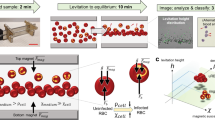
Abstract
The mechanical retention of rigid erythrocytes in the spleen is central in major hematological diseases such as hereditary spherocytosis, sickle-cell disease and malaria. Here, we describe the use of microsphiltration (microsphere filtration) to assess erythrocyte deformability in hundreds to thousands of samples in parallel, by filtering them through microsphere layers in 384-well plates adapted for the discovery of compounds that stiffen Plasmodium falciparum gametocytes, with the aim of interrupting malaria transmission. Compound-exposed gametocytes are loaded into microsphiltration plates, filtered and then transferred to imaging plates for analysis. High-content imaging detects viable gametocytes upstream and downstream from filters and quantifies spleen-like retention. This screening assay takes 3–4 d. Unlike currently available methods used to assess red blood cell (RBC) deformability, microsphiltration enables high-throughput pharmacological screening (tens of thousands of compounds tested in a matter of months) and involves a cell mechanical challenge that induces a physiologically relevant dumbbell-shape deformation. It therefore directly assesses the ability of RBCs to cross inter-endothelial splenic slits in vivo. This protocol has potential applications in quality control for transfusion and in determination of phenotypic markers of erythrocytes in hematological diseases.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Pivkin, I.V. et al. Biomechanics of red blood cells in human spleen and consequences for physiology and disease. Proc. Natl. Acad. Sci. USA 113, 7804–7809 (2016).
Brousse, V., Buffet, P. & Rees, D. The spleen and sickle cell disease: the sick(led) spleen. Br. J. Haematol. 166, 165–176 (2014).
Oliveira, M.C.L.A. et al. Clinical course of 63 children with hereditary spherocytosis: a retrospective study. Rev. Bras Hematol. Hemoter. 34, 9–13 (2012).
Dondorp, A.M. et al. Red cell deformability, splenic function and anaemia in thalassaemia. Br. J. Haematol. 105, 505–508 (1999).
Dinkla, S. et al. Inflammation-associated changes in lipid composition and the organization of the erythrocyte membrane. BBA Clin. 5, 186–192 (2016).
Badr, B.M. et al. Increased levels of type 1 interferon in a type 1 diabetic mouse model induce the elimination of B cells from the periphery by apoptosis and increase their retention in the spleen. Cell. Physiol. Biochem. 35, 137–147 (2015).
Zaets, S.B. et al. Burn-induced red blood cell deformability and shape changes are modulated by sex hormones. Am. J. Surg. 186, 540–546 (2003).
Buffet, P.A., Safeukui, I., Milon, G., Mercereau-Puijalon, O. & David, P.H. Retention of erythrocytes in the spleen: a double-edged process in human malaria. Curr. Opin. Hematol. 16, 157–164 (2009).
Lavazec, C. et al. Microsphiltration: a microsphere matrix to explore erythrocyte deformability. Methods Mol. Biol. 923, 291–297 (2013).
Deplaine, G. et al. The sensing of poorly deformable red blood cells by the human spleen can be mimicked in vitro. Blood 117, e88–e95 (2011).
Ndour, P.A. et al. Role of the spleen in human malaria. in Encyclopedia of Malaria (eds. Hommel M. & Kremsner P.G.) 1–24 (Springer, 2015).
Buffet, P.A. et al. Ex vivo perfusion of human spleens maintains clearing and processing functions. Blood 107, 3745–3752 (2006).
Dearnley, M. et al. Reversible host cell remodeling underpins deformability changes in malaria parasite sexual blood stages. Proc. Natl. Acad. Sci. USA 113, 4800–4805 (2016).
Tibúrcio, M. et al. A switch in infected erythrocyte deformability at the maturation and blood circulation of Plasmodium falciparum transmission stages. Blood 119, e172–e180 (2012).
Naissant, B. et al. Plasmodium falciparum STEVOR phosphorylation regulates host erythrocyte deformability enabling malaria parasite transmission. Blood 127, e42–e53 (2016).
Ramdani, G. et al. cAMP-signalling regulates gametocyte-infected erythrocyte deformability required for malaria parasite transmission. PLoS Pathog. 11, e1004815 (2015).
Sanyal, S. et al. Plasmodium falciparum STEVOR proteins impact erythrocyte mechanical properties. Blood 119, e1–e8 (2012).
Safeukui, I. et al. Surface area loss and increased sphericity account for the splenic entrapment of subpopulations of Plasmodium falciparum ring-infected erythrocytes. PLoS One 8, e60150 (2013).
Ndour, P.A. et al. Plasmodium falciparum clearance is rapid and pitting independent in immune Malian children treated with artesunate for malaria. J. Infect. Dis. 211, 290–297 (2015).
Diakité, S.A.S. et al. Stage-dependent fate of Plasmodium falciparum-infected red blood cells in the spleen and sickle-cell trait-related protection against malaria. Malar. J. 15, 482 (2016).
Svelc, T. & Svetina, S. Stress-free state of the red blood cell membrane and the deformation of its skeleton. Cell. Mol. Biol. Lett. 17, 217–227 (2012).
Agrawal, R. et al. Assessment of red blood cell deformability in type 2 diabetes mellitus and diabetic retinopathy by dual optical tweezers stretching technique. Sci. Rep. 6, 5873 (2016).
Rabai, M. et al. Deformability analysis of sickle blood using ektacytometry. Biorheology 51, 159–170 (2014).
Picot, J. et al. A biomimetic microfluidic chip to study the circulation and mechanical retention of red blood cells in the spleen. Am. J. Hematol. 90, 339–345 (2015).
Rigat-Brugarolas, L.G. et al. A functional microengineered model of the human splenon-on-a-chip. Lab Chip 14, 1715–1724 (2014).
Gambhire, P. et al. High aspect ratio sub-micrometer channels using wet etching: application to the dynamics of red blood cell transiting through biomimetic splenic slits. Small http://dx.doi.org/10.1002/smll.201700967 (2017).
Tomaiuolo, G. Biomechanical properties of red blood cells in health and disease towards microfluidics. Biomicrofluidics 8, 051501 (2014).
Duez, J. et al. Splenic retention of Plasmodium falciparum gametocytes to block the transmission of malaria. Antimicrob. Agents Chemother. 59, 4206–4214 (2015).
Henry, B. et al. Red blood cell deformability, age, ethnicity and susceptibility to malaria in Africa. Blood 128, 2441–2441 (2016).
Duez, J. et al. Mechanical clearance of red blood cells by the human spleen: potential therapeutic applications of a biomimetic RBC filtration method. Transfus. Clin. Biol. 22, 151–157 (2015).
Roussel, C. et al. Spherocytic shift of red blood cells during storage provides a quantitative whole cell-based marker of the storage lesion. Transfusion 57, 1007–1018 (2017).
Lucantoni, L. & Avery, V. Whole-cell in vitro screening for gametocytocidal compounds. Future Med. Chem. 4, 2337–2360 (2012).
Zhang, W. et al. Microfluidics separation reveals the stem-cell–like deformability of tumor-initiating cells. Proc. Natl. Acad. Sci. USA 109, 18707–18712 (2012).
Chen, J. et al. Efficient extravasation of tumor-repopulating cells depends on cell deformability. Sci. Rep. 6, 19304 (2016).
Allard, W.J. et al. Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin. Cancer Res. 10, 6897–6904 (2004).
Nishino, M. et al. Serial changes in leukocyte deformability and whole blood rheology in patients with sepsis or trauma. J. Trauma 59, 1425–1431 (2005).
Inoue, Y. et al. A neutrophil elastase inhibitor, sivelestat, improves leukocyte deformability in patients with acute lung injury. J. Trauma 60, 936–943 discussion 943 (2006).
Delves, M.J. et al. Routine in vitro culture of P. falciparum gametocytes to evaluate novel transmission-blocking interventions. Nat. Protoc. 11, 1668–1680 (2016).
Duffy, S. & Avery, V.M. Identification of inhibitors of Plasmodium falciparum gametocyte development. Malar. J. 12, 408 (2013).
Sosa, J.M., Nielsen, N.D., Vignes, S.M., Chen, T.G. & Shevkoplyas, S.S. The relationship between red blood cell deformability metrics and perfusion of an artificial microvascular network. Clin. Hemorheol. Microcirc. 57, 275–289 (2014).
Zhang, R. et al. A basis for rapid clearance of circulating ring-stage malaria parasites by the spiroindolone KAE609. J. Infect. Dis. 213, 100–104 (2016).
Duffy, S., Loganathan, S., Holleran, J.P. & Avery, V.M. Large-scale production of Plasmodium falciparum gametocytes for malaria drug discovery. Nat. Protoc. 11, 976–992 (2016).
Acknowledgements
We thank the Bill & Melinda Gates Foundation (BMGF; grant OPP1123683), GlaxoSmithKline's (GSK's) Tres Cantos and the GSK Tres Cantos Open Lab Foundation (TCOLF) for funding this project. We gratefully acknowledge the Human Resources Department of INTS, including C. Pille, for the administrative support provided. We are indebted to O. Vandal from BMGF for the constant trust he placed in this HTS project. We express our sincere gratitude to S. Ruchaud for writing support. We thank HRA Pharma Paris, as well as V.M. Avery and J.P. Holleran, for their initial involvement in the microsphiltration project.
Author information
Authors and Affiliations
Contributions
P.B. supervised the overall project as principal investigator and supported the design and analysis of experiments. J.D., M. Carucci and I.G.-B. designed and analyzed the experiments under the supervision of P.B., F.-J.G., L.S., N.B.R. and P.A.N. J.D. wrote the paper with support provided by P.B. M. Carucci and I.G.-B. supported the preparation of figures and tables. C.R. and B.H. performed the microsphiltration of RBC samples from human donors and prepared Supplementary Figure 6. M. Corral. and O.P. ensured automation maintenance, provided technical support and machined the platform used to confine the Biomek dispenser. J.L.P. provided expertise for data analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Retention rates of P. falciparum ring asexual stages in microsphere filters when exposed to 0.05% DMSO or 250 nM cipargamin (NITD609) over 2 h.
Quantification of retention rates using flow cytometry involved the staining of ring-infected RBC samples with Sybr Green I and mitotracker deep red FM before proceeding to microsphiltration. To quantify retention rates using high content imaging, samples were stained with Syto 40 blue, mitotracker deep red FM and Cellmask orange. Cipargamin 250 nM increases the retention of rings up to an average of 60%, what may define a hit threshold for screening activities. Screening on rings would then use glutaraldehyde-fixed RBC as positive control to define the highest retention rate of the assay.
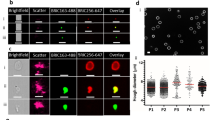
Supplementary Figure 2 High-content imaging of mature gametocyte-infected RBCs from control wells in upstream imaging plates (= aspect before filtration; 60× objective).
i-ii/ Elongated and metabolically active gametocytes in DMSO and calyculin wells. iii/ Swollen mature gametocytes in cipargamin (NITD609, KAE609) wells. iv/ Dead gametocytes in Gentian Violet wells (Mitotracker signal low or absent).
Supplementary Figure 3 Testing different batches of microspheres is essential to setting up a microsphiltration assay.
Retention rates of mature gametocytes exposed to DMSO 0.05% or calyculin A 50-nM during 2-hours and tested in microsphiltration using various sphere batches from two international microsphere providers. Here, IPS (Industrie des Poudres Sphériques, France) sphere batch #1 was selected because it produced the highest retention differential between DMSO and calyculin A wells, with optimal repetitivity.
Supplementary Figure 4 Assembly of the Beckman vacuum manifold.
a = manifold base; b = square well collar; c = polyethylene block; d = reservoir plate; e = microsphiltration plate.
Supplementary Figure 5 Sealing the deficient wells of a microsphiltration plate.
A soldering iron is used to quickly squeeze the director nozzle of deficient filter wells where a microsphere leak is detected. Those wells are identified by the presence of microspheres in the director nozzle.
Supplementary Figure 6 Microsphiltration to analyze the deformability of RBCs in blood donors.
RBC from 11 donors were tested using microplate-based microsphiltration. a/ Unstained RBC from each donor were mixed in CFSE-stained RBC from a reference donor in a 5/95 ratio, then filtered using microsphiltration plate. Quantification of the proportion of unstained / stained RBC was performed by flow cytometry, enabling the determination of retention rates. Retention values are positive when retention was observed (like with glutaraldehyde-fixed RBC, positive control), while negative values correspond to an enrichment of test RBC in the downstream sample, suggesting that RBC from donors were mildly to moderately more deformable than control RBC. Negative control corresponds to unstained RBC from the reference donor diluted in stained RBC from this same donor. The deformability of red blood cells was determined from 4 replicates and 12 replicates per donor in Experiment 1 and Experiment 2, respectively. b/ Significant inter-individual differences were observed in a global test involving all donors (double-tailed Kruskall-Walis test p < 0.0001). When paired comparisons (each involving 2 donors) were performed, 23 of 30 comparisons showed a significant difference (double-tailed Kruskall-Walis test < 0.03). In this preliminary set of results, Donor 1 was tested twice in 2 experiments performed one month apart, and showed stable retention rates (Mean retention = -6.99 versus -1.65%, no significant difference with p = 0.1429 [Mann Whitney test]).
Supplementary Figure 7 Gating strategy used to quantify retention rates of Plasmodium falciparum asexual stage–infected RBCs stained with Sybr Green I using flow cytometry.
As an example, samples of RBC containing 2-5% of ring-stage infected RBC were filtered across microsphiltration plates. Filtered samples (Downstream sample) and their associated control (Upstream sample; unfiltered cell population) were then immediately stained using Sybr green I at 1X final concentration and placed into the incubator for 15 minutes for subsequent analysis with BD Accuri C6 flow cytometer (BD Biosciences) using a 488nm laser. The RBC selection involved the exclusion of debris using the forward scatter and side scatter (FSC-A/SSC-A) profile (Plot 1). A second round of selection using the FSC-A/FSC-H profile was used to exclude RBC doublets (Plot 2). Finally, the SSC-A/FL1-Sybr green I profile (Plot 3) was used to select (Gate G3) and quantify the population of parasitized RBC stained with Sybr green I, thereby enabling the quantification of retention rates. The gate G3 was defined using an uninfected RBC sample stained with Sybr green I (negative control).
Supplementary Figure 8 Gating strategy used to quantify retention rates of CFSE-stained RBCs with flow cytometry.
Samples of RBC containing 2-5% of uninfected RBC (normal, heat-stiffened or glutaraldehyde-fixed) stained with CFSE were filtered across microsphiltration plates. Filtered samples (Downstream sample) and their associated control (Upstream sample; unfiltered cell population) were then immediately analysed with BD Accuri C6 flow cytometer (BD Biosciences) using a 488nm laser. The RBC selection involved the exclusion of debris using the forward scatter and side scatter (FSC-A/SSC-A) profile (Plot 1). A second round of selection using the FSC-A/FSC-H profile was used to exclude RBC doublets (Plot 2). Finally, the SSC-A/FL1-CFSE profile (Plot 3) was used to select (Gate P3) and quantify the population of CFSE-stained RBC, thereby enabling the quantification of retention rates. The gate P3 was defined using unstained RBC sample (negative control).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8. (PDF 1394 kb)
Rights and permissions
About this article
Cite this article
Duez, J., Carucci, M., Garcia-Barbazan, I. et al. High-throughput microsphiltration to assess red blood cell deformability and screen for malaria transmission–blocking drugs. Nat Protoc 13, 1362–1376 (2018). https://doi.org/10.1038/nprot.2018.035
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2018.035
This article is cited by
-
Safe drugs with high potential to block malaria transmission revealed by a spleen-mimetic screening
Nature Communications (2023)
-
Hypothermal opto-thermophoretic tweezers
Nature Communications (2023)
-
Sorting of circulating tumor cells based on the microfluidic device of a biomimetic splenic interendothelial slit array
Microfluidics and Nanofluidics (2021)
-
Plasmodium asexual growth and sexual development in the haematopoietic niche of the host
Nature Reviews Microbiology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.