Abstract
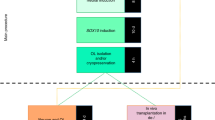
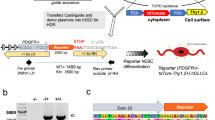
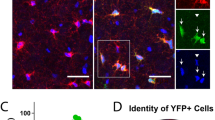
In the CNS, oligodendrocytes act as the myelinating cells. Oligodendrocytes have been identified to be key players in several neurodegenerative disorders. This protocol describes a robust, fast and reproducible differentiation protocol to generate human oligodendrocytes from pluripotent stem cells (PSCs) using a chemically defined, growth factor–rich medium. Within 8 d, PSCs differentiate into paired box 6–positive (PAX6+) neural stem cells, which give rise to OLIG2+ progenitors by day 12. Oligodendrocyte lineage transcription factor 2–positive (OLIG2+) cells begin to express the transcription factor NKX2.2 around day 18, followed by SRY-box 10 (SOX10) around day 40. Oligodendrocyte progenitor cells (OPCs) that are positive for the cell surface antigen recognized by the O4 antibody (O4+) appear around day 50 and reach, on average, 43% of the cell population after 75 d of differentiation. O4+ OPCs can be isolated by cell sorting for myelination studies, or they can be terminally differentiated to myelin basic protein–positive (MBP+) oligodendrocytes. This protocol also describes an alternative strategy for markedly reducing the length and the costs of the differentiation and generating ∼30% O4+ cells after only 55 d of culture.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kassmann, C.M. et al. Axonal loss and neuroinflammation caused by peroxisome-deficient oligodendrocytes. Nat. Genet. 39, 969–976 (2007).
Bartzokis, G. Brain myelination in prevalent neuropsychiatric developmental disorders: primary and comorbid addiction. Adolesc. Psychiatry 29, 55–96 (2005).
Irion, S., Nostro, M.C., Kattman, S.J. & Keller, G.M. Directed differentiation of pluripotent stem cells: from developmental biology to therapeutic applications. Cold Spring Harb. Symp. Quant. Biol. 73, 101–110 (2008).
Murry, C.E. & Keller, G. Differentiation of embryonic stem cells to clinically relevant populations: lessons from embryonic development. Cell 132, 661–680 (2008).
Wichterle, H., Lieberam, I., Porter, J.A. & Jessell, T.M. Directed differentiation of embryonic stem cells into motor neurons. Cell 110, 385–397 (2002).
Nistor, G.I., Totoiu, M.O., Haque, N., Carpenter, M.K. & Keirstead, H.S. Human embryonic stem cells differentiate into oligodendrocytes in high purity and myelinate after spinal cord transplantation. Glia 49, 385–396 (2005).
Izrael, M. et al. Human oligodendrocytes derived from embryonic stem cells: effect of noggin on phenotypic differentiation in vitro and on myelination in vivo. Mol. Cell. Neurosci. 34, 310–323 (2007).
Hu, B.Y., Du, Z.W., Li, X.J., Ayala, M. & Zhang, S.C. Human oligodendrocytes from embryonic stem cells: conserved SHH signaling networks and divergent FGF effects. Development 136, 1443–1452 (2009).
Hu, B.Y., Du, Z.W. & Zhang, S.C. Differentiation of human oligodendrocytes from pluripotent stem cells. Nat. Protoc. 4, 1614–1622 (2009).
Numasawa-Kuroiwa, Y. et al. Involvement of ER stress in dysmyelination of Pelizaeus-Merzbacher disease with PLP1 missense mutations shown by iPSC-derived oligodendrocytes. Stem Cell Rep. 2, 648–661 (2014).
Stacpoole, S.R. et al. High yields of oligodendrocyte lineage cells from human embryonic stem cells at physiological oxygen tensions for evaluation of translational biology. Stem Cell Rep. 1, 437–450 (2013).
Wang, S. et al. Human iPSC-derived oligodendrocyte progenitor cells can myelinate and rescue a mouse model of congenital hypomyelination. Cell Stem Cell 12, 252–264 (2013).
Liu, Y., Jiang, P. & Deng, W. OLIG gene targeting in human pluripotent stem cells for motor neuron and oligodendrocyte differentiation. Nat. Protoc. 6, 640–655 (2011).
Sim, F.J. et al. CD140a identifies a population of highly myelinogenic, migration-competent and efficiently engrafting human oligodendrocyte progenitor cells. Nat. Biotechnol. 29, 934–941 (2011).
Boulting, G.L. et al. A functionally characterized test set of human induced pluripotent stem cells. Nat. Biotechnol. 29, 279–286 (2011).
Douvaras, P. et al. Efficient generation of myelinating oligodendrocytes from primary progressive multiple sclerosis patients by induced pluripotent stem cells. Stem Cell Rep. 3, 250–259 (2014).
Kang, J. et al. Granulocyte colony-stimulating factor minimizes negative remodeling of decellularized small diameter vascular graft conduits but not medial degeneration. Ann. Vasc. Surg. 27, 487–496 (2013).
Fennema-Notestine, C. et al. In vivo evidence of cerebellar atrophy and cerebral white matter loss in Huntington disease. Neurology 63, 989–995 (2004).
Behrendt, G. et al. Dynamic changes in myelin aberrations and oligodendrocyte generation in chronic amyloidosis in mice and men. Glia 61, 273–286 (2013).
Bernstein, H.G., Steiner, J., Guest, P.C., Dobrowolny, H. & Bogerts, B. Glial cells as key players in schizophrenia pathology: recent insights and concepts of therapy. Schizophr. Res. 161, 4–18 (2015).
Chambers, S.M. et al. Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nat. Biotechnol. 27, 275–280 (2009).
Patani, R. et al. Retinoid-independent motor neurogenesis from human embryonic stem cells reveals a medial columnar ground state. Nat. Commun. 2, 214 (2011).
Miller, R.H. et al. Patterning of spinal cord oligodendrocyte development by dorsally derived BMP4. J. Neurosci. Res. 76, 9–19 (2004).
Tomassy, G.S. & Fossati, V. How big is the myelinating orchestra? Cellular diversity within the oligodendrocyte lineage: facts and hypotheses. Front. Cell. Neurosci. 8, 201 (2014).
Acknowledgements
We thank M. Zimmer for excellent assistance with cell sorting and J. Goldman (Columbia University) for providing the O4 antibody. We are thankful to D. Paul and B. Corneo for their insightful comments. This work was supported by a New York Stem Cell Foundation (NYSCF)-Helmsley Early Career Investigator Award, The NYCSF, and The Leona M. and Harry B. Helmsley Charitable Trust. P.D. is a NYSCF-Druckenmiller postdoctoral fellow.
Author information
Authors and Affiliations
Contributions
P.D. performed the experiments. P.D. and V.F. designed the protocol, analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Douvaras, P., Fossati, V. Generation and isolation of oligodendrocyte progenitor cells from human pluripotent stem cells. Nat Protoc 10, 1143–1154 (2015). https://doi.org/10.1038/nprot.2015.075
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2015.075
This article is cited by
-
Directional induction of neural stem cells, a new therapy for neurodegenerative diseases and ischemic stroke
Cell Death Discovery (2023)
-
Identifying Genes that Affect Differentiation of Human Neural Stem Cells and Myelination of Mature Oligodendrocytes
Cellular and Molecular Neurobiology (2023)
-
OCT4-induced oligodendrocyte progenitor cells promote remyelination and ameliorate disease
npj Regenerative Medicine (2022)
-
Promoting Oligodendrocyte Differentiation from Human Induced Pluripotent Stem Cells by Activating Endocannabinoid Signaling for Treating Spinal Cord Injury
Stem Cell Reviews and Reports (2022)
-
Recent Advances in 3D-Cultured Brain Tissue Models Derived from Human iPSCs
BioChip Journal (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.