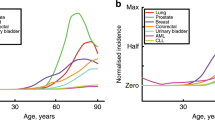
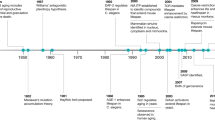
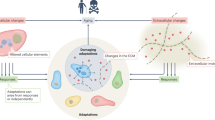
Key Points
-
Cancer is a problem that affects organisms with renewable tissues; these have evolved tumour-suppressor mechanisms to suppress the development of cancer.
-
Tumour-suppressor genes act to prevent or repair genomic damage (caretakers), or inhibit the propagation of potential cancer cells (gatekeepers) by permanently arresting their growth (cellular senescence) or inducing cell death (apoptosis).
-
Some caretaker tumour suppressors seem to postpone the development of ageing phenotypes, and so are also longevity-assurance genes.
-
The gatekeeper tumour-suppressor mechanisms (apoptosis and cellular senescence), by contrast, might promote certain ageing phenotypes.
-
Apoptosis and cellular senescence are controlled by the p53 and RB tumour-suppressor pathways, components of which are evolutionarily conserved among multicellular organisms.
-
The evolutionary hypothesis of antagonistic pleiotropy predicts that some processes that benefit young organisms (by suppressing cancer, for example) can have detrimental effects later in life and would therefore contribute to ageing.
-
Both apoptosis and cellular senescence might be antagonistically pleiotropic, promoting ageing by exhausting progenitor or stem cells. Additionally, senescent cells secrete factors that can disrupt tissue integrity and function, and even promote the progression of late-life cancers.
-
Recent studies on p53 provide a molecular basis for how tumour suppression and ageing might be intertwined.
Abstract
Organisms with renewable tissues use a network of genetic pathways and cellular responses to prevent cancer. The main mammalian tumour-suppressor pathways evolved from ancient mechanisms that, in simple post-mitotic organisms, act predominantly to regulate embryogenesis or to protect the germline. The shift from developmental and/or germline maintenance in simple organisms to somatic maintenance in complex organisms might have evolved at a cost. Recent evidence indicates that some mammalian tumour-suppressor mechanisms contribute to ageing. How might this have happened, and what are its implications for our ability to control cancer and ageing?
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Miller, R. A. Gerontology as oncology: research on aging as a key to the understanding of cancer. Cancer 68, 2496–2501 (1991).
DePinho, R. A. The age of cancer. Nature 408, 248–254 (2000).
Balducci, L. & Beghe, C. Cancer and age in the USA. Crit. Rev. Oncol. Hematol. 37, 137–145 (2001).
Kinzler, K. W. & Vogelstein, B. Cancer susceptibility genes: gatekeepers and caretakers. Nature 386, 761–763 (1997).
Barzilai, N. & Shuldiner, A. R. Searching for human longevity genes: the future history of gerontology in the post-genomic era. J Gerontol A Biol Sci Med Sci 56, M83–M87 (2001).
Bookstein, R. & Lee, W. H. Molecular genetics of the retinoblastoma tumor suppressor gene. Crit Rev Oncog 2, 211–227 (1991).
Hollstein, M., Sidransky, D., Vogelstein, B. & Harris, C. C. p53 mutation in human cancer. Science 253, 49–53 (1991).
Bishop, J. M. Cancer: the rise of the genetic paradigm. Genes Dev. 9, 1309–1315 (1995).
Wu, X. & Pandolfi, P. Mouse models for multistep tumorigenesis. Trends Cell Biol. 11, S2–S9 (2001).
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000).
Compagni, A. & Christofori, G. Recent advances in research on multistage tumorigenesis. Br. J. Cancer. 83, 1–5 (2000).
Macleod, K. Tumor suppressor genes. Curr. Opin. Genet. Dev. 10, 81–93 (2000).
Weinberg, R. A. How cancer arises. Sci. Am. 275, 62–70 (1996).
Ghebranious, N. & Donehower, L. A. Mouse models in tumor suppression. Oncogene 17, 3385–3400 (1998).
Knudson, A. G. Chasing the cancer demon. Annu. Rev. Genet. 34, 1–19 (2000).
Hakem, R. & Mak, T. W. Animal models of tumor-suppressor genes. Annu. Rev. Genet. 35, 209–241 (2001).
Hasty, P., Campisi, J., Hoeijmakers, J., van Steeg, H. & Vijg, J. Aging and genome maintenance: lessons from the mouse? Science 299, 1355–1359 (2003).
Vijg, J. & Dolle, M. E. Large genome rearrangements as a primary cause of aging. Mech. Ageing Dev. 123, 907–915 (2002).
Rose, M. R. The Evolutionary Biology of Aging (Oxford Univ. Press, Oxford, 1991).
Kirkwood, T. B. & Austad, S. N. Why do we age? Nature 408, 233–238 (2000).
Umezu, K., Nakayama, K. & Nakayama, H. Escherichia coli RecQ protein is a DNA helicase. Proc. Natl Acad. Sci. USA 87, 5363–5367 (1990).
Gangloff, S., McDonald, J. P., Bendixen, C., Arthur, L. & Rothstein, R. The yeast type I topoisomerase Top3 interacts with Sgs1, a DNA helicase homolog: a potential eukaryotic reverse gyrase. Mol. Cell. Biol. 14, 8391–8398 (1994).
Stewart, E., Chapman, C. R., Al-Khodairy, F., Carr, A. M. & Enoch, T. Rqh1+, a fission yeast gene related to the Bloom's and Werner's syndrome genes, is required for reversible S phase arrest. EMBO J. 16, 2682–2692 (1997).
Frei, C. & Gasser, S. M. RecQ-like helicases: the DNA replication checkpoint connection. J. Cell Sci. 113, 2641–2646 (2000).
van Brabant, A. J., Stan, R. & Ellis, N. A. DNA helicases, genomic instability, and human genetic disease. Annu Rev Genomics Hum Genet 1, 409–459 (2000).
Chakraverty, R. K. & Hickson, I. D. Defending genome integrity during DNA replication: a proposed role for RecQ family helicases. Bioessays 21, 286–294 (1999).
Goto, M. Hierarchical deterioration of body systems in Werner's syndrome: implications for normal ageing. Mech. Ageing Dev. 98, 239–254 (1997).
Martin, G. M., Oshima, J., Gray, M. D. & Poot, M. What geriatricians should know about the Werner Syndrome. J. Am. Geriatr. Soc. 47, 1136–1144 (1999).
Ellis, N. A. & German, J. Molecular genetics of Bloom's syndrome. Hum. Mol. Genet. 5, 1457–1463 (1996).
Vennos, E. M. & James, W. D. Rothmund-Thomson syndrome. Dermatol. Clin. 13, 143–150 (1995).
Mohaghegh, P. & Hickson, I. D. DNA helicase deficiencies associated with cancer predisposition and premature ageing disorders. Hum. Mol. Genet. 10, 741–746 (2001).
German, J. Bloom's syndrome. Dermatol. Clin. 13, 7–18 (1995).
Oshima, J. The Werner syndrome protein: an update. Bioessays 22, 894–901 (2000).
Hickson, I. D. RecQ helicases: caretakers of the genome. Nature Rev. Cancer 3, 169–178 (2003).
Chen, L. & Oshima, J. Werner Syndrome. J. Biomed. Biotechnol. 2, 46–54 (2002).
Fukuchi, K., Martin, G. M. & Monnat, R. J. Mutator phenotype of Werner syndrome is characterized by extensive deletions. Proc. Natl Acad. Sci. USA 86, 5893–5897 (1989).
Lebel, M. Increased frequency of DNA deletions in pink-eyed unstable mice carrying a mutation in the Werner syndrome gene homologue. Carcinogenesis 23, 213–216 (2002).
Oshima, J., Huang, S., Pae, C., Campisi, J. & Schiestl, R. H. Lack of WRN results in extensive deletion at nonhomologous joining ends. Cancer Res. 62, 547–551 (2002).
Prince, P. R., Emond, M. J. & Monnat, R. J. Loss of Werner syndrome protein function promotes aberrant mitotic recombination. Genes Dev. 15, 933–938 (2001).
Martin, G. M. Somatic mutagenesis and antimutagenesis in aging research. Mutat. Res. 350, 35–41 (1996).
Modrich, P. Mismatch repair, genetic stability, and cancer. Science 266, 1959–1960 (1994).
de Boer, J. & Hoeijmakers, J. Cancer from the outside, aging from the inside: mouse models to study the consequences of defective nucleotide excision repair. Biochimie 81, 127–137 (1999).
Lieber, M. R. Pathological and physiological double-strand breaks: roles in cancer, aging, and the immune system. Am. J. Pathol. 153, 1323–1332 (1998).
Modesti, M. & Kanaar, R. Homologous recombination: from model organisms to human disease. Genome Biol. 2, 1014 (2001).
Eisen, J. A. & Hanawalt, P. C. A phylogenomic study of DNA repair genes, proteins, and processes. Mutat. Res. 435, 171–213 (1999).
Burkle, A. Physiology and pathophysiology of poly(ADP-ribosyl)ation. Bioessays 23, 795–806 (2001).
Friedberg, E. C. How nucleotide excision repair protects against cancer. Nature Rev. Cancer 1, 22–33 (2001).
Pierce, A. et al. Double-strand breaks and tumorigenesis. Trends Cell Biol. 11, S52–S59 (2001).
Vogel, H., Lim, D. S., Karsenty, G., Finegold, M. & Hasty, P. Deletion of Ku86 causes early onset of senescence in mice. Proc. Natl Acad. Sci. USA 96, 10770–10775 (1999). Describes the premature ageing phenotypes of mice that are deficient in a protein required for repairing double-strand breaks in DNA.
Berneburg, M. & Lehmann, A. R. Xeroderma pigmentosum and related disorders: defects in DNA repair and transcription. Adv. Genet. 43, 71–102 (2001).
de Boer, J. et al. Premature aging in mice deficient in DNA repair and transcription. Science 296, 1276–1279 (2002). This paper describes the premature ageing phenotypes of mice deficient in a protein required for reparing damaged nucleotides in DNA.
Thompson, L. H. & Schild, D. Recombinational DNA repair and human disease. Mutat. Res. 509, 49–78 (2002).
Cao, L., Li, W., Kim, S., Brodie, S. G. & Deng, C. X. Senescence, aging, and malignant transformation mediated by p53 in mice lacking the Brca1 full-length isoform. Genes Dev. 17, 201–213 (2003).
Arends, M. J. & Wyllie, A. H. Apoptosis: mechanisms and roles in pathology. Int. Rev. Exp. Pathol. 32, 223–254 (1991).
Ellis, R. E., Yuan, J. Y. & Horvitz, H. R. Mechanisms and functions of cell death. Annu. Rev. Cell Biol. 7, 663–698 (1991).
Vaux, D. L. & Korsmeyer, S. J. Cell death in development. Cell 96, 245–254 (1999).
Sinha Hakim, A. P. & Swerdloff, R. S. Hormonal and genetic control of germ cell apoptosis in the testis. Rev. Reprod. 4, 38–47 (1999).
Laun, P. et al. Aged mother cells of Saccharomyces cerevisiae show markers of oxidative stress and apoptosis. Mol. Microbiol. 39, 1166–1173 (2001).
Reed, J. C. Mechanisms of apoptosis in avoidance of cancer. Curr. Opin. Oncol. 11, 68–75 (1999).
Green, D. R. & Evan, G. I. A matter of life and death. Cancer Cell 1, 19–30 (2002).
Campisi, J. Cellular senescence as a tumor-suppressor mechanism. Trends Cell Biol. 11, S27–S31 (2001).
Kim, S. H., Kaminker, P. & Campisi, J. Telomeres, aging and cancer: in search of a happy ending. Oncogene 21, 503–511 (2002).
Chiu, C. P. & Harley, C. B. Replicative senescence and cell immortality: the role of telomeres and telomerase. Proc. Soc. Exp. Biol. Med. 214, 99–106 (1997).
Campisi, J. Cancer, aging and cellular senescence. In Vivo 14, 183–188 (2000).
Serrano, M. & Blasco, M. A. Putting the stress on senescence. Curr. Opin. Cell Biol. 13, 748–753 (2001).
Campisi, J., Dimri, G. P. & Hara, E. in Handbook of the Biology of Aging (eds Schneider, E. & Rowe, J.) 121–149 (Academic Press, New York, 1996).
Linskens, M. H. K. et al. Cataloging altered gene expression in young and senescent cells using enhanced differential display. Nucleic Acids Res 23, 3244–3251 (1995).
Shelton, D. N., Chang, E., Whittier, P. S., Choi, D. & Funk, W. D. Microarray analysis of replicative senescence. Curr. Biol. 9, 939–945 (1999). This paper uses microarrays to compare the senescent phenotype of replicatively senescent human fibroblasts with human fibroblasts that are induced to 'prematurely' senesce owing to non-telomeric events.
Campisi, J. From cells to organisms: can we learn about aging from cells in culture? Exp. Gerontol. 36, 607–618 (2001).
Margolis, J. & Spradling, A. Identification and behavior of epithelial stem cells in the Drosophila ovary. Development 121, 3797–3807 (1995).
Jazwinski, S. M. Longevity, Genes and Aging. Science 273, 54–59 (1996).
Sager, R. Senescence as a mode of tumor suppression. Environ Health Perspect 93, 59–62 (1991).
Smith, J. R. & Pereira-Smith, O. M. Replicative senescence: implications for in vivo aging and tumor suppression. Science 273, 63–67 (1996).
Wright, W. E. & Shay, J. W. Cellular senescence as a tumor-protection mechanism: the essential role of counting. Curr. Opin. Genet. Dev. 11, 98–103 (2001).
Yeager, T. R. et al. Overcoming cellular senescence in human cancer pathogenesis. Genes Dev. 12, 163–174 (1998).
Serrano, M. et al. Role of the INK4A locus in tumor suppression and cell mortality. Cell 85, 27–37 (1996).
Sharpless, N. E. et al. Loss of p16Ink4a with retention of p19Arf predisposes mice to tumorigenesis. Nature 413, 86–91 (2001).
Harvey, M. et al. In vitro growth characteristics of embryo fibroblasts isolated from p53-deficient mice. Oncogene 8, 2457–2467 (1993).
Donehower, L. A. et al. Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumors. Nature 356, 215–221 (1992).
Hara, E. et al. Regulation of p16/CDKN2 expression and its implications for cell immortalization and senescence. Mol. Cell. Biol. 16, 859–867 (1996).
McConnell, B. B., Starborg, M., Brookes, S. & Peters, G. Inhibitors of cyclin-dependent kinases induce features of replicative senescence in early passage human diploid fibroblasts. Curr. Biol. 8, 351–354 (1998).
Serrano, M., Lin, A. W., McCurrach, M. E., Beach, D. & Lowe, S. W. Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 88, 593–602 (1997).
Alcorta, D. A. et al. Involvement of the cyclin-dependent kinase inhibitor p16 (INK4a) in replicative senescence of normal human fibroblasts. Proc. Natl Acad. Sci. USA 93, 13742–13747 (1996).
Dimri, G. P., Itahana, K., Acosta, M. & Campisi, J. Regulation of a senescence checkpoint response by the E2F1 transcription factor and p14/ARF tumor suppressor. Mol. Cell. Biol. 20, 273–285 (2000).
Stein, G. H., Beeson, M. & Gordon, L. Failure to phosphorylate the retinoblastoma gene product in senescent human fibroblasts. Science 249, 666–669 (1990).
Futreal, P. A. & Barrett, J. C. Failure of senescent cells to phosphorylate the RB protein. Oncogene 6, 1109–1113 (1991).
Atadja, P., Wong, H., Garkavstev, I., Veillette, C. & Riabowol, K. Increased activity of p53 in senescing fibroblasts. Proc. Natl Acad. Sci. USA 92, 8348–8352 (1995).
DiLeonardo, A., Linke, S. P., Clarkin, K. & Wahl, G. M. DNA damage triggers a prolonged p53-dependent G1 arrest and long-term induction of Cip1 in normal human fibroblasts. Genes Dev. 8, 2540–2551 (1994).
Chen, Q. et al. Molecular analysis of H2O2-induced senescent-like growth arrest in normal human fibroblasts: p53 and Rb control G(1) arrest but not cell replication. Biochem. J. 332, 43–50 (1998).
Hara, E., Tsuri, H., Shinozaki, S. & Oda, K. Cooperative effect of antisense-Rb and antisense-p53 oligomers on the extension of lifespan in human diploid fibroblasts, TIG-1. Biochem. Biophys. Res. Comm. 179, 528–534 (1991).
Shay, J. W., Pereira-Smith, O. M. & Wright, W. E. A role for both Rb and p53 in the regulation of human cellular senescence. Exp. Cell Res. 196, 33–39 (1991).
Gire, V. & Wynford-Thomas, D. Reinitiation of DNA synthesis and cell division in senescent human fibroblasts by microinjection of anti-p53 antibodies. Mol. Cell. Biol. 18, 1611–1621 (1998).
Lundberg, A. S., Hahn, W. C., Gupta, P. & Weinberg, R. A. Genes involved in senescence and immortalization. Curr. Opin. Cell Biol. 12, 705–709 (2000).
Bringold, F. & Serrano, M. Tumor suppressors and oncogenes in cellular senescence. Exp. Gerontol. 35, 317–329 (2000).
Itahana, K., Dimri, G. & Campisi, J. Regulation of cellular senescence by p53. Eur. J. Biochem. 268, 2784–2791 (2001).
Sharpless, N. E. & DePinho, R. A. The INK4A/ARF locus and its two gene products. Curr. Opin. Genet. Dev. 9, 22–30 (1999).
Dai, C. Y. & Enders, G. H. p16 INK4a can initiate an autonomous senescence program. Oncogene 19, 1613–1622 (2000).
Sugrue, M. M., Shin, D. Y., Lee, S. W. & Aaronson, S. A. Wild-type p53 triggers a rapid senescence program in human tumor cells lacking functional p53. Proc. Natl Acad. Sci. USA 94, 9648–9653 (1997).
Xu, H. J. et al. Reexpression of the retinoblastoma protein in tumor cells induces senescence and telomerase inhibition. Oncogene 15, 2589–2596 (1997).
Amundson, S. A., Myers, T. G. & Fornace, A. J. Roles for p53 in growth arrest and apoptosis: putting on the brakes after genotoxic stress. Oncogene 17, 3287–3299 (1998).
Prives, C. & Hall, P. A. The p53 pathway. J. Pathol. 187, 112–126 (1999).
Bargonetti, J. & Manfredi, J. J. Multiple roles of the tumor suppressor p53. Curr. Opin. Oncol. 14, 86–91 (2002).
Wahl, G. M. & Carr, A. M. The evolution of diverse biological responses to DNA damage: insights from yeast and p53. Nature Cell Biol. 3, E277–E286 (2001).
Lu, X. & Horvitz, H. R. lin-35 and lin–53, two genes that antagonize a C. elegans Ras pathway, encode proteins similar to Rb and its binding protein RbbAp48. Cell 95, 981–991 (1998).
Du, W., Vidal, M., Xie, J. E. & Dyson, N. RBF, a novel RB-related gene that regulates E2F activity and interacts with cyclin E in Drosophila. Genes Dev. 10, 1206–1218 (1996).
Derry, W. B., Putzke, A. P. & Rothman, J. H. Caenorhabditis elegans p53: role in apoptosis, meiosis, and stress resistance. Science 294, 591–595 (2001).
Schumacher, B., Hoffman, K., Boulton, S. & Gartner, A. The C. elegans homolog of the p53 tumor suppressor is required for DNA damage-induced apoptosis. Curr. Biol. 11, 1722–1727 (2001).
Brodsky, M. H. et al. Drosophila p53 binds a damage response element at the reaper locus. Cell 101, 103–113 (2000).
Ollmann, M. et al. Drosophila p53 is a structural and functional homolog of the tumor suppressor p53. Cell 101, 91–101 (2000).
Jin, S. et al. Identification and characterization of a p53 homologue in Drosophila melanogaster. Proc. Natl Acad. Sci. USA 97, 7301–7306 (2000). References 106–110 describe the conserved sequence and functions of invertebrate ( C. elegans and D. melanogaster ) p53.
Finch, C. R. Longevity, Senescence and the Genome (Univ. Chicago Press, Chicago, 1991).
Williams, G. C. Pleiotropy, natural selection, and the evolution of senescence. Evolution 11, 398–411 (1957).
Thompson, C. B. Apoptosis in the pathogenesis and treatment of disease. Science 267, 1456–1462 (1995).
Fadeel, B., Orrenius, S. & Zhivotovsky, B. Apoptosis in human disease: a new skin for an old ceremony? Biochem. Biophys. Res. Comm. 266, 699–717 (1999).
Martin, L. J. Neuronal cell death in nervous system development, disease, and injury. Int. J. Mol. Med. 7, 455–478 (2001).
Almeida-Porada, G., Porada, C. & Zanjani, E. D. Adult stem cell plasticity and methods of detection. Rev. Clin. Exp. Hematol. 5, 26–41 (2001).
Weissman, I. L., Anderson, D. J. & Gage, F. Stem and progenitor cells: origins, phenotypes, lineage commitments, and transdifferentiations. Annu. Rev. Cell Dev. Biol. 17, 387–403 (2001).
Weinstein, B. S. & Ciszek, D. The reserve capacity hypothesis: evolutionary origins and modern implications between tumor suppression and tissue repair. Exp. Gerontol. 37, 615–627 (2002).
Donehower, L. A. Does p53 affect organismal aging? J. Cell Physiol. 192, 23–33 (2002).
Krtolica, A. & Campisi, J. Cancer and aging: a model for the cancer promoting effects of the aging stroma. Int. J. Biochem. Cell Biol. 34, 1401–1414 (2002).
Dimri, G. P. et al. A novel biomarker identifies senescent human cells in culture and in aging skin in vivo. Proc. Natl Acad. Sci. USA 92, 9363–9367 (1995).
Choi, J. et al. Expression of senescence-associated beta-galactosidase in enlarged prostates from men with benign prostatic hyperplasia. Urology 56, 160–166 (2000).
Paradis, V. et al. Replicative senescence in normal liver, chronic hepatitis C, and hepatocellular carcinomas. Hum. Pathol. 32, 327–332 (2001).
Vasile, E., Tomita, Y., Brown, L. F., Kocher, O. & Dvorak, H. F. Differential expression of thymosin beta-10 by early passage and senescent vascular endothelium is modulated by VPF/VEGF: evidence for senescent endothelial cells in vivo at sites of atherosclerosis. FASEB J. 15, 458–466 (2001). References 121–124 describe some of the evidence that senescent cells exist, accumulate with age and contribute to age-related pathology in vivo.
Fusenig, N. E. & Boukamp, P. Multiple stages and genetic alterations in immortalization, malignant transformation, and tumor progression of human skin keratinocytes. Mol. Carcinog. 23, 144–158 (1998).
Olumi, A. F. et al. Carcinoma-associated fibroblasts direct tumor progression of initiated human prostatic epithelium. Cancer Res. 59, 5002–5011 (1999). Shows that fibroblasts, if appropriately stimulated, can facilitate the neoplastic progression of epithelial cells.
Krtolica, A., Parrinello, S., Lockett, S., Desprez, P. & Campisi, J. Senescent fibroblasts promote epithelial cell growth and tumorigenesis: a link between cancer and aging. Proc. Natl Acad. Sci. USA 98, 12072–12077 (2001). Shows that senescent human fibroblasts can promote the neoplastic progression of preneoplastic epithelial cells.
Tyner, S. D. et al. p53 mutant mice that display early aging-associated phenotypes. Nature 415, 45–53 (2002). Shows that increased p53 activity suppresses the development of cancer in mice, but also promotes the premature development of ageing phenotypes.
Garcia-Cao, I. et al. 'Super p53' mice exhibit enhanced DNA damage response, are tumor resistant and age normally. EMBO J. 21, 6225–6235 (2002).
Davenport, J. Tumor-free but not in the clear. Science, SAGE–KE, 2002.
Gray, J. W. & Collins, C. Genome changes and gene expression in human solid tumors. Carcinogenesis 21, 443–452 (2000).
Jonason, A. S. et al. Frequent clones of p53-mutated keratinocytes in normal human skin. Proc. Natl Acad. Sci. USA 93, 14025–14029 (1996). Shows that potentially oncogenic cells — in this case, harbouring TP53 mutations — are present in apparently normal young human tissue.
Aubele, M. et al. Extensive ductal carcinoma in situ with small foci of invasive ductal carcinoma: evidence of genetic resemblance by CGH. Int. J. Cancer 85, 82–86 (2000).
Deng, G., Lu, Y., Zlotnikov, G., Thor, A. D. & Smith, H. S. Loss of heterozygosity in normal tissue adjacent to breast carcinomas. Science 274, 2057–2059 (1996). Shows that apparently normal human tissue harbours potentially oncogenic mutations.
Umayahara, K. et al. Comparative genomic hybridization detects genetic alterations during early stages of cervical cancer progression. Genes Chromosom. Cancer 33, 98–102 (2002).
Ilmensee, K. Reversion of malignancy and normalized differentiation of teratocarcinoma cells in chimeric mice. Basic Life Sci. 12, 3–25 (1978). Shows that potentially malignant cells can fail to express their neoplastic properties when placed in a normal tissue microenvironment.
Liotta, L. A. & Kohn, E. C. The microenvironment of the tumour-host interface. Nature 411, 375–379 (2001).
Park, C. C., Bissell, M. J. & Barcellos-Hoff, M. H. The influence of the microenvironment on the malignant phenotype. Mol Med Today 6, 324–329 (2000).
Yap, D. B., Hsieh, J. K., Chan, F. S. & Lu, X. Mdm2: a bridge over the two tumour suppressors, p53 and Rb. Oncogene 18, 7681–7689 (1999).
Sherr, C. J. & Roberts, J. M. CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev. 13, 1501–1512 (1999).
Dyson, N. The regulation of E2F by pRB-family proteins. Genes Dev. 12, 2245–2262 (1998).
Author information
Authors and Affiliations
Related links
Related links
DATABASES
FlyBase
LocusLink
OMIM
Saccharomyces Genome Database
WormBase
Glossary
- COMPLEX ORGANISMS
-
Multicellular organisms that are composed of both post-mitotic and renewable (mitotic) somatic tissues.
- SIMPLE ORGANISMS
-
Multicellular organisms that are composed entirely or largely of post-mitotic somatic cells.
- CARETAKERS
-
Tumour-suppressor genes or proteins that act to protect the genome from damage or mutations. Many caretaker genes encode proteins that recognize or repair DNA damage.
- GATEKEEPERS
-
Tumour-suppressor genes or proteins that regulate cellular responses that prevent the survival or proliferation of potential cancer cells. These responses are known as apoptosis and cellular senescence, respectively.
- NUCLEOTIDE EXCISION REPAIR
-
A DNA-repair pathway that removes and replaces damaged nucleotides, particularly those that distort the DNA helix.
- TELOMERES
-
The DNA–protein structure that stabilizes the ends of linear chromosomes and protects them from degradation or fusion. In vertebrates, telomeres are composed of several-kilobase pairs of the sequence TTTAGGG and several associated proteins.
- APOPTOSIS
-
Ordered, genetically programmed cell death triggered by both physiological stimuli and cellular damage. Apoptosis avoids cell lysis and subsequent inflammation.
- CELLULAR SENESCENCE
-
The essentially irreversible loss of cell division potential and the associated functional changes that are triggered by damage and other potential cancer-causing stimuli.
- LONGEVITY
-
Average or maximum lifespan of a cohort of organisms.
- AGEING
-
The decline in organismal fitness that occurs with increasing age.
- AGEING PHENOTYPES
-
The specific physiological manifestations of ageing.
- ANTAGONISTIC PLEIOTROPY
-
The hypothesis that genes or processes that were selected to benefit the health and fitness of young organisms can have unselected deleterious effects that are manifest in older organisms and thereby contribute to ageing.
- MISMATCH REPAIR
-
A DNA-repair pathway that removes and replaces nucleotides that have been misrepaired by DNA polymerases during DNA replication.
- BASE EXCISION REPAIR
-
A DNA-repair pathway that excises and replaces damaged DNA bases.
- NON-HOMOLOGOUS END-JOINING REPAIR
-
A relatively error-prone pathway that repairs double-strand breaks by ligating non-homologous DNA ends.
- HOMOLOGOUS RECOMBINATIONAL REPAIR
-
A relatively error-free pathway that repairs DNA double-strand breaks using an undamaged sister chromatid or homologous chromosome as a template.
- XERODERMA PIGMENTOSUM
-
A group of cancer-prone syndromes in humans that are caused by defects in the nucleotide excision repair genes.
- NECROSIS
-
Passive or unregulated cell death, in which cells lyse and deposit degradative and antigenic cell constituents into the surrounding tissue. Necrotic cell death, in contrast to apoptosis, often provokes an inflammation reaction.
Rights and permissions
About this article
Cite this article
Campisi, J. Cancer and ageing: rival demons?. Nat Rev Cancer 3, 339–349 (2003). https://doi.org/10.1038/nrc1073
Issue Date:
DOI: https://doi.org/10.1038/nrc1073
This article is cited by
-
Biomarkers of UVB radiation-related senescent fibroblasts
Scientific Reports (2024)
-
Coloured Noises Induced Regime Shift Yet Energy-Consuming in an E2F/Myc Genetic Circuit Involving miR-17-92
Journal of Statistical Physics (2023)
-
Association between gut microbiota and longevity: a genetic correlation and mendelian randomization study
BMC Microbiology (2022)
-
Geotemporospatial and causal inferential epidemiological overview and survey of USA cannabis, cannabidiol and cannabinoid genotoxicity expressed in cancer incidence 2003–2017: part 1 – continuous bivariate analysis
Archives of Public Health (2022)
-
Biologia Futura: four questions about ageing and the future of relevant animal models
Biologia Futura (2022)