Abstract
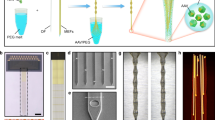
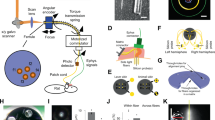
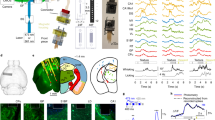
In vivo optogenetic strategies have redefined our ability to assay how neural circuits govern behavior. Although acutely implanted optical fibers have previously been used in such studies, long-term control over neuronal activity has been largely unachievable. Here we describe a method to construct implantable optical fibers to readily manipulate neural circuit elements with minimal tissue damage or change in light output over time (weeks to months). Implanted optical fibers readily interface with in vivo electrophysiological arrays or electrochemical detection electrodes. The procedure described here, from implant construction to the start of behavioral experimentation, can be completed in approximately 2–6 weeks. Successful use of implantable optical fibers will allow for long-term control of mammalian neural circuits in vivo, which is integral to the study of the neurobiology of behavior.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Claridge-Chang, A. et al. Writing memories with light-addressable reinforcement circuitry. Cell 139, 405–415 (2009).
Guo, Z.V., Hart, A.C. & Ramanathan, S. Optical interrogation of neural circuits in Caenorhabditis elegans. Nat. Methods 6, 891–896 (2009).
Liewald, J.F. et al. Optogenetic analysis of synaptic function. Nat. Methods 5, 895–902 (2008).
Zhu, P. et al. Optogenetic dissection of neuronal circuits in zebrafish using viral gene transfer and the Tet system. Front. Neural Circuits 3, 21 (2009).
Tsai, H.C. et al. Phasic firing in dopaminergic neurons is sufficient for behavioral conditioning. Science (New York, NY) 324, 1080–1084 (2009).
Adamantidis, A.R., Zhang, F., Aravanis, A.M., Deisseroth, K. & de Lecea, L. Neural substrates of awakening probed with optogenetic control of hypocretin neurons. Nature 450, 420–424 (2007).
Kravitz, A.V. et al. Regulation of parkinsonian motor behaviours by optogenetic control of basal ganglia circuitry. Nature 466, 622–626 (2010).
Tye, K.M. et al. Amygdala circuitry mediating reversible and bidirectional control of anxiety. Nature 471, 358–362 (2011).
Lin, D. et al. Functional identification of an aggression locus in the mouse hypothalamus. Nature 470, 221–226 (2011).
Witten, I.B. et al. Cholinergic interneurons control local circuit activity and cocaine conditioning. Science (New York, NY) 330, 1677–1681 (2010).
Zhang, F. et al. Optogenetic interrogation of neural circuits: technology for probing mammalian brain structures. Nat. Protoc. 5, 439–456 (2010).
Covington, H.E., III et al. Antidepressant effect of optogenetic stimulation of the medial prefrontal cortex. J. Neurosci. 30, 16082–16090 (2010).
Han, X. et al. Millisecond-timescale optical control of neural dynamics in the nonhuman primate brain. Neuron 62, 191–198 (2009).
Diester, I. et al. An optogenetic toolbox designed for primates. Nat. Neurosci. 14, 387–397 (2011).
Stuber, G.D. et al. Amygdala to nucleus accumbens excitatory transmission facilitates reward seeking. Nature 475, 377–380 (2011).
Cardin, J.A. et al. Driving fast-spiking cells induces gamma rhythm and controls sensory responses. Nature 459, 663–667 (2009).
Kravitz, A.V. & Kreitzer, A.C. Optogenetic manipulation of neural circuitry in vivo. Curr. Opin. Neurobiol. 21, 433–439 (2011).
Martin-Garcia, E. et al. New operant model of reinstatement of food-seeking behavior in mice. Psychopharmacology 215, 49–70 (2010).
Olsen, C.M. & Winder, D.G. Operant sensation seeking engages similar neural substrates to operant drug seeking in C57 mice. Neuropsychopharmacology 34, 1685–1694 (2009).
Aravanis, A.M. et al. An optical neural interface: in vivo control of rodent motor cortex with integrated fiberoptic and optogenetic technology. J. Neural Eng. 4, S143–S156 (2007).
Tian, L. et al. Imaging neural activity in worms, flies and mice with improved GCaMP calcium indicators. Nat. Methods 6, 875–881 (2009).
Cardin, J.A. et al. Targeted optogenetic stimulation and recording of neurons in vivo using cell-type-specific expression of Channelrhodopsin-2. Nat. Protoc. 5, 247–254 (2010).
Acknowledgements
We thank A. Kravitz, M. Patel, J. Smithius, M. Weber and D. Albaugh for discussion and assistance. This study was supported by funds from the National Institute on Alcohol Abuse and Alcoholism (NIAA) (F32AA018610 to D.R.S.), the National Alliance for Research on Schizophrenia and Depression (NARSAD), The Whitehall Foundation, the Foundation for Alcohol Research (ABMRF), the Foundation of Hope, the National Institute on Drug Abuse (DA029325) and startup funds provided by the Department of Psychiatry at the University of North Carolina at Chapel Hill (G.D.S.).
Author information
Authors and Affiliations
Contributions
D.R.S., A.M.S., J.L.P., N.H., R.v.Z. and G.D.S. performed the experiments. D.R.S., A.M.S., J.L.P., N.H. and G.D.S. developed the protocol. D.R.S. and G.D.S wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Sparta, D., Stamatakis, A., Phillips, J. et al. Construction of implantable optical fibers for long-term optogenetic manipulation of neural circuits. Nat Protoc 7, 12–23 (2012). https://doi.org/10.1038/nprot.2011.413
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2011.413
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.