Key Points
-
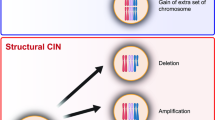
Aneuploidy, or abnormal chromosome content, is the most common characteristic of human solid tumours. Aneuploidy might contribute to tumour formation and is associated with acquired resistance to some chemotherapeutics.
-
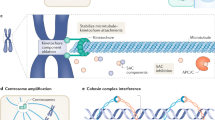
Tumour cells become aneuploid as a result of aberrant mitotic divisions. These aberrant divisions are caused by divisions with a multipolar spindle as a result of previous defects in cytokinesis or centrosome amplification, by defects in chromosome cohesion, by spindle attachment defects, or by impairment of the mitotic checkpoint response.
-
The mitotic checkpoint is a signalling cascade that arrests the cell cycle in mitosis when even a single chromosome is not properly attached to the mitotic spindle. This arrest is achieved by inhibiting the anaphase-promoting complex/cyclosome (APC/C), an E3 ubiquitin ligase that is essential for mitotic progression.
-
Many tumour cells have a diminished, but not absent, mitotic checkpoint response. Mouse models in which mitotic checkpoint signalling is decreased show an increase in spontaneous or carcinogen-induced tumour formation.
-
Mutations in mitotic checkpoint genes themselves are not a common mechanism of checkpoint impairment in human tumour cells.
-
Mitotic checkpoint impairment and aneuploidy in human tumour cells are often associated with changes in the protein levels of mitotic checkpoint proteins. In some tumour cells, these changes occur through altered transcriptional regulation by tumour suppressors or oncogene products.
-
Complete inactivation of mitotic checkpoint signalling causes cell-autonomous lethality. Drugs that specifically and efficiently interfere with mitotic checkpoint signalling could therefore be useful as anticancer agents.
Abstract
Abnormal chromosome content — also known as aneuploidy — is the most common characteristic of human solid tumours. It has therefore been proposed that aneuploidy contributes to, or even drives, tumour development. The mitotic checkpoint guards against chromosome mis-segregation by delaying cell-cycle progression through mitosis until all chromosomes have successfully made spindle-microtubule attachments. Defects in the mitotic checkpoint generate aneuploidy and might facilitate tumorigenesis, but more severe disabling of checkpoint signalling is a possible anticancer strategy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Flemming, W. Zellsubstanz, kern und zelltheilung. FCW Vogel, Leipzig, (1882).
Boveri, T. Über mehrpolige Mitosen als Mittel zur analyse des zellkerns. Verh. d. phys.med. Ges. Würzburg N. F. 35, 67–90 (1902).
von Hansemann, D. Ueber asymmetrische Zellheilteilung in epithelkrebsen und deren biologische bedeutung. Virschows Arch. Pathol. Anat. 119, 299–326 (1890).
Boveri, T. Zur Frage der Entstehung maligner tumoren. Jena:Gustav Fischer Verlag (1914).
Cancer Cytogenetics (eds Heim, S. & Mitelman, F.) (Wiley Liss Inc., New York, 1995).
Lengauer, C., Kinzler, K. W. & Vogelstein, B. Genetic instability in colorectal cancers. Nature 386, 623–627 (1997).
Kops, G. J., Foltz, D. R. & Cleveland, D. W. Lethality to human cancer cells through massive chromosome loss by inhibition of the mitotic checkpoint. Proc. Natl Acad. Sci. USA 101, 8699–8704 (2004).
Michel, L. et al. Complete loss of the tumor suppressor MAD2 causes premature cyclin B degradation and mitotic failure in human somatic cells. Proc. Natl Acad. Sci. USA 101, 4459–4464 (2004). References 7 and 8 show that the complete inhibition of the mitotic checkpoint causes death in human tumour cells.
Duesberg, P. et al. How aneuploidy may cause cancer and genetic instability. Anticancer Res. 19, 4887–4906 (1999).
Wang, S. I. et al. Somatic mutations of PTEN in glioblastoma multiforme. Cancer Res. 57, 4183–4186 (1997).
Fodde, R. & Smits, R. Cancer biology. A matter of dosage. Science 298, 761–763 (2002).
Wang, T. L. et al. Digital karyotyping identifies thymidylate synthase amplification as a mechanism of resistance to 5-fluorouracil in metastatic colorectal cancer patients. Proc. Natl Acad. Sci. USA 101, 3089–3094 (2004).
Sawyers, C. L. Research on resistance to cancer drug Gleevec. Science 294, 1834 (2001).
Storchova, Z. & Pellman, D. From polyploidy to aneuploidy, genome instability and cancer. Nature Rev. Mol. Cell Biol. 5, 45–54 (2004).
Nigg, E. A. Centrosome aberrations: cause or consequence of cancer progression? Nature Rev. Cancer 2, 815–825 (2002).
Zhou, H. et al. Tumour amplified kinase STK15/BTAK induces centrosome amplification, aneuploidy and transformation. Nature Genet. 20, 189–193 (1998).
Fukasawa, K., Choi, T., Kuriyama, R., Rulong, S. & Vande Woude, G. F. Abnormal centrosome amplification in the absence of p53. Science 271, 1744–1747 (1996).
Deng, C. X. Roles of BRCA1 in centrosome duplication. Oncogene 21, 6222–6227 (2002).
Pei, L. & Melmed, S. Isolation and characterization of a pituitary tumor-transforming gene (PTTG). Mol. Endocrinol 11, 433–441 (1997).
McGrew, J. T., Goetsch, L., Byers, B. & Baum, P. Requirement for ESP1 in the nuclear division of Saccharomyces cerevisiae. Mol. Biol. Cell 3, 1443–1454 (1992).
Uzawa, S., Samejima, I., Hirano, T., Tanaka, K. & Yanagida, M. The fission yeast cut1+ gene regulates spindle pole body duplication and has homology to the budding yeast ESP1 gene. Cell 62, 913–925 (1990).
Yamamoto, A., Guacci, V. & Koshland, D. Pds1p is required for faithful execution of anaphase in the yeast, Saccharomyces cerevisiae. J. Cell Biol. 133, 85–97 (1996).
Jallepalli, P. V. et al. Securin is required for chromosomal stability in human cells. Cell 105, 445–457 (2001).
Zhang, X. et al. Structure, expression, and function of human pituitary tumor-transforming gene (PTTG). Mol. Endocrinol. 13, 156–166 (1999).
Zhang, X. et al. Pituitary tumor transforming gene (PTTG) expression in pituitary adenomas. J. Clin. Endocrinol. Metab. 84, 761–767 (1999).
Cimini, D. et al. Merotelic kinetochore orientation is a major mechanism of aneuploidy in mitotic mammalian tissue cells. J. Cell Biol. 153, 517–527 (2001).
Gassmann, R. et al. Borealin: a novel chromosomal passenger required for stability of the bipolar mitotic spindle. J. Cell Biol. 166, 179–191 (2004).
Kaplan, K. B. et al. A role for the adenomatous polyposis coli protein in chromosome segregation. Nature Cell Biol. 3, 429–432 (2001).
Fodde, R. et al. Mutations in the APC tumour suppressor gene cause chromosomal instability. Nature Cell Biol. 3, 433–438 (2001).
Fodde, R., Smits, R. & Clevers, H. APC, signal transduction and genetic instability in colorectal cancer. Nature Rev. Cancer 1, 55–67 (2001).
Green, R. A. & Kaplan, K. B. Chromosome instability in colorectal tumor cells is associated with defects in microtubule plus-end attachments caused by a dominant mutation in APC. J. Cell Biol 163, 949–961 (2003).
Shin, H. J. et al. Dual roles of human BubR1, a mitotic checkpoint kinase, in the monitoring of chromosomal instability. Cancer Cell 4, 483–497 (2003).
Taylor, S. S. & McKeon, F. Kinetochore localization of murine Bub1 is required for normal mitotic timing and checkpoint response to spindle damage. Cell 89, 727–735 (1997).
Peters, J. M. The anaphase-promoting complex: proteolysis in mitosis and beyond. Mol. Cell 9, 931–943 (2002).
Cleveland, D. W., Mao, Y. & Sullivan, K. F. Centromeres and kinetochores. From epigenetics to mitotic checkpoint signaling. Cell 112, 407–421 (2003).
Rieder, C. L., Schultz, A., Cole, R. & Sluder, G. Anaphase onset in vertebrate somatic cells is controlled by a checkpoint that monitors sister kinetochore attachment to the spindle. J. Cell Biol. 127, 1301–1310 (1994).
Weiss, E. & Winey, M. The Saccharomyces cerevisiae spindle pole body duplication gene MPS1 is part of a mitotic checkpoint. J. Cell Biol. 132, 111–123 (1996).
Li, R. & Murray, A. W. Feedback control of mitosis in budding yeast. Cell 66, 519–531 (1991).
Hoyt, M. A., Totis, L. & Roberts, B. T. S. cerevisiae genes required for cell cycle arrest in response to loss of microtubule function. Cell 66, 507–517 (1991). References 38 and 39 reported the identification of the molecular components of the mitotic checkpoint in budding yeast.
Abrieu, A. et al. Mps1 is a kinetochore-associated kinase essential for the vertebrate mitotic checkpoint. Cell 106, 83–93 (2001).
Jin, D. Y., Spencer, F. & Jeang, K. T. Human T cell leukemia virus type 1 oncoprotein Tax targets the human mitotic checkpoint protein MAD1. Cell 93, 81–91 (1998).
Li, Y. & Benezra, R. Identification of a human mitotic checkpoint gene: hsMAD2. Science 274, 246–248 (1996).
Taylor, S. S., Ha, E. & McKeon, F. The human homologue of Bub3 is required for kinetochore localization of Bub1 and a Mad3/Bub1-related protein kinase. J. Cell Biol. 142, 1–11 (1998).
Abrieu, A., Kahana, J. A., Wood, K. W. & Cleveland, D. W. CENP-E as an essential component of the mitotic checkpoint in vitro. Cell 102, 817–826 (2000).
Chan, G. K., Jablonski, S. A., Sudakin, V., Hittle, J. C. & Yen, T. J. Human BUBR1 is a mitotic checkpoint kinase that monitors CENP-E functions at kinetochores and binds the cyclosome/APC. J. Cell Biol. 146, 941–954 (1999).
Chan, G. K., Jablonski, S. A., Starr, D. A., Goldberg, M. L. & Yen, T. J. Human Zw10 and ROD are mitotic checkpoint proteins that bind to kinetochores. Nature Cell Biol. 2, 944–947 (2000).
Mao, Y., Abrieu, A. & Cleveland, D. W. Activating and silencing the mitotic checkpoint through CENP-E-dependent activation/inactivation of BubR1. Cell 114, 87–98 (2003). This study shows how a lack of attachment at the kinetochore can be converted into a catalytic activity that sustains mitotic checkpoint signalling. The microtubule-binding protein CENPE, when unbound by microtubules, directly activates the checkpoint kinase BUBR1, which is an essential mediator of the mitotic checkpoint response.
Minshull, J., Sun, H., Tonks, N. K. & Murray, A. W. A MAP kinase-dependent spindle assembly checkpoint in Xenopus egg extracts. Cell 79, 475–486 (1994).
Shah, J. V. et al. Dynamics of centromere and kinetochore proteins; implications for checkpoint signaling and silencing. Curr. Biol. 14, 942–952 (2004).
Howell, B. J. et al. Spindle checkpoint protein dynamics at kinetochores in living cells. Curr. Biol. 14, 953–964 (2004).
Fang, G., Yu, H. & Kirschner, M. W. The checkpoint protein MAD2 and the mitotic regulator CDC20 form a ternary complex with the anaphase-promoting complex to control anaphase initiation. Genes Dev. 12, 1871–1883 (1998).
Luo, X. et al. The Mad2 spindle checkpoint protein has two distinct natively folded states. Nature Struct. Mol. Biol. 11, 338–345 (2004).
De Antoni, A. et al. The Mad1/Mad2 complex as a template for Mad2 activation in the spindle assembly checkpoint. Curr. Biol. 15, 214–225 (2005).
Fang, G. Checkpoint protein BubR1 acts synergistically with Mad2 to inhibit anaphase-promoting complex. Mol. Biol. Cell 13, 755–766 (2002).
Tang, Z., Bharadwaj, R., Li, B. & Yu, H. Mad2-Independent inhibition of APCCdc20 by the mitotic checkpoint protein BubR1. Dev. Cell 1, 227–237 (2001).
Sudakin, V., Chan, G. K. & Yen, T. J. Checkpoint inhibition of the APC/C in HeLa cells is mediated by a complex of BUBR1, BUB3, CDC20, and MAD2. J. Cell Biol. 154, 925–936 (2001). References 55 and 56 challenged the view that MAD2 alone was the APC/C inhibitor. Although reference 55 showed BUBR1 could directly inhibit the APC/C better than MAD2 could, reference 56 showed that in mitotic HeLa cells, both BUBR1 and MAD2, together with BUB3, were found in complex with CDC20, and that this complex was a very potent inhibitor of the APC/C.
Hwang, L. H. et al. Budding yeast Cdc20: a target of the spindle checkpoint. Science 279, 1041–1044 (1998).
Kallio, M., Weinstein, J., Daum, J. R., Burke, D. J. & Gorbsky, G. J. Mammalian p55CDC mediates association of the spindle checkpoint protein Mad2 with the cyclosome/anaphase-promoting complex, and is involved in regulating anaphase onset and late mitotic events. J. Cell Biol. 141, 1393–1406 (1998).
Chen, R. H. Phosphorylation and activation of Bub1 on unattached chromosomes facilitate the spindle checkpoint. EMBO J. 23, 3113–3121 (2004).
Ditchfield, C. et al. Aurora B couples chromosome alignment with anaphase by targeting BubR1, Mad2, and Cenp-E to kinetochores. J. Cell Biol. 161, 267–280 (2003).
Camasses, A., Bogdanova, A., Shevchenko, A. & Zachariae, W. The CCT chaperonin promotes activation of the anaphase-promoting complex through the generation of functional Cdc20. Mol. Cell 12, 87–100 (2003).
Lens, S. M. et al. Survivin is required for a sustained spindle checkpoint arrest in response to lack of tension. EMBO J. 22, 2934–2947 (2003).
Sassoon, I. et al. Regulation of Saccharomyces cerevisiae kinetochores by the type 1 phosphatase Glc7p. Genes Dev. 13, 545–555 (1999).
Hauf, S. et al. The small molecule hesperadin reveals a role for Aurora B in correcting kinetochore-microtubule attachment and in maintaining the spindle assembly checkpoint. J. Cell Biol. 161, 281–294 (2003).
Tanaka, T. U. et al. Evidence that the Ipl1–Sli15 (aurora kinase–INCENP) complex promotes chromosome bi-orientation by altering kinetochore–spindle pole connections. Cell 108, 317–329 (2002).
Lampson, M. A. & Kapoor, T. M. The human mitotic checkpoint protein BubR1 regulates chromosome–spindle attachments. Nature Cell Biol. 7, 93–98 (2005).
Putkey, F. R. et al. Unstable kinetochore-microtubule capture and chromosomal instability following deletion of CENP-E. Dev. Cell 3, 351–365 (2002).
Weaver, B. A. et al. Centromere-associated protein-E is essential for the mammalian mitotic checkpoint to prevent aneuploidy due to single chromosome loss. J. Cell Biol. 162, 551–563 (2003).
Habu, T., Kim, S. H., Weinstein, J. & Matsumoto, T. Identification of a MAD2-binding protein, CMT2, and its role in mitosis. EMBO J. 21, 6419–6428 (2002).
Xia, G. et al. Conformation-specific binding of p31(comet) antagonizes the function of Mad2 in the spindle checkpoint. EMBO J. 23, 3133–3143 (2004).
Kienitz, A., Vogel, C., Morales, I., Muller, R. & Bastians, H. Partial downregulation of MAD1 causes spindle checkpoint inactivation and aneuploidy, but does not confer resistance towards taxol. Oncogene 24, 4301–4310 (2005).
Cahill, D. P. et al. Mutations of mitotic checkpoint genes in human cancers. Nature 392, 300–303 (1998). First study to identify mutant alleles of the mitotic checkpoint proteins BUB1 and BUBR1 in human colorectal cancer cell lines.
Dai, W. et al. Slippage of mitotic arrest and enhanced tumor development in mice with BubR1 haploinsufficiency. Cancer Res. 64, 440–445 (2004).
Babu, J. R. et al. Rae1 is an essential mitotic checkpoint regulator that cooperates with Bub3 to prevent chromosome missegregation. J. Cell Biol. 160, 341–353 (2003).
Michel, M. L. et al. MAD2 haplo-insufficiency causes premature anaphase and chromosome instability in mammalian cells. Nature 409, 355–359 (2001). References 74 and 75 provided proof-of-principle that reduced levels of a mitotic checkpoint protein can cause checkpoint malfunction and CIN, and thereby contribute to tumour formation.
Baker, D. J. et al. BubR1 insufficiency causes early onset of aging-associated phenotypes and infertility in mice. Nature Genet. 36, 744–749 (2004).
Musio, A. et al. Inhibition of BUB1 results in genomic instability and anchorage-independent growth of normal human fibroblasts. Cancer Res. 63, 2855–2863 (2003).
Wang, Q. et al. BUBR1 deficiency results in abnormal megakaryopoiesis. Blood 103, 1278–1285 (2004).
Myung, K., Smith, S. & Kolodner, R. D. Mitotic checkpoint function in the formation of gross chromosomal rearrangements in Saccharomyces cerevisiae. Proc. Natl Acad. Sci. USA 101, 15980–15985 (2004).
Iouk, T., Kerscher, O., Scott, R. J., Basrai, M. A. & Wozniak, R. W. The yeast nuclear pore complex functionally interacts with components of the spindle assembly checkpoint. J. Cell Biol. 159, 807–819 (2002).
Campbell, M. S., Chan, G. K. & Yen, T. J. Mitotic checkpoint proteins HsMAD1 and HsMAD2 are associated with nuclear pore complexes in interphase. J. Cell. Sci. 114, 953–963 (2001).
Rao, C. V. et al. Colonic tumorigenesis in BubR1+/−ApcMin/+ compound mutant mice is linked to premature separation of sister chromatids and enhanced genomic instability. Proc. Natl Acad. Sci. USA 102, 4365–4370 (2005).
Connor, F. et al. Tumorigenesis and a DNA repair defect in mice with a truncating Brca2 mutation. Nature Genet. 17, 423–430 (1997).
Friedman, L. S. et al. Thymic lymphomas in mice with a truncating mutation in Brca2. Cancer Res. 58, 1338–1343 (1998).
Lee, H. et al. Mitotic checkpoint inactivation fosters transformation in cells lacking the breast cancer susceptibility gene, Brca2. Mol. Cell 4, 1–10 (1999).
Rieder, C. L. & Maiato, H. Stuck in division or passing through: what happens when cells cannot satisfy the spindle assembly checkpoint. Dev. Cell 7, 637–651 (2004).
Ouyang, B., Knauf, J. A., Ain, K., Nacev, B. & Fagin, J. A. Mechanisms of aneuploidy in thyroid cancer cell lines and tissues: evidence for mitotic checkpoint dysfunction without mutations in BUB1 and BUBR1. Clin. Endocrinol. (Oxf.) 56, 341–350 (2002).
Takahashi, T. et al. Identification of frequent impairment of the mitotic checkpoint and molecular analysis of the mitotic checkpoint genes, hsMAD2 and p55CDC, in human lung cancers. Oncogene 18, 4295–4300 (1999).
Wang, X. et al. Significance of MAD2 expression to mitotic checkpoint control in ovarian cancer cells. Cancer Res. 62, 1662–1668 (2002).
Tighe, A., Johnson, V. L., Albertella, M. & Taylor, S. S. Aneuploid colon cancer cells have a robust spindle checkpoint. EMBO Rep. 2, 609–614 (2001).
Saeki, A. et al. Frequent impairment of the spindle assembly checkpoint in hepatocellular carcinoma. Cancer 94, 2047–2054 (2002).
Yoon, D. S. et al. Variable levels of chromosomal instability and mitotic spindle checkpoint defects in breast cancer. Am. J. Pathol. 161, 391–397 (2002).
Grigorova, M., Staines, J. M., Ozdag, H., Caldas, C. & Edwards, P. A. Possible causes of chromosome instability: comparison of chromosomal abnormalities in cancer cell lines with mutations in BRCA1, BRCA2, CHK2 and BUB1. Cytogenet. Genome Res. 104, 333–340 (2004).
Gemma, A. et al. Somatic mutation of the hBUB1 mitotic checkpoint gene in primary lung cancer. Genes Chromosomes Cancer 29, 213–218 (2000).
Hempen, P. M., Kurpad, H., Calhoun, E. S., Abraham, S. & Kern, S. E. A double missense variation of the BUB1 gene and a defective mitotic spindle checkpoint in the pancreatic cancer cell line Hs766T. Hum. Mutat. 21, 445 (2003).
Hernando, E. et al. Molecular analyses of the mitotic checkpoint components hsMAD2, hBUB1 and hBUB3 in human cancer. Int. J. Cancer 95, 223–227 (2001).
Imai, Y., Shiratori, Y., Kato, N., Inoue, T. & Omata, M. Mutational inactivation of mitotic checkpoint genes, hsMAD2 and hBUB1, is rare in sporadic digestive tract cancers. Jpn. J. Cancer Res. 90, 837–840 (1999).
Ohshima, K. et al. Mutation analysis of mitotic checkpoint genes (hBUB1 and hBUBR1) and microsatellite instability in adult T-cell leukemia/lymphoma. Cancer Lett. 158, 141–150 (2000).
Nomoto, S. et al. Search for in vivo somatic mutations in the mitotic checkpoint gene, hMAD1, in human lung cancers. Oncogene 18, 7180–7183 (1999).
Percy, M. J. et al. Expression and mutational analyses of the human MAD2L1 gene in breast cancer cells. Genes Chromosomes Cancer 29, 356–362 (2000).
Tsukasaki, K. et al. Mutations in the mitotic check point gene, MAD1L1, in human cancers. Oncogene 20, 3301–3305 (2001).
Shichiri, M., Yoshinaga, K., Hisatomi, H., Sugihara, K. & Hirata, Y. Genetic and epigenetic inactivation of mitotic checkpoint genes hBUB1 and hBUBR1 and their relationship to survival. Cancer Res. 62, 13–17 (2002).
Wang, Z. et al. Three classes of genes mutated in colorectal cancers with chromosomal instability. Cancer Res. 64, 2998–3001 (2004).
Hanks, S. et al. Constitutional aneuploidy and cancer predisposition caused by biallelic mutations in BUB1B. Nature Genet. 36, 1159–1161 (2004).
Haruki, N. et al. Molecular analysis of the mitotic checkpoint genes BUB1, BUBR1 and BUB3 in human lung cancers. Cancer Lett. 162, 201–205 (2001).
Olesen, S. H., Thykjaer, T. & Orntoft, T. F. Mitotic checkpoint genes hBUB1, hBUB1B, hBUB3 and TTK in human bladder cancer, screening for mutations and loss of heterozygosity. Carcinogenesis 22, 813–815 (2001).
Cahill, D. P. et al. Characterization of MAD2B and other mitotic spindle checkpoint genes. Genomics 58, 181–187 (1999).
Myrie, K. A., Percy, M. J., Azim, J. N., Neeley, C. K. & Petty, E. M. Mutation and expression analysis of human BUB1 and BUB1B in aneuploid breast cancer cell lines. Cancer Lett. 152, 193–199 (2000).
Scolnick, D. M. & Halazonetis, T. D. Chfr defines a mitotic stress checkpoint that delays entry into metaphase. Nature 406, 430–435 (2000).
Tao, W. et al. Human homologue of the Drosophila melanogaster lats tumour suppressor modulates CDC2 activity. Nature Genet. 21, 177–181 (1999).
Song, M. S. et al. The tumour suppressor RASSF1A regulates mitosis by inhibiting the APC–Cdc20 complex. Nature Cell Biol. 6, 129–137 (2004).
Grabsch, H. et al. Overexpression of the mitotic checkpoint genes BUB1, BUBR1, and BUB3 in gastric cancer — association with tumour cell proliferation. J. Pathol. 200, 16–22 (2003).
Hernando, E. et al. Rb inactivation promotes genomic instability by uncoupling cell cycle progression from mitotic control. Nature 430, 797–802 (2004). This study shows how a tumour suppressor can manipulate the mitotic checkpoint to create CIN. Aneuploidy occurs in RB-negative tumor cells via increased levels of MAD2. MAD2L1 is a target gene of the RB-controlled transcription factor E2F.
Cheung, H. W. et al. Mitotic arrest deficient 2 expression induces chemosensitization to a DNA-damaging agent, cisplatin, in nasopharyngeal carcinoma cells. Cancer Res. 65, 1450–1458 (2005).
Wang, R. H., Yu, H. & Deng, C. X. A requirement for breast-cancer-associated gene 1 (BRCA1) in the spindle heckpoint. Proc. Natl Acad. Sci. USA 101, 17108–17113 (2004).
van't Veer, L. J. et al. Gene expression profiling predicts clinical outcome of breast cancer. Nature 415, 530–536 (2002).
Ren, B. et al. E2F integrates cell cycle progression with DNA repair, replication, and G(2)/M checkpoints. Genes Dev. 16, 245–256 (2002).
Chun, A. C. & Jin, D. Y. Transcriptional regulation of mitotic checkpoint gene MAD1 by p53. J. Biol. Chem. 278, 37439–37450 (2003).
Iwanaga, Y. & Jeang, K. T. Expression of mitotic spindle checkpoint protein hsMAD1 correlates with cellular proliferation and is activated by a gain-of-function p53 mutant. Cancer Res. 62, 2618–2624 (2002).
Gupta, A., Inaba, S., Wong, O. K., Fang, G. & Liu, J. Breast cancer-specific gene 1 interacts with the mitotic checkpoint kinase BubR1. Oncogene 22, 7593–7599 (2003).
Jordan, M. A. & Wilson, L. Microtubules as a target for anticancer drugs. Nature Rev. Cancer 4, 253–265 (2004).
Keen, N. & Taylor, S. Aurora-kinase inhibitors as anticancer agents. Nature Rev. Cancer 4, 927–936 (2004).
Harrington, E. A. et al. VX-680, a potent and selective small-molecule inhibitor of the Aurora kinases, suppresses tumor growth in vivo. Nature Med. 10, 262–267 (2004).
Dorer, R. K. et al. A small-molecule inhibitor of Mps1 blocks the spindle-checkpoint response to a lack of tension on mitotic chromosomes. Curr. Biol. 15, 1070–1076 (2005).
Larsen, N. A. & Harrison, S. C. Crystal structure of the spindle assembly checkpoint protein Bub3. J. Mol. Biol. 344, 885–892 (2004).
Kops, G. J. et al. ZW10 links mitotic checkpoint signaling to the structural kinetochore. J. Cell Biol. 169, 49–60 (2005).
Luo, X. et al. Structure of the Mad2 spindle assembly checkpoint protein and its interaction with Cdc20. Nature Struct. Biol. 7, 224–229 (2000).
Tang, Z., Shu, H., Oncel, D., Chen, S. & Yu, H. Phosphorylation of Cdc20 by Bub1 provides a catalytic mechanism for APC/C inhibition by the spindle checkpoint. Mol. Cell 16, 387–397 (2004).
Chen, R. H., Shevchenko, A., Mann, M. & Murray, A. W. Spindle checkpoint protein Xmad1 recruits Xmad2 to unattached kinetochores. J. Cell Biol. 143, 283–295 (1998).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- ADENOMATOSIS POLYPOSIS COLI
-
Tumour-suppressor protein that, in a mutated, defective form, causes familial adenomatous polyposis (FAP), a rare hereditary disease in which patients have thousands of colorectal polyps that develop into tumours. Most sporadic colorectal tumours harbour mutations in both APC alleles.
- LOSS OF HETEROZYGOSITY
-
Following acquisition of a deleterious mutation in one of the two copies of a specific gene, loss of heterozygosity occurs from subsequent loss of, or mutation in, the normal allele.
- NUDE MICE
-
Strains of athymic mice bearing the recessive allele nu/nu, which are largely hairless and lack all, or most, of the T-cell population. Nude mice can accept either allografts or xenografts. nu/nu alleles on some backgrounds have near-normal numbers of T-cells.
- KINETOCHORE
-
A multiprotein structure, positioned at the central constriction of each chromosome (centromere), which is responsible for chromosome attachment to the mitotic spindle, chromosome segregation during anaphase and mitotic checkpoint activity.
- ANAPHASE PROMOTING COMPLEX/CYCLOSOME
-
Multisubunit E3 ubiquitin ligase required for mitotic progression by targeting key mitotic regulators such as cyclin B1 and securin for destruction through direct poly-ubiquitylation. Note that the nomenclature can be confusing here: the APC/C is completely distinct from the APC associated with β-catenin signalling and colorectal cancer.
- MICROTUBULE CAPTURE
-
Process in prometaphase in which unattached kinetochores each interact with one or more microtubules that emanate from one spindle pole. The interaction is mediated by microtubule-binding proteins that stably associate with the kinetochore.
- MEGAKARYOPOIESIS
-
Process that leads to the production of megakaryocytes, the polyploid precursor to platelets. These precursors develop from haematopoietic stem cells by executing several cell-cycles in which cytokinesis is skipped (known as endoreduplication).
- DOMINANT-INTERFERING MUTANT
-
Non-functional mutant protein that inhibits the function of the endogenous wild-type protein. These mutants often work by occupying subcellular binding sites required for the activation or correct subcellular positioning of the wild-type protein.
- NOCODAZOLE
-
Synthetic compound that binds tubulin dimers, thereby causing inibition of microtubule polymerization. Because of the highly dynamic nature of spindle microtubules in mitosis, this results in complete depolymerization of the cellular microtubule network.
- CELL-AUTONOMOUS LETHAL
-
The situation in which a deleterious subcellular environment (such as a genetic lesion that inactivates the mitotic checkpoint) causes cell death from damage solely developed within that cell. Surrounding cells have no influence on this process.
Rights and permissions
About this article
Cite this article
Kops, G., Weaver, B. & Cleveland, D. On the road to cancer: aneuploidy and the mitotic checkpoint. Nat Rev Cancer 5, 773–785 (2005). https://doi.org/10.1038/nrc1714
Issue Date:
DOI: https://doi.org/10.1038/nrc1714
This article is cited by
-
Profilin 1 deficiency drives mitotic defects and reduces genome stability
Communications Biology (2023)
-
Principles and dynamics of spindle assembly checkpoint signalling
Nature Reviews Molecular Cell Biology (2023)
-
Tracking circulating PD-L1-positive cells to monitor the outcome of patients with gastric cancer receiving anti-HER2 plus anti-PD-1 therapy
Human Cell (2023)
-
Potential antitumor activity of garlic against colorectal cancer: focus on the molecular mechanisms of action
European Journal of Nutrition (2023)
-
Heterogeneous circulating tumor cells correlate with responses to neoadjuvant chemotherapy and prognosis in patients with locally advanced breast cancer
Breast Cancer Research and Treatment (2023)