Key Points
-
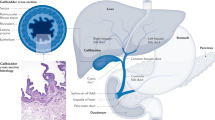
Gallbladder cancer is a relatively rare neoplasm worldwide, but shows significant geographic variation in incidence, being particularly common in certain native American populations. The prognosis for patients with this neoplasm is poor, as diagnosis is often at late, untreatable stages of the disease.
-
A unique combination of predisposing factors makes gallbladder carcinoma a unique tumour and offers potential for understanding cancer pathogenesis. These factors include ethnicity, genetic predisposition, geographic location, female gender, chronic inflammation and congenital developmental abnormalities.
-
Two main pathways of gallbladder carcinoma pathogenesis have been identified. The most common is associated with gallstones and chronic inflammation of the gallbladder, whereas a second, less frequent pathway is associated with a congenital abnormality of the pancreatic bile-duct junction, which is particularly common in Japan.
-
A multistage sequence of histopathological and molecular changes has been identified for gallbladder carcinoma, which is especially well-defined for tumorigenesis associated with gallstones. Molecular abnormalities commence in normal-appearing epithelium in chronically inflamed gallbladders.
-
TP53 inactivation has an important and early role in gallbladder carcinoma associated with gallstones and chronic inflammation. Different patterns of TP53 mutation have been detected in the two main types of gallbladder carcinomas that have been identified.
-
Although KRAS mutations are rarely detected in gallbladder carcinomas associated with gallstones, they are frequent and early events in tumours associated with congenital abnormality of the pancreatic bile-duct junction.
-
Relatively little is known about gallbladder cancer, and a significant influx of research funding is required for this to be remedied. In particular, the identification of susceptibility genes, elucidation of the role of inflammation and an increased understanding of the molecular changes that occur during multistage pathogenesis should be important goals for the future.
Abstract
Gallbladder cancer is a relatively rare form of malignancy about which our knowledge is scant. However, a unique combination of predisposing factors — including genetic predisposition, geographic distribution, female gender bias, chronic inflammation and congenital developmental abnormalities — makes this type of cancer unique and offers potential for understanding cancer pathogenesis in general. An understanding of how these risk factors contribute to the molecular basis of the disease is essential for understanding the origins of this unusual cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Misra, S., Chaturvedi, A., Misra, N. C. & Sharma, I. D. Carcinoma of the gallbladder. Lancet Oncol. 4, 167–176 (2003). Comprehensive review of clinical presentation, treatment and prognosis of gallbladder carcinoma.
Carriaga, M. T. & Henson, D. E. Liver, gallbladder, extrahepatic bile ducts, and pancreas. Cancer 75, 171–190 (1995).
Roa, I., Araya, J. C., de Aretxabala, X., Salinas, C. & Wistuba, I. Gallbladder pathology in Temuco, IX Region. Rev. Med. Chil. 117, 889–894 (1989).
Serra, I. in Epidemiology of Gallbladder and Bile Duct Cancers (eds Yamamoto, H., Serra, I., Endoh, K. & Ogoshi, K.) 95–97 (Nishimura and Smith-Gordon, London, 1999).
de Aretxabala, X. et al. Gallbladder cancer in Chile. A report on 54 potentially resectable tumors. Cancer 69, 60–65 (1992).
de Aretxabala, X., Roa, I. & Burgos, L. Gallbladder cancer, management of early tumors. Hepatogastroenterology 46, 1547–1551 (1999).
Taner, C. B., Nagorney, D. M. & Donohue, J. H. Surgical treatment of gallbladder cancer. J. Gastrointest. Surg. 8, 83–89 (2004). Supports radical surgical resection for the treatment of gallbladder cancer to improve patient survival.
Kiguchi, K. et al. Constitutive expression of ErbB-2 in gallbladder epithelium results in development of adenocarcinoma. Cancer Res. 61, 6971–6976 (2001).
Ku, J. L. et al. Establishment and characterisation of six human biliary tract cancer cell lines. Br. J. Cancer 87, 187–193 (2002).
Ghosh, M. et al. Establishment and characterization of unique human gallbladder cancer cell lines. Int. J. Oncol. 24, 1189–1196 (2004).
Cancer Incidence in Five Continents: Vol. VII. Parkin, D. M., Whelan, S. L., Ferlay, J., Raymond, L. & Young, J. (IARC Scientific, Lyon, 1997).
Lazcano-Ponce, E. C. et al. Epidemiology and molecular pathology of gallbladder cancer. CA Cancer J. Clin. 51, 349–364 (2001). Detailed epidemiological analysis of the worldwide geographical distribution of GBC and gallstones. Some comments on perspectives for prevention are also provided.
O'Brien, K. et al. Cancer statistics for Hispanics, 2003. CA Cancer J. Clin. 53, 208–226 (2003).
Black, W. C., Key, C. R., Carmany, T. B. & Herman, D. Carcinoma of the gallbladder in a population of Southwestern American Indians. Cancer 39, 1267–1279 (1977).
Wagener, D. K. & McDonald, M. Increased gallbladder-related mortality among Hispanics: does education play a role? Ethn. Health 1, 197–205 (1996).
Boss, L. P., Lanier, A. P., Dohan, P. H. & Bender, T. R. Cancers of the gallbladder and biliary tract in Alaskan natives: 1970—79. J. Natl Cancer Inst. 69, 1005–1007 (1982).
Dhir, V. & Mohandas, K. M. Epidemiology of digestive tract cancers in India IV. Gall bladder and pancreas. Indian J. Gastroenterol. 18, 24–28 (1999).
Ferrecio, C., Chianale, J., Gonzalez, C. & Nervi, F. Epidemiologia descriptiva del cancer digestivo en Chile (1982–1991): Una aproximacion desde la Mortalidad (Alfa Beta, Santiago, Chile, 1995).
Miquel, J. F. et al. Genetic epidemiology of cholesterol cholelithiasis among Chilean Hispanics, Amerindians, and Maoris. Gastroenterology 115, 937–946 (1998).
Strom, B. L. et al. Risk factors for gallbladder cancer. An international collaborative case-control study. Cancer 76, 1747–1756 (1995).
Jemal, A. et al. Cancer statistics, 2004. CA Cancer J. Clin. 54, 8–29 (2004).
Balkwill, F. & Mantovani, A. Inflammation and cancer: back to Virchow? Lancet 357, 539–545 (2001).
Coussens, L. M. & Werb, Z. Inflammation and cancer. Nature 420, 860–867 (2002).
Zatonski, W. A. et al. Epidemiologic aspects of gallbladder cancer: a case-control study of the SEARCH Program of the International Agency for Research on Cancer. J. Natl Cancer Inst. 89, 1132–1138 (1997).
Lowenfels, A. B., Maisonneuve, P., Boyle, P. & Zatonski, W. A. Epidemiology of gallbladder cancer. Hepatogastroenterology 46, 1529–1532 (1999).
Nervi, F. et al. Frequency of gallbladder cancer in Chile, a high-risk area. Int. J. Cancer 41, 657–660 (1988).
de Aretxabala, X. et al. Gallbladder cancer. Case-control study. Rev. Med. Chil. 123, 581–586 (1995).
Roa, I. et al. Gallbladder cancer in a high risk area: morphological features and spread patterns. Hepatogastroenterology 46, 1540–1546 (1999).
Diehl, A. K. Gallstone size and the risk of gallbladder cancer. JAMA 250, 2323–2326 (1983).
Zanlungo, S. & Nervi, F. The molecular and metabolic basis of biliary cholesterol secretion and gallstone disease. Front Biosci. 8, S1166–S1174 (2003).
Apstein, M. D. & Carey, M. C. Pathogenesis of cholesterol gallstones: a parsimonious hypothesis. Eur. J. Clin. Invest. 26, 343–352 (1996).
Tazuma, S. & Kajiyama, G. Carcinogenesis of malignant lesions of the gall bladder. The impact of chronic inflammation and gallstones. Langenbecks Arch. Surg. 386, 224–229 (2001).
Maclure, K. M., Hayes, K. C., Colditz, G. A., Stampfer, M. J. & Willett, W. C. Dietary predictors of symptom-associated gallstones in middle-aged women. Am. J. Clin. Nutr. 52, 916–922 (1990).
Everson, G. T., McKinley, C., Lawson, M., Johnson, M. & Kern, F. Jr. Gallbladder function in the human female: effect of the ovulatory cycle, pregnancy, and contraceptive steroids. Gastroenterology 82, 711–719 (1982).
Scragg, R. K., McMichael, A. J. & Seamark, R. F. Oral contraceptives, pregnancy, and endogenous oestrogen in gall stone disease — a case-control study. Br. Med. J. (Clin. Res. Ed.) 288, 1795–1799 (1984).
Lambe, M. et al. Parity and cancers of the gall bladder and the extrahepatic bile ducts. Int. J. Cancer 54, 941–944 (1993).
Bertomeu, A. et al. Apolipoprotein E polymorphism and gallstones. Gastroenterology 111, 1603–1610 (1996).
Fernandez, E., La Vecchia, C., D'Avanzo, B., Negri, E. & Franceschi, S. Family history and the risk of liver, gallbladder, and pancreatic cancer. Cancer Epidemiol. Biomarkers Prev. 3, 209–212 (1994).
Hemminki, K. & Li, X. Familial liver and gall bladder cancer: a nationwide epidemiological study from Sweden. Gut 52, 592–596 (2003).
Stephen, A. E. & Berger, D. L. Carcinoma in the porcelain gallbladder: a relationship revisited. Surgery 129, 699–703 (2001).
Dutta, U., Garg, P. K., Kumar, R. & Tandon, R. K. Typhoid carriers among patients with gallstones are at increased risk for carcinoma of the gallbladder. Am. J. Gastroenterol. 95, 784–787 (2000).
Nath, G., Singh, H. & Shukla, V. K. Chronic typhoid carriage and carcinoma of the gallbladder. Eur. J. Cancer Prev. 6, 557–559 (1997).
Roa, I. et al. Microbiological study of gallbladder bile in a high risk zone for gallbladder cancer. Rev. Med. Chil. 127, 1049–1055 (1999).
Leong, R. W. & Sung, J. J. Helicobacter species and hepatobiliary diseases. Aliment. Pharmacol. Ther. 16, 1037–1045 (2002).
Chijiiwa, K., Kimura, H. & Tanaka, M. Malignant potential of the gallbladder in patients with anomalous pancreaticobiliary ductal junction. The difference in risk between patients with and without choledochal cyst. Int. Surg. 80, 61–64 (1995).
Hasumi, A. et al. Precancerous conditions of biliary tract cancer in patients with pancreaticobiliary maljunction: reappraisal of nationwide survey in Japan. J. Hepatobiliary Pancreat. Surg. 7, 551–555 (2000).
Hu, B., Gong, B. & Zhou, D. Y. Association of anomalous pancreaticobiliary ductal junction with gallbladder carcinoma in Chinese patients: an ERCP study. Gastrointest. Endosc. 57, 541–545 (2003).
Matsumoto, Y. et al. Recent advances in pancreaticobiliary maljunction. J. Hepatobiliary Pancreat. Surg. 9, 45–54 (2002).
Kimura, K. et al. Association of gallbladder carcinoma and anomalous pancreaticobiliary ductal union. Gastroenterology 89, 1258–1265 (1985).
Kowalewski, K. & Todd, E. F. Carcinoma of the gallbladder induced in hamsters by insertion of cholesterol pellets and feeding dimethylnitrosamine. Proc. Soc. Exp. Biol. Med. 136, 482–486 (1971).
Enomoto, M. et al. Carcinogenesis in extrahepatic bile duct and gallbladder —carcinogenic effect of N-hydroxy-2-acetamidofluorene in mice fed a 'gallstone-inducing' diet. Jpn J. Exp. Med. 44, 37–54 (1974).
Darby, S. C. et al. Radon and cancers other than lung cancer in underground miners: a collaborative analysis of 11 studies. J. Natl Cancer Inst. 87, 378–384 (1995).
Serra, I. et al. Association of chili pepper consumption, low socioeconomic status and longstanding gallstones with gallbladder cancer in a Chilean population. Int. J. Cancer 102, 407–411 (2002).
Sasatomi, E., Tokunaga, O. & Miyazaki, K. Precancerous conditions of gallbladder carcinoma: overview of histopathologic characteristics and molecular genetic findings. J. Hepatobiliary Pancreat. Surg. 7, 556–567 (2000). Premalignant epithelial lesions associated with both main types of GBC are described, including histopathology and molecular genetic findings.
Hanada, K. et al. Gene mutations of K-ras in gallbladder mucosae and gallbladder carcinoma with an anomalous junction of the pancreaticobiliary duct. Am. J. Gastroenterol. 94, 1638–1642 (1999).
Masuhara, S. et al. Relation between K-ras codon 12 mutation and p53 protein overexpression in gallbladder cancer and biliary ductal epithelia in patients with pancreaticobiliary maljunction. J. Hepatobiliary Pancreat. Surg. 7, 198–205 (2000).
Wistuba, I. I. et al. Allele-specific mutations involved in the pathogenesis of endemic gallbladder carcinoma in Chile. Cancer Res. 55, 2511–2515 (1995).
Wistuba, I. I., Gazdar, A. F., Roa, I. & Albores-Saavedra, J. p53 protein overexpression in gallbladder carcinoma and its precursor lesions: an immunohistochemical study. Hum. Pathol. 27, 360–365 (1996).
Albores-Saavedra, J. & Henson, D. E. Tumors of gallbladder and extrahepatic bile ducts. Fascicle 23, 3rd ed. (Armed Forces Institute of Pathology, Washington, DC, 2000).
Vogelstein, B. et al. Genetic alterations during colorectal-tumor development. N. Engl. J. Med. 319, 525–532 (1988).
Wistuba, I. I., Miquel, J. F., Gazdar, A. F. & Albores-Saavedra, J. Gallbladder adenomas have molecular abnormalities different from those present in gallbladder carcinomas. Hum. Pathol. 30, 21–25 (1999).
Roa, I. et al. Preneoplastic lesions and gallbladder cancer: an estimate of the period required for progression. Gastroenterology 111, 232–236 (1996). Shows that the period required to progress from dysplasia to advanced GBC is ∼15 years.
Hanada, K. et al. Pathology and cellular kinetics of gallbladder with an anomalous junction of the pancreaticobiliary duct. Am. J. Gastroenterol. 91, 1007–1011 (1996).
Tanno, S. et al. Epithelial hyperplasia of the gallbladder in children with anomalous pancreaticobiliary ductal union. Hepatogastroenterology 46, 3068–3073 (1999).
Dawes, L. G. Gallbladder cancer. Cancer Treat. Res. 109, 145–155 (2001).
Levin, B. Gallbladder carcinoma. Ann. Oncol. 10 (Suppl. 4), 129–130 (1999).
Wistuba, I. I. & Albores-Saavedra, J. Genetic abnormalities involved in the pathogenesis of gallbladder carcinoma. J. Hepatobiliary Pancreat. Surg. 6, 237–244 (1999).
Almoguera, C. et al. Most human carcinomas of the exocrine pancreas contain mutant c-K-ras genes. Cell 53, 549–554 (1988).
Tada, M., Yokosuka, O., Omata, M., Ohto, M. & Isono, K. Analysis of ras gene mutations in biliary and pancreatic tumors by polymerase chain reaction and direct sequencing. Cancer 66, 930–935 (1990).
Yoshida, S. et al. Mutations of p16Ink4/CDKN2 and p15Ink4B/MTS2 genes in biliary tract cancers. Cancer Res. 55, 2756–2760 (1995).
Ajiki, T. et al. K-ras gene mutation in gall bladder carcinomas and dysplasia. Gut 38, 426–429 (1996).
Hanada, K. et al. K-ras and p53 mutations in stage I gallbladder carcinoma with an anomalous junction of the pancreaticobiliary duct. Cancer 77, 452–458 (1996).
Matsubara, T. et al. K-ras point mutations in cancerous and noncancerous biliary epithelium in patients with pancreaticobiliary maljunction. Cancer 77, 1752–1757 (1996). KRAS mutations are reported as early and frequent events in the pathogenesis of GBC associated with APBDJ.
Iwase, T. et al. Ras gene point mutations in gallbladder lesions associated with anomalous connection of pancreatobiliary ducts. Hepatogastroenterology 44, 1457–1462 (1997).
Cowgill, S. M. & Muscarella, P. The genetics of pancreatic cancer. Am. J. Surg. 186, 279–286 (2003).
Suzuki, T., Takano, Y., Kakita, A. & Okudaira, M. An immunohistochemical and molecular biological study of c-erbB-2 amplification and prognostic relevance in gallbladder cancer. Pathol. Res. Pract. 189, 283–292 (1993).
Chow, N. H. et al. Significance of c-erbB-2 expression in normal and neoplastic epithelium of biliary tract. Anticancer Res. 15, 1055–1059 (1995).
Kim, Y. W. et al. Expression of the c-erb-B2 and p53 protein in gallbladder carcinomas. Oncol. Rep. 8, 1127–1132 (2001).
Hui, A. M. et al. Cyclin D1 overexpression is a critical event in gallbladder carcinogenesis and independently predicts decreased survival for patients with gallbladder carcinoma. Clin. Cancer Res. 6, 4272–4277 (2000).
Eguchi, N. et al. Cyclin E overexpression in human gallbladder carcinomas. Oncol. Rep. 6, 93–96 (1999).
Pfeifer, G. P. p53 mutational spectra and the role of methylated CpG sequences. Mutat. Res. 450, 155–166 (2000).
Fujii, K. et al. High frequency of p53 gene mutation in adenocarcinomas of the gallbladder. Cancer Epidemiol. Biomarkers Prev. 5, 461–466 (1996).
Hanada, K. et al. TP53 mutations in stage I gallbladder carcinoma with special attention to growth patterns. Eur. J. Cancer 33, 1136–1140 (1997).
Itoi, T. et al. Correlation of p53 protein expression with gene mutation in gall-bladder carcinomas. Pathol. Int. 47, 525–530 (1997).
Yokoyama, N. et al. Mutations of p53 in gallbladder carcinomas in high-incidence areas of Japan and Chile. Cancer Epidemiol. Biomarkers Prev. 7, 297–301 (1998).
Olivier, M. et al. The IARC TP53 Database: new online mutation analysis and recomendations to users. Hum. Mutat. 19, 607–614 (2002).
Roa, I. et al. p53 tumour suppressor gene protein expression in early and advanced gallbladder carcinoma. Histopathology 31, 226–230 (1997).
Kim, Y. T. et al. Genetic alterations in gallbladder adenoma, dysplasia and carcinoma. Cancer Lett. 169, 59–68 (2001).
Parwani, A. V. et al. Immunohistochemical and genetic analysis of non-small cell and small cell gallbladder carcinoma and their precursor lesions. Mod. Pathol. 16, 299–308 (2003).
House, M. G. et al. Progression of gene hypermethylation in gallstone disease leading to gallbladder cancer. Ann. Surg. Oncol. 10, 882–889 (2003).
Ueki, T. et al. Alterations of p16 and prognosis in biliary tract cancers from a population-based study in China. Clin. Cancer Res. 10, 1717–1725 (2004).
Shi, Y. Z., Hui, A. M., Li, X., Takayama, T. & Makuuchi, M. Overexpression of retinoblastoma protein predicts decreased survival and correlates with loss of p16INK4 protein in gallbladder carcinomas. Clin. Cancer Res. 6, 4096–4100 (2000).
Li, X., Hui, A. M., Shi, Y. Z., Takayama, T. & Makuuchi, M. Reduced p21(WAF1/CIP1) expression is an early event in gallbladder carcinogenesis and is of prognostic significance for patients with carcinomas of the gallbladder. Hum. Pathol. 32, 771–777 (2001).
Wistuba, I. I. et al. Genome-wide allelotyping analysis reveals multiple sites of allelic loss in gallbladder carcinoma. Cancer Res. 61, 3795–3800 (2001).
Nakayama, K. et al. Allelotype analysis of gallbladder carcinoma associated with anomalous junction of pancreaticobiliary duct. Cancer Lett. 166, 135–141 (2001).
Yanagisawa, N., Mikami, T., Saegusa, M. & Okayasu, I. More frequent β-catenin exon 3 mutations in gallbladder adenomas than in carcinomas indicate different lineages. Cancer Res. 61, 19–22 (2001).
Yoshida, T. et al. Microsatellite instability in gallbladder carcinoma: two independent genetic pathways of gallbladder carcinogenesis. J. Gastroenterol. 35, 768–774 (2000).
Matsuo, K., Kuroki, T., Kitaoka, F., Tajima, Y. & Kanematsu, T. Loss of heterozygosity of chromosome 16q in gallbladder carcinoma. J. Surg. Res. 102, 133–136 (2002).
Wistuba, I. I. et al. High resolution chromosome 3p, 8p, 9q and 22q allelotyping analysis in the pathogenesis of gallbladder carcinoma. Br. J. Cancer 87, 432–440 (2002). LOH study showing that genetic abnormalities affecting different chromosomal sites are detected as early events (in normal appearing epithelium and dysplasia) in the multistage pathogenesis of GBC.
Zabarovsky, E. R., Lerman, M. I. & Minna, J. D. Tumor suppressor genes on chromosome 3p involved in the pathogenesis of lung and other cancers. Oncogene 21, 6915–6935 (2002).
Wistuba, I. I. et al. Fragile histidine triad gene abnormalities in the pathogenesis of gallbladder carcinoma. Am. J. Pathol. 160, 2073–2079 (2002).
Pekarsky, Y., Zanesi, N., Palamarchuk, A., Huebner, K. & Croce, C. M. FHIT: from gene discovery to cancer treatment and prevention. Lancet Oncol. 3, 748–754 (2002).
Rashid, A. et al. β-catenin mutations in biliary tract cancers: a population-based study in China. Cancer Res. 61, 3406–3409 (2001).
Chang, H. J., Jee, C. D. & Kim, W. H. Mutation and altered expression of β-catenin during gallbladder carcinogenesis. Am. J. Surg. Pathol. 26, 758–766 (2002).
Rashid, A. et al. K-ras mutation, p53 overexpression, and microsatellite instability in biliary tract cancers: a population-based study in China. Clin. Cancer Res. 8, 3156–3163 (2002).
Yanagisawa, N., Mikami, T., Yamashita, K. & Okayasu, I. Microsatellite instability in chronic cholecystitis is indicative of an early stage in gallbladder carcinogenesis. Am. J. Clin. Pathol. 120, 413–417 (2003).
Campomenosi, P. et al. p53 is frequently mutated in Barrett's metaplasia of the intestinal type. Cancer Epidemiol. Biomarkers Prev. 5, 559–565 (1996).
Hussain, S. P. et al. Increased p53 mutation load in noncancerous colon tissue from ulcerative colitis: a cancer-prone chronic inflammatory disease. Cancer Res. 60, 3333–3337 (2000).
Parsons, T. J. et al. A high observed substitution rate in the human mitochondrial DNA control region. Nature Genet. 15, 363–368 (1997).
Sanchez-Cespedes, M. et al. Identification of a mononucleotide repeat as a major target for mitochondrial DNA alterations in human tumors. Cancer Res. 61, 7015–7019 (2001).
Mambo, E. et al. Electrophile and oxidant damage of mitochondrial DNA leading to rapid evolution of homoplasmic mutations. Proc. Natl Acad. Sci. USA 100, 1838–1843 (2003).
Tang, M. et al. Mitochondrial DNA mutation at the D310 (displacement loop) mononucleotide sequence in the pathogenesis of gallbladder carcinoma. Clin. Cancer Res. 10, 1041–1046 (2004). Mitochondrial DNA mutations affecting the D310 sequence are relatively frequent events in GBC and to occur start at the chronic cholecystitis stage.
Hwang, D., Scollard, D., Byrne, J. & Levine, E. Expression of cyclooxygenase-1 and cyclooxygenase-2 in human breast cancer. J. Natl Cancer Inst. 90, 455–460 (1998).
Hayashi, N. et al. Differential expression of cyclooxygenase-2 (COX-2) in human bile duct epithelial cells and bile duct neoplasm. Hepatology 34, 638–650 (2001).
Buskens, C. J., Ristimaki, A., Offerhaus, G. J., Richel, D. J. & van Lanschot, J. J. Role of cyclooxygenase-2 in the development and treatment of oesophageal adenocarcinoma. Scand. J. Gastroenterol. Suppl. 239, 87–93 (2003).
Sinicrope, F. A. & Gill, S. Role of cyclooxygenase-2 in colorectal cancer. Cancer Metastasis Rev. 23, 63–75 (2004).
Grossman, E. M., Longo, W. E., Panesar, N., Mazuski, J. E. & Kaminski, D. L. The role of cyclooxygenase enzymes in the growth of human gall bladder cancer cells. Carcinogenesis 21, 1403–1409 (2000).
Tsujii, M., Kawano, S. & DuBois, R. N. Cyclooxygenase-2 expression in human colon cancer cells increases metastatic potential. Proc. Natl Acad. Sci USA 94, 3336–3340 (1997).
Asano, T. et al. Expressions of cyclooxygenase-2 and prostaglandin E-receptors in carcinoma of the gallbladder: crucial role of arachidonate metabolism in tumor growth and progression. Clin. Cancer Res. 8, 1157–1167 (2002).
Kawamoto, T. et al. Expression of cyclooxygenase-2 in the subserosal layer correlates with postsurgical prognosis of pathological tumor stage 2 carcinoma of the gallbladder. Int. J. Cancer 98, 427–434 (2002).
Fumino, S., Tokiwa, K., Ono, S. & Iwai, N. Cyclooxygenase-2 expression in the gallbladder of patients with anomalous arrangement of the pancreaticobiliary duct. J. Pediatr. Surg. 38, 585–589 (2003).
Longo, W. E., Panesar, N., Mazuski, J. E. & Kaminski, D. Synthetic pathways of gallbladder mucosal prostanoids: the role of cyclooxygenase-1 and 2. Prostaglandins Leukot. Essent. Fatty Acids 60, 77–85 (1999).
Kanoh, K. et al. Significance of contracted cholecystitis lesions as high risk for gallbladder carcinogenesis. Cancer Lett. 169, 7–14 (2001).
Tanno, S. et al. Proliferative potential and K-ras mutation in epithelial hyperplasia of the gallbladder in patients with anomalous pancreaticobiliary ductal union. Cancer 83, 267–275 (1998).
Church, R. D., Fleshman, J. W. & McLeod, H. L. Cyclo-oxygenase 2 inhibition in colorectal cancer therapy. Br. J. Surg. 90, 1055–1067 (2003).
Lane, D. Curing cancer with p53. N. Engl. J. Med. 350, 2711–2712 (2004).
Gallstones and laparoscopic cholecystectomy. NIH Consens. Statement 10, 1–28 (1992).
Crosby, C. G. & DuBois, R. N. The cyclooxygenase-2 pathway as a target for treatment or prevention of cancer. Expert Opin. Emerg. Drugs 8, 1–7 (2003).
Acknowledgements
Work in the authors laboratory is supported by grants from FONDECYT (Fondo Nacional de Desarrollo Cientifico y Tecnologico Chile) and the Early Detection Research Network, National Cancer Institute. We thank I. Roa for his assistance with the histopathology pictures in figure 5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
DATABASES
Cancer.gov
Entrez Gene
FURTHER INFORMATION
Glossary
- CHOLECYSTECTOMY
-
Surgical removal of the gallbladder.
- CHRONIC CHOLECYSTITIS
-
Chronic inflammation of the gallbladder wall that is almost always associated with the presence of gallstones and is thought to result from repeated bouts of acute or subacute cholecystitis.
- MICELLE
-
A globule of lipid molecules in water where the non-polar ends face inward and the polar tails face outward.
- MAPUCHE INDIANS
-
The main American Indian tribe of Chile, which has very high incidences of cholelithiasis and gallbladder cancer. Much of our knowledge of gallbladder cancer is derived from studying this subpopulation.
- ODDS RATIO
-
The odds of developing a particular disease in the exposed group divided by the odds of developing it in an unexposed group. Usually derived from a case–control study.
- LITHOGENIC GENES
-
Genes associated with an increased incidence of gallstone formation and, directly or indirectly, of gallbladder cancer.
- CALCIUM BILIRUBINATE
-
A salt composed of calcium and insoluble bilirrubin that results in the precipitation of pigmented gallstones.
- HYPERLIPIDAEMIA
-
General term for increased concentrations of any or all of the lipids in the plasma, such as cholesterol, triglycerides and lipoproteins.
- GALLBLADDER HYPOMOTILITY
-
A reduction in the contractile activity of the gallbladder required to evacuate bile content into the duodenum.
- STASIS
-
Accumulation of bile content of the gallbladder producing dilatation and an increase in bile viscosity.
- ACUTE CHOLECYSTITIS
-
Acute inflammation of the gallbladder wall that can be evoked by mechanical, chemical or bacterial factors.
- DYSPLASIA
-
An epithelial change that frequently precedes the development of invasive carcinoma. It is characterized by mild to severe cytological and structural abnormalities.
- CARCINOMA IN SITU
-
Pre-invasive epithelial precursor of invasive carcinomas, without disruption of the basement membrane and characterized by severe cytological changes, similar to those detected in the invasive counterpart.
- HYPERCHROMATIC
-
Showing dark staining of the nucleus, indicating increased chromatin content.
- ALLELOTYPING STUDIES
-
Analyses of loss of heterozygosity (LOH) — a hallmark of tumour-suppressor-gene inactivation — that is performed using microsatellite markers.
- MICROSATELLITE MARKERS
-
Polymorphic tandem-repeat sequences distributed throughout the genome that have been used to detect loss of heterozygosity.
Rights and permissions
About this article
Cite this article
Wistuba, I., Gazdar, A. Gallbladder cancer: lessons from a rare tumour. Nat Rev Cancer 4, 695–706 (2004). https://doi.org/10.1038/nrc1429
Issue Date:
DOI: https://doi.org/10.1038/nrc1429
This article is cited by
-
Intratumoural microbiota: from theory to clinical application
Cell Communication and Signaling (2023)
-
Induction chemotherapy and hepatic artery embolization followed by extended resection for locally advanced gallbladder cancer: a case report
Surgical Case Reports (2023)
-
GBCdb: RNA expression landscapes and ncRNA–mRNA interactions in gallbladder carcinoma
BMC Bioinformatics (2023)
-
Updated epidemiology of gastrointestinal cancers in East Asia
Nature Reviews Gastroenterology & Hepatology (2023)
-
Axillary nodal metastasis of operated gallbladder carcinoma: remote site of aggression—a case report
BMC Surgery (2022)