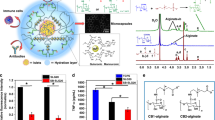
Abstract
Cell therapy has the potential to treat or cure a wide variety of diseases. Non-invasive cell tracking techniques are, however, necessary to translate this approach to the clinical setting. This protocol details methods to create microcapsules that are visible by X-ray, ultrasound (US) or magnetic resonance (MR) for the encapsulation and immunoisolation of cellular therapeutics. Three steps are generally used to encapsulate cellular therapeutics in an alginate matrix: (i) droplets of cell-containing liquid alginate are extruded, using an electrostatic generator, through a needle tip into a solution containing a dissolved divalent cation salt to form a solid gel; (ii) the resulting gelled spheres are coated with polycations as a cross-linker; and (iii) these complexes are then incubated in a second solution of alginate to form a semipermeable membrane composed of an inner and an outer layer of alginate. The microcapsules can be rendered visible during the first step by adding contrast agents to the primary alginate layer. Such contrast agents include superparamagnetic iron oxide for detection by 1H MR imaging (MRI); the radiopaque agents barium or bismuth sulfate for detection by X-ray modalities; or perfluorocarbon emulsions for multimodal detection by 19F MRI, X-ray and US imaging. The entire synthesis can be completed within 2 h.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Mullen, Y., Maruyama, M. & Smith, C.V. Current progress and perspectives in immunoisolated islet transplantation. J. Hepatobiliary Pancreat. Surg. 7, 347–357 (2000).
Schneider, S. et al. Long-term graft function of adult rat and human islets encapsulated in novel alginate-based microcapsules after transplantation in immunocompetent diabetic mice. Diabetes 54, 687–693 (2005).
Luca, G. et al. Xenograft of microencapsulated sertoli cells reverses T1DM in NOD mice by inducing neogenesis of beta-cells. Transplantation 90, 1352–1357 (2010).
Soon-Shiong, P. et al. Insulin independence in a type 1 diabetic patient after encapsulated islet transplantation. Lancet 343, 950–951 (1994).
Tuch, B.E. et al. Safety and viability of microencapsulated human islets transplanted into diabetic humans. Diabetes Care 32, 1887–1889 (2009).
Tibell, A. et al. Survival of macroencapsulated allogeneic parathyroid tissue one year after transplantation in nonimmunosuppressed humans. Cell Transplant. 10, 591–599 (2001).
Borlongan, C.V. et al. Intracerebral transplantation of porcine choroid plexus provides structural and functional neuroprotection in a rodent model of stroke. Stroke 35, 2206–2210 (2004).
Visted, T. et al. Prospects for delivery of recombinant angiostatin by cell-encapsulation therapy. Hum. Gene. Ther. 14, 1429–1440 (2003).
Mai, G. et al. Treatment of fulminant liver failure by transplantation of microencapsulated primary or immortalized xenogeneic hepatocytes. Transplant Proc. 37, 527–529 (2005).
Barnett, B.P. et al. Radiopaque alginate microcapsules for X-ray visualization and immunoprotection of cellular therapeutics. Mol. Pharm. 3, 531–538 (2006).
Barnett, B.P. et al. Magnetic resonance-guided, real-time targeted delivery and imaging of magnetocapsules immunoprotecting pancreatic islet cells. Nat. Med. 13, 986–991 (2007).
Barnett, B.P. et al. Fluorocapsules for improved function, immunoprotection, and visualization of cellular therapeutics with MR, US, and CT imaging. Radiology 258, 182–191 (2011).
Kim, J. et al. Multifunctional capsule-in-capsules for immunoprotection and trimodal imaging. Angew. Chem. Int. Ed. Engl. 50, 2317–2321 (2011).
Bulte, J.W. et al. Magnetodendrimers allow endosomal magnetic labeling and in vivo tracking of stem cells. Nat. Biotechnol. 19, 1141–1147 (2001).
Ahrens, E.T., Flores, R., Xu, H. & Morel, P.A. In vivo imaging platform for tracking immunotherapeutic cells. Nat. Biotechnol. 23, 983–987 (2005).
Gilad, A.A. et al. Artificial reporter gene providing MRI contrast based on proton exchange. Nat. Biotechnol. 25, 217–219 (2007).
Kostura, L., Kraitchman, D.L., Mackay, A.M., Pittenger, M.F. & Bulte, J.W. Feridex labeling of mesenchymal stem cells inhibits chondrogenesis but not adipogenesis or osteogenesis. NMR Biomed. 17, 513–517 (2004).
de Vos, P., Faas, M.M., Strand, B. & Calafiore, R. Alginate-based microcapsules for immunoisolation of pancreatic islets. Biomaterials 27, 5603–5617 (2006).
Kakizawa, Y. et al. Controlled release of protein drugs from newly developed amphiphilic polymer-based microparticles composed of nanoparticles. J. Control. Release 142, 8–13 (2010).
Meunier, C.F., Dandoy, P. & Su, B.L. Encapsulation of cells within silica matrixes: towards a new advance in the conception of living hybrid materials. J. Colloid Interface Sci. 342, 211–224 (2010).
Zustiak, S.P. & Leach, J.B. Hydrolytically degradable poly(ethylene glycol) hydrogel scaffolds with tunable degradation and mechanical properties. Biomacromolecules 11, 1348–1357 (2010).
Orive, G. & Pedraz, J.L. Highlights and trends in cell encapsulation. Adv. Exp. Med. Biol. 670, 1–4 (2010).
Gobin, Y.P., Vinuela, F., Vinters, H.V., Ji, C. & Chow, K. Embolization with radiopaque microbeads of polyacrylonitrile hydrogel: evaluation in swine. Radiology 214, 113–119 (2000).
Horak, D. et al. Hydrogels in endovascular embolization. III. Radiopaque spherical particles, their preparation and properties. Biomaterials 8, 142–145 (1987).
Jayakrishnan, A., Thanoo, B.C., Rathinam, K. & Mohanty, M. Preparation and evaluation of radiopaque hydrogel microspheres based on PHEMA/iothalamic acid and PHEMA/iopanoic acid as particulate emboli. J. Biomed. Mater. Res. 24, 993–1004 (1990).
Megibow, A.J. & Bosniak, M.A. Dilute barium as a contrast agent for abdominal CT. AJR Am. J. Roentgenol. 134, 1273–1274 (1980).
Rabin, O., Manuel Perez, J., Grimm, J., Wojtkiewicz, G. & Weissleder, R. An X-ray computed tomography imaging agent based on long-circulating bismuth sulphide nanoparticles. Nat. Mater. 5, 118–122 (2006).
Ponce, S. et al. Chemistry and the biological response against immunoisolating alginate-polycation capsules of different composition. Biomaterials 27, 4831–4839 (2006).
Morch, Y.A., Donati, I., Strand, B.L. & Skjak-Braek, G. Effect of Ca2+, Ba2+, and Sr2+ on alginate microbeads. Biomacromolecules 7, 1471–1480 (2006).
Haque, T. et al. Superior cell delivery features of poly(ethylene glycol) incorporated alginate, chitosan, and poly-L-lysine microcapsules. Mol. Pharm. 2, 29–36 (2005).
Omer, A. et al. Long-term normoglycemia in rats receiving transplants with encapsulated islets. Transplantation 79, 52–58 (2005).
Bulte, J.W. Hot spot MRI emerges from the background. Nat. Biotechnol. 23, 945–946 (2005).
Stuber, M. et al. Positive contrast visualization of iron oxide-labeled stem cells using inversion-recovery with ON-resonant water suppression (IRON). Magn. Reson. Med. 58, 1072–1077 (2007).
Dufrane, D., Goebbels, R.M., Saliez, A., Guiot, Y. & Gianello, P. Six-month survival of microencapsulated pig islets and alginate biocompatibility in primates: proof of concept. Transplantation 81, 1345–1353 (2006).
Acknowledgements
The development of this synthesis protocol was supported by a Howard Hughes Medical Institute Research Training Grant (B.P.B.), NIH grants K08 EB004348 (A.A.), R01 HL073223 (D.L.K.), R01 EB007825, U54CA151838 (J.W.M.B.) and RO1 HL084186 (M.S.).
Author information
Authors and Affiliations
Contributions
B.P.B., A.A., D.R.A., M.S., D.L.K. and J.W.M.B. designed the experiments and analyzed the data. B.P.B., A.A., M.S. and D.L.K. conducted the experiments and collected data. B.P.B. and J.W.M.B. wrote the article.
Corresponding author
Ethics declarations
Competing interests
The authors have a patent pending that describes the methods of composition and use of these capsules.
Rights and permissions
About this article
Cite this article
Barnett, B., Arepally, A., Stuber, M. et al. Synthesis of magnetic resonance–, X-ray– and ultrasound-visible alginate microcapsules for immunoisolation and noninvasive imaging of cellular therapeutics. Nat Protoc 6, 1142–1151 (2011). https://doi.org/10.1038/nprot.2011.352
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2011.352
This article is cited by
-
Synthesis of hollow magnetic carbon microbeads using iron oleate@alginate core-shell hydrogels and their application to magnetic separation of organic dye
Korean Journal of Chemical Engineering (2020)
-
Quantitative CT and 19F-MRI tracking of perfluorinated encapsulated mesenchymal stem cells to assess graft immunorejection
Magnetic Resonance Materials in Physics, Biology and Medicine (2019)
-
Tracking of stem cells in vivo for cardiovascular applications
Journal of Cardiovascular Magnetic Resonance (2014)
-
Imaging macrophages with nanoparticles
Nature Materials (2014)
-
Endogenous release of female hormones from co-microencapsulated rat granulosa and theca cells
Biomedical Microdevices (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.