Abstract
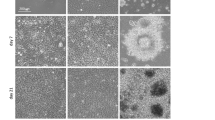
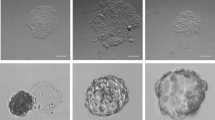
The culture of human embryonic stem (hES) cells in defined and xenogeneic-free conditions will contribute substantially to future biotechnological and medical applications. To achieve this goal, we developed the first fully defined synthetic polymer coating poly[2-(methacryloyloxy)ethyl dimethyl-(3-sulfopropyl)ammonium hydroxide] (PMEDSAH) that sustains long-term growth of hES cells in different culture media. Here we describe a detailed protocol for the reproducible fabrication of PMEDSAH coating on tissue culture polystyrene dishes, and for the feeder-free culture of hES cells on PMEDSAH coating in defined culture medium. This culture system represents a key step toward the fully defined and xenogeneic-free culture of hES cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Thomson, J.A. et al. Embryonic stem cell lines derived from human blastocysts. Science 282, 1145–1147 (1998).
Xu, C. et al. Feeder-free growth of undifferentiated human embryonic stem cells. Nat. Biotechnol. 19, 971–974 (2001).
Martin, M.J., Muotri, A., Gage, F. & Varki, A. Human embryonic stem cells express an immunogenic nonhuman sialic acid. Nat. Med. 11, 228–232 (2005).
Ludwig, T.E. et al. Derivation of human embryonic stem cells in defined conditions. Nat. Biotechnol. 24, 185–187 (2006).
Wang, L. et al. Self-renewal of human embryonic stem cells requires insulin-like growth factor-1 receptor and ERBB2 receptor signaling. Blood 110, 4111–4119 (2007).
Genbacev, O. et al. Serum-free derivation of human embryonic stem cell lines on human placental fibroblast feeders. Fertil. Steril. 83, 1517–1529 (2005).
Li, Y., Powell, S., Brunette, E., Lebkowski, J. & Mandalam, R. Expansion of human embryonic stem cells in defined serum-free medium devoid of animal-derived products. Biotechnol. Bioeng. 91, 688–698 (2005).
Villa-Diaz, L.G. et al. Synthetic polymer coatings for long-term growth of human embryonic stem cells. Nat. Biotechnol. 28, 581–583 (2010).
Nagaoka, M., Si-Tayeb, K., Akaike, T. & Duncan, S.A. Culture of human pluripotent stem cells using completely defined conditions on a recombinant E-cadherin substratum. BMC Dev. Biol. 10, 60 (2010).
Rodin, S. et al. Long-term self-renewal of human pluripotent stem cells on human recombinant laminin-511. Nat. Biotechnol. 28, 611–615 (2010).
Braam, S.R. et al. Recombinant vitronectin is a functionally defined substrate that supports human embryonic stem cell self-renewal via alphavbeta5 integrin. Stem Cells 26, 2257–2265 (2008).
Klim, J.R., Li, L., Wrighton, P.J., Piekarczyk, M.S. & Kiessling, L.L. A defined glycosaminoglycan-binding substratum for human pluripotent stem cells. Nat. Methods 7, 989–994 (2010).
Melkoumian, Z. et al. Synthetic peptide-acrylate surfaces for long-term self-renewal and cardiomyocyte differentiation of human embryonic stem cells. Nat. Biotechnol. 28, 606–610 (2010).
Brimble, S. et al. Karyotypic stability, genotyping, differentiation, feeder-free maintenance, and gene expression sampling in three human embryonic stem cell lines derived prior to August 9, 2001. Stem Cells Dev. 13, 585–597 (2004).
Maitra, A. et al. Genomic alterations in cultured human embryonic stem cells. Nat. Genet. 37, 1099–1103 (2005).
Baker, D.E.C. et al. Adaptation to culture of human embryonic stem cells and oncogenesis in vivo. Nat. Biotechnol. 25, 207–215 (2007).
Wu, J.M. et al. A surface-modified sperm sorting device with long-term stability. Biomed. Microdevices 8, 99–107 (2006).
Ware, C.B., Nelson, A.M. & Blau, C.A. A comparison of NIH-approved human ESC lines. Stem Cells 24, 2677–2684 (2006).
Villa-Diaz, L.G. et al. Analysis of the factors that limit the ability of feeder-cells to maintain the undifferentiated state of human embryonic stem cells. Stem Cells Dev. 18, 641–651 (2008).
Acknowledgements
H.N. acknowledges funding from the University of Michigan Rackham Predoctoral Fellowship. J.L. gratefully acknowledges support from the NSF in form of a CAREER grant and funding from the NSF under the MRI program. This research was supported by NIH grants P20 GM-069985, R01 DE016530 and the NIDCR T32 Tissue Engineering and Regeneration Training Program.
Author information
Authors and Affiliations
Contributions
This protocol was developed and written by H.N. and L.G.V.-D. after obtaining the material from all authors. H.N. designed the 'Fabrication of PMEDSAH-coated TCPS dishes' portion of the protocol; L.G.V.-D. designed the 'Establishment of hES cell cultures on PMEDSAH-coated TCPS dishes' portion of the protocol. K.S.O., G.D.S., P.H.K. and J.L. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Nandivada, H., Villa-Diaz, L., O'Shea, K. et al. Fabrication of synthetic polymer coatings and their use in feeder-free culture of human embryonic stem cells. Nat Protoc 6, 1037–1043 (2011). https://doi.org/10.1038/nprot.2011.342
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2011.342
This article is cited by
-
Synthetic alternatives to Matrigel
Nature Reviews Materials (2020)
-
Human embryonic stem cell cultivation: historical perspective and evolution of xeno-free culture systems
Reproductive Biology and Endocrinology (2015)
-
Long-term xeno-free culture of human pluripotent stem cells on hydrogels with optimal elasticity
Scientific Reports (2015)
-
Production of Human Pluripotent Stem Cell Therapeutics under Defined Xeno-free Conditions: Progress and Challenges
Stem Cell Reviews and Reports (2015)
-
Materials as stem cell regulators
Nature Materials (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.