Key Points
-
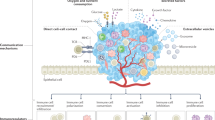
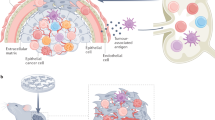
The pathological interactions between cancer cells and host immune cells in the tumour microenvironment create an immunosuppressive network that promotes tumour growth, protects the tumour from immune attack and attenuates immunotherapeutic efficacy.
-
Poor tumour-associated antigen (TAA)-specific immunity is not simply due to a passive process whereby adaptive immunity is shielded from detecting TAAs. There is an active process of 'tolerization' taking place in the tumour microenvironment.
-
Tumour tolerization is the result of imbalances in the tumour microenvironment, including alterations in antigen-presenting-cell subsets, co-stimulatory and co-inhibitory molecule alterations and altered ratios of effector T cells and regulatory T cells.
-
Human tumorigenesis is a slow process that can occur over several years and in this respect is similar to chronic infection. The lack of an acute phase in the course of tumorigenesis might profoundly shape T-cell immune responses, including the quality of antigen release, T-cell priming and activation.
-
Current immunotherapies often target patients with advanced-stage tumours, which have high levels of inflammatory molecules, cytokines, chemokines, tumour-infiltrating T cells, dendritic cells and macrophages. It is arguable whether we need to incorporate more of these components into tumour treatments.
-
Immune tolerization is predominant in the immune system in patients with advanced-stage tumours. It is time to consider combinatorial tumour therapies, including those that subvert the immune-tolerizing conditions within the tumour.
Abstract
It is well known that many tumours are potentially immunogenic, as corroborated by the presence of tumour-specific immune responses in vivo. Nonetheless, spontaneous clearance of established tumours by endogenous immune mechanisms is rare. Therefore, the focus of most cancer immunotherapies is to supplement essential immunogenic elements to boost tumour-specific immunity. Why then has tumour immunotherapy resulted in a generally poor clinical efficiency? The reason might lie in the increasingly documented fact that tumours develop diverse strategies that escape tumour-specific immunity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Dunn, G. P., Bruce, A. T., Ikeda, H., Old, L. J. & Schreiber, R. D. Cancer immunoediting: from immunosurveillance to tumor escape. Nature Immunol. 3, 991–998 (2002).
Smyth, M. J., Godfrey, D. I. & Trapani, J. A. A fresh look at tumor immunosurveillance and immunotherapy. Nature Immunol. 2, 293–299 (2001). References 1 and 2 re-visit and refine the concept of tumour immune surveillance. The authors provide their own opinions and views on this subject.
Melief, C. J. et al. Strategies for immunotherapy of cancer. Adv. Immunol. 75, 235–282 (2000).
Pardoll, D. Does the immune system see tumors as foreign or self? Annu. Rev. Immunol. 21, 807–839 (2003).
Finn, O. J. Cancer vaccines: between the idea and the reality. Nature Rev. Immunol. 3, 630–641 (2003).
Khong, H. T. & Restifo, N. P. Natural selection of tumor variants in the generation of 'tumor escape' phenotypes. Nature Immunol. 3, 999–1005 (2002).
Gilboa, E. The promise of cancer vaccines. Nature Rev. Cancer 4, 401–411 (2004).
Schuler, G., Schuler-Thurner, B. & Steinman, R. M. The use of dendritic cells in cancer immunotherapy. Curr. Opin. Immunol. 15, 138–147 (2003).
Yee, C. & Greenberg, P. Modulating T-cell immunity to tumours: new strategies for monitoring T-cell responses. Nature Rev. Cancer 2, 409–419 (2002).
O'Neill, D. W., Adams, S. & Bhardwaj, N. Manipulating dendritic cell biology for the active immunotherapy of cancer. Blood 104, 2235–2246 (2004).
Knutson, K. L., Schiffman, K. & Disis, M. L. Immunization with a HER-2/neu helper peptide vaccine generates HER-2/neu CD8 T-cell immunity in cancer patients. J. Clin. Invest. 107, 477–484 (2001).
Hwu, P. & Freedman, R. S. The immunotherapy of patients with ovarian cancer. J. Immunother. 25, 189–201 (2002).
Figdor, C. G., de Vries, I. J., Lesterhuis, W. J. & Melief, C. J. Dendritic cell immunotherapy: mapping the way. Nature Med. 10, 475–480 (2004).
Melief, C. J. Tumor eradication by adoptive transfer of cytotoxic T lymphocytes. Adv. Cancer Res. 58, 143–175 (1992).
Rosenberg, S. A. et al. Use of tumor-infiltrating lymphocytes and interleukin-2 in the immunotherapy of patients with metastatic melanoma. A preliminary report. N. Engl. J. Med. 319, 1676–1680 (1988).
Dudley, M. E. et al. Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science 298, 850–854 (2002).
Pamer, E. & Cresswell, P. Mechanisms of MHC class I-restricted antigen processing. Annu. Rev. Immunol. 16, 323–358 (1998).
Itano, A. A. & Jenkins, M. K. Antigen presentation to naive CD4 T cells in the lymph node. Nature Immunol. 4, 733–739 (2003).
Mueller, S. N., Jones, C. M., Smith, C. M., Heath, W. R. & Carbone, F. R. Rapid cytotoxic T lymphocyte activation occurs in the draining lymph nodes after cutaneous herpes simplex virus infection as a result of early antigen presentation and not the presence of virus. J. Exp. Med. 195, 651–656 (2002).
Kaech, S. M. & Ahmed, R. Memory CD8+ T cell differentiation: initial antigen encounter triggers a developmental program in naive cells. Nature Immunol. 2, 415–422 (2001).
van Stipdonk, M. J., Lemmens, E. E. & Schoenberger, S. P. Naive CTLs require a single brief period of antigenic stimulation for clonal expansion and differentiation. Nature Immunol. 2, 423–429 (2001).
Wong, P. & Pamer, E. G. CD8 T cell responses to infectious pathogens. Annu. Rev. Immunol. 21, 29–70 (2003).
Degli-Esposti, M. A. & Smyth, M. J. Close encounters of different kinds: dendritic cells and NK cells take centre stage. Nature Rev. Immunol. 5, 112–124 (2005).
Spiotto, M. T. et al. Increasing tumor antigen expression overcomes 'ignorance' to solid tumors via crosspresentation by bone marrow-derived stromal cells. Immunity 17, 737–747 (2002).
Ochsenbein, A. F. et al. Roles of tumour localization, second signals and cross priming in cytotoxic T-cell induction. Nature 411, 1058–1064 (2001).
Ochsenbein, A. F. et al. Immune surveillance against a solid tumor fails because of immunological ignorance. Proc. Natl Acad. Sci. USA 96, 2233–2238 (1999).
Palmowski, M., Salio, M., Dunbar, R. P. & Cerundolo, V. The use of HLA class I tetramers to design a vaccination strategy for melanoma patients. Immunol. Rev. 188, 155–163 (2002).
Romero, P. et al. Ex vivo staining of metastatic lymph nodes by class I major histocompatibility complex tetramers reveals high numbers of antigen-experienced tumor-specific cytolytic T lymphocytes. J. Exp. Med. 188, 1641–1650 (1998).
Yu, P. et al. Priming of naive T cells inside tumors leads to eradication of established tumors. Nature Immunol. 5, 141–149 (2004). Demonstrates that efficient TAA-specific priming in the tumour microenvironment is possible.
Feuerer, M. et al. Bone marrow as a priming site for T-cell responses to blood-borne antigen. Nature Med. 9, 1151–1157 (2003).
Banchereau, J. & Steinman, R. M. Dendritic cells and the control of immunity. Nature 392, 245–252 (1998).
Lanzavecchia, A. & Sallusto, F. The instructive role of dendritic cells on T cell responses: lineages, plasticity and kinetics. Curr. Opin. Immunol. 13, 291–298 (2001).
Colonna, M., Trinchieri, G. & Liu, Y. J. Plasmacytoid dendritic cells in immunity. Nature Immunol. 5, 1219–1226 (2004).
Cerundolo, V., Hermans, I. F. & Salio, M. Dendritic cells: a journey from laboratory to clinic. Nature Immunol. 5, 7–10 (2004).
Zou, W. et al. Stromal-derived factor-1 in human tumors recruits and alters the function of plasmacytoid precursor dendritic cells. Nature Med. 7, 1339–1346 (2001). The first functional study of human tumour-associated plasmacytoid DCs. This paper shows that tumour-derived CXCL12 attracts plasmacytoid DCs and that tumour plasmacytoid DCs induce IL-10+ suppressive T cells.
Curiel, T. J. et al. Blockade of B7-H1 improves myeloid dendritic cell-mediated antitumor immunity. Nature Med. 9, 562–567 (2003).
Curiel, T. J. et al. Dendritic cell subsets differentially regulate angiogenesis in human ovarian cancer. Cancer Res. 64, 5535–5538 (2004).
Gabrilovich, D. I. et al. Production of vascular endothelial growth factor by human tumors inhibits the functional maturation of dendritic cells. Nature Med. 2, 1096–1103 (1996). The first study showing that VEGF inhibits DC differentiation from CD34+ progenitor cells.
Munn, D. H. & Mellor, A. L. IDO and tolerance to tumors. Trends Mol. Med. 10, 15–18 (2004).
Bell, D. et al. In breast carcinoma tissue, immature dendritic cells reside within the tumor, whereas mature dendritic cells are located in peritumoral areas. J. Exp. Med. 190, 1417–1426 (1999).
Iwamoto, M. et al. Prognostic value of tumor-infiltrating dendritic cells expressing CD83 in human breast carcinomas. Int. J. Cancer 104, 92–97 (2003).
Troy, A., Davidson, P., Atkinson, C. & Hart, D. Phenotypic characterisation of the dendritic cell infiltrate in prostate cancer. J. Urol. 160, 214–219 (1998).
Troy, A. J., Summers, K. L., Davidson, P. J., Atkinson, C. H. & Hart, D. N. Minimal recruitment and activation of dendritic cells within renal cell carcinoma. Clin. Cancer Res. 4, 585–593 (1998).
Kryczek, I. et al. CXCL12 and vascular endothelial growth factor synergistically induce neoangiogenesis in human ovarian cancers. Cancer Res. 65, 465–472 (2005).
Carmeliet, P. & Jain, R. K. Angiogenesis in cancer and other diseases. Nature 407, 249–257 (2000).
Menetrier-Caux, C. et al. Inhibition of the differentiation of dendritic cells from CD34+ progenitors by tumor cells: role of interleukin-6 and macrophage colony-stimulating factor. Blood 92, 4778–4791 (1998).
Kryczek, I., Grybos, M., Karabon, L., Klimczak, A. & Lange, A. IL-6 production in ovarian carcinoma is associated with histiotype and biological characteristics of the tumour and influences local immunity. Br. J. Cancer 82, 621–628 (2000).
Freedman, R. S., Deavers, M., Liu, J. & Wang, E. Peritoneal inflammation — a microenvironment for Epithelial Ovarian Cancer (EOC). J. Transl. Med. 2, 23 (2004).
Pasare, C. & Medzhitov, R. Toll pathway-dependent blockade of CD4+CD25+ T cell-mediated suppression by dendritic cells. Science 299, 1033–1036 (2003).
Shono, T., Tofilon, P. J., Bruner, J. M., Owolabi, O. & Lang, F. F. Cyclooxygenase-2 expression in human gliomas: prognostic significance and molecular correlations. Cancer Res. 61, 4375–4381 (2001).
Wolff, H. et al. Expression of cyclooxygenase-2 in human lung carcinoma. Cancer Res. 58, 4997–5001 (1998).
Joki, T. et al. Expression of cyclooxygenase 2 (COX-2) in human glioma and in vitro inhibition by a specific COX-2 inhibitor, NS-398. Cancer Res. 60, 4926–4931 (2000).
Kalinski, P., Schuitemaker, J. H., Hilkens, C. M. & Kapsenberg, M. L. Prostaglandin E2 induces the final maturation of IL-12-deficient CD1a+CD83+ dendritic cells: the levels of IL-12 are determined during the final dendritic cell maturation and are resistant to further modulation. J. Immunol. 161, 2804–2809 (1998).
Sombroek, C. C. et al. Prostanoids play a major role in the primary tumor-induced inhibition of dendritic cell differentiation. J. Immunol. 168, 4333–4343 (2002).
Jozefowski, S., Bobek, M. & Marcinkiewicz, J. Exogenous but not endogenous prostanoids regulate cytokine secretion from murine bone marrow dendritic cells: EP2, DP, and IP but not EP1, EP3, and FP prostanoid receptors are involved. Int. Immunopharmacol. 3, 865–878 (2003).
Akasaki, Y. et al. Induction of a CD4+ T regulatory type 1 response by cyclooxygenase-2-overexpressing glioma. J. Immunol. 173, 4352–4359 (2004).
Shurin, G. V. et al. Neuroblastoma-derived gangliosides inhibit dendritic cell generation and function. Cancer Res. 61, 363–369 (2001).
Peguet-Navarro, J. et al. Gangliosides from human melanoma tumors impair dendritic cell differentiation from monocytes and induce their apoptosis. J. Immunol. 170, 3488–3494 (2003).
Labeur, M. S. et al. Generation of tumor immunity by bone marrow-derived dendritic cells correlates with dendritic cell maturation stage. J. Immunol. 162, 168–175 (1999).
Dhodapkar, M. V. et al. Rapid generation of broad T-cell immunity in humans after a single injection of mature dendritic cells. J. Clin. Invest. 104, 173–180 (1999).
Jonuleit, H., Schmitt, E., Schuler, G., Knop, J. & Enk, A. H. Induction of interleukin 10-producing, nonproliferating CD4+ T cells with regulatory properties by repetitive stimulation with allogeneic immature human dendritic cells. J. Exp. Med. 192, 1213–1222 (2000).
Dhodapkar, M. V., Steinman, R. M., Krasovsky, J., Munz, C. & Bhardwaj, N. Antigen-specific inhibition of effector T cell function in humans after injection of immature dendritic cells. J. Exp. Med. 193, 233–238 (2001).
Hawiger, D. et al. Dendritic cells induce peripheral T cell unresponsiveness under steady state conditions in vivo. J. Exp. Med. 194, 769–779 (2001).
Choi, I. H. et al. Genomic organization and expression analysis of B7-H4, an immune inhibitory molecule of the B7 family. J. Immunol. 171, 4650–4654 (2003).
Curiel, T. J. et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nature Med. 10, 942–949 (2004). Provides a direct functional and clinical link between regulatory T cells and human tumour immunopathogenesis.
Chen, L. Co-inhibitory molecules of the B7-CD28 family in the control of T-cell immunity. Nature Rev. Immunol. 4, 336–347 (2004).
Brown, J. A. et al. Blockade of programmed death-1 ligands on dendritic cells enhances T cell activation and cytokine production. J. Immunol. 170, 1257–1266 (2003).
Dong, H. et al. Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nature Med. 8, 793–800 (2002).
Strome, S. E. et al. B7-H1 blockade augments adoptive T-cell immunotherapy for squamous cell carcinoma. Cancer Res. 63, 6501–6505 (2003).
Iwai, Y. et al. Involvement of PD-L1 on tumor cells in the escape from host immune system and tumor immunotherapy by PD-L1 blockade. Proc. Natl Acad. Sci. USA 99, 12293–12297 (2002).
Iwai, Y., Terawaki, S. & Honjo, T. PD-1 blockade inhibits hematogenous spread of poorly immunogenic tumor cells by enhanced recruitment of effector T cells. Int. Immunol. 17, 133–144 (2005). Reference 36 and 68–71 report that B7-H1 or PD-1 in the tumour microenvironment attenuates tumour-specific immunity and that blocking B7-H1 improves tumour immunity in vivo.
Sica, G. L. et al. B7-H4, a molecule of the B7 family, negatively regulates T cell immunity. Immunity 18, 849–861 (2003).
Prasad, D. V., Richards, S., Mai, X. M. & Dong, C. B7S1, a novel B7 family member that negatively regulates T cell activation. Immunity 18, 863–873 (2003).
Zang, X. et al. B7x: a widely expressed B7 family member that inhibits T cell activation. Proc. Natl Acad. Sci. USA 100, 10388–10392 (2003).
Tamura, H. et al. B7-H1 costimulation preferentially enhances CD28-independent T-helper cell function. Blood 97, 1809–1816 (2001).
Subudhi, S. K. et al. Local expression of B7-H1 promotes organ-specific autoimmunity and transplant rejection. J. Clin. Invest. 113, 694–700 (2004).
Munn, D. H. et al. Potential regulatory function of human dendritic cells expressing indoleamine 2,3-dioxygenase. Science 297, 1867–1870 (2002).
Uyttenhove, C. et al. Evidence for a tumoral immune resistance mechanism based on tryptophan degradation by indoleamine 2,3-dioxygenase. Nature Med. 9, 1269–1274 (2003). Reference 77 and 78 document the relevance of IDO-expressing dendritic cells or tumours in tumour immune responses.
Grohmann, U. et al. CTLA-4-Ig regulates tryptophan catabolism in vivo. Nature Immunol. 3, 1097–1101 (2002).
Salio, M. et al. Plasmacytoid dendritic cells prime IFN-γ-secreting melanoma-specific CD8 lymphocytes and are found in primary melanoma lesions. Eur. J. Immunol. 33, 1052–1062 (2003).
Hartmann, E. et al. Identification and functional analysis of tumor-infiltrating plasmacytoid dendritic cells in head and neck cancer. Cancer Res. 63, 6478–6487 (2003).
Moseman, E. A. et al. Human plasmacytoid dendritic cells activated by CpG oligodeoxynucleotides induce the generation of CD4+CD25+ regulatory T cells. J. Immunol. 173, 4433–4442 (2004).
Gilliet, M. & Liu, Y. J. Generation of human CD8 T regulatory cells by CD40 ligand-activated plasmacytoid dendritic cells. J. Exp. Med. 195, 695–704 (2002).
Conejo-Garcia, J. R. et al. Tumor-infiltrating dendritic cell precursors recruited by a β-defensin contribute to vasculogenesis under the influence of Vegf-A. Nature Med. 10, 950–958 (2004).
Kusmartsev, S. A., Li, Y. & Chen, S. H. Gr-1+ myeloid cells derived from tumor-bearing mice inhibit primary T cell activation induced through CD3/CD28 costimulation. J. Immunol. 165, 779–785 (2000).
Mazzoni, A. et al. Myeloid suppressor lines inhibit T cell responses by an NO-dependent mechanism. J. Immunol. 168, 689–695 (2002).
Schmielau, J. & Finn, O. J. Activated granulocytes and granulocyte-derived hydrogen peroxide are the underlying mechanism of suppression of T-cell function in advanced cancer patients. Cancer Res. 61, 4756–4760 (2001).
Almand, B. et al. Increased production of immature myeloid cells in cancer patients: a mechanism of immunosuppression in cancer. J. Immunol. 166, 678–689 (2001).
Bronte, V., Serafini, P., Apolloni, E. & Zanovello, P. Tumor-induced immune dysfunctions caused by myeloid suppressor cells. J. Immunother. 24, 431–446 (2001).
Saio, M., Radoja, S., Marino, M. & Frey, A. B. Tumor-infiltrating macrophages induce apoptosis in activated CD8+ T cells by a mechanism requiring cell contact and mediated by both the cell-associated form of TNF and nitric oxide. J. Immunol. 167, 5583–5593 (2001).
Rodriguez, P. C. et al. L-arginine consumption by macrophages modulates the expression of CD3 ζ chain in T lymphocytes. J. Immunol. 171, 1232–1239 (2003).
Rodriguez, P. C. et al. Arginase I production in the tumor microenvironment by mature myeloid cells inhibits T-cell receptor expression and antigen-specific T-cell responses. Cancer Res. 64, 5839–5849 (2004).
Matzinger, P. The danger model: a renewed sense of self. Science 296, 301–305 (2002).
Fuchs, E. J. & Matzinger, P. Is cancer dangerous to the immune system? Semin. Immunol. 8, 271–280 (1996).
Vakkila, J. & Lotze, M. T. Inflammation and necrosis promote tumour growth. Nature Rev. Immunol. 4, 641–648 (2004).
Balkwill, F. & Mantovani, A. Inflammation and cancer: back to Virchow? Lancet 357, 539–545 (2001).
Jacoby, R. F., Seibert, K., Cole, C. E., Kelloff, G. & Lubet, R. A. The cyclooxygenase-2 inhibitor celecoxib is a potent preventive and therapeutic agent in the min mouse model of adenomatous polyposis. Cancer Res. 60, 5040–5044 (2000).
Levin, G., Kariv, N., Khomiak, E. & Raz, A. Indomethacin inhibits the accumulation of tumor cells in mouse lungs and subsequent growth of lung metastases. Chemotherapy 46, 429–437 (2000).
Steinbach, G. et al. The effect of celecoxib, a cyclooxygenase-2 inhibitor, in familial adenomatous polyposis. N. Engl. J. Med. 342, 1946–1952 (2000).
Duperron, C. & Castonguay, A. Chemopreventive efficacies of aspirin and sulindac against lung tumorigenesis in A/J mice. Carcinogenesis 18, 1001–1006 (1997).
Leahy, K. M. et al. Cyclooxygenase-2 inhibition by celecoxib reduces proliferation and induces apoptosis in angiogenic endothelial cells in vivo. Cancer Res. 62, 625–631 (2002).
Tsubouchi, Y. et al. Meloxicam inhibits the growth of non-small cell lung cancer. Anticancer Res. 20, 2867–2872 (2000).
Choy, H. & Milas, L. Enhancing radiotherapy with cyclooxygenase-2 enzyme inhibitors: a rational advance? J. Natl Cancer Inst. 95, 1440–1452 (2003).
Dong, H., Zhu, G., Tamada, K. & Chen, L. B7-H1, a third member of the B7 family, co-stimulates T-cell proliferation and interleukin-10 secretion. Nature Med. 5, 1365–1369 (1999).
Hussain, S. F. & Paterson, Y. CD4+CD25+ regulatory T cells that secrete TGFβ and IL-10 are preferentially induced by a vaccine vector. J. Immunother. 27, 339–346 (2004).
Chakraborty, N. G., Chattopadhyay, S., Mehrotra, S., Chhabra, A. & Mukherji, B. Regulatory T-cell response and tumor vaccine-induced cytotoxic T lymphocytes in human melanoma. Hum. Immunol. 65, 794–802 (2004).
van Mierlo, G. J. et al. Activation of dendritic cells that cross-present tumor-derived antigen licenses CD8+ CTL to cause tumor eradication. J. Immunol. 173, 6753–6759 (2004).
Yamazaki, S. et al. Direct expansion of functional CD25+CD4+ regulatory T cells by antigen-processing dendritic cells. J. Exp. Med. 198, 235–247 (2003).
Donzella, G. A. et al. AMD3100, a small molecule inhibitor of HIV-1 entry via the CXCR4 co-receptor. Nature Med. 4, 72–77 (1998).
Muller, A. et al. Involvement of chemokine receptors in breast cancer metastasis. Nature 410, 50–56 (2001).
Le Bon, A. et al. Cross-priming of CD8+ T cells stimulated by virus-induced type I interferon. Nature Immunol. 4, 1009–1015 (2003).
Klinman, D. M. Immunotherapeutic uses of CpG oligodeoxynucleotides. Nature Rev. Immunol. 4, 249–258 (2004).
Krieg, A. M. CpG motifs: the active ingredient in bacterial extracts? Nature Med. 9, 831–835 (2003).
Shevach, E. M. Fatal attraction: tumors beckon regulatory T cells. Nature Med. 10, 900–901 (2004).
Shevach, E. M. CD4+ CD25+ suppressor T cells: more questions than answers. Nature Rev. Immunol. 2, 389–400 (2002).
Von Herrath, M. G. & Harrison, L. C. Regulatory lymphocytes: antigen-induced regulatory T cells in autoimmunity. Nature Rev. Immunol. 3, 223–232 (2003).
Sakaguchi, S. et al. Immunologic tolerance maintained by CD25+ CD4+ regulatory T cells: their common role in controlling autoimmunity, tumor immunity, and transplantation tolerance. Immunol. Rev. 182, 18–32 (2001).
Homann, D. et al. Autoreactive CD4+ T cells protect from autoimmune diabetes via bystander suppression using the IL-4/Stat6 pathway. Immunity 11, 463–472 (1999).
Bluestone, J. A. & Abbas, A. K. Opinion-regulatory lymphocytes: natural versus adaptive regulatory T cells. Nature Rev. Immunol. 3, 253–257 (2003). Discusses the concept of natural versus adaptive regulatory T cells.
Barrat, F. J. et al. In vitro generation of interleukin 10-producing regulatory CD4+ T cells is induced by immunosuppressive drugs and inhibited by T helper type 1 (Th1)- and Th2-inducing cytokines. J. Exp. Med. 195, 603–616 (2002).
Groux, H. et al. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature 389, 737–742 (1997). References 120 and 121 provide evidence that T Reg cells can be induced and that they can be functionally suppressive in vivo.
Woo, E. Y. et al. Regulatory CD4+CD25+ T cells in tumors from patients with early-stage non-small cell lung cancer and late-stage ovarian cancer. Cancer Res. 61, 4766–4772 (2001).
Woo, E. Y. et al. Regulatory T cells from lung cancer patients directly inhibit autologous T cell proliferation. J. Immunol. 168, 4272–4276 (2002).
Liyanage, U. K. et al. Prevalence of regulatory T cells is increased in peripheral blood and tumor microenvironment of patients with pancreas or breast adenocarcinoma. J. Immunol. 169, 2756–2761 (2002).
Onizuka, S. et al. Tumor rejection by in vivo administration of anti-CD25 (interleukin-2 receptor α) monoclonal antibody. Cancer Res. 59, 3128–3133 (1999).
Shimizu, J., Yamazaki, S. & Sakaguchi, S. Induction of tumor immunity by removing CD25+CD4+ T cells: a common basis between tumor immunity and autoimmunity. J. Immunol. 163, 5211–5218 (1999). References 125 and 126 were the first to report that depletion of CD25-expressing cells promotes tumour regression.
van Elsas, A. et al. Elucidating the autoimmune and antitumor effector mechanisms of a treatment based on cytotoxic T lymphocyte antigen-4 blockade in combination with a B16 melanoma vaccine: comparison of prophylaxis and therapy. J. Exp. Med. 194, 481–489 (2001).
Steitz, J., Bruck, J., Lenz, J., Knop, J. & Tuting, T. Depletion of CD25+ CD4+ T cells and treatment with tyrosinase-related protein 2-transduced dendritic cells enhance the interferon α-induced, CD8+ T-cell-dependent immune defense of B16 melanoma. Cancer Res. 61, 8643–8646 (2001).
Takahashi, T. et al. Immunologic self-tolerance maintained by CD25+CD4+ regulatory T cells constitutively expressing cytotoxic T lymphocyte-associated antigen 4. J. Exp. Med. 192, 303–310 (2000).
Read, S., Malmstrom, V. & Powrie, F. Cytotoxic T lymphocyte-associated antigen 4 plays an essential role in the function of CD25+CD4+ regulatory cells that control intestinal inflammation. J. Exp. Med. 192, 295–302 (2000).
Sutmuller, R. P. et al. Synergism of cytotoxic T lymphocyte-associated antigen 4 blockade and depletion of CD25+ regulatory T cells in antitumor therapy reveals alternative pathways for suppression of autoreactive cytotoxic T lymphocyte responses. J. Exp. Med. 194, 823–832 (2001).
Turk, M. J., Guevara-Patino, J. A., Rizzuto, G. A., Engelhorn, M. E. & Houghton, A. N. Concomitant tumor immunity to a poorly immunogenic melanoma is prevented by regulatory T cells. J. Exp. Med. 200, 771–782 (2004).
Mendez, S., Reckling, S. K., Piccirillo, C. A., Sacks, D. & Belkaid, Y. Role for CD4+ CD25+ regulatory T cells in reactivation of persistent leishmaniasis and control of concomitant immunity. J. Exp. Med. 200, 201–210 (2004).
Coussens, L. M. & Werb, Z. Inflammation and cancer. Nature 420, 860–867 (2002).
Kuper, H., Adami, H. O. & Trichopoulos, D. Infections as a major preventable cause of human cancer. J. Intern. Med. 248, 171–183 (2000).
Leach, D. R., Krummel, M. F. & Allison, J. P. Enhancement of antitumor immunity by CTLA-4 blockade. Science 271, 1734–1736 (1996). The first published study to show that CTLA4 blockade enhances tumour immunity. Human clinical trials from this group and others further prove this concept.
Hurwitz, A. A., Yu, T. F., Leach, D. R. & Allison, J. P. CTLA-4 blockade synergizes with tumor-derived granulocyte-macrophage colony-stimulating factor for treatment of an experimental mammary carcinoma. Proc. Natl Acad. Sci. USA 95, 10067–10071 (1998).
Hodi, F. S. et al. Biologic activity of cytotoxic T lymphocyte-associated antigen 4 antibody blockade in previously vaccinated metastatic melanoma and ovarian carcinoma patients. Proc. Natl Acad. Sci. USA 100, 4712–4717 (2003).
Phan, G. Q. et al. Cancer regression and autoimmunity induced by cytotoxic T lymphocyte-associated antigen 4 blockade in patients with metastatic melanoma. Proc. Natl Acad. Sci. USA 100, 8372–8377 (2003).
Foss, F. M. DAB(389)IL-2 (ONTAK): a novel fusion toxin therapy for lymphoma. Clin. Lymphoma 1, 110–106 (2000).
Wang, T. et al. Regulation of the innate and adaptive immune responses by Stat-3 signaling in tumor cells. Nature Med. 10, 48–54 (2004).
Shen, L., Evel-Kabler, K., Strube, R. & Chen, S. Y. Silencing of SOCS1 enhances antigen presentation by dendritic cells and antigen-specific anti-tumor immunity. Nature Biotechnol. 22, 1546–1553 (2004).
Kwon, E. D. et al. Elimination of residual metastatic prostate cancer after surgery and adjunctive cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) blockade immunotherapy. Proc. Natl Acad. Sci. USA 96, 15074–15079 (1999).
Acknowledgements
I would thank my father, Y. Zou for his precious support, M. von Herrath, V. Cerundolo and C.J.M. Melief for critical reading the manuscript, and the collaborators for their intellectual input and hard work. The review focuses on human tumour immunopathogenesis; owing to the plethora of literature related to this topic, a complete and extensive review is extremely challenging. I apologize in advance for any inadvertent omission. The work described in this review was supported by grants from the United States National Institutes of Health and the Department of Defense.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Related links
Related links
DATABASES
Entrez Gene
National Cancer Institute
FURTHER INFORMATION
Glossary
- TOLERANCE
-
A state in which the immune system does not mount effective immune responses against specific antigens.
- ANTIGEN-PRESENTING CELLS
-
Cells that uptake, process and present antigen to other immune cells to initiate and activate immune responses. Monocytes, macrophages, dendritic cells and B cells are antigen-presenting cells. Dendritic cells are the most potent of these.
- EFFECTOR T CELLS
-
T cells that exert a cytolytic function following engagement of their T-cell antigen receptor on target cells. CTLs express the co-receptor CD8 and recognize antigenic peptides that are presented by human leukocyte antigen class I molecules.
- NATURAL KILLER CELLS
-
Natural killer cells are a type of cytotoxic lymphocyte that can be distinguished from CD8+ T cells by their lack of rearrangement of T-cell receptor genes. They have abundant granule-containing cytoplasm, induce target-cell death through direct contact or by cytokine production, and confer innate immunity.
- MYELOID DENDRITIC CELLS
-
A subset of dendritic cells that are lineage-negative CD11c+HLA-DR+ mononuclear cells with a monocytoid appearance. Human myeloid dendritic cells might differentiate from myeloid precursors (for example, monocytes, macrophages and CD11c+ precursors).
- PLASMACYTOID DENDRITIC CELLS
-
A subset of dendritic cells that are lineage negative HLA-DR+CD11c− mononuclear cells with a microscopic appearance similar to plasmablasts. Plasmacytoid dendritic cells are the main producers of type I IFN.
- B7-H1
-
A recently defined B7 family member that is found to be expressed in human epithelial tumours, and can be induced in antigen-presenting cells and in non-lymphoid organs. PD-1 is the identified receptor. Experimental evidence indicates the existence of an unidentified receptor for B7-H1. B7-H1 can mediate an inhibitory role as well as stimulatory role in T-cell-mediated immune responses.
- INDOLEAMINE 2,3-DIOXYGENASE
-
An intracellular haeme-containing enzyme that catalyses oxidative catabolism of tryptophan.
- T HELPER 1 CELLS AND T HELPER 2 CELLS
-
Two functionally defined CD4+ T-cell subsets. T helper 1 (TH1) cells predominantly produce interferon-γ, and support cellular immunity. TH2 cells predominantly produce interleukin-4, and support humoral immunity.
- TUMOUR ASCITES
-
An accumulated fluid in the peritoneum due to cancer. In some cancers, the ascites fluid contains viable tumour cells, immune cells and soluble factors. Tumour ascites is an accessible tumour environment for research.
- REGULATORY T CELLS
-
A T-cell population that can functionally suppress an immune response by influencing the activity of another cell type. There might exist several phenotypically distinct regulatory T cells. The classic ones are CD4+CD25+FOXP3+ T cells.
- NOD/SCID MICE
-
Mice that do not have T cells or B cells. Tumour cells can be grown in these mice without rejection.
- B7-H4
-
A newly defined B7 family member that is found to be expressed in human ovarian epithelial tumours, non-lymphoid organs and antigen-presenting cells within the tumour microenvironment. The receptor remains to be identified. The B7-H4 fusion protein can mediate a profound inhibitory role in T-cell-mediated immune responses.
- ANERGY
-
A state in which lymphocytes can not respond to antigen-specific and non-specific stimulation.
- CYTOTOXIC T-LYMPHOCYTE-ASSOCIATED PROTEIN 4
-
Following engagement by B7.1 or B7.2 on antigen-presenting cells, cytotoxic T-lymphocyte-associated protein 4 (CTLA4) signalling in activated T cells induces cell-cycle arrest, and reduces cytokine production, and diminishes T-cell responses. CD4+CD25+ regulatory T cells constitutively express CTLA4.
- ALLOGENEIC
-
Allogeneic tissues or cells are genetically different from the host and can elicit an immune response when transplanted into immune-competent hosts.
- DANGER SIGNALS
-
Triggers that are associated with host-cell damage. These 'danger signals' induce the activation of antigen-presenting cells. The danger signals could be intracellular components that are released when cells are damaged, such as DNA and proteins, or inflammatory molecules and cytokines, such as interferon-γ.
- CpG OLIGONUCLEOTIDES
-
Synthetic oligodeoxynucleotides (CpG-ODNs) that contain CpG motifs that are similar to those found in bacterial DNA that can stimulate an immune response. These CpG-ODNs have various potential therapeutic uses.
Rights and permissions
About this article
Cite this article
Zou, W. Immunosuppressive networks in the tumour environment and their therapeutic relevance. Nat Rev Cancer 5, 263–274 (2005). https://doi.org/10.1038/nrc1586
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc1586
This article is cited by
-
A novel PDPN antagonist peptide CY12-RP2 inhibits melanoma growth via Wnt/β-catenin and modulates the immune cells
Journal of Experimental & Clinical Cancer Research (2024)
-
An engineered influenza virus to deliver antigens for lung cancer vaccination
Nature Biotechnology (2024)
-
Promising dawn in tumor microenvironment therapy: engineering oral bacteria
International Journal of Oral Science (2024)
-
Discovery of novel indoleamine 2,3-dioxygenase-1 (IDO-1) inhibitors: pharmacophore-based 3D-QSAR, Gaussian field-based 3D-QSAR, docking, and binding free energy studies
Structural Chemistry (2024)
-
HCMV IE1/IE1mut Therapeutic Vaccine Induces Tumor Regression via Intratumoral Tertiary Lymphoid Structure Formation and Peripheral Immunity Activation in Glioblastoma Multiforme
Molecular Neurobiology (2024)