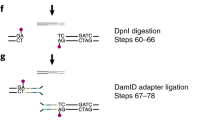
Abstract
This protocol is an extension to: Nat. Protoc. 2, 1467–1478 (2007); doi:10.1038/nprot.2007.148; published online 7 June 2007
The ability to profile transcription and chromatin binding in a cell-type-specific manner is a powerful aid to understanding cell-fate specification and cellular function in multicellular organisms. We recently developed targeted DamID (TaDa) to enable genome-wide, cell-type-specific profiling of DNA- and chromatin-binding proteins in vivo without cell isolation. As a protocol extension, this article describes substantial modifications to an existing protocol, and it offers additional applications. TaDa builds upon DamID, a technique for detecting genome-wide DNA-binding profiles of proteins, by coupling it with the GAL4 system in Drosophila to enable both temporal and spatial resolution. TaDa ensures that Dam-fusion proteins are expressed at very low levels, thus avoiding toxicity and potential artifacts from overexpression. The modifications to the core DamID technique presented here also increase the speed of sample processing and throughput, and adapt the method to next-generation sequencing technology. TaDa is robust, reproducible and highly sensitive. Compared with other methods for cell-type-specific profiling, the technique requires no cell-sorting, cross-linking or antisera, and binding profiles can be generated from as few as 10,000 total induced cells. By profiling the genome-wide binding of RNA polymerase II (Pol II), TaDa can also identify transcribed genes in a cell-type-specific manner. Here we describe a detailed protocol for carrying out TaDa experiments and preparing the material for next-generation sequencing. Although we developed TaDa in Drosophila, it should be easily adapted to other organisms with an inducible expression system. Once transgenic animals are obtained, the entire experimental procedure—from collecting tissue samples to generating sequencing libraries—can be accomplished within 5 d.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Southall, T.D. et al. Cell-type-specific profiling of gene expression and chromatin binding without cell isolation: assaying RNA Pol II occupancy in neural stem cells. Dev. Cell. 26, 101–112 (2013).
van Steensel, B. & Henikoff, S. Identification of in vivo DNA targets of chromatin proteins using tethered dam methyltransferase. Nat. Biotechnol. 18, 424–428 (2000).
Vogel, M.J., Peric-Hupkes, D. & van Steensel, B. Detection of in vivo protein-DNA interactions using DamID in mammalian cells. Nat. Protoc. 2, 1467–1478 (2007).
Brand, A.H. & Perrimon, N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118, 401–415 (1993).
Luukkonen, B.G., Tan, W. & Schwartz, S. Efficiency of reinitiation of translation on human immunodeficiency virus type 1 mRNAs is determined by the length of the upstream open reading frame and by intercistronic distance. J. Virol. 69, 4086–4094 (1995).
Van Blokland, H.J.M., Hoeksema, F., Siep, M., Otte, A.P. & Verhees, J.A. Methods to create a stringent selection system for mammalian cell lines. Cytotechnology 63, 371–384 (2011).
Marshall, O.J. & Brand, A.H. damidseq_pipeline: an automated pipeline for processing DamID sequencing datasets. Bioinformatics 31, 3371–3373 (2015).
Filion, G.J. et al. Systematic protein location mapping reveals five principal chromatin types in Drosophila cells. Cell 143, 212–224 (2010).
Woolcock, K.J., Gaidatzis, D., Punga, T. & Bühler, M. Dicer associates with chromatin to repress genome activity in Schizosaccharomyces pombe. Nat. Struct. Mol. Biol. 18, 94–99 (2011).
Germann, S., Juul-Jensen, T., Letarnec, B. & Gaudin, V. DamID, a new tool for studying plant chromatin profiling in vivo, and its use to identify putative LHP1 target loci. Plant J. 48, 153–163 (2006).
Schuster, E. et al. DamID in C. elegans reveals longevity-associated targets of DAF-16/FoxO. Mol. Syst. Biol. 6, 399 (2010).
Guelen, L. et al. Domain organization of human chromosomes revealed by mapping of nuclear lamina interactions. Nature 453, 948–951 (2008).
Vogel, M. et al. Human heterochromatin proteins form large domains containing KRAB-ZNF genes. Genome Res. 16, 1493–1504 (2006).
Bryant, Z. et al. Characterization of differentially expressed genes in purified Drosophila follicle cells: toward a general strategy for cell type-specific developmental analysis. Proc. Natl. Acad. Sci. USA 96, 5559–5564 (1999).
Bonn, S. et al. Tissue-specific analysis of chromatin state identifies temporal signatures of enhancer activity during embryonic development. Nat. Genet. 44, 148–156 (2012).
Bonn, S. et al. Cell type-specific chromatin immunoprecipitation from multicellular complex samples using BiTS-ChIP. Nat. Protoc. 7, 978–994 (2012).
Deal, R.B. & Henikoff, S. A simple method for gene expression and chromatin profiling of individual cell types within a tissue. Dev. Cell 18, 1030–1040 (2010).
Henry, G.L., Davis, F.P., Picard, S. & Eddy, S.R. Cell type-specific genomics of Drosophila neurons. Nucleic Acids Res. 40, 9691–9704 (2012).
Steiner, F.A., Talbert, P.B., Kasinathan, S., Deal, R.B. & Henikoff, S. Cell-type-specific nuclei purification from whole animals for genome-wide expression and chromatin profiling. Genome Res. 22, 766–777 (2012).
Schauer, T. et al. CAST-ChIP maps cell-type-specific chromatin states in the Drosophila central nervous system. Cell Rep. 5, 271–282 (2013).
Otsuki, L., Cheetham, S.W. & Brand, A.H. Freedom of expression: cell-type-specific gene profiling. Wiley Interdiscip. Rev. Dev. Biol. 3, 429–443 (2014).
Roy, P.J., Stuart, J.M., Lund, J. & Kim, S.K. Chromosomal clustering of muscle-expressed genes in Caenorhabditis elegans. Nature 418, 975–979 (2002).
Thomas, A. et al. A versatile method for cell-specific profiling of translated mRNAs in Drosophila. PLoS ONE 7, e40276 (2012).
Miller, M.R., Robinson, K.J., Cleary, M.D. & Doe, C.Q. TU-tagging: cell type-specific RNA isolation from intact complex tissues. Nat. Methods 6, 439–441 (2009).
Modrich, P. & Lahue, R. Mismatch repair in replication fidelity, genetic recombination, and cancer biology. Annu. Rev. Biochem. 65, 101–133 (1996).
Baena-Lopez, L.A., Alexandre, C., Mitchell, A., Pasakarnis, L. & Vincent, J.-P. Accelerated homologous recombination and subsequent genome modification in Drosophila. Development 140, 4818–4825 (2013).
Venken, K.J.T. et al. MiMIC: a highly versatile transposon insertion resource for engineering Drosophila melanogaster genes. Nat. Methods 8, 737–743 (2011).
Luan, H., Peabody, N.C., Vinson, C. & White, B.H. Refined spatial manipulation of neuronal function by combinatorial restriction of transgene expression. Neuron 52, 425–436 (2006).
Matsumoto, K., Toh-e, A. & Oshima, Y. Genetic control of galactokinase synthesis in Saccharomyces cerevisiae: evidence for constitutive expression of the positive regulatory gene gal4. J. Bacteriol. 134, 446–457 (1978).
McGuire, S.E., Le, P.T., Osborn, A.J., Matsumoto, K. & Davis, R.L. Spatiotemporal rescue of memory dysfunction in Drosophila. Science 302, 1765–1768 (2003).
Osterwalder, T., Yoon, K.S., White, B.H. & Keshishian, H.A Conditional tissue-specific transgene expression system using inducible GAL4. Proc. Natl. Acad. Sci. USA 98, 12596–12601 (2001).
Rohland, N. & Reich, D. Cost-effective, high-throughput DNA sequencing libraries for multiplexed target capture. Genome Res. 22, 939–946 (2012).
Acknowledgements
This work was funded by a Wellcome Trust Senior Investigator Award (103792), Wellcome Trust Programme Grant (092545) and BBSRC Project Grant (BB/L00786X/1) to A.H.B. A.H.B. acknowledges core funding to the Gurdon Institute from the Wellcome Trust (092096) and CRUK (C6946/A14492).
Author information
Authors and Affiliations
Contributions
O.J.M., T.D.S., S.W.C. and A.H.B. designed the research and analyzed the data; T.D.S., S.W.C. and O.J.M. developed the protocols; and O.J.M., T.D.S., S.W.C. and A.H.B. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Details of the pUAST-attB-LT3-NDam vector used for cloning Dam-fusion proteins for TaDa
(A) Schematic vector map illustrating the key features of the vector. (B) Sequence details of the linker and MCS regions of the vector; translated codons in frame with Dam are shaded. Vector map created using SnapGene software.
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1 (PDF 389 kb)
Supplementary Table 1
Sequencing adaptor oligos (DST purity) (XLSX 10 kb)
Rights and permissions
About this article
Cite this article
Marshall, O., Southall, T., Cheetham, S. et al. Cell-type-specific profiling of protein–DNA interactions without cell isolation using targeted DamID with next-generation sequencing. Nat Protoc 11, 1586–1598 (2016). https://doi.org/10.1038/nprot.2016.084
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2016.084
This article is cited by
-
Long range inter-chromosomal interaction of Oct4 distal enhancer loci regulates ESCs pluripotency
Cell Death Discovery (2023)
-
Dynamic adult tracheal plasticity drives stem cell adaptation to changes in intestinal homeostasis in Drosophila
Nature Cell Biology (2021)
-
A transposon expression burst accompanies the activation of Y-chromosome fertility genes during Drosophila spermatogenesis
Nature Communications (2021)
-
Methods for mapping 3D chromosome architecture
Nature Reviews Genetics (2020)
-
Promoter-proximal pausing mediated by the exon junction complex regulates splicing
Nature Communications (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.