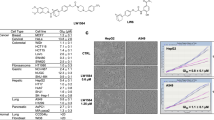
Abstract
Regions of insufficient oxygen supply—hypoxia—occur in diverse contexts across biology in both healthy and diseased organisms. The difference in the chemical environment between a hypoxic biological system and one with normal oxygen levels provides an opportunity for targeting compound delivery to hypoxic regions by using bioreductive prodrugs. Here we detail a protocol for the efficient synthesis of (1-methyl-2-nitro-1H-imidazol-5-yl)methanol, which is a key intermediate that can be converted into a range of 1-methyl-2-nitro-1H-imidazole–based precursors of bioreductive prodrugs. We outline methods for attaching the bioreductive group to a range of functionalities, and we discuss the strategy for positioning of the group on the biologically active parent compound. We have used two parent checkpoint kinase 1 (Chk1) inhibitors to exemplify the protocol. The PROCEDURE also describes a suite of reduction assays, of increasing biological relevance, to validate the bioreductive prodrug. These assays are applied to an exemplar compound, CH-01, which is a bioreductive Chk1 inhibitor. This protocol has broad applications to the development of hypoxia-targeted compounds.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Hammond, E.M. et al. The meaning, measurement and modification of hypoxia in the laboratory and the clinic. Clin. Oncol. (R. Coll. Radiol.) 26, 277–288 (2014).
McKeown, S.R. Defining normoxia, physoxia and hypoxia in tumours—implications for treatment response. Br. J. Radiol. 87, 20130676 (2014).
Wilson, W.R. & Hay, M.P. Targeting hypoxia in cancer therapy. Nat. Rev. Cancer 11, 393–410 (2011).
Thomlinson, R.H. & Gray, L.H. The histological structure of some human lung cancers and the possible implications for radiotherapy. Br. J. Cancer 9, 539–549 (1955).
Vaupel, P. The role of hypoxia-induced factors in tumor progression. Oncologist 9, 10–17 (2004).
Yasui, H. et al. Low-field magnetic resonance imaging to visualize chronic and cycling hypoxia in tumor-bearing mice 70, 6427–6436 (2010).
Nordsmark, M. et al. Prognostic value of tumor oxygenation in 397 head and neck tumors after primary radiation therapy. An international multi-center study. Radiother. Oncol. 77, 18–24 (2005).
Begg, A.C., Stewart, F.A. & Vens, C. Strategies to improve radiotherapy with targeted drugs. Nat. Rev. Cancer 11, 239–253 (2011).
Cazares-Korner, C. et al. CH-01 is a hypoxia-activated prodrug that sensitizes cells to hypoxia/reoxygenation through inhibition of Chk1 and Aurora A. ACS Chem. Biol. 8, 1451–1459 (2013).
O'Connor, L.J. et al. Efficient synthesis of 2-nitroimidazole derivatives and the bioreductive clinical candidate evofosfamide (TH-302). Org. Chem. Front. 2, 1026–1029 (2015).
Cavalleri, B., Ballotta, R. & Lancini, G. Synthesis of 1-alkyl-2-nitroimidazole-5-carboxaldehydes. J. Heterocycl. Chem. 9, 979–984 (1972).
Matteucci, M. et al. Phosphoramidate alkylator prodrugs. Patent no. WO 2007/002931 A2 (2007).
Parveen, I., Naughton, D.P., Whish, W. & Threadgill, M.D. 2-Nitroimidazol-5-ylmethyl as a potential bioreductively activated prodrug system: reductively triggered release of the PARP inhibitor 5-bromoisoquinolinone. Bioorg. Med. Chem. Lett. 9, 2031–2036 (1999).
Damen, E.W.P., Nevalainen, T.J., van den Bergh, T.J.M., de Groot, F.M.H. & Scheeren, H.W. Synthesis of novel paclitaxel prodrugs designed for bioreductive activation in hypoxic tumour tissue. Bioorg. Med. Chem. 10, 71–77 (2002).
Ferrer, S., Naughton, D.P. & Threadgill, M.D. H-1 NMR studies on the reductively triggered release of heterocyclic and steroid drugs from 4,7-dioxoindole-3-methyl prodrugs. Tetrahedron 59, 3445–3454 (2003).
Zhang, Z., Tanabe, K., Hatta, H. & Nishimoto, S.-I. Bioreduction activated prodrugs of camptothecin: molecular design, synthesis, activation mechanism and hypoxia selective cytotoxicity. Org. Biomol. Chem. 3, 1905–1910 (2005).
Hay, M.P., Wilson, W.R. & Denny, W.A. Nitroarylmethylcarbamate prodrugs of doxorubicin for use with nitroreductase gene-directed enzyme prodrug therapy. Bioorg. Med. Chem. 13, 4043–4055 (2005).
Thomson, P. et al. Hypoxia-driven elimination of thiopurines from their nitrobenzyl prodrugs. Bioorg. Med. Chem. Lett. 17, 4320–4322 (2007).
Granchi, C. et al. Bioreductively activated lysyl oxidase inhibitors against hypoxic tumours. ChemMedChem 4, 1590–1594 (2009).
Zhu, R. et al. 4-Nitrobenzyloxycarbonyl derivatives of O(6)-benzylguanine as hypoxia-activated prodrug inhibitors of O(6)-alkylguanine-DNA alkyltransferase (AGT), which produces resistance to agents targeting the O-6 position of DNA guanine. J. Med. Chem. 54, 7720–7728 (2011).
Lindquist, K.E. et al. Selective radiosensitization of hypoxic cells using BCCA621C: a novel hypoxia activated prodrug targeting DNA-dependent protein kinase. Tumor Microenviron. Ther. 1, 46–55 (2013).
Couch, G.D., Burke, P.J., Knox, R.J. & Moody, C.J. Synthesis of 2-aryl-6-methyl-5-nitroquinoline derivatives as potential prodrug systems for reductive activation. Tetrahedron 64, 2816–2823 (2008).
Cui, L. et al. A new prodrug-derived ratiometric fluorescent probe for hypoxia: high selectivity of nitroreductase and imaging in tumor cell. Org. Lett. 13, 928–931 (2011).
Blanche, E.A., Maskell, L., Colucci, M.A., Whatmore, J.L. & Moody, C.J. Synthesis of potential prodrug systems for reductive activation. Prodrugs for anti-angiogenic isoflavones and VEGF receptor tyrosine kinase inhibitory oxindoles. Tetrahedron 65, 4894–4903 (2009).
Naughton, D.P. Drug targeting to hypoxic tissue using self-inactivating bioreductive delivery systems. Adv. Drug Deliv. Rev. 53, 229–233 (2001).
Wardman, P. Some reactions and properties of nitro radical-anions important in biology and medicine. Environ. Health Perspect. 64, 309–320 (1985).
Foloppe, N. et al. Structure-based design of novel Chk1 inhibitors: insights into hydrogen bonding and protein-ligand affinity. J. Med. Chem. 48, 4332–4345 (2005).
Larock, R.C. Comprehensive Organic Transformations (Wiley-VCH, 1999).
Acknowledgements
S.J.C., E.M.H. and L.J.O.C. thank the UK Medical Research Council (MRC) for the award of a studentship to L.J.O.C. S.J.C., E.M.H. and C.C.-K. thank Cancer Research UK (CRUK) for the award of a studentship to C.C.-K. S.J.C., E.M.H. and C.N.G.E. thank CRUK and the UK Engineering and Physical Sciences Research Council (EPSRC) for the award of a studentship to C.N.G.E., through the CRUK and EPSRC Cancer Imaging Centre in Oxford (OCIC). S.J.C. and J.S. thank the European Commission for the award of a Marie Curie Fellowship to J.S. (PIIF-GA-2012-331327, Hypoxprobe). E.M.H. thanks CRUK for research funding. S.J.C. thanks St. Hugh's College, Oxford, for research funding.
Author information
Authors and Affiliations
Contributions
The study was conceived and planned by S.J.C. and E.M.H., and they also supervised the in vitro bioreduction studies. The chemical synthesis was conducted by L.J.O.C., C.C.-K., J.S. and C.N.G.E. The chemical synthesis was supervised by S.J.C. The in vitro bioreduction studies and the cellular bioreduction studies were performed by L.J.O.C., C.C.-K. and M.R.L.S. The cellular bioreduction studies were supervised by E.M.H. All authors interpreted the data. The manuscript was written by L.J.O.C., J.S., E.M.H. and S.J.C. All authors commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Data (PDF 402 kb)
Rights and permissions
About this article
Cite this article
O'Connor, L., Cazares-Körner, C., Saha, J. et al. Design, synthesis and evaluation of molecularly targeted hypoxia-activated prodrugs. Nat Protoc 11, 781–794 (2016). https://doi.org/10.1038/nprot.2016.034
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2016.034
This article is cited by
-
Therapeutic targeting of the hypoxic tumour microenvironment
Nature Reviews Clinical Oncology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.