Abstract
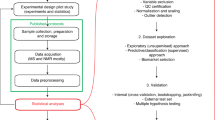
The role of urinary metabolic profiling in systems biology research is expanding. This is because of the use of this technology for clinical diagnostic and mechanistic studies and for the development of new personalized health care and molecular epidemiology (population) studies. The methodologies commonly used for metabolic profiling are NMR spectroscopy, liquid chromatography mass spectrometry (LC/MS) and gas chromatography–mass spectrometry (GC/MS). In this protocol, we describe urine collection and storage, GC/MS and data preprocessing methods, chemometric data analysis and urinary marker metabolite identification. Results obtained using GC/MS are complementary to NMR and LC/MS. Sample preparation for GC/MS analysis involves the depletion of urea via treatment with urease, protein precipitation with methanol, and trimethylsilyl derivatization. The protocol described here facilitates the metabolic profiling of ∼400–600 metabolites in 120 urine samples per week.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Wishart, D.S. et al. HMDB: the human metabolome database. Nucleic Acids Res. 35, D521–D526 (2007).
Nicholson, J.K. & Lindon, J.C. Systems biology: metabonomics. Nature 455, 1054–1056 (2008).
Lenz, E.M. & Wilson, I.D. Analytical strategies in metabonomics. J. Proteome Res. 6, 443–458 (2007).
Nicholson, J.K., Lindon, J.C. & Holmes, E. 'Metabonomics': understanding the metabolic responses of living systems to pathophysiological stimuli via multivariate statistical analysis of biological NMR spectroscopic data. Xenobiotica 29, 1181–1189 (1999).
Dettmer, K., Aronov, P.A. & Hammock, B.D. Mass spectrometry-based metabolomics. Mass Spectrom. Rev. 26, 51–78 (2007).
Dunn, W.B., Bailey, N.J. & Johnson, H.E. Measuring the metabolome: current analytical technologies. Analyst 130, 606–625 (2005).
Sreekumar, A. et al. Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature 457, 910–914 (2009).
Spratlin, J.L., Serkova, N.J. & Eckhardt, S.G. Clinical applications of metabolomics in oncology: a review. Clin. Cancer Res. 15, 431–440 (2009).
Pasikanti, K.K. et al. Noninvasive urinary metabonomic diagnosis of human bladder cancer. J. Proteome Res. 9, 2988–2995 (2010).
Nordstrom, A. & Lewensohn, R. Metabolomics: moving to the clinic. J. Neuroimmune Pharmacol. 5, 4–17 (2010).
Keun, H.C. Metabonomic modeling of drug toxicity. Pharmacol. Ther. 109, 92–106 (2006).
Lindon, J.C. et al. Contemporary issues in toxicology the role of metabonomics in toxicology and its evaluation by the COMET project. Toxicol. Appl. Pharmacol. 187, 137–146 (2003).
Lanza, I.R. et al. Quantitative metabolomics by H-NMR and LC-MS/MS confirms altered metabolic pathways in diabetes. PLoS One 5, e10538 (2010).
McClay, J.L. et al. 1H nuclear magnetic resonance metabolomics analysis identifies novel urinary biomarkers for lung function. J. Proteome Res. 9, 3083–3090 (2010).
Lenz, E.M. & Wilson, I.D. Analytical strategies in metabonomics. J. Proteome Res. 6, 443–458 (2007).
Kind, T., Tolstikov, V., Fiehn, O. & Weiss, R.H. A comprehensive urinary metabolomic approach for identifying kidney cancerr. Anal. Biochem. 363, 185–195 (2007).
Issaq, H.J. et al. Detection of bladder cancer in human urine by metabolomic profiling using high performance liquid chromatography/mass spectrometry. J. Urol. 179, 2422–2426 (2008).
Kim, K. et al. Urine metabolomics analysis for kidney cancer detection and biomarker discovery. Mol. Cell Proteomics 8, 558–570 (2009).
Lindon, J.C., Holmes, E. & Nicholson, J.K. So what's the deal with metabonomics? Anal. Chem. 75, 384A–391A (2003).
Walsh, M.C., Brennan, L., Malthouse, J.P.G., Roche, H.M. & Gibney, M.J. Effect of acute dietary standardization on the urinary, plasma, and salivary metabolomic profiles of healthy humans. Am. J. Clin. Nutr. 84, 531–539 (2006).
Pasikanti, K.K., Ho, P.C. & Chan, E.C. Development and validation of a gas chromatography/mass spectrometry metabonomic platform for the global profiling of urinary metabolites. Rapid Commun. Mass Spectrom. 22, 2984–2992 (2008).
Assfalg, M. et al. Evidence of different metabolic phenotypes in humans. Proc. Natl. Acad. Sci. USA 105, 1420–1424 (2008).
Pasikanti, K.K., Ho, P.C. & Chan, E.C. Gas chromatography/mass spectrometry in metabolic profiling of biological fluids. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 871, 202–211 (2008).
Kopka, J. et al. GMD@CSB.DB: the Golm metabolome database. Bioinformatics 21, 1635–1638 (2005).
Griffin, J.L. The Cinderella story of metabolic profiling: does metabolomics get to go to the functional genomics ball? Philos. Trans. R Soc. Lond. B Biol. Sci. 361, 147–161 (2006).
Ng, D., Pasikanti, K. & Chan, E. Trend analysis of metabonomics and systematic review of metabonomics-derived cancer marker metabolites. Metabolomics 7, 155–178 (2011).
Xiayan, L. & Legido-Quigley, C. Advances in separation science applied to metabonomics. Electrophoresis 29, 3724–3736 (2008).
Beckonert, O. et al. Metabolic profiling, metabolomic and metabonomic procedures for NMR spectroscopy of urine, plasma, serum and tissue extracts. Nat. Protoc. 2, 2692–2703 (2007).
Rooney, O.M., Troke, J., Nicholson, J.K. & Griffin, J.L. High-resolution diffusion and relaxation-edited magic angle spinning 1H NMR spectroscopy of intact liver tissue. Magn. Reson. Med. 50, 925–930 (2003).
Wang, Y. et al. Spectral editing and pattern recognition methods applied to high-resolution magic-angle spinning 1H nuclear magnetic resonance spectroscopy of liver tissues. Anal. Biochem. 323, 26–32 (2003).
Atherton, H.J. et al. A combined 1H-NMR spectroscopy- and mass spectrometry-based metabolomic study of the PPAR-alpha null mutant mouse defines profound systemic changes in metabolism linked to the metabolic syndrome. Physiol. Genomics 27, 178–186 (2006).
Want, E.J., Nordstrom, A., Morita, H. & Siuzdak, G. From exogenous to endogenous: the inevitable imprint of mass spectrometry in metabolomics. J. Proteome Res. 6, 459–468 (2007).
Halket, J.M. et al. Chemical derivatization and mass spectral libraries in metabolic profiling by GC/MS and LC/MS/MS. J. Exp. Bot. 56, 219–243 (2005).
Allwood, J.W. et al. Inter-laboratory reproducibility of fast gas chromatography-electron impact-time of flight mass spectrometry (GC-EI-TOF/MS) based plant metabolomics. Metabolomics 5, 479–496 (2009).
Lisec, J., Schauer, N., Kopka, J., Willmitzer, L. & Fernie, A.R. Gas chromatography mass spectrometry-based metabolite profiling in plants. Nat. Protoc. 1, 387–396 (2006).
Koh, Y., Pasikanti, K.K., Yap, C.W. & Chan, E.C. Comparative evaluation of software for retention time alignment of gas chromatography/time-of-flight mass spectrometry-based metabonomic data. J. Chromatogr. A 1217, 8308–8316 (2010).
Pasikanti, K.K. et al. Metabolic footprinting of tumorigenic and nontumorigenic uroepithelial cells using two-dimensional gas chromatography time-of-flight mass spectrometry. Anal. Bioanal. Chem. 398, 1285–1293 (2010).
Law, W.S. et al. Metabonomics investigation of human urine after ingestion of green tea with gas chromatography/mass spectrometry, liquid chromatography/mass spectrometry and (1)H NMR spectroscopy. Rapid. Commun. Mass Spectrom. 22, 2436–2446 (2008).
Smart, K.F., Aggio, R.B., Van Houtte, J.R. & Villas-Boas, S.G. Analytical platform for metabolome analysis of microbial cells using methyl chloroformate derivatization followed by gas chromatography-mass spectrometry. Nat. Protoc. 5, 1709–1729 (2010).
Want, E.J. et al. Global metabolic profiling procedures for urine using UPLC-MS. Nat. Protoc. 5, 1005–1018 (2010).
Lauridsen, M., Hansen, S.H., Jaroszewski, J.W. & Cornett, C. Human urine as test material in 1H NMR-based metabonomics: recommendations for sample preparation and storage. Anal. Chem. 79, 1181–1186 (2007).
Gika, H.G., Theodoridis, G.A. & Wilson, I.D. Liquid chromatography and ultra-performance liquid chromatography-mass spectrometry fingerprinting of human urine: sample stability under different handling and storage conditions for metabonomics studies. J. Chromatogr. A 1189, 314–322 (2008).
Saude, E. & Sykes, B. Urine stability for metabolomic studies: effects of preparation and storage. Metabolomics 3, 19–27 (2007).
Kuhara, T. Diagnosis and monitoring of inborn errors of metabolism using urease-pretreatment of urine, isotope dilution, and gas chromatography-mass spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 781, 497–517 (2002).
Chen, M., Zhao, L. & Jia, W. Metabonomic study on the biochemical profiles of a hydrocortisone-induced animal model. J. Proteome Res. 4, 2391–2396 (2005).
Lee, S.H. et al. Metabolomic approach to evaluate the toxicological effects of nonylphenol with rat urine. Anal. Chem. 79, 6102–6110 (2007).
Little, J.L. Artifacts in trimethylsilyl derivatization reactions and ways to avoid them. J. Chromatogr. A 844, 1–22 (1999).
Gika, H.G., Theodoridis, G.A., Wingate, J.E. & Wilson, I.D. Within-day reproducibility of an HPLC-MS-based method for metabonomic analysis: application to human urine. J. Proteome Res. 6, 3291–3303 (2007).
Trygg, J., Holmes, E. & Lundstedt, T. Chemometrics in metabonomics. J. Proteome Res. 6, 469–479 (2007).
Lu, H.M., Dunn, W.B., Shen, H.L., Kell, D.B. & Liang, Y.Z. Comparative evaluation of software for deconvolution of metabolomics data based on GC-TOF-MS. Trac.-Trend. Anal. Chem. 27, 215–227 (2008).
Lommen, A. MetAlign: interface-driven, versatile metabolomics tool for hyphenated full-scan mass spectrometry data preprocessing. Anal. Chem. 81, 3079–3086 (2009).
Pluskal, T., Castillo, S., Villar-Briones, A. & Oresic, M. MZmine 2: modular framework for processing, visualizing, and analyzing mass spectrometry-based molecular profile data. BMC Bioinformatics 11, 395 (2010).
Styczynski, M.P. et al. Systematic identification of conserved metabolites in GC/MS data for metabolomics and biomarker discovery. Anal. Chem. 79, 966–973 (2007).
Tikunov, Y. et al. A novel approach for nontargeted data analysis for metabolomics. Large-scale profiling of tomato fruit volatiles. Plant Physiol. 139, 1125–1137 (2005).
Eriksson, L. & Umetrics, A.B. Multi- and Megavariate Data Analysis Edn. 2nd rev. and enl. (Umetrics AB, 2006).
Madsen, R., Lundstedt, T. & Trygg, J. Chemometrics in metabolomics—a review in human disease diagnosis. Anal. Chim. Acta. 659, 23–33 (2010).
Pierce, K.M., Hoggard, J.C., Mohler, R.E. & Synovec, R.E. Recent advancements in comprehensive two-dimensional separations with chemometrics. J. Chromatogr. A 1184, 341–352 (2008).
Xia, J., Psychogios, N., Young, N. & Wishart, D.S. MetaboAnalyst: a web server for metabolomic data analysis and interpretation. Nucleic Acids Res. 37, W652–W660 (2009).
Chae, M., Shmookler Reis, R.J. & Thaden, J.J. An iterative block-shifting approach to retention time alignment that preserves the shape and area of gas chromatography-mass spectrometry peaks. BMC Bioinformatics 9 (Suppl 9): S15 (2008).
Wiklund, S. et al. Visualization of GC/TOF-MS-based metabolomics data for identification of biochemically interesting compounds using OPLS class models. Anal. Chem. 80, 115–122 (2008).
Smit, S. et al. Assessing the statistical validity of proteomics based biomarkers. Anal. Chim. Acta. 592, 210–217 (2007).
Wiklund, S. et al. A randomization test for PLS component selection. J. Chemometrics 21, 427–439 (2007).
Mahadevan, S., Shah, S.L., Marrie, T.J. & Slupsky, C.M. Analysis of metabolomic data using support vector machines. Anal. Chem. 80, 7562–7570 (2008).
Vandendool, H. & Kratz, P.D. A generalization of the retention index system including linear temperature programmed gas-liquid partition chromatography. J. Chromatogr. 11, 463–471 (1963).
Kind, T. et al. FiehnLib: mass spectral and retention index libraries for metabolomics based on quadrupole and time-of-flight gas chromatography/mass spectrometry. Anal. Chem. 81, 10038–10048 (2009).
Mendes, P. Emerging bioinformatics for the metabolome. Brief Bioinform. 3, 134–145 (2002).
Xu, F.G., Zou, L. & Ong, C.N. Experiment-originated variations, and multi-peak and multi-origination phenomena in derivatization-based GC-MS metabolomics. Trac.-Trend. Anal. Chem. 29, 269–280 (2010).
Acknowledgements
GC/TOFMS method development was supported by the National University of Singapore (NUS) grant R-148-000-100-112 and National Medical Research Council grant R-176-000-119-213 provided to E.C.Y.C. GC/TOFMS was sponsored by the NUS grant R-279-000-249-646. P.K.K. is supported by a NUS President's Graduate fellowship.
Author information
Authors and Affiliations
Contributions
E.C.Y.C. and P.K.K. designed the protocol and conducted the initial study; E.C.Y.C., P.K.K. and J.K.N. wrote the manuscript; J.K.N. gave conceptual advice.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Chan, E., Pasikanti, K. & Nicholson, J. Global urinary metabolic profiling procedures using gas chromatography–mass spectrometry. Nat Protoc 6, 1483–1499 (2011). https://doi.org/10.1038/nprot.2011.375
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2011.375
This article is cited by
-
Comparative metabolomics of root-tips reveals distinct metabolic pathways conferring drought tolerance in contrasting genotypes of rice
BMC Genomics (2023)
-
Optimising a urinary extraction method for non-targeted GC–MS metabolomics
Scientific Reports (2023)
-
Characterization of d-amino acids in colostral, transitional, and mature preterm human milk
Amino Acids (2023)
-
Non-invasive metabolomics biomarkers of production efficiency and beef carcass quality traits
Scientific Reports (2022)
-
Alterations in Microbial-Associated Fecal Metabolites in Relation to Arsenic Exposure Among Infants
Exposure and Health (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.