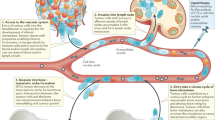
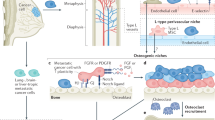
Key Points
-
Several tumours either develop in the skeleton, such as multiple myeloma, or metastasize to bone, such as breast and prostate cancers.
-
The skeleton provides a unique microenvironment that supports the development of these tumours, although the nature of this microenvironment has until recently been poorly defined.
-
Our understanding of the early, crucial events of tumour cell colonization, survival and dormancy, and reactivation of dormant cancer cells has been limited. However, progress in single cell imaging and molecular techniques has provided new insights.
-
Tumour cell dormancy in the skeleton is induced by interactions with specific cells in the local bone microenvironment. Cells of the osteoblast lineage, present on the endosteal bone surface, provide a supportive niche to keep tumour cells in a dormant state.
-
Reactivation of dormant tumour cells is mediated by extrinsic changes in the bone microenvironment. Osteoclasts, by remodelling the endosteal bone surface, represent one mechanism by which dormant cells can be reactivated.
-
Our improved understanding of the control of tumour dormancy in the skeleton has revealed new therapeutic opportunities. These include using bone-active drugs to promote long-term tumour cell dormancy, or conversely, promoting reactivation and targeting dormant cells to eradicate them and 'cure' tumours that develop in bone.
Abstract
During the past decade preclinical studies have defined many of the mechanisms used by tumours to hijack the skeleton and promote bone metastasis. This has led to the development and widespread clinical use of bone-targeted drugs to prevent skeletal-related events. This understanding has also identified a critical dependency between colonizing tumour cells and the cells of bone. This is particularly important when tumour cells first arrive in bone, adapt to their new microenvironment and enter a long-lived dormant state. In this Review, we discuss the role of different bone cell types in supporting disseminated tumour cell dormancy and reactivation, and highlight the new opportunities this provides for targeting the bone microenvironment to control dormancy and bone metastasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Fidler, I. J. The pathogenesis of cancer metastasis: the 'seed and soil' hypothesis revisited. Nat. Rev. Cancer 3, 453–458 (2003).
Mundy, G. R. & Martin, T. J. in Physiology and Pharmacology of Bone (ed. Mundy, G. R. & Martin, T. J.) 641–671 (Springer-Verlag, 1993).
Paget, S. The distribution of secondary growths in cancer of the breast. Lancet 1, 571–573 (1889). The classic description of the predilection of breast cancer cells to metastasize to the skeleton.
Fidler, I. J. & Kripke, M. L. The challenge of targeting metastasis. Cancer Metastasis Rev. 34, 635–641 (2015).
Weilbaecher, K. N., Guise, T. A. & McCauley, L. K. Cancer to bone: a fatal attraction. Nat. Rev. Cancer 11, 411–425 (2011).
Sterling, J. A., Edwards, J. R., Martin, T. J. & Mundy, G. R. Advances in the biology of bone metastasis: how the skeleton affects tumor behavior. Bone 48, 6–15 (2011).
Chambers, A. F., Groom, A. C. & MacDonald, I. C. Dissemination and growth of cancer cells in metastatic sites. Nat. Rev. Cancer 2, 563–572 (2002).
Cameron, M. D. et al. Temporal progression of metastasis in lung: cell survival, dormancy, and location dependence of metastatic inefficiency. Cancer Res. 60, 2541–2546 (2000).
Luzzi, K. J. et al. Multistep nature of metastatic inefficiency: dormancy of solitary cells after successful extravasation and limited survival of early micrometastases. Am. J. Pathol. 153, 865–873 (1998).
Ottewell, P. D. et al. Zoledronic acid has differential antitumor activity in the pre- and postmenopausal bone microenvironment in vivo. Clin. Cancer Res. 20, 2922–2932 (2014). One of the first studies to show that the bone microenvironment can control metastasis development.
Ottewell, P. D. et al. Castration-induced bone loss triggers growth of disseminated prostate cancer cells in bone. Endocr. Relat. Cancer 21, 769–781 (2014).
Wang, N. et al. Prostate cancer cells preferentially home to osteoblast-rich areas in the early stages of bone metastasis — evidence from in vivo models. J. Bone Miner. Res. 29, 2688–2696 (2014).
Lawson, M. A. et al. Osteoclasts control reactivation of dormant myeloma cells by remodeling the endosteal niche. Nat. Commun. 6, 8983 (2015). A demonstration of dormant cancer cells in the skeleton and the importance of extrinsic microenvironmental changes in promoting dormant cell reactivation.
Fortunato, L. et al. Positive bone marrow biopsy is associated with a decreased disease-free survival in patients with operable breast cancer. Ann. Surg. Oncol. 16, 3010–3019 (2009).
Tjensvoll, K. et al. Persistent tumor cells in bone marrow of non-metastatic breast cancer patients after primary surgery are associated with inferior outcome. BMC Cancer 12, 190 (2012).
Domschke, C. et al. Prognostic value of disseminated tumor cells in the bone marrow of patients with operable primary breast cancer: a long-term follow-up study. Ann. Surg. Oncol. 20, 1865–1871 (2013).
Boyle, W. J., Simonet, W. S. & Lacey, D. L. Osteoclast differentiation and activation. Nature 423, 337–342 (2003). A review of the discovery of osteoclast formation and activity by the tumour necrosis factor (TNF) receptor and TNF ligand family of molecules.
Suda, T. et al. Modulation of osteoclast differentiation and function by the new members of the tumor necrosis factor receptor and ligand families. Endocr. Rev. 20, 345–357 (1999).
Baron, R. Molecular mechanisms of bone resorption by the osteoclast. Anat. Rec. 224, 317–324 (1989).
Andersen, T. L. et al. Understanding coupling between bone resorption and formation: are reversal cells the missing link? Am. J. Pathol. 183, 235–246 (2013).
Delaisse, J. M. The reversal phase of the bone-remodeling cycle: cellular prerequisites for coupling resorption and formation. Bonekey Rep. 3, 561 (2014).
Kassem, M. & Bianco, P. Skeletal stem cells in space and time. Cell 160, 17–19 (2015). A succinct and authoritative summary of the differentiation of osteoblasts from stem cell precursors.
Kim, S. W. et al. Intermittent parathyroid hormone administration converts quiescent lining cells to active osteoblasts. J. Bone Miner. Res. 27, 2075–2084 (2012).
Mizoguchi, T. et al. Osterix marks distinct waves of primitive and definitive stromal progenitors during bone marrow development. Dev. Cell 29, 340–349 (2014).
Martin, T. J. & Sims, N. A. Coupling the activities of bone formation and resorption: a multitude of signals within the basic multicellular unit. BoneKey Rep. 3, 215 (2014).
Sims, N. A. & Martin, T. J. Coupling signals between the osteoclast and osteoblast: how are messages transmitted between these temporary visitors to the bone surface? Front. Endocrinol. (Lausanne) 6, 41 (2015). A critical review of current ideas behind the mechanisms coupling bone formation and bone resorption.
Martin, T. J., Ng, K. W. & Sims, N. A. in Translational Endocrinology of Bone (ed. Karsenty, G.) 5–26 (Elsevier Inc, 2013).
Kalajzic, Z. et al. Use of an alpha-smooth muscle actin GFP reporter to identify an osteoprogenitor population. Bone 43, 501–510 (2008).
Acar, M. et al. Deep imaging of bone marrow shows non-dividing stem cells are mainly perisinusoidal. Nature 526, 126–130 (2015).
Hattner, R., Epker, B. N. & Frost, H. M. Suggested sequential mode of control of changes in cell behaviour in adult bone remodelling. Nature 206, 489–490 (1965).
Rodan, G. A. & Martin, T. J. Role of osteoblasts in hormonal control of bone resorption—a hypothesis. Calcif. Tissue Int. 33, 349–351 (1981).
Parfitt, A. M. The coupling of bone formation to bone resorption: a critical analysis of the concept and of its relevance to the pathogenesis of osteoporosis. Metab. Bone Dis. Relat. Res. 4, 1–6 (1982).
Martin, T. J. & Sims, N. A. Osteoclast-derived activity in the coupling of bone formation to resorption. Trends Mol. Med. 11, 76–81 (2005).
Xian, L. et al. Matrix IGF-1 maintains bone mass by activation of mTOR in mesenchymal stem cells. Nat. Med. 18, 1095–1101 (2012).
Weivoda, M. M. et al. Osteoclast TGF-β receptor signaling induces Wnt1 secretion and couples bone resorption to bone formation. J. Bone Miner. Res. 31, 76–85 (2015).
Mundy, G. R. Mechanisms of bone metastasis. Cancer 80, 1546–1556 (1997).
Kozlow, W. & Guise, T. A. Breast cancer metastasis to bone: mechanisms of osteolysis and implications for therapy. J. Mammary Gland Biol. Neoplasia 10, 169–180 (2005).
Andersen, T. L. et al. A physical mechanism for coupling bone resorption and formation in adult human bone. Am. J. Pathol. 174, 239–247 (2009).
Kristensen, H. B., Andersen, T. L., Marcussen, N., Rolighed, L. & Delaisse, J. M. Increased presence of capillaries next to remodeling sites in adult human cancellous bone. J. Bone Miner. Res. 28, 574–585 (2013). A detailed description of BRCs in human bone and how blood vessels participate.
Hauge, E. M., Qvesel, D., Eriksen, E. F., Mosekilde, L. & Melsen, F. Cancellous bone remodeling occurs in specialized compartments lined by cells expressing osteoblastic markers. J. Bone Miner. Res. 16, 1575–1582 (2001).
Kristensen, H. B., Andersen, T. L., Marcussen, N., Rolighed, L. & Delaisse, J. M. Osteoblast recruitment routes in human cancellous bone remodeling. Am. J. Pathol. 184, 778–789 (2014).
Eriksen, E. F., Eghbali-Fatourechi, G. Z. & Khosla, S. Remodeling and vascular spaces in bone. J. Bone Miner. Res. 22, 1–6 (2007).
Andersen, T. L., Soe, K., Sondergaard, T. E., Plesner, T. & Delaisse, J. M. Myeloma cell-induced disruption of bone remodelling compartments leads to osteolytic lesions and generation of osteoclast-myeloma hybrid cells. Br. J. Haematol. 148, 551–561 (2010).
Yamaguchi, K., Croucher, P. I. & Compston, J. E. Comparison between the lengths of individual osteoid seams and resorption cavities in human iliac crest cancellous bone. Bone Miner. 23, 27–33 (1993).
Chang, M. K. et al. Osteal tissue macrophages are intercalated throughout human and mouse bone lining tissues and regulate osteoblast function in vitro and in vivo. J. Immunol. 181, 1232–1244 (2008).
Schofield, R. The relationship between the spleen colony-forming cell and the haemopoietic stem cell. Blood Cells 4, 7–25 (1978).
Nilsson, S. K., Johnston, H. M. & Coverdale, J. A. Spatial localization of transplanted hemopoietic stem cells: inferences for the localization of stem cell niches. Blood 97, 2293–2299 (2001).
Calvi, L. M. et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature 425, 841–846 (2003).
Zhang, J. et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nature 425, 836–841 (2003).
Bianco, P. Minireview: The stem cell next door: skeletal and hematopoietic stem cell “niches” in bone. Endocrinology 152, 2957–2962 (2011).
Askmyr, M., Sims, N. A., Martin, T. J. & Purton, L. E. What is the true nature of the osteoblastic hematopoietic stem cell niche? Trends Endocrinol. Metab. 20, 303–309 (2009).
Bianco, P. Bone and the hematopoietic niche: a tale of two stem cells. Blood 117, 5281–5288 (2011).
Doherty, M. J. et al. Vascular pericytes express osteogenic potential in vitro and in vivo. J. Bone Miner. Res. 13, 828–838 (1998).
Sacchetti, B. et al. Self-renewing osteoprogenitors in bone marrow sinusoids can organize a hematopoietic microenvironment. Cell 131, 324–336 (2007).
Xie, Y. et al. Detection of functional haematopoietic stem cell niche using real-time imaging. Nature 457, 97–101 (2009).
Lo Celso, C. et al. Live-animal tracking of individual haematopoietic stem/progenitor cells in their niche. Nature 457, 92–96 (2009).
Chan, C. K. et al. Endochondral ossification is required for haematopoietic stem-cell niche formation. Nature 457, 490–494 (2009).
Kode, A. et al. Leukaemogenesis induced by an activating β-catenin mutation in osteoblasts. Nature 506, 240–244 (2014).
Ding, L. & Morrison, S. J. Haematopoietic stem cells and early lymphoid progenitors occupy distinct bone marrow niches. Nature 495, 231–235 (2013).
Greenbaum, A. et al. CXCL12 in early mesenchymal progenitors is required for haematopoietic stem-cell maintenance. Nature 495, 227–230 (2013).
Van 't Veer, L. J. et al. Gene expression profiling predicts clinical outcome of breast cancer. Nature 415, 530–536 (2002).
Van de Vijver, M. J. et al. A gene-expression signature as a predictor of survival in breast cancer. N. Engl. J. Med. 347, 1999–2009 (2002).
Weigelt, B. et al. Gene expression profiles of primary breast tumors maintained in distant metastases. Proc. Natl Acad. Sci. USA 100, 15901–15905 (2003).
Kang, Y. et al. A multigenic program mediating breast cancer metastasis to bone. Cancer Cell 3, 537–549 (2003).
Zhang, X. H. et al. Latent bone metastasis in breast cancer tied to Src-dependent survival signals. Cancer Cell 16, 67–78 (2009).
Padua, D. et al. TGFβ primes breast tumors for lung metastasis seeding through angiopoietin-like 4. Cell 133, 66–77 (2008).
Zhang, X. H. et al. Selection of bone metastasis seeds by mesenchymal signals in the primary tumor stroma. Cell 154, 1060–1073 (2013).
Jung, Y. et al. Recruitment of mesenchymal stem cells into prostate tumours promotes metastasis. Nat. Commun. 4, 1795 (2013).
Bidwell, B. N. et al. Silencing of Irf7 pathways in breast cancer cells promotes bone metastasis through immune escape. Nat. Med. 18, 1224–1231 (2012).
Vecchi, M. et al. Breast cancer metastases are molecularly distinct from their primary tumors. Oncogene 27, 2148–2158 (2008).
Ki, D. H. et al. Whole genome analysis for liver metastasis gene signatures in colorectal cancer. Int. J. Cancer 121, 2005–2012 (2007).
Stoecklein, N. H. & Klein, C. A. Genetic disparity between primary tumours, disseminated tumour cells, and manifest metastasis. Int. J. Cancer 126, 589–598 (2010).
Klein, C. A. Parallel progression of primary tumours and metastases. Nat. Rev. Cancer 9, 302–312 (2009).
Hong, M. K. et al. Tracking the origins and drivers of subclonal metastatic expansion in prostate cancer. Nat. Commun. 6, 6605 (2015). A study using next-generation sequencing to define the origins of individual bone metastases in men with prostate cancer.
Gundem, G. et al. The evolutionary history of lethal metastatic prostate cancer. Nature 520, 353–357 (2015).
Butcher, D. T., Alliston, T. & Weaver, V. M. A tense situation: forcing tumour progression. Nat. Rev. Cancer 9, 108–122 (2009).
Paszek, M. J. & Weaver, V. M. The tension mounts: mechanics meets morphogenesis and malignancy. J. Mammary Gland Biol. Neoplasia 9, 325–342 (2004).
Guelcher, S. A., Dumas, J. Srinivasan, A., Didier, J. E. & Hollinger, J. O. Synthesis, mechanical properties, biocompatibility, and biodegradation of polyurethane networks from lysine polyisocyanates. Biomaterials 29, 1762–1775 (2008).
Johnson, R. W. et al. TGF-β promotion of Gli2-induced expression of parathyroid hormone-related protein, an important osteolytic factor in bone metastasis, is independent of canonical Hedgehog signaling. Cancer Res. 71, 822–831 (2011).
Ruppender, N. S. et al. Matrix rigidity induces osteolytic gene expression of metastatic breast cancer cells. PLoS ONE 5, e15451 (2010).
Muller, A. et al. Involvement of chemokine receptors in breast cancer metastasis. Nature 410, 50–56 (2001).
Taichman, R. S. et al. Use of the stromal cell-derived factor-1/CXCR4 pathway in prostate cancer metastasis to bone. Cancer Res. 62, 1832–1837 (2002).
Corcoran, K. E. et al. Mesenchymal stem cells in early entry of breast cancer into bone marrow. PLoS ONE 3, e2563 (2008).
Correa, D., Somoza, R. A., Lin, P., Schiemann, W. P. & Caplan, A. I. Mesenchymal stem cells regulate melanoma cancer cells extravasation to bone and liver at their perivascular niche. Int. J. Cancer 138, 417–427 (2015).
Loibl, M. et al. Direct cell–cell contact between mesenchymal stem cells and endothelial progenitor cells induces a pericyte-like phenotype in vitro. Biomed. Res. Int. 2014, 395781 (2014).
Parmo-Cabanas, M. et al. Integrin α4β1 involvement in stromal cell-derived factor-1α-promoted myeloma cell transendothelial migration and adhesion: role of cAMP and the actin cytoskeleton in adhesion. Exp. Cell Res. 294, 571–580 (2004).
Sanz-Rodriguez, F., Hidalgo, A. & Teixido, J. Chemokine stromal cell-derived factor-1α modulates VLA-4 integrin-mediated multiple myeloma cell adhesion to CS-1/fibronectin and VCAM-1. Blood 97, 346–351 (2001).
Abe, M., Hiura, K., Ozaki, S., Kido, S. & Matsumoto, T. Vicious cycle between myeloma cell binding to bone marrow stromal cells via VLA-4–VCAM-1 adhesion and macrophage inflammatory protein-1α and MIP-1β production. J. Bone Miner. Metab. 27, 16–23 (2009).
Choi, S. J. et al. Macrophage inflammatory protein 1-α is a potential osteoclast stimulatory factor in multiple myeloma. Blood 96, 671–675 (2000).
Oyajobi, B. O. et al. Dual effects of macrophage inflammatory protein-1α on osteolysis and tumor burden in the murine 5TGM1 model of myeloma bone disease. Blood 102, 311–319 (2003).
Azab, A. K. et al. CXCR4 inhibitor AMD3100 disrupts the interaction of multiple myeloma cells with the bone marrow microenvironment and enhances their sensitivity to therapy. Blood 113, 4341–4351 (2009).
Domanska, U. M. et al. CXCR4 inhibition with AMD3100 sensitizes prostate cancer to docetaxel chemotherapy. Neoplasia 14, 709–718 (2012).
Xiang, J. et al. CXCR4 protein epitope mimetic antagonist POL5551 disrupts metastasis and enhances chemotherapy effect in triple-negative breast cancer. Mol. Cancer Ther. 14, 2473–2485 (2015).
Liapis, H., Flath, A. & Kitazawa, S. Integrin αVβ3 expression by bone-residing breast cancer metastases. Diagn. Mol. Pathol. 5, 127–135 (1996).
Carlson, C. S. et al. Immunolocalization of noncollagenous bone matrix proteins in lumbar vertebrae from intact and surgically menopausal cynomolgus monkeys. J. Bone Miner. Res. 8, 71–81 (1993).
Grzesik, W. J. & Robey, P. G. Bone matrix RGD glycoproteins: immunolocalization and interaction with human primary osteoblastic bone cells in vitro. J. Bone Miner. Res. 9, 487–496 (1994).
Wang, H. et al. The osteogenic niche promotes early-stage bone colonization of disseminated breast cancer cells. Cancer Cell 27, 193–210 (2015).
Beausoleil, M. S., Schulze, E. B., Goodale, D., Postenka, C. O. & Allan, A. L. Deletion of the thrombin cleavage domain of osteopontin mediates breast cancer cell adhesion, proteolytic activity, tumorgenicity, and metastasis. BMC Cancer 11, 25 (2011).
Klevesath, M. B. et al. Patterns of metastatic spread in early breast cancer. Breast 22, 449–454 (2013).
Sung, V., Stubbs, J. T. 3rd, Fisher, L., Aaron, A. D. & Thompson, E. W. Bone sialoprotein supports breast cancer cell adhesion proliferation and migration through differential usage of the αvβ3 and αvβ5 integrins. J. Cell. Physiol. 176, 482–494 (1998).
Adwan, H. et al. Decreased levels of osteopontin and bone sialoprotein II are correlated with reduced proliferation, colony formation, and migration of GFP-MDA-MB-231 cells. Int. J. Oncol. 24, 1235–1244 (2004).
Wang, J. et al. BSP gene silencing inhibits migration, invasion, and bone metastasis of MDA-MB-231BO human breast cancer cells. PLoS ONE 8, e62936 (2013).
Uchiyama, H., Barut, B. A., Chauhan, D., Cannistra, S. A. & Anderson, K. C. Characterization of adhesion molecules on human myeloma cell lines. Blood 80, 2306–2314 (1992).
Mori, Y. et al. Anti-α4 integrin antibody suppresses the development of multiple myeloma and associated osteoclastic osteolysis. Blood 104, 2149–2154 (2004).
Jones, D. H. et al. Regulation of cancer cell migration and bone metastasis by RANKL. Nature 440, 692–696 (2006).
Thomas, R. J. et al. Breast cancer cells interact with osteoblasts to support osteoclast formation. Endocrinology 140, 4451–4458 (1999).
Nannuru, K. C. et al. Enhanced expression and shedding of receptor activator of NF-κB ligand during tumor-bone interaction potentiates mammary tumor-induced osteolysis. Clin. Exp. Metastasis 26, 797–808 (2009).
Mundy, G. R. Metastasis to bone: causes, consequences and therapeutic opportunities. Nat. Rev. Cancer 2, 584–593 (2002).
Pearse, R. N. et al. Multiple myeloma disrupts the TRANCE/osteoprotegerin cytokine axis to trigger bone destruction and promote tumor progression. Proc. Natl Acad. Sci. USA 98, 11581–11586 (2001).
Zhang, J. et al. Osteoprotegerin inhibits prostate cancer-induced osteoclastogenesis and prevents prostate tumor growth in the bone. J. Clin. Invest. 107, 1235–1244 (2001).
Croucher, P. I. et al. Osteoprotegerin inhibits the development of osteolytic bone disease in multiple myeloma. Blood 98, 3534–3540 (2001).
Canon, J. R. et al. Inhibition of RANKL blocks skeletal tumor progression and improves survival in a mouse model of breast cancer bone metastasis. Clin. Exp. Metastasis 25, 119–129 (2008).
Armstrong, A. P. et al. RANKL acts directly on RANK-expressing prostate tumor cells and mediates migration and expression of tumor metastasis genes. Prostate 68, 92–104 (2008).
Campbell, J. P. et al. Stimulation of host bone marrow stromal cells by sympathetic nerves promotes breast cancer bone metastasis in mice. PLoS Biol. 10, e1001363 (2012).
Shiozawa, Y. et al. Human prostate cancer metastases target the hematopoietic stem cell niche to establish footholds in mouse bone marrow. J. Clin. Invest. 121, 1298–1312 (2011). A key study highlighting the potential of colonizing tumour cells to hijack the HSC niche in bone.
Vanderkerken, K. et al. Selective initial in vivo homing pattern of 5T2 multiple myeloma cells in the C57BL/KalwRij mouse. Br. J. Cancer 82, 953–959 (2000).
Chen, Z., Orlowski, R. Z., Wang, M., Kwak, L. & McCarty, N. Osteoblastic niche supports the growth of quiescent multiple myeloma cells. Blood 123, 2204–2208 (2014).
Karadag, A., Oyajobi, B. O., Apperley, J. F., Russell, R. G. G. & Croucher, P. I. Human myeloma cells promote the production of interleukin-6 by primary human osteoblasts. Br. J. Haematol. 108, 383–390 (2000).
Ro, T. B. et al. Bone morphogenetic protein-5, -6 and -7 inhibit growth and induce apoptosis in human myeloma cells. Oncogene 23, 3024–3032 (2004).
Hjertner, O. et al. Bone morphogenetic protein-4 inhibits proliferation and induces apoptosis of multiple myeloma cells. Blood 97, 516–522 (2001).
Kawamura, C. et al. Bone morphogenetic protein-2 induces apoptosis in human myeloma cells with modulation of STAT3. Blood 96, 2005–2011 (2000).
Holien, T. & Sundan, A. The role of bone morphogenetic proteins in myeloma cell survival. Cytokine Growth Factor Rev. 25, 343–350 (2014).
Dosen, G. et al. Wnt expression and canonical Wnt signaling in human bone marrow B lymphopoiesis. BMC Immunol. 7, 13 (2006).
Li, X., Pennisi, A. & Yaccoby, S. Role of decorin in the antimyeloma effects of osteoblasts. Blood 112, 159–168 (2008).
D'Souza, S. et al. Annexin II interactions with the annexin II receptor enhance multiple myeloma cell adhesion and growth in the bone marrow microenvironment. Blood 119, 1888–1896 (2012).
Holen, I., Croucher, P. I., Hamdy, F. C. & Eaton, C. L. Osteoprotegerin (OPG) is a survival factor for human prostate cancer cells. Cancer Res. 62, 1619–1623 (2002).
Shipman, C. M. & Croucher, P. I. Osteoprotegerin is a soluble decoy receptor for tumour necrosis factor-related apoptosis-inducing ligand/Apo2 ligand and can function as a paracrine survival factor for human myeloma cells. Cancer Res. 63, 912–916 (2003).
Nyambo, R. et al. Human bone marrow stromal cells protect prostate cancer cells from TRAIL-induced apoptosis. J. Bone Miner. Res. 19, 1712–1721 (2004).
Neville-Webbe, H. L. et al. Osteoprotegerin (OPG) produced by bone marrow stromal cells protects breast cancer cells from TRAIL-induced apoptosis. Breast Cancer Res. Treat. 86, 269–279 (2004).
Vitovski, S., Phillips, J. S., Sayers, J. & Croucher, P. I. Investigating the interaction between osteoprotegerin and receptor activator of NF-κB or tumor necrosis factor-related apoptosis-inducing ligand: evidence for a pivotal role for osteoprotegerin in regulating two distinct pathways. J. Biol. Chem. 282, 31601–31609 (2007).
Schlimok, G. et al. Micrometastatic cancer cells in bone marrow: in vitro detection with anti-cytokeratin and in vivo labeling with anti-17-1A monoclonal antibodies. Proc. Natl Acad. Sci. USA 84, 8672–8676 (1987).
Cote, R. J. et al. Monoclonal antibodies detect occult breast carcinoma metastases in the bone marrow of patients with early stage disease. Am. J. Surg. Pathol. 12, 333–340 (1988).
Pantel, K. et al. Frequency and prognostic significance of isolated tumour cells in bone marrow of patients with non-small-cell lung cancer without overt metastases. Lancet 347, 649–653 (1996).
Engel, J. et al. The process of metastasisation for breast cancer. Eur. J. Cancer 39, 1794–1806 (2003).
Husemann, Y. et al. Systemic spread is an early step in breast cancer. Cancer Cell 13, 58–68 (2008).
Shiozawa, Y. et al. GAS6/AXL axis regulates prostate cancer invasion, proliferation, and survival in the bone marrow niche. Neoplasia 12, 116–127 (2010). Evidence that AXL may be a key molecule in controlling prostate cancer cell survival in the skeleton.
Jung, Y. et al. Prevalence of prostate cancer metastases after intravenous inoculation provides clues into the molecular basis of dormancy in the bone marrow microenvironment. Neoplasia 14, 429–439 (2012).
Taichman, R. S. et al. GAS6 receptor status is associated with dormancy and bone metastatic tumor formation. PLoS ONE 8, e61873 (2013).
Mishra, A. et al. Hypoxia stabilizes GAS6/Axl signaling in metastatic prostate cancer. Mol. Cancer Res. 10, 703–712 (2012).
Kusumbe, A. P., Ramasamy, S. K. & Adams, R. H. Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone. Nature 507, 323–328 (2014).
Spencer, J. A. et al. Direct measurement of local oxygen concentration in the bone marrow of live animals. Nature 508, 269–273 (2014).
Ghajar, C. M. Metastasis prevention by targeting the dormant niche. Nat. Rev. Cancer 15, 238–247 (2015).
Ghajar, C. M. et al. The perivascular niche regulates breast tumour dormancy. Nat. Cell Biol. 15, 807–817 (2013).
Horiuchi, K. et al. Identification and characterization of a novel protein, periostin, with restricted expression to periosteum and periodontal ligament and increased expression by transforming growth factor β. J. Bone Miner. Res. 14, 1239–1249 (1999).
Merle, B., Bouet, G., Rousseau, J. C., Bertholon, C. & Garnero, P. Periostin and transforming growth factor β-induced protein (TGFβIp) are both expressed by osteoblasts and osteoclasts. Cell Biol. Int. 38, 398–404 (2014).
Malanchi, I. et al. Interactions between cancer stem cells and their niche govern metastatic colonization. Nature 481, 85–89 (2012).
Lee, Y. J. et al. Periostin-binding DNA aptamer inhibits breast cancer growth and metastasis. Mol. Ther. 21, 1004–1013 (2013).
Yin, J. J. et al. TGF-β signaling blockade inhibits PTHrP secretion by breast cancer cells and bone metastases development. J. Clin. Invest. 103, 197–206 (1999).
Kakonen, S. M. et al. Transforming growth factor-β stimulates parathyroid hormone-related protein and osteolytic metastases via Smad and mitogen-activated protein kinase signaling pathways. J. Biol. Chem. 277, 24571–24578 (2002).
Fournier, P. G. et al. The TGF-β signaling regulator PMEPA1 suppresses prostate cancer metastases to bone. Cancer Cell 27, 809–821 (2015).
Pettit, A. R., Chang, M. K., Hume, D. A. & Raggatt, L. J. Osteal macrophages: a new twist on coupling during bone dynamics. Bone 43, 976–982 (2008).
Parfitt, A. M. Osteonal and hemi-osteonal remodeling: the spatial and temporal framework for signal traffic in adult human bone. J. Cell Biochem. 55, 273–286 (1994).
Lu, X. et al. VCAM-1 promotes osteolytic expansion of indolent bone micrometastasis of breast cancer by engaging α4β1-positive osteoclast progenitors. Cancer Cell 20, 701–714 (2011).
Chen, Q., Zhang, X. H. & Massague, J. Macrophage binding to receptor VCAM-1 transmits survival signals in breast cancer cells that invade the lungs. Cancer Cell 20, 538–549 (2011).
Hynes, R. O. Metastatic cells will take any help they can get. Cancer Cell 20, 689–690 (2011).
Giancotti, F. G. Mechanisms governing metastatic dormancy and reactivation. Cell 155, 750–764 (2013).
Guise, T. A. et al. Evidence for a causal role of parathyroid hormone-related protein in the pathogenesis of human breast cancer-mediated osteolysis. J. Clin. Invest. 98, 1544–1549 (1996).
Zheng, Y. et al. Bone resorption increases tumour growth in a mouse model of osteosclerotic breast cancer metastasis. Clin. Exp. Metastasis 25, 559–567 (2008).
Zheng, Y. et al. Vitamin D deficiency promotes prostate cancer growth in bone. Prostate 71, 1012–1021 (2011).
Ooi, L. L. et al. Vitamin D deficiency promotes growth of MCF-7 human breast cancer in a rodent model of osteosclerotic bone metastasis. Bone 47, 795–803 (2010).
Ooi, L. L. et al. Vitamin D deficiency promotes human breast cancer growth in a murine model of bone metastasis. Cancer Res. 70, 1835–1844 (2010).
Corey, E. et al. Osteoprotegerin in prostate cancer bone metastasis. Cancer Res. 65, 1710–1718 (2005).
Corey, E. et al. Zoledronic acid exhibits inhibitory effects on osteoblastic and osteolytic metastases of prostate cancer. Clin. Cancer Res. 9, 295–306 (2003).
Kiefer, J. A. et al. The effect of osteoprotegerin administration on the intra-tibial growth of the osteoblastic LuCaP 23.1 prostate cancer xenograft. Clin. Exp. Metastasis 21, 381–387 (2004).
Ottewell, P. D. et al. OPG-Fc inhibits ovariectomy-induced growth of disseminated breast cancer cells in bone. Int. J. Cancer 137, 968–977 (2015).
Libouban, H., Moreau, M. F., Basle, M. F., Bataille, R. & Chappard, D. Increased bone remodeling due to ovariectomy dramatically increases tumoral growth in the 5T2 multiple myeloma mouse model. Bone 33, 283–292 (2003).
Croucher, P. I. et al. Zoledronic acid treatment of 5T2MM-bearing mice inhibits the development of myeloma bone disease: evidence for decreased osteolysis, tumor burden and angiogenesis, and increased survival. J. Bone Miner. Res. 18, 482–492 (2003).
Vanderkerken, K. et al. Recombinant osteoprotegerin decreases tumor burden and increases survival in a murine model of multiple myeloma. Cancer Res. 63, 287–289 (2003).
Yaccoby, S. et al. Myeloma interacts with the bone marrow microenvironment to induce osteoclastogenesis and is dependent on osteoclast activity. Br. J. Haematol. 116, 278–290 (2002).
Oyajobi, B. O. & Mundy, G. R. Receptor activator of NF-κB ligand, macrophage inflammatory protein-1α, and the proteasome: novel therapeutic targets in myeloma. Cancer 97, 813–817 (2003).
Kollet, O. et al. Osteoclasts degrade endosteal components and promote mobilization of hematopoietic progenitor cells. Nat. Med. 12, 657–664 (2006).
Bataille, R., Chappard, D. & Basle, M. F. Quantifiable excess of bone resorption in monoclonal gammopathy is an early symptom of malignancy: a prospective study of 87 bone biopsies. Blood 87, 4762–4769 (1996).
Lund, T., Abildgaard, N., Andersen, T. L., Delaisse, J. M. & Plesner, T. Multiple myeloma: changes in serum C-terminal telopeptide of collagen type I and bone-specific alkaline phosphatase can be used in daily practice to detect imminent osteolysis. Eur. J. Haematol. 84, 412–420 (2010).
Michaelson, M. D., Marujo, R. M. & Smith, M. R. Contribution of androgen deprivation therapy to elevated osteoclast activity in men with metastatic prostate cancer. Clin. Cancer Res. 10, 2705–2708 (2004).
Smith, M. R. et al. Pamidronate to prevent bone loss during androgen-deprivation therapy for prostate cancer. N. Engl. J. Med. 345, 948–955 (2001).
Smith, M. R. et al. Randomized controlled trial of zoledronic acid to prevent bone loss in men receiving androgen deprivation therapy for nonmetastatic prostate cancer. J. Urol. 169, 2008–2012 (2003).
Greenspan, S. L., Nelson, J. B., Trump, D. L. & Resnick, N. M. Effect of once-weekly oral alendronate on bone loss in men receiving androgen deprivation therapy for prostate cancer: a randomized trial. Ann. Intern. Med. 146, 416–424 (2007).
Klotz, L. H. et al. A phase 3, double-blind, randomised, parallel-group, placebo-controlled study of oral weekly alendronate for the prevention of androgen deprivation bone loss in nonmetastatic prostate cancer: the Cancer and Osteoporosis Research with Alendronate and Leuprolide (CORAL) study. Eur. Urol. 63, 927–935 (2013).
Smith, M. R. et al. Denosumab in men receiving androgen-deprivation therapy for prostate cancer. N. Engl. J. Med. 361, 745–755 (2009).
Smith, M. R. et al. Denosumab and changes in bone turnover markers during androgen deprivation therapy for prostate cancer. J. Bone Miner. Res. 26, 2827–2833 (2011).
Nguyen, P. L. et al. Adverse effects of androgen deprivation therapy and strategies to mitigate them. Eur. Urol. 67, 825–836 (2015).
Perez, E. A. & Weilbaecher, K. Aromatase inhibitors and bone loss. Oncol. (Williston Park) 20, 1029–1039; discussion 1039–1048 (2006).
Llombart, A. et al. Immediate administration of zoledronic acid reduces aromatase inhibitor-associated bone loss in postmenopausal women with early breast cancer: 12-month analysis of the E-ZO-FAST trial. Clin. Breast Cancer 12, 40–48 (2012).
Silbermann, R. & Roodman, G. D. Bone effects of cancer therapies: pros and cons. Curr. Opin. Support. Palliat. Care 5, 251–257 (2011).
Oyajobi, B. O. et al. Stimulation of new bone formation by the proteasome inhibitor, bortezomib: implications for myeloma bone disease. Br. J. Haematol. 139, 434–438 (2007).
Giuliani, N. et al. The proteasome inhibitor bortezomib affects osteoblast differentiation in vitro and in vivo in multiple myeloma patients. Blood 110, 334–338 (2007).
Pennisi, A. et al. The proteasome inhibitor, bortezomib suppresses primary myeloma and stimulates bone formation in myelomatous and nonmyelomatous bones in vivo. Am. J. Hematol. 84, 6–14 (2009).
Garcia-Gomez, A. et al. Preclinical activity of the oral proteasome inhibitor MLN9708 in myeloma bone disease. Clin. Cancer Res. 20, 1542–1554 (2014).
Scullen, T. et al. Lenalidomide in combination with an activin A-neutralizing antibody: preclinical rationale for a novel anti-myeloma strategy. Leukemia 27, 1715–1721 (2013).
Bolomsky, A. et al. Immunomodulatory drugs thalidomide and lenalidomide affect osteoblast differentiation of human bone marrow stromal cells in vitro. Exp. Hematol. 42, 516–525 (2014).
Breitkreutz, I. et al. Lenalidomide inhibits osteoclastogenesis, survival factors and bone-remodeling markers in multiple myeloma. Leukemia 22, 1925–1932 (2008).
Munemasa, S. et al. Osteoprogenitor differentiation is not affected by immunomodulatory thalidomide analogs but is promoted by low bortezomib concentration, while both agents suppress osteoclast differentiation. Int. J. Oncol. 33, 129–136 (2008).
Bolzoni, M. et al. Immunomodulatory drugs lenalidomide and pomalidomide inhibit multiple myeloma-induced osteoclast formation and the RANKL/OPG ratio in the myeloma microenvironment targeting the expression of adhesion molecules. Exp. Hematol. 41, 387–397 (2013).
Friedlaender, G. E., Tross, R. B., Doganis, A. C., Kirkwood, J. M. & Baron, R. Effects of chemotherapeutic agents on bone. I. Short-term methotrexate and doxorubicin (adriamycin) treatment in a rat model. J. Bone Joint Surg. Am. 66, 602–607 (1984).
Van Leeuwen, B. L. et al. Effect of single chemotherapeutic agents on the growing skeleton of the rat. Ann. Oncol. 11, 1121–1126 (2000).
Rana, T., Chakrabarti, A., Freeman, M. & Biswas, S. Doxorubicin-mediated bone loss in breast cancer bone metastases is driven by an interplay between oxidative stress and induction of TGFβ. PLoS ONE 8, e78043 (2013).
Van der Kraan, A. G. et al. HSP90 inhibitors enhance differentiation and MITF (microphthalmia transcription factor) activity in osteoclast progenitors. Biochem. J. 451, 235–244 (2013).
Chai, R. C. et al. Molecular stress-inducing compounds increase osteoclast formation in a heat shock factor 1 protein-dependent manner. J. Biol. Chem. 289, 13602–13614 (2014).
King, T. J. et al. Methotrexate chemotherapy promotes osteoclast formation in the long bone of rats via increased pro-inflammatory cytokines and enhanced NF-κB activation. Am. J. Pathol. 181, 121–129 (2012).
Early Breast Cancer Trialists' Collaborative Group. Adjuvant bisphosphonate treatment in early breast cancer: meta-analyses of individual patient data from randomised trials. Lancet 386, 1353–1361 (2015). A meta-analysis demonstrating the potential survival benefit of early bisphosphonate treatment in individuals with breast cancer.
Gnant, M. et al. Adjuvant denosumab in breast cancer (ABCSG-18): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet 386, 433–443 (2015).
Fizazi, K. et al. Denosumab versus zoledronic acid for treatment of bone metastases in men with castration-resistant prostate cancer: a randomised, double-blind study. Lancet 377, 813–822 (2011).
Saad, F. et al. A randomized, placebo-controlled trial of zoledronic acid in patients with hormone-refractory metastatic prostate carcinoma. J. Natl Cancer Inst. 94, 1458–1468 (2002).
Morgan, G. J. et al. First-line treatment with zoledronic acid as compared with clodronic acid in multiple myeloma (MRC Myeloma IX): a randomised controlled trial. Lancet 376, 1989–1999 (2010).
Morgan, G. J. et al. Long-term follow-up of MRC Myeloma IX trial: survival outcomes with bisphosphonate and thalidomide treatment. Clin. Cancer Res. 19, 6030–6038 (2013).
Coleman, R., Gnant, M., Morgan, G. & Clezardin, P. Effects of bone-targeted agents on cancer progression and mortality. J. Natl Cancer Inst. 104, 1059–1067 (2012).
Coleman, R. E. et al. Breast-cancer adjuvant therapy with zoledronic acid. N. Engl. J. Med. 365, 1396–1405 (2011).
Smith, M. R. et al. Denosumab and bone-metastasis-free survival in men with castration-resistant prostate cancer: results of a phase 3, randomised, placebo-controlled trial. Lancet 379, 39–46 (2012).
Nozawa, M. et al. Phase II trial of zoledronic acid combined with androgen-deprivation therapy for treatment-naive prostate cancer with bone metastasis. Int. J. Clin. Oncol. 19, 693–701 (2014).
Okegawa, T. et al. Zoledronic acid improves clinical outcomes in patients with bone metastatic hormone-naive prostate cancer in a multicenter clinical trial. Anticancer Res. 34, 4415–4420 (2014).
Smith, M. R. et al. Randomized controlled trial of early zoledronic acid in men with castration-sensitive prostate cancer and bone metastases: results of CALGB 90202 (alliance). J. Clin. Oncol. 32, 1143–1150 (2014).
Bone, H. G. et al. Odanacatib, a cathepsin-K inhibitor for osteoporosis: a two-year study in postmenopausal women with low bone density. J. Bone Miner. Res. 25, 937–947 (2010).
Lotinun, S. et al. Osteoclast-specific cathepsin K deletion stimulates S1P-dependent bone formation. J. Clin. Invest. 123, 666–681 (2013).
Duong, L. T., Wesolowski, G. A., Leung, P., Oballa, R. & Pickarski, M. Efficacy of a cathepsin K inhibitor in a preclinical model for prevention and treatment of breast cancer bone metastasis. Mol. Cancer Ther. 13, 2898–2909 (2014).
Power, C. A. et al. A novel model of bone-metastatic prostate cancer in immunocompetent mice. Prostate 69, 1613–1623 (2009).
Godebu, E. et al. PCSD1, a new patient-derived model of bone metastatic prostate cancer, is castrate-resistant in the bone-niche. J. Transl Med. 12, 275 (2014).
Valta, M. P. et al. Development of a realistic in vivo bone metastasis model of human renal cell carcinoma. Clin. Exp. Metastasis 31, 573–584 (2014).
Sipkins, D. A. et al. In vivo imaging of specialized bone marrow endothelial microdomains for tumour engraftment. Nature 435, 969–973 (2005).
Runnels, J. M. et al. Optical techniques for tracking multiple myeloma engraftment, growth, and response to therapy. J. Biomed. Opt. 16, 011006 (2011).
Conway, J. R., Carragher, N. O. & Timpson, P. Developments in preclinical cancer imaging: innovating the discovery of therapeutics. Nat. Rev. Cancer 14, 314–328 (2014).
Chtanova, T. et al. Real-time interactive two-photon photoconversion of recirculating lymphocytes for discontinuous cell tracking in live adult mice. J. Biophoton. 7, 425–433 (2014).
Acknowledgements
P.I.C. acknowledges the support of Mrs Janice Gibson and the Ernest Heine Family Foundation, the Cancer Council New South Wales, the Prostate Cancer Foundation of Australia, the Wellcome Trust, Leukaemia and Lymphoma Research and Cancer Research UK. T.J.M. acknowledges research support from the National Health and Medical Research Council (Australia) and the Victorian Government OIS Program.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Bone resorption
-
The process by which bone-resorbing cells break down bone.
- Osteoblast lineage
-
The lineage of cells responsible for making new bone.
- Osteoprogenitors
-
Cells that are derived from skeletal stem cells with the ability to transition through the osteogenic lineage.
- Preosteoblasts
-
Cells that are committed to forming functional bone-forming osteoblasts.
- Mineralization
-
The process by which mineral, including hydroxyapatite, is precipitated and deposited on a collagen matrix.
- Pericytes
-
Cells located adjacent to endothelial cells of capillaries.
- Osteomacs
-
Macrophages associated with the bone surface.
- Osteogenic lineage
-
The lineage that gives rise to cells that can make new bone; these include bone lining cells, osteoblasts and osteocytes.
- Basic multicellular units
-
The cells responsible for the coordinated removal and replacement of a quantum of bone in bone remodelling.
- Hyperparathyroidism
-
A disorder of the parathyroid gland resulting in increased concentrations of circulating parathyroid hormone.
- Bone marrow stromal progenitor cells
-
A population of stromal cells found in the bone marrow that can differentiate into a range of cell types including cells of the osteogenic lineage.
- Reversal phase
-
The phase of bone remodelling between bone formation and bone resorption.
- Endochondral ossification
-
One of the essential processes by which bone is formed via a cartilage cell precursor.
- Canaliculi
-
The microscopic canals that link the lacunae occupied by osteocytes.
- Innate immune system
-
The cells and mechanisms present in readiness to fight microorganisms.
- Elastic modulus
-
A measure of the elasticity or stiffness of a material.
- Metaphyseal region
-
The section of a long bone between the diaphysis (shaft) and epiphysis (end).
- Diaphyseal region
-
The midsection or shaft of a long bone.
- Metastasis initiating cells
-
Cancer cells with the ability to give rise to overt metastasis in distant organs.
- Cancer stem cells
-
Cancer cells with self-renewal and tumour-initiating abilities.
- Monoclonal gammopathy of unknown significance
-
(MGUS). A disorder characterized by the presence in the blood of an abnormal protein that is produced by plasma cells that in some individuals can transition to multiple myeloma.
- Perivascular niche
-
A specialized microenvironment located adjacent to blood vessels that supports the long-term survival of specific cell types, including stem or progenitor cells.
- Calvaria
-
The bones of the skull (cranium) that protect the brain.
- Two-photon photoconversion
-
The use of two-photon excitation light to convert the emission spectra of a photoconvertible fluorescent probe from one colour (for example, green) to another (for example, red).
Rights and permissions
About this article
Cite this article
Croucher, P., McDonald, M. & Martin, T. Bone metastasis: the importance of the neighbourhood. Nat Rev Cancer 16, 373–386 (2016). https://doi.org/10.1038/nrc.2016.44
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc.2016.44
This article is cited by
-
Procoxacin bidirectionally inhibits osteoblastic and osteoclastic activity in bone and suppresses bone metastasis of prostate cancer
Journal of Experimental & Clinical Cancer Research (2023)
-
Skull metastasis is a poor prognostic factor for prostate cancer patients with bone metastasis: a retrospective study based on a Chinese population
BMC Urology (2023)
-
Myeloid-like tumor hybrid cells in bone marrow promote progression of prostate cancer bone metastasis
Journal of Hematology & Oncology (2023)
-
Bone-matrix mineralization dampens integrin-mediated mechanosignalling and metastatic progression in breast cancer
Nature Biomedical Engineering (2023)
-
The role of integrin family in bone metabolism and tumor bone metastasis
Cell Death Discovery (2023)