Key Points
-
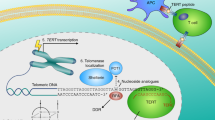
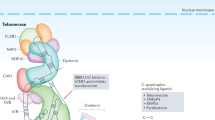
Telomerase is a ribonucleoprotein complex responsible for maintaining chromosome-end structures called telomeres. Telomerase activity enables unlimited cell proliferation (immortality) in diverse human cancers.
-
Despite the cloning of the catalytic component of telomerase approximately 20 years ago and the central role of telomerase in diverse cancers, only one small-molecule telomerase inhibitor, imetelstat, has been tested in clinical trials.
-
Components of the telomerase complex have extra-telomeric activities that may contribute to cancer cell survival, proliferation and malignancy.
-
Recent advances in drug discovery technology may be leveraged in the identification and development of novel small-molecule inhibitors that simultaneously disrupt telomere maintenance and the non-canonical activities of telomerase components.
-
This Review outlines strategies for harnessing state-of-the-art drug discovery techniques to find novel cancer therapeutics that exploit multifaceted biological functions of core components of telomerase.
Abstract
Telomerase activity is responsible for the maintenance of chromosome end structures (telomeres) and cancer cell immortality in most human malignancies, making telomerase an attractive therapeutic target. The rationale for targeting components of the telomerase holoenzyme has been strengthened by accumulating evidence indicating that these molecules have extra-telomeric functions in tumour cell survival and proliferation. This Review discusses current knowledge of the biogenesis, structure and multiple functions of telomerase-associated molecules intertwined with recent advances in drug discovery approaches. We also describe the fertile ground available for the pursuit of next-generation small-molecule inhibitors of telomerase.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Shay, J. W. & Bacchetti, S. A survey of telomerase activity in human cancer. Eur. J. Cancer 33, 787–791 (1997).
Kim, N. W. et al. Specific association of human telomerase activity with immortal cells and cancer. Science 266, 2011–2015 (1994).
Greider, C. W. & Blackburn, E. H. Identification of a specific telomere terminal transferase activity in Tetrahymena extracts. Cell 43, 405–413 (1985). A description of the discovery of the enzymatic activity responsible for the synthesis of telomeric DNA and its necessity for chromosome end replication.
Levy, M. Z., Allsopp, R. C., Futcher, A. B., Greider, C. W. & Harley, C. B. Telomere end-replication problem and cell aging. J. Mol. Biol. 225, 951–960 (1992).
Harley, C. B. Telomerase and cancer therapeutics. Nat. Rev. Cancer 8, 167–179 (2008).
Akiyama, M. et al. Effects of oligonucleotide N3′→P5′ thio-phosphoramidate (GRN163) targeting telomerase RNA in human multiple myeloma cells. Cancer Res. 63, 6187–6194 (2003).
Asai, A. et al. A novel telomerase template antagonist (GRN163) as a potential anticancer agent. Cancer Res. 63, 3931–3939 (2003).
Wang, E. S. et al. Telomerase inhibition with an oligonucleotide telomerase template antagonist: in vitro and in vivo studies in multiple myeloma and lymphoma. Blood 103, 258–266 (2004). Demonstrates that a TR agonist, a precursor of imetelstat, has more potent effects on tumour cells with short telomeres than on those with long telomeres.
Baerlocher, G. M. et al. Telomerase inhibitor imetelstat in patients with essential thrombocythemia. N. Engl. J. Med. 373, 920–928 (2015).
Tefferi, A. et al. A pilot study of the telomerase inhibitor imetelstat for myelofibrosis. N. Engl. J. Med. 373, 908–919 (2015).
Chiappori, A. A. et al. A randomized phase II study of the telomerase inhibitor imetelstat as maintenance therapy for advanced non-small-cell lung cancer. Ann. Oncol. 26, 354–362 (2015). Shows that imetelstat has limited efficacy in patients with lung cancer in a phase II trial.
Harley, C. B., Futcher, A. B. & Greider, C. W. Telomeres shorten during ageing of human fibroblasts. Nature 345, 458–460 (1990).
D'Adda di Fagagna, F. et al. A DNA damage checkpoint response in telomere-initiated senescence. Nature 426, 194–198 (2003).
Bodnar, A. G. et al. Extension of life-span by introduction of telomerase into normal human cells. Science 279, 349–352 (1998).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Feng, J. et al. The RNA component of human telomerase. Science 269, 1236–1241 (1995). Reports the cloning and characterization of the human TR gene.
Meyerson, M. et al. hEST2, the putative human telomerase catalytic subunit gene, is up-regulated in tumor cells and during immortalization. Cell 90, 785–795 (1997).
Weinrich, S. L. et al. Reconstitution of human telomerase with the template RNA component hTR and the catalytic protein subunit hTRT. Nat. Genet. 17, 498–502 (1997).
Kilian, A. et al. Isolation of a candidate human telomerase catalytic subunit gene, which reveals complex splicing patterns in different cell types. Hum. Mol. Genet. 6, 2011–2019 (1997).
Harrington, L. et al. Human telomerase contains evolutionarily conserved catalytic and structural subunits. Genes Dev. 11, 3109–3115 (1997).
Nakamura, T. M. et al. Telomerase catalytic subunit homologs from fission yeast and human. Science 277, 955–959 (1997).
Cohen, S. B. et al. Protein composition of catalytically active human telomerase from immortal cells. Science 315, 1850–1853 (2007). Demonstrates the purification of catalytically active telomerase enzyme complexes comprising TERT, TR and dyskerin from human cells.
Mitchell, J. R., Wood, E. & Collins, K. A telomerase component is defective in the human disease dyskeratosis congenita. Nature 402, 551–555 (1999). The first study to show that dyskerin associates with the human telomerase RNA component.
Fu, D. & Collins, K. Purification of human telomerase complexes identifies factors involved in telomerase biogenesis and telomere length regulation. Mol. Cell 28, 773–785 (2007). Demonstrates the affinity purification of human telomerase complexes containing proteins known to bind H/ACA box-containing snoRNAs.
Sauerwald, A. et al. Structure of active dimeric human telomerase. Nat. Struct. Mol. Biol. 20, 454–460 (2013).
Miracco, E. J., Jiang, J., Cash, D. D. & Feigon, J. Progress in structural studies of telomerase. Curr. Opin. Struct. Biol. 24, 115–124 (2014).
Jiang, J. et al. The architecture of Tetrahymena telomerase holoenzyme. Nature 496, 187–192 (2013).
Maritz, M. F., Richards, L. A. & Mackenzie, K. L. Assessment and quantification of telomerase enzyme activity. Methods Mol. Biol. 965, 215–231 (2013).
Damm, K. et al. A highly selective telomerase inhibitor limiting human cancer cell proliferation. EMBO J. 20, 6958–6968 (2001). Identifies a small-molecule compound that specifically inhibits telomerase-mediated telomere maintenance.
Seimiya, H. et al. Telomere shortening and growth inhibition of human cancer cells by novel synthetic telomerase inhibitors MST-312, MST-295, and MST-1991. Mol. Cancer Ther. 1, 657–665 (2002).
Naasani, I., Seimiya, H., Yamori, T. & Tsuruo, T. FJ5002: a potent telomerase inhibitor identified by exploiting the disease-oriented screening program with COMPARE analysis. Cancer Res. 59, 4004–4011 (1999).
Hayakawa, N. et al. Isothiazolone derivatives selectively inhibit telomerase from human and rat cancer cells in vitro. Biochemistry 38, 11501–11507 (1999).
Yaswen, P. et al. Therapeutic targeting of replicative immortality. Semin. Cancer Biol. 5 (Suppl.) S104–S128 (2015).
Jagadeesh, S., Kyo, S. & Banerjee, P. P. Genistein represses telomerase activity via both transcriptional and posttranslational mechanisms in human prostate cancer cells. Cancer Res. 66, 2107–2115 (2006).
Ramachandran, C., Fonseca, H. B., Jhabvala, P., Escalon, E. A. & Melnick, S. J. Curcumin inhibits telomerase activity through human telomerase reverse transcritpase in MCF-7 breast cancer cell line. Cancer Lett. 184, 1–6 (2002).
Wong, L. H. et al. A yeast chemical genetic screen identifies inhibitors of human telomerase. Chem. Biol. 20, 333–340 (2013).
Strahl, C. & Blackburn, E. H. Effects of reverse transcriptase inhibitors on telomere length and telomerase activity in two immortalized human cell lines. Mol. Cell. Biol. 16, 53–65 (1996).
Shin-ya, K. et al. Telomestatin, a novel telomerase inhibitor from Streptomyces anulatus. J. Am. Chem. Soc. 123, 1262–1263 (2001).
Izbicka, E. et al. Telomere-interactive agents affect proliferation rates and induce chromosomal destabilization in sea urchin embryos. Anticancer Drug Des. 14, 355–365 (1999).
Tauchi, T. et al. Telomerase inhibition with a novel G-quadruplex-interactive agent, telomestatin: in vitro and in vivo studies in acute leukemia. Oncogene 25, 5719–5725 (2006).
Tauchi, T. et al. Activity of a novel G-quadruplex-interactive telomerase inhibitor, telomestatin (SOT-095), against human leukemia cells: involvement of ATM-dependent DNA damage response pathways. Oncogene 22, 5338–5347 (2003).
Maritz, M. F., Napier, C. E., Wen, V. W. & MacKenzie, K. L. Targeting telomerase in hematologic malignancy. Future Oncol. 6, 769–789 (2010).
Herbert, B. S. et al. Lipid modification of GRN163, an N3′→P5′ thio-phosphoramidate oligonucleotide, enhances the potency of telomerase inhibition. Oncogene 24, 5262–5268 (2005).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT00124189 (2015).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01273090 (2014).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02598661 (2016).
Thompson, P. A. et al. A phase I trial of imetelstat in children with refractory or recurrent solid tumors: a Children's Oncology Group Phase I Consortium Study (ADVL1112). Clin. Cancer Res. 19, 6578–6584 (2013).
Engelhardt, M., Mackenzie, K., Drullinsky, P., Silver, R. T. & Moore, M. A. Telomerase activity and telomere length in acute and chronic leukemia, pre- and post-ex vivo culture. Cancer Res. 60, 610–617 (2000).
Schuller, C. E., Jankowski, K. & Mackenzie, K. L. Telomere length of cord blood-derived CD34+ progenitors predicts erythroid proliferative potential. Leukemia 21, 983–991 (2007).
Jackson, S. R. et al. Antiadhesive effects of GRN163L — an oligonucleotide N3′→P5′ thio-phosphoramidate targeting telomerase. Cancer Res. 67, 1121–1129 (2007).
Frazier, K. S. Antisense oligonucleotide therapies: the promise and the challenges from a toxicologic pathologist's perspective. Toxicol. Pathol. 43, 78–89 (2015).
Yi, X., Shay, J. W. & Wright, W. E. Quantitation of telomerase components and hTERT mRNA splicing patterns in immortal human cells. Nucleic Acids Res. 29, 4818–4825 (2001).
Ducrest, A. L., Szutorisz, H., Lingner, J. & Nabholz, M. Regulation of the human telomerase reverse transcriptase gene. Oncogene 21, 541–552 (2002).
Counter, C. M. et al. Telomerase activity is restored in human cells by ectopic expression of hTERT (hEST2), the catalytic subunit of telomerase. Oncogene 16, 1217–1222 (1998).
Vaziri, H. & Benchimol, S. Reconstitution of telomerase activity in normal human cells leads to elongation of telomeres and extended replicative life span. Curr. Biol. 8, 279–282 (1998).
MacKenzie, K. L., Franco, S., May, C., Sadelain, M. & Moore, M. A. Mass cultured human fibroblasts overexpressing hTERT encounter a growth crisis following an extended period of proliferation. Exp. Cell Res. 259, 336–350 (2000).
MacKenzie, K. L. et al. Multiple stages of malignant transformation of human endothelial cells modelled by co-expression of telomerase reverse transcriptase, SV40 T antigen and oncogenic N-ras. Oncogene 21, 4200–4211 (2002).
Kiyono, T. et al. Both Rb/p16INK4a inactivation and telomerase activity are required to immortalize human epithelial cells. Nature 396, 84–88 (1998).
Xie, M., Podlevsky, J. D., Qi, X., Bley, C. J. & Chen, J. J. A novel motif in telomerase reverse transcriptase regulates telomere repeat addition rate and processivity. Nucleic Acids Res. 38, 1982–1996 (2010).
Peng, Y., Mian, I. S. & Lue, N. F. Analysis of telomerase processivity: mechanistic similarity to HIV-1 reverse transcriptase and role in telomere maintenance. Mol. Cell 7, 1201–1211 (2001).
Gillis, A. J., Schuller, A. P. & Skordalakes, E. Structure of the Tribolium castaneum telomerase catalytic subunit TERT. Nature 455, 633–637 (2008).
Jacobs, S. A., Podell, E. R. & Cech, T. R. Crystal structure of the essential N-terminal domain of telomerase reverse transcriptase. Nat. Struct. Mol. Biol. 13, 218–225 (2006).
Lingner, J. et al. Reverse transcriptase motifs in the catalytic subunit of telomerase. Science 276, 561–567 (1997). Identifies a eukaryotic TERT protein and characterizes reverse transcriptase motifs required for telomere maintenance.
Stewart, S. A. et al. Telomerase contributes to tumorigenesis by a telomere length-independent mechanism. Proc. Natl Acad. Sci. USA 99, 12606–12611 (2002). Demonstrates that ectopic expression of TERT transforms immortalized cells via an ALT mechanism.
Fleisig, H. B. & Wong, J. M. Telomerase promotes efficient cell cycle kinetics and confers growth advantage to telomerase-negative transformed human cells. Oncogene 31, 954–965 (2012).
Sharma, G. G. et al. hTERT associates with human telomeres and enhances genomic stability and DNA repair. Oncogene 22, 131–146 (2003).
Gonzalez, O. G. et al. Telomerase stimulates ribosomal DNA transcription under hyperproliferative conditions. Nat. Commun. 5, 4599 (2014).
Smith, L. L., Coller, H. A. & Roberts, J. M. Telomerase modulates expression of growth-controlling genes and enhances cell proliferation. Nat. Cell Biol. 5, 474–479 (2003).
Mukherjee, S., Firpo, E. J., Wang, Y. & Roberts, J. M. Separation of telomerase functions by reverse genetics. Proc. Natl Acad. Sci. USA 108, E1363–E1371 (2011). Describes the use of a panel of TERT mutants to functionally demonstrate multiple aspects of telomerase function that are distinct from telomere maintenance.
Ludwig, A. et al. Ribozyme cleavage of telomerase mRNA sensitizes breast epithelial cells to inhibitors of topoisomerase. Cancer Res. 61, 3053–3061 (2001).
Cao, Y., Li, H., Deb, S. & Liu, J. P. TERT regulates cell survival independent of telomerase enzymatic activity. Oncogene 21, 3130–3138 (2002).
Luiten, R. M., Pene, J., Yssel, H. & Spits, H. Ectopic hTERT expression extends the life span of human CD4+ helper and regulatory T-cell clones and confers resistance to oxidative stress-induced apoptosis. Blood. 101, 4512–4519 (2003).
Dudognon, C. et al. Death receptor signaling regulatory function for telomerase: hTERT abolishes TRAIL-induced apoptosis, independently of telomere maintenance. Oncogene 23, 7469–7474 (2004).
Saretzki, G., Ludwig, A., von Zglinicki, T. & Runnebaum, I. B. Ribozyme-mediated telomerase inhibition induces immediate cell loss but not telomere shortening in ovarian cancer cells. Cancer Gene Ther. 8, 827–834 (2001).
Kraemer, K. et al. Antisense-mediated hTERT inhibition specifically reduces the growth of human bladder cancer cells. Clin. Cancer Res. 9, 3794–3800 (2003).
Gandellini, P. et al. Down-regulation of human telomerase reverse transcriptase through specific activation of RNAi pathway quickly results in cancer cell growth impairment. Biochem. Pharmacol. 73, 1703–1714 (2007).
Baird, D. M. et al. Telomere instability in the male germline. Hum. Mol. Genet. 15, 45–51 (2006).
Xu, L. & Blackburn, E. H. Human cancer cells harbor T-stumps, a distinct class of extremely short telomeres. Mol. Cell 28, 315–327 (2007).
Choi, J. et al. TERT promotes epithelial proliferation through transcriptional control of a Myc- and Wnt-related developmental program. PLoS Genet. 4, e10 (2008).
Maida, Y. et al. An RNA-dependent RNA polymerase formed by TERT and the RMRP RNA. Nature 461, 230–235 (2009).
Koh, C. M. et al. Telomerase regulates MYC-driven oncogenesis independent of its reverse transcriptase activity. J. Clin. Invest. 125, 2109–2122 (2015). Demonstrates that TERT stabilizes MYC on chromatin to facilitate activation of transcriptional programmes involved in tumorigenesis.
Ghosh, A. et al. Telomerase directly regulates NF-κB-dependent transcription. Nat. Cell Biol. 14, 1270–1281 (2012).
Park, J. I. et al. Telomerase modulates Wnt signalling by association with target gene chromatin. Nature 460, 66–72 (2009).
Listerman, I., Gazzaniga, F. S. & Blackburn, E. H. An investigation of the effects of the core protein telomerase reverse transcriptase on Wnt signaling in breast cancer cells. Mol. Cell. Biol. 34, 280–289 (2014).
Strong, M. A. et al. Phenotypes in mTERT+/− and mTERT−/− mice are due to short telomeres, not telomere-independent functions of telomerase reverse transcriptase. Mol. Cell. Biol. 31, 2369–2379 (2011).
Luo, J., Solimini, N. L. & Elledge, S. J. Principles of cancer therapy: oncogene and non-oncogene addiction. Cell 136, 823–837 (2009).
Kondo, Y. et al. Inhibition of telomerase increases the susceptibility of human malignant glioblastoma cells to cisplatin-induced apoptosis. Oncogene 16, 2243–2248 (1998).
Zhang, P., Chan, S. L., Fu, W., Mendoza, M. & Mattson, M. P. TERT suppresses apoptotis at a premitochondrial step by a mechanism requiring reverse transcriptase activity and 14-3-3 protein-binding ability. FASEB J. 17, 767–769 (2003).
Shin, K. H. et al. Introduction of human telomerase reverse transcriptase to normal human fibroblasts enhances DNA repair capacity. Clin. Cancer Res. 10, 2551–2560 (2004).
Masutomi, K. et al. The telomerase reverse transcriptase regulates chromatin state and DNA damage responses. Proc. Natl Acad. Sci. USA 102, 8222–8227 (2005).
Lue, N. F. et al. Telomerase can act as a template- and RNA-independent terminal transferase. Proc. Natl Acad. Sci. USA 102, 9778–9783 (2005).
Xi, L. & Cech, T. R. Inventory of telomerase components in human cells reveals multiple subpopulations of hTR and hTERT. Nucleic Acids Res. 42, 8565–8577 (2014).
Akincilar, S. C. et al. Quantitative assessment of telomerase components in cancer cell lines. FEBS Lett. 589, 974–984 (2015).
Kellermann, G. et al. Identification of human telomerase assembly inhibitors enabled by a novel method to produce hTERT. Nucleic Acids Res. 43, e99 (2015). Describes a method for producing purified TERT in high quantities that could be applied to in vitro biochemical assays and small-molecule HTS.
Higueruelo, A. P., Jubb, H. & Blundell, T. L. Protein–protein interactions as druggable targets: recent technological advances. Curr. Opin. Pharmacol. 13, 791–796 (2013). A concise review of novel assay formats and molecular libraries that can be used to identify small-molecule inhibitors of molecular targets previously considered undruggable.
Folini, M. et al. Antisense oligonucleotide-mediated inhibition of hTERT, but not hTERC, induces rapid cell growth decline and apoptosis in the absence of telomere shortening in human prostate cancer cells. Eur. J. Cancer 41, 624–634 (2005).
Zaffaroni, N., Pennati, M. & Folini, M. Validation of telomerase and survivin as anticancer therapeutic targets using ribozymes and small-interfering RNAs. Methods Mol. Biol. 361, 239–263 (2007).
Takakura, M. et al. Cloning of human telomerase catalytic subunit (hTERT) gene promoter and identification of proximal core promoter sequences essential for transcriptional activation in immortalized and cancer cells. Cancer Res. 59, 551–557 (1999).
Greenberg, R. A. et al. Telomerase reverse transcriptase gene is a direct target of c-Myc but is not functionally equivalent in cellular transformation. Oncogene 18, 1219–1226 (1999).
Horikawa, I., Cable, P. L., Afshari, C. & Barrett, J. C. Cloning and characterization of the promoter region of human telomerase reverse transcriptase gene. Cancer Res. 59, 826–830 (1999).
Grasselli, A. et al. Estrogen receptor-α and endothelial nitric oxide synthase nuclear complex regulates transcription of human telomerase. Circ. Res. 103, 34–42 (2008).
Lin, S. Y. & Elledge, S. J. Multiple tumor suppressor pathways negatively regulate telomerase. Cell 113, 881–889 (2003).
Maida, Y. et al. Direct activation of telomerase by EGF through Ets-mediated transactivation of TERT via MAP kinase signaling pathway. Oncogene 21, 4071–4079 (2002).
Cong, Y. S., Wen, J. & Bacchetti, S. The human telomerase catalytic subunit hTERT: organization of the gene and characterization of the promoter. Hum. Mol. Genet. 8, 137–142 (1999).
Moon, D. O., Kim, M. O., Lee, J. D., Choi, Y. H. & Kim, G. Y. Butein suppresses c-Myc-dependent transcription and Akt-dependent phosphorylation of hTERT in human leukemia cells. Cancer Lett. 286, 172–179 (2009).
Guo, Q. L. et al. Inhibition of human telomerase reverse transcriptase gene expression by gambogic acid in human hepatoma SMMC-7721 cells. Life Sci. 78, 1238–1245 (2006).
Bilsland, A. E. et al. Dynamic telomerase gene suppression via network effects of GSK3 inhibition. PLoS ONE 4, e6459 (2009). Demonstrates the power of cell-based assays for identifying small-molecule inhibitors of pathways required for transcription from the TERT promoter.
Bilsland, A. E. et al. Mathematical model of a telomerase transcriptional regulatory network developed by cell-based screening: analysis of inhibitor effects and telomerase expression mechanisms. PLoS Comput. Biol. 10, e1003448 (2014).
Horn, S. et al. TERT promoter mutations in familial and sporadic melanoma. Science 339, 959–961 (2013).
Killela, P. J. et al. TERT promoter mutations occur frequently in gliomas and a subset of tumors derived from cells with low rates of self-renewal. Proc. Natl Acad. Sci. USA 110, 6021–6026 (2013).
Li, Y. et al. Non-canonical NF-κB signalling and ETS1/2 cooperatively drive C250T mutant TERT promoter activation. Nat. Cell Biol. 17, 1327–1338 (2015).
Avilion, A. A. et al. Human telomerase RNA and telomerase activity in immortal cell lines and tumor tissues. Cancer Res. 56, 645–650 (1996).
Bachand, F. & Autexier, C. Functional regions of human telomerase reverse transcriptase and human telomerase RNA required for telomerase activity and RNA–protein interactions. Mol. Cell. Biol. 21, 1888–1897 (2001).
Mitchell, J. R. & Collins, K. Human telomerase activation requires two independent interactions between telomerase RNA and telomerase reverse transcriptase. Mol. Cell 6, 361–371 (2000).
Fu, D. & Collins, K. Distinct biogenesis pathways for human telomerase RNA and H/ACA small nucleolar RNAs. Mol. Cell 11, 1361–1372 (2003).
Mitchell, J. R., Cheng, J. & Collins, K. A box H/ACA small nucleolar RNA-like domain at the human telomerase RNA 3′ end. Mol. Cell. Biol. 19, 567–576 (1999).
Rangarajan, S. & Friedman, S. H. Design, synthesis, and evaluation of phenanthridine derivatives targeting the telomerase RNA/DNA heteroduplex. Bioorg. Med. Chem. Lett. 17, 2267–2273 (2007).
Dominick, P. K., Keppler, B. R., Legassie, J. D., Moon, I. K. & Jarstfer, M. B. Nucleic acid-binding ligands identify new mechanisms to inhibit telomerase. Bioorg. Med. Chem. Lett. 14, 3467–3471 (2004).
Booy, E. P. et al. The RNA helicase RHAU (DHX36) unwinds a G4-quadruplex in human telomerase RNA and promotes the formation of the P1 helix template boundary. Nucleic Acids Res. 40, 4010–4024 (2012).
Lattmann, S., Stadler, M. B., Vaughn, J. P., Akman, S. A. & Nagamine, Y. The DEAH-box RNA helicase RHAU binds an intramolecular RNA G-quadruplex in TERC and associates with telomerase holoenzyme. Nucleic Acids Res. 39, 9390–9404 (2011).
Shadrick, W. R. et al. Discovering new medicines targeting helicases: challenges and recent progress. J. Biomol. Screen. 18, 761–781 (2013).
Nguyen, G. H. et al. A small molecule inhibitor of the BLM helicase modulates chromosome stability in human cells. Chem. Biol. 20, 55–62 (2013).
Keppler, B. R. & Jarstfer, M. B. Inhibition of telomerase activity by preventing proper assemblage. Biochemistry 43, 334–343 (2004).
Katunaric, M. & Zamolo, G. Modulating telomerase activity in tumor patients by targeting dyskerin binding site for hTR. Med. Hypotheses 79, 319–320. (2012).
Keppler, B. R. & Jarstfer, M. B. A high-throughput assay for a human telomerase protein–human telomerase RNA interaction. Anal. Biochem. 353, 75–82 (2006).
Mills, N. L., Shelat, A. A. & Guy, R. K. Assay optimization and screening of RNA–protein interactions by AlphaScreen. J. Biomol. Screen. 12, 946–955 (2007). Describes the development of AlphaScreen-based detection assays for the identification of small-molecule inhibitors of biomolecular interactions.
Li, S., Crothers, J., Haqq, C. M. & Blackburn, E. H. Cellular and gene expression responses involved in the rapid growth inhibition of human cancer cells by RNA interference-mediated depletion of telomerase RNA. J. Biol. Chem. 280, 23709–23717 (2005).
Kedde, M. et al. Telomerase-independent regulation of ATR by human telomerase RNA. J. Biol. Chem. 281, 40503–40514 (2006).
Kang, S., Im, K., Baek, J., Yoon, S. & Min, H. Macro and small over micro: macromolecules and small molecules that regulate microRNAs. Chembiochem 15, 1071–1078 (2014).
Penchovsky, R. & Stoilova, C. C. Riboswitch-based antibacterial drug discovery using high-throughput screening methods. Expert Opin. Drug Discov. 8, 65–82 (2013).
Ecker, D. J. & Griffey, R. H. RNA as a small-molecule drug target: doubling the value of genomics. Drug Discov. Today 4, 420–429 (1999).
Wilson, W. D. & Li, K. Targeting RNA with small molecules. Curr. Med. Chem. 7, 73–98 (2000).
Xavier, K. A., Eder, P. S. & Giordano, T. RNA as a drug target: methods for biophysical characterization and screening. Trends Biotechnol. 18, 349–356 (2000).
Tran, T. & Disney, M. D. Identifying the preferred RNA motifs and chemotypes that interact by probing millions of combinations. Nat. Commun. 3, 1125 (2012). Describes a novel combinatorial platform to find small-molecule chemotypes that interact with specific RNA motifs to permit the design of small molecules that modulate RNA function.
Baugh, C., Wang, S., Li, B., Appleman, J. R. & Thompson, P. A. SCAN — a high-throughput assay for detecting small molecule binding to RNA targets. J. Biomol. Screen. 14, 219–229 (2009).
Pinto, I. G., Guilbert, C., Ulyanov, N. B., Stearns, J. & James, T. L. Discovery of ligands for a novel target, the human telomerase RNA, based on flexible-target virtual screening and NMR. J. Med. Chem. 51, 7205–7215 (2008).
Irwin, J. J. & Shoichet, B. K. ZINC — a free database of commercially available compounds for virtual screening. J. Chem. Inf. Model. 45, 177–182 (2005).
Venteicher, A. S., Meng, Z., Mason, P. J., Veenstra, T. D. & Artandi, S. E. Identification of ATPases pontin and reptin as telomerase components essential for holoenzyme assembly. Cell 132, 945–957 (2008).
Heiss, N. S. et al. X-linked dyskeratosis congenita is caused by mutations in a highly conserved gene with putative nucleolar functions. Nat. Genet. 19, 32–38 (1998).
Meier, U. T. & Blobel, G. NAP57, a mammalian nucleolar protein with a putative homolog in yeast and bacteria. J. Cell Biol. 127, 1505–1514 (1994).
Hamma, T. & Ferre-D'Amare, A. R. Pseudouridine synthases. Chem. Biol. 13, 1125–1135 (2006).
Kim, N. K., Theimer, C. A., Mitchell, J. R., Collins, K. & Feigon, J. Effect of pseudouridylation on the structure and activity of the catalytically essential P6.1 hairpin in human telomerase RNA. Nucleic Acids Res. 38, 6746–6756 (2010).
Mochizuki, Y., He, J., Kulkarni, S., Bessler, M. & Mason, P. J. Mouse dyskerin mutations affect accumulation of telomerase RNA and small nucleolar RNA, telomerase activity, and ribosomal RNA processing. Proc. Natl Acad. Sci. USA 101, 10756–10761 (2004).
Watkins, N. J. et al. Cbf5p, a potential pseudouridine synthase, and Nhp2p, a putative RNA-binding protein, are present together with Gar1p in all H BOX/ACA-motif snoRNPs and constitute a common bipartite structure. RNA 4, 1549–1568 (1998).
Jack, K. et al. rRNA pseudouridylation defects affect ribosomal ligand binding and translational fidelity from yeast to human cells. Mol. Cell 44, 660–666 (2011).
Montanaro, L. et al. Novel dyskerin-mediated mechanism of p53 inactivation through defective mRNA translation. Cancer Res. 70, 4767–4777 (2010).
Montanaro, L. Dyskerin and cancer: more than telomerase. The defect in mRNA translation helps in explaining how a proliferative defect leads to cancer. J. Pathol. 222, 345–349 (2010).
Alawi, F. & Lin, P. Dyskerin is required for tumor cell growth through mechanisms that are independent of its role in telomerase and only partially related to its function in precursor rRNA processing. Mol. Carcinog. 50, 334–345 (2011).
Ruggero, D. et al. Dyskeratosis congenita and cancer in mice deficient in ribosomal RNA modification. Science 299, 259–262 (2003).
Ge, J. et al. Dyskerin ablation in mouse liver inhibits rRNA processing and cell division. Mol. Cell. Biol. 30, 413–422 (2010).
Pereboom, T. C., van Weele, L. J., Bondt, A. & MacInnes, A. W. A zebrafish model of dyskeratosis congenita reveals hematopoietic stem cell formation failure resulting from ribosomal protein-mediated p53 stabilization. Blood 118, 5458–5465 (2011).
Boon, K. et al. N-myc enhances the expression of a large set of genes functioning in ribosome biogenesis and protein synthesis. EMBO J. 20, 1383–1393 (2001).
Iritani, B. M. & Eisenman, R. N. c-Myc enhances protein synthesis and cell size during B lymphocyte development. Proc. Natl Acad. Sci. USA 96, 13180–13185 (1999).
O'Brien, R. et al. MYC-driven neuroblastomas are addicted to a telomerase-independent function of dyskerin. Cancer Res. 76, 2206–2218 (2016). Describes the telomerase-independent function and potential of dyskerin as a therapeutic target in neuroblastoma.
Rocchi, L., Barbosa, A. J., Onofrillo, C., Del Rio, A. & Montanaro, L. Inhibition of human dyskerin as a new approach to target ribosome biogenesis. PLoS ONE 9, e101971 (2014).
Heiss, N. S. et al. Gene structure and expression of the mouse dyskeratosis congenita gene, Dkc1. Genomics 67, 153–163 (2000).
He, J. et al. Targeted disruption of Dkc1, the gene mutated in X-linked dyskeratosis congenita, causes embryonic lethality in mice. Oncogene 21, 7740–7744 (2002).
Westin, E. R. et al. Telomere restoration and extension of proliferative lifespan in dyskeratosis congenita fibroblasts. Aging Cell 6, 383–394 (2007).
Knight, S. et al. Dyskeratosis Congenita (DC) Registry: identification of new features of DC. Br. J. Haematol. 103, 990–996 (1998).
Von Stedingk, K. et al. snoRNPs regulate telomerase activity in neuroblastoma and are associated with poor prognosis. Transl Oncol. 6, 447–457 (2013).
Alawi, F. & Lin, P. Loss of dyskerin reduces the accumulation of a subset of H/ACA snoRNA-derived miRNA. Cell Cycle 9, 2467–2469 (2010).
Montanaro, L. et al. Dyskerin expression influences the level of ribosomal RNA pseudo-uridylation and telomerase RNA component in human breast cancer. J. Pathol. 210, 10–18 (2006).
Sieron, P. et al. DKC1 overexpression associated with prostate cancer progression. Br. J. Cancer 101, 1410–1416 (2009).
Kiss, T., Fayet-Lebaron, E. & Jady, B. E. Box H/ACA small ribonucleoproteins. Mol. Cell 37, 597–606 (2010). A review of the biogenesis, intracellular trafficking, structure and function of H/ACA RNPs.
Dez, C., Noaillac-Depeyre, J., Caizergues-Ferrer, M. & Henry, Y. Naf1p, an essential nucleoplasmic factor specifically required for accumulation of box H/ACA small nucleolar RNPs. Mol. Cell. Biol. 22, 7053–7065 (2002).
Darzacq, X. et al. Stepwise RNP assembly at the site of H/ACA RNA transcription in human cells. J. Cell Biol. 173, 207–218 (2006).
Fatica, A., Dlakic, M. & Tollervey, D. Naf1 p is a box H/ACA snoRNP assembly factor. RNA 8, 1502–1514 (2002).
Grozdanov, P. N., Roy, S., Kittur, N. & Meier, U. T. SHQ1 is required prior to NAF1 for assembly of H/ACA small nucleolar and telomerase RNPs. RNA 15, 1188–1197 (2009).
Holt, S. E. et al. Functional requirement of p23 and Hsp90 in telomerase complexes. Genes Dev. 13, 817–826 (1999).
Ivanov, A. A., Khuri, F. R. & Fu, H. Targeting protein–protein interactions as an anticancer strategy. Trends Pharmacol. Sci. 34, 393–400 (2013).
Osterwald, S. et al. A three-dimensional colocalization RNA interference screening platform to elucidate the alternative lengthening of telomeres pathway. Biotechnol. J. 7, 103–116 (2012).
Stern, J. L., Zyner, K. G., Pickett, H. A., Cohen, S. B. & Bryan, T. M. Telomerase recruitment requires both TCAB1 and Cajal bodies independently. Mol. Cell. Biol. 32, 2384–2395 (2012).
Cristofari, G. et al. Human telomerase RNA accumulation in Cajal bodies facilitates telomerase recruitment to telomeres and telomere elongation. Mol. Cell 27, 882–889 (2007). Demonstrates the functional importance of telomerase precursors localized in Cajal bodies.
Venteicher, A. S. et al. A human telomerase holoenzyme protein required for Cajal body localization and telomere synthesis. Science 323, 644–648 (2009).
Tang, M. et al. Disease mutant analysis identifies a new function of DAXX in telomerase regulation and telomere maintenance. J. Cell Sci. 128, 331–341 (2015).
Soohoo, C. Y. et al. Telomerase inhibitor PinX1 provides a link between TRF1 and telomerase to prevent telomere elongation. J. Biol. Chem. 286, 3894–3906 (2011).
Zhou, X. Z. & Lu, K. P. The Pin2/TRF1-interacting protein PinX1 is a potent telomerase inhibitor. Cell 107, 347–359 (2001).
Yoo, J. E., Park, Y. N. & Oh, B. K. PinX1, a telomere repeat-binding factor 1 (TRF1)-interacting protein, maintains telomere integrity by modulating TRF1 homeostasis, the process in which human telomerase reverse transcriptase (hTERT) plays dual roles. J. Biol. Chem. 289, 6886–6898 (2014).
Banik, S. S. & Counter, C. M. Characterization of interactions between PinX1 and human telomerase subunits hTERT and hTR. J. Biol. Chem. 279, 51745–51748 (2004).
Zhong, F. L. et al. TPP1 OB-fold domain controls telomere maintenance by recruiting telomerase to chromosome ends. Cell 150, 481–494 (2012).
Abreu, E. et al. TIN2-tethered TPP1 recruits human telomerase to telomeres in vivo. Mol. Cell. Biol. 30, 2971–2982 (2010).
Chen, L. Y., Redon, S. & Lingner, J. The human CST complex is a terminator of telomerase activity. Nature 488, 540–544 (2012).
Brenke, R. et al. Fragment-based identification of druggable 'hot spots' of proteins using Fourier domain correlation techniques. Bioinformatics 25, 621–627 (2009).
Bilsland, A. E., Cairney, C. J. & Keith, W. N. Targeting the telomere and shelterin complex for cancer therapy: current views and future perspectives. J. Cell. Mol. Med. 15, 179–186 (2011).
Sfeir, A. & de Lange, T. Removal of shelterin reveals the telomere end-protection problem. Science 336, 593–597 (2012).
Bywater, M. J. et al. Inhibition of RNA polymerase I as a therapeutic strategy to promote cancer-specific activation of p53. Cancer Cell 22, 51–65 (2012).
Quin, J. E. et al. Targeting the nucleolus for cancer intervention. Biochim. Biophys. Acta 1842, 802–816 (2014).
Engelhardt, M. et al. Telomerase regulation, cell cycle, and telomere stability in primitive hematopoietic cells. Blood 90, 182–193 (1997).
Wright, W. E., Piatyszek, M. A., Rainey, W. E., Byrd, W. & Shay, J. W. Telomerase activity in human germline and embryonic tissues and cells. Dev. Genet. 18, 173–179 (1996).
Samper, E. et al. Long-term repopulating ability of telomerase-deficient murine hematopoietic stem cells. Blood 99, 2767–2775 (2002).
Imamura, S. et al. A non-canonical function of zebrafish telomerase reverse transcriptase is required for developmental hematopoiesis. PLoS ONE 3, e3364 (2008).
Sarin, K. Y. et al. Conditional telomerase induction causes proliferation of hair follicle stem cells. Nature 436, 1048–1052 (2005).
Drygin, D., O'Brien, S. E., Hannan, R. D., McArthur, G. A. & Von Hoff, D. D. Targeting the nucleolus for cancer-specific activation of p53. Drug Discov. Today 19, 259–265 (2014).
Henras, A. et al. Nhp2p and Nop10p are essential for the function of H/ACA snoRNPs. EMBO J. 17, 7078–7090 (1998).
Pogacic, V. D., F. & Filipowicz, W. Human H/ACA small nucleolar RNPs and telomerase share evolutionarily conserved proteins NHP2 and NOP10. Mol. Cell. Biol. 20, 9028–9040 (2000).
Girard, J. P. et al. GAR1 is an essential small nucleolar RNP protein required for pre-rRNA processing in yeast. EMBO J. 11, 673–682 (1992).
Liu, D. et al. PTOP interacts with POT1 and regulates its localization to telomeres. Nat. Cell Biol. 6, 673–680 (2004).
Xin, H. et al. TPP1 is a homologue of ciliate TEBP-β and interacts with POT1 to recruit telomerase. Nature 445, 559–562 (2007).
Nandakumar, J. et al. The TEL patch of telomere protein TPP1 mediates telomerase recruitment and processivity. Nature 492, 285–289 (2012).
Tang, J. et al. Critical role for Daxx in regulating Mdm2. Nat. Cell Biol. 8, 855–862 (2006).
Hollenbach, A. D., McPherson, C. J., Mientjes, E. J., Iyengar, R. & Grosveld, G. Daxx and histone deacetylase II associate with chromatin through an interaction with core histones and the chromatin-associated protein Dek. J. Cell Sci. 115, 3319–3330 (2002).
Forsythe, H. L., Jarvis, J. L., Turner, J. W., Elmore, L. W. & Holt, S. E. Stable association of hsp90 and p23, but Not hsp70, with active human telomerase. J. Biol. Chem. 276, 15571–15574 (2001).
Isaac, C., Yang, Y. & Meier, U. T. Nopp140 functions as a molecular link between the nucleolus and the coiled bodies. J. Cell Biol. 142, 319–329 (1998).
Yang, Y. et al. Conserved composition of mammalian box H/ACA and box C/D small nucleolar ribonucleoprotein particles and their interaction with the common factor Nopp140. Mol. Biol. Cell 11, 567–577 (2000).
Renvoise, B. et al. The loss of the snoRNP chaperone Nopp140 from Cajal bodies of patient fibroblasts correlates with the severity of spinal muscular atrophy. Hum. Mol. Genet. 18, 1181–1189 (2009).
Gallant, P. Control of transcription by Pontin and Reptin. Trends Cell Biol. 17, 187–192 (2007).
Zhu, Q. et al. GNL3L stabilizes the TRF1 complex and promotes mitotic transition. J. Cell Biol. 185, 827–839 (2009).
Du, X. et al. The homologous putative GTPases Grn1p from fission yeast and the human GNL3L are required for growth and play a role in processing of nucleolar pre-rRNA. Mol. Biol. Cell 17, 460–474 (2006).
Sexton, A. N. & Collins, K. The 5′ guanosine tracts of human telomerase RNA are recognized by the G-quadruplex binding domain of the RNA helicase DHX36 and function to increase RNA accumulation. Mol. Cell. Biol. 31, 736–743 (2011).
Snow, B. E. et al. Functional conservation of the telomerase protein Est1p in humans. Curr. Biol. 13, 698–704 (2003).
Reichenbach, P. et al. A human homolog of yeast Est1 associates with telomerase and uncaps chromosome ends when overexpressed. Curr. Biol. 13, 568–574 (2003).
Page, M. F., Carr, B., Anders, K. R., Grimson, A. & Anderson, P. SMG-2 is a phosphorylated protein required for mRNA surveillance in Caenorhabditis elegans and related to Upf1p of yeast. Mol. Cell. Biol. 19, 5943–5951 (1999).
Chawla, R. et al. Human UPF1 interacts with TPP1 and telomerase and sustains telomere leading-strand replication. EMBO J. 30, 4047–4058 (2011).
Fu, W., Lu, C. & Mattson, M. P. Telomerase mediates the cell survival-promoting actions of brain-derived neurotrophic factor and secreted amyloid precursor protein in developing hippocampal neurons. J. Neurosci. 22, 10710–10719 (2002).
Zhu, J., Wang, H., Bishop, J. M. & Blackburn, E. H. Telomerase extends the lifespan of virus-transformed human cells without net telomere lengthening. Proc. Natl Acad. Sci. USA 96, 3723–3728 (1999).
Li, S., Ferguson, M. J., Hawkins, C. J., Smith, C. & Elwood, N. J. Human telomerase reverse transcriptase protects hematopoietic progenitor TF-1 cells from death and quiescence induced by cytokine withdrawal. Leukemia 20, 1270–1278 (2006).
Nitta, E. et al. Telomerase reverse transcriptase protects ATM-deficient hematopoietic stem cells from ROS-induced apoptosis through a telomere-independent mechanism. Blood 117, 4169–4180 (2011).
Gomez, D. E., Armando, R. G. & Alonso, D. F. AZT as a telomerase inhibitor. Front. Oncol. 2, 113 (2012).
Hukezalie, K. R., Thumati, N. R., Cote, H. C. & Wong, J. M. In vitro and ex vivo inhibition of human telomerase by anti-HIV nucleoside reverse transcriptase inhibitors (NRTIs) but not by non-NRTIs. PLoS ONE 7, e47505 (2012).
El-Daly, H. et al. Selective cytotoxicity and telomere damage in leukemia cells using the telomerase inhibitor BIBR1532. Blood 105, 1742–1749 (2005).
Naasani, I., Seimiya, H. & Tsuruo, T. Telomerase inhibition, telomere shortening, and senescence of cancer cells by tea catechins. Biochem. Biophys. Res. Commun. 249, 391–396 (1998).
Gowan, S. M., Heald, R., Stevens, M. F. & Kelland, L. R. Potent inhibition of telomerase by small-molecule pentacyclic acridines capable of interacting with G-quadruplexes. Mol. Pharmacol. 60, 981–988 (2001).
Villa, R. et al. Inhibition of telomerase activity by geldanamycin and 17-allylamino, 17-demethoxygeldanamycin in human melanoma cells. Carcinogenesis 24, 851–859 (2003).
Acknowledgements
The Children's Cancer Institute Australia is affiliated with University of New South Wales, Australia, and the Sydney Children's Hospitals Network. The authors acknowledge financial support from the Cancer Council New South Wales (RG 15-16), the Australian Cancer Research Foundation, the Cancer Institute New South Wales (15/RIG/1-01), Therapeutic Innovation Australia (NCRIS 2015) and Cancer Therapeutics CRC.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Alternative lengthening of telomeres
-
A recombination-based mechanism of telomere lengthening that operates in cancer cells in the absence of telomerase.
- High-content imaging
-
Automated imaging of fluorescent stains or tags used to quantify changes in specific cellular processes, phenotypes or subcellular components.
- G-quadruplex
-
A nucleic acid secondary structure that forms in guanine-rich regions involving the stacking of square planar structures each formed by four guanines.
- Essential thrombocythaemia
-
A rare blood disorder characterized by the overproduction of platelets by megakaryocytes in the bone marrow.
- Myelofibrosis
-
A rare clonal bone marrow disorder in which haematopoiesis is overtaken by the production of fibrous tissue.
- Thrombocytopenia
-
A disorder featuring abnormally low platelet counts.
- Megakaryocyte
-
A bone marrow cell that produces small cell bodies called platelets that are involved in blood clotting.
- Nucleotide excision repair
-
A multistep DNA repair mechanism that removes and repairs DNA damage caused by radiation and chemicals.
- Terminal transferase
-
A template-independent polymerase that adds deoxynucleotides to the 3′ hydroxyl terminus of DNA.
- AlphaScreen technology
-
(amplified luminescent proximity homogeneous assay screen). A homogeneous bead-based proximity assay used to measure the interaction between two biological molecules.
- Small nucleolar RNAs
-
(snoRNAs). A class of small RNA molecules localized in nucleoli that guide chemical modifications of other RNAs, such as rRNAs and tRNAs.
- Small Cajol body-specific RNAs
-
(scaRNAs). Small RNAs that specifically localize to Cajal bodies and function to guide the modification (methylation and pseudouridinylation) of spliceosomal RNAs.
- Cajal bodies
-
Nuclear organelles adjacent to nucleoli in proliferating cells that are involved in the biogenesis of small nuclear ribonucleoproteins.
- Solution-based two-dimensional combinatorial screening
-
An array-based method for identifying interactions between specific RNA motifs and small molecules.
- Dyskeratosis congenita
-
A rare inherited disorder caused by mutation in the DKC1 gene (which encodes dyskerin) and characterized by multiple organ failure.
- Structure-based design
-
A form of rational drug design that relies on the three-dimensional structure of a specific biomolecular target.
Rights and permissions
About this article
Cite this article
Arndt, G., MacKenzie, K. New prospects for targeting telomerase beyond the telomere. Nat Rev Cancer 16, 508–524 (2016). https://doi.org/10.1038/nrc.2016.55
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc.2016.55
This article is cited by
-
The regulations of telomerase reverse transcriptase (TERT) in cancer
Cell Death & Disease (2024)
-
A ratiometric fluorescent nanoprobe for signal amplification monitoring of intracellular telomerase activity
Analytical and Bioanalytical Chemistry (2022)
-
Heparanase and the hallmarks of cancer
Journal of Translational Medicine (2020)
-
Emerging roles of telomeric chromatin alterations in cancer
Journal of Experimental & Clinical Cancer Research (2019)
-
Fundamental insights into autosomal dominant polycystic kidney disease from human-based cell models
Pediatric Nephrology (2019)