by decreased bone density and increased risk of bone fracture.
- LIPID RAFTS
-
Cholesterol-rich areas of the cell membrane.
- SEVERE COMBINED IMMUNODEFICIENT MOUSE
-
(SCID mouse). Mice with this defect in their immune system do not have B cells or T cells. Therefore, they can accept tumour cells from another species without rejection.
- C-REACTIVE PROTEIN
-
(CRP). An inflammatory mediator produced by the liver in response to pro-inflammatory signals. Raised levels of CRP correlate with cardiovascular disease risk and are thought to indicate instability of inflamed atherosclerotic plaques.
- AZOXYMETHANE
-
A potent carcinogen that is used to induce colon cancer in rats and mice. Treatment with azoxymethane activates the epidermal growth factor receptor and stimulates the synthesis of transforming growth factor-α.
- ABERRANT CRYPT FOCI
-
A pre-cancerous change that represents early clonal precursors of colorectal neoplasia; presumed to precede microadenomas.
- PERILLYL ALCOHOL
-
A naturally occurring isoprenoid.
- NON-STEROIDAL ANTI-INFLAMMATORY DRUGS
-
Include aspirin, ibuprofen, celecoxib, and many others. Associated with the decreased occurence of colon cancer and colon polyps.
- CYCLOOXYGENASE 2 INHIBITORS
-
Drugs that specifically inhibit the cyclooxygenase 2 enzyme (for example, rofecoxib), which have been associated with decreased colon polyps cancer. A subset of NSAIDs.
- C3 EXOENZYME
-
ADP-ribosyltransferase isolated from Clostridium botulinum. Substrates include the Rho proteins. ADP-ribosylation inactivates these signalling proteins.
- OSTEOBLASTS
-
Cells originating in the bone marrow that generate new bone. These cells are stimulated by statins, which might explain the decreased risk of fracture in statin users.
- TPRAS
-
A transgenic mouse model that contains a mutated human T24 HRAS gene driven by a 2.5 kb promoter region from the mouse tyrosinase gene that enables expression within melanocytes.
- ISOPRENYLATION
-
Post-translational covalent addition of a farnesyl (15-mer) or a geranylgeranyl (20-mer) moiety to proteins that lack transmembrane domains, which allows them to localize to membranes and perform their usual function.
- RELATIVE RISK
-
A measure of the comparative risk of developing a disease or condition. Statistically, relative risk is the chance that a person receiving an exposure (statins) will develop a condition (cancer) compared with the chance that a non-exposed person will develop the same condition.
- HYPERCHOLESTEROLAEMIA
-
Increased cholesterol in the blood, associated with heart disease and stroke.
- ODDS RATIO
-
The odds ratio is a way of comparing whether the probability of a certain event is the same for two groups, and is calculated using a 2×2 table. An odds ratio of one implies that an event is equally likely in both groups. An odds ratio greater than one implies that an event is more likely in the first group. An odds ratio less than one implies that the event is less likely in the first group.
- META-ANALYSIS
-
A statistical practice of combining the results of a number of studies to overcome the problem of reduced statistical power in studies with small sample sizes; analysing the results from a group of studies can allow a more accurate estimation of effects.
Rights and permissions
About this article
Cite this article
Demierre, MF., Higgins, P., Gruber, S. et al. Statins and cancer prevention. Nat Rev Cancer 5, 930–942 (2005). https://doi.org/10.1038/nrc1751
Issue Date:
DOI: https://doi.org/10.1038/nrc1751
This article is cited by
-
Statin use in relation to long-term survival after gastrectomy for gastric adenocarcinoma: a Swedish population-based cohort study
Gastric Cancer (2024)
-
Therapeutic potential of ADAM10 modulation in Alzheimer’s disease: a review of the current evidence
Cell Communication and Signaling (2023)
-
Impact of oral statin therapy on clinical outcomes in patients with cT1 breast cancer
BMC Cancer (2023)
-
Effect of statin use on head and neck cancer prognosis in a multicenter study using a Common Data Model
Scientific Reports (2023)
-
Associations of genetically proxied inhibition of HMG-CoA reductase, NPC1L1, and PCSK9 with breast cancer and prostate cancer
Breast Cancer Research (2022)